Book - Physiology of the Fetus 11
| Embryology - 28 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Windle WF. Physiology of the Fetus. (1940) Saunders, Philadelphia.
1940 Physiology of the Fetus: 1 Introduction | 2 Heart | 3 Circulation | 4 Blood | 5 Respiration | 6 Respiratory Movements | 7 Digestive | 8 Renal - Skin | 9 Muscles | 10 Neural Genesis | 11 Neural Activity | 12 Motor Reactions and Reflexes | 13 Senses | 14 Endocrine | 15 Nutrition and Metabolism | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter XI Conditions Regulating Fetal Nervous Activity
There is no doubt that the progressive development of nervous function is related to difkerentiation of structure in the fetal nervous System. 1t was hoped that many correlations like those discussed in Chapter X could be drawn between specific responses and the appearance histologically of new neura1 connections. This is not an easy task because structural growth proceeds with great rapidity and results in the establishment very early of complexities defying microscopic ana1ysis. 0n the other hand, function in the central nervous system of the fetus is not regulated solely by structural factors. 0ne must not lose sight of metabolic influencesz it is quite clear that variations in fetal activities are closely related to changing respiratory conditions. The parts played by the blood, the heart and vascular system and even the endocrine glands have to be determinedh Many problems in physiology of the fetal nervous system await solution, but it will be well to view some of them, even though questions raised thereby can not be given satisfactory answers.
The Plan of Structural Development of the Fetal Brain
Knowledge of development of intrinsic brain structure is far from complete, for it ha’s been within the past few years only that systematic studies in specimens prepared by adequate methods were undertaken. The« usual histologic procedures are unsatisfactory to demonstrate embryonic nerve übers, their terminations and relations to one another inside the brain and spinal cord. Cajal’s silver stains, especially the Ranson modification, bring out details of this nature incomparably better than any other known techniqueJ Procedures of this type are being used extensively for studying the prenatal mammalian nervous system in this2-I2 and the Madrid13-I7 laboratories, as well as in a few other places.18-22
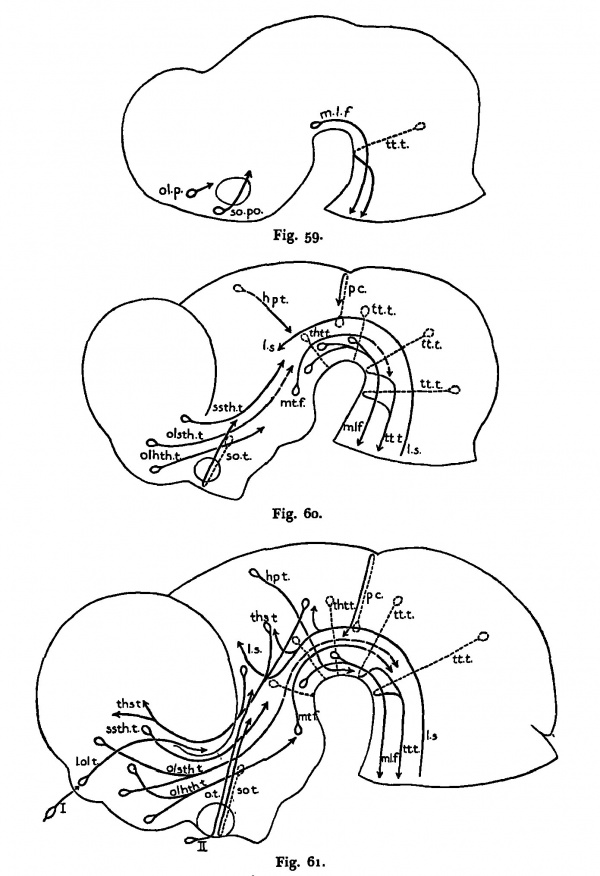
By the time fetal movements can be elicited for the Hrst time in any species, a surprisingly exfensive organization ok neuron groups and fiber tracts has already formed within the central nervous System. We have discussed certain correlations between intrinsic spinal connections and the first· forelimb reilexes. In the brain equally specific correlations have not been worlced out completely. Most of the tracts and nuclei have been identilied in cat embryos and those that are present at the time« behavior has its genesis are listed in Table 2 I. Figss 59 to 61 illustrate diagrammatically and incomp1etely this extensive development of tracts ich the rostral portions of the embryonic brain.
Figs. 59-61. Diagrams of the brains of cat embryos 7 mm. (Fig. 59) , 10 mm. (Fig. 60) and 15 mm. (Fig. 61) C. R. length showing the principal über tracts present in each. Crossing neurons are dotted lines. Questionable courses: dash lines.
| Name of fibre group | Abbreviations used in figs. 59-61 | Size of smallest embryo (mm) in which it was found |
|---|---|---|
| Medial longitudinal fascicle | m.l.f. | 5.0 |
| Supraoptic system: direct preoptic component | so. po. | 6.0 |
| Supraoptic system: commissural component | so. t. | 7.0 |
| Olfacto-hypothalamic fibers | olhth. t. | 7.0 |
| Olfacto-subthalamic fibers | olsth. t. | 8.0 |
| Strio-subthalamic fibers | ssth. t. | 8.0 |
| Direct subthalamo-tegmental fibers (diffuse) | 8.0 | |
| Crossed pretecto-tegmental and thalamo-tegmental fibers (ventral commissure) | tho t. j tht. t. | 8.0 |
| Lemniscus system | l. s. | 8.0 |
| Terminal nerve fibers | 10.0 | |
| Posterior commissure fibers | p. c. | 10.0 |
| Habenulo-peduncular fibers | hp. t. | 10.0 |
| Lateral olfactory tract fibers | l. ol. t. | 11.5 |
| Mammillo-tegmental tract fibers | mt. f. | 11.5 |
| Optic nerve fibers | II | 11.5 |
| Thalamo-strial and thalamo-cortical fibers | ths. t. | 11.5 |
| Mammillo-thalamic fibers | 13.5 | |
| Olfactory nerve fibers | I | 13.0 |
(a) Order of Development in Functional Systems
Within the growing tangle of nerve iibers it is possible to distinguish several systems of functionally related tracts. They appear to be growing in anticipation of the time they will conduct impulses to distant elfector organs. such systems of tracts are laid down economicallyz for the most part they pursue the shortest possible courses from place to place. 0ne .of the most interesting features of growth of each System is the order in which its component tracts are formed. This is from motor toward SenSory side.
The very Erst neurons which can be recognized in the embryo are primary motor e1ements of the spinal and cranial nerveS, present in rat embryos only z mm. long and appearing in the other species at comparable Stages. These are final common path neurons over which impulses must ultimately pass to bring about responses in the muscleS of. the body. They are the first, local connecting neurons are second and the primary afferent tracts appear third.
The earliest secondary connecting neurons make their appearance in 17 day old cat embryos in the portion of the medulla oblongata which will become the reticular formation. Growth very soon spreads into the upper spinal cord segmentS. There, nerve cells give rise to crossed and direct axons which make local connections with primary motor elementS. Some in the medulla oblongata form the earliest reticuloSpinal übers, but they do not extend their procesSes far along the nerve axis. secondary elementS of the spinal cord enter into the formation of local conduction Systems, all components of which are not present how— ever until primary afferent neurons begin to build the sensory tracts. Even then, the local Systems do not become functional retlex mechanisms until Synaptic relations between the three elements-—motor, connector and sensory—have been eStablished on the 24th day in the cat embryos. similar reflex arcs are completed during the 8th week in man.
Almost simu1taneously with the appearance of secondary neurons in the medulla oblongata, another group begins to be laid down in front of the mesencephalon. FiberS from this source descend to the lower part of the brain stem without crossing and form one component of- the medial longitudina1 kascicle. 1t seems probable that the early secondary neurons are related to more than one baSic rellex System. Just as the lower motor neurons are not an exclusive component of any one System, so these interneurons may be shared by several.
Secondary neurons malce their appearance in still another location in .the early embryo. crossing and direct ftbers arise in the mesencephalic tectum and form the tectobulbar and tectospinal tracts. Thus we lind essential1y four groups of secondary neurons undergoing developrnent in the Central nervous system between the 18th and 2oth days-of gestation in the cat and during the 5th weelc in man. These are (a) local intraseginental and intersegmental elements, such as later constitute the ground bundles of the spinal cord, (b) reticulospinal tracts, (c) medial longitudinal fascic1es and (d) tectobulbar and tectospinal tracts. Few übers course farther caudally than the lower end of the medulla oblongata until a day or so after initiation of the first individual head and forelimb ref1exes· However, it can not be doubted that one or more of these tracts play a part in the earliest cephalocaudal integration of movements which occurs on the 25th day in cat embryos.
Secondary neurons of the spinal cord, especially commissural, course rostrally in the ventral and lateral funiculi. These are present before a signiiicant number of descending iibers from the brain reach the cord. They account for conduction of impulses from caudal to rostral segments and explain the observation of forelimb and head reflexes which sometimes follow stimulation of more caudal structures.
(b) Growth of Other Circuits for Reflexes and Higher Integration
Just as the intrasegmental neurons of the spinal cord enter into the structure of basic short reflex circuits, reticulospinah tectospinal and medial iongitudinal iibers constitute Iinlcs in Ionger circuits which becotne functional much later than the local ones. Examples of such- systems are the olfactory and optic reflex mechanisms of the early embryo. Analysis of the growth of these systems shows that theircomponent tracts too are laid down from efferent to afkerent side. For example, during development of the optioreflex conduction mechanism the primary spinal motor and the oculomotor neurons begin to form Erst, secondary neurons represented by the medial longitudinal and tectospinal tracts are next, the posterior commissure follows, the optic tract itseIf is fourth, and linally the retinal bipolar neurons and visua1 cells appear. similar sequential development seems to be the rule in other reiiex conduction systems.
Within any conduction system such as the optic or olfactory, the simplest reflex pathways are« formt-d« before those which have to do with perception and higher integrative activities. Neurofibrillar development is late in the cerebellum, corpus striatum and cerebral cortex. A primitive secondary aiferent tract, medial lemniscus in part, reaches the thalamus of cat embryos from spinal and bulbar centers by the 2 ist day of gestation. Fibers begin to pass from the thalamus to the cerebrum on the 22nd day, but not until late in fetal life does the cerebral cortex exert any influence over lower motor parts of the nervous System. The pyramidal tracts are last to form.
Correlated with the tardy development of cerebral cortex, it has been determined that cerebral electric potentials are absent throughout most of prenatal life. Even in the guinea pig, an animal which is much more mature than the rat, cat or man at the time of birth, they do not manifest themselves until about 2 or z weelcs before the end of the gestation periodks A cortical control of the motor mechanism for forelimbs has been established at the time of birth in the cat, but the hind limbs laclc it until 16 days later.24
(c) Myelogeny as Related to Function in the Nervous System
Many neurologists adhere to the theory that the initiation and maturation of function of the nervous system depends upon the formation of myelin sheaths. FlechsigV called attention to the fact that the progress of deposition of myelin is orderly and that tracts having definite functions become myelinated at different times in the human infant’s brain. Some investigatorsW who have studied the course of development of behavior in lcittens and have attempted to correlate it with myelin formation have suggested functional relationships. 0thers24-27-31 have carried out similar studies in pouch-young opossum, Icitten and human infant brains as well as in the fetal nervous system of the cat and man. In a general way it seemed that maturation of behavior and the acquisition of myelin sheaths of certain fiber tracts were related, but it was impossible to draw specific correlations in all cases. For example, the corticospinal tract is still unmyelinated at birth but cortical areas for control of forelimbs are electrically excitable.
They can be a great deal of well organized activity in the brain before any nerve libers become myelinated« In the cat, myelin is present neither in the peripheral nerve roots nor in the tracts of the spinal cord and brain before the 42nd day of fetal life.33 But 30 day old cat· fetuses can execute rhythmical respiratory and other coordinated reflex movements similar to those employing myelinated tracts at a later time. The early behavioral reactions of the rat are certainly executed in the absence of myelinated nerve fibers.32- 34
An attempt was made to correlate specific righting reflexes of cat fetuses with development of myelin sheaths on the nerve fibers which were involvedks It was demonstrated that the vestibular righting reaction appears coincidentally with sheaths upon the iibers of the vestibular nerve and that conduction pathways used in the reaction are partially myelinated when the reflex first occurs. However, the righting response to sensory impulses from the slcin and deep tissuesof the body, i. e» a body righting reflex, is manifested before that employing the labyrinthine apparatus. Its neural mechanism is incompletely myelinated at the time.
It is possible that myelination is more closely related to the order of development of tracts in the embryonic nervous system than to specific functional activities. Thus we find parts of the medial longitudinal and reticulospinal systems of neurons appearing early and receiving their myelin sheaths first. But the correlation is not absolute and many discrepancies can be ob— served. About all that one can say is that the first tracts to develop in the embryo are the first to begin to be myelinated and the last to form in the late fetus are the last to receive sheaths. Some tracts never develop signiftcant numbers of myelin sheaths. It is quite probable that conduction of impulses may ,be improved with the acquisition of» myelin, but myelination is certainly not an essential corollary of function. With increasing fetal si2e, distances between points in the nervous system become greaten Perhaps myelin is laid down to compensate by increasing the conduction speed of the iibers.
Factors Other than Structural Growth
(a) The Quiescence of Intra-Uterine Life
It is relatively easy to elicit nervous activities throughout the greater part of prenatal life in the guinea pig, cat, sheep and man under certain experimental conditions. But it should not be assumed that all responses which can— sbe induced occur spontaneeously within the uterus of the normal intact individual. As a matter of fact, there is scanty evidence that any of them occur normally during the early part of the gestation period.
Human fetal movements can be detected as early as the 14th weelc by means of a stethoscope, but the mother is usually unable to feel them before the 17th weelc of gestation. It is difhcult to diagnose them accurately much before the latter time without considerable experiencttz for they are often confused with the sounds produced by movements of intestinal gases. Fetuses of experimental animals appear to be singularly quiet until late in prenatal life when occasional quiclc jerlcs or twitches can be ob— served upon the maternal abdomen. surprisingly little fetal activity is seen even when the thin-wal1ed uterus is delivered under local anesthesia.
Although the few fetal movements which are readily visib1e in the intact individual seem to be purposeless, we know that some well coordinated and useful activities do talce place normally during the second half of prenata1 life. For examplq intrauterine swallowing has been proved to be a normal physiologic func— tionks This is an activity engaged in with great regularity during the last third of gestation in the guinea pig. We do not lcnow its cause (see Chapter VII) .
(b) Afferent Stimulation In Utero
The relative quiescence of the normal fetus in utero is somewhat surprising when one considers all the activities of which the growing specixnen is capable when removed from the uterus. The reason seems to be at least twofold: laclc of adequate stimulation and high thresholds in the fetal central nervous System. The fetus is adequately nourished and warmed in a medium laclcing practically all the stimulating iniluences of the environment with which it will have to cope later on. No signiftcant excitation of the external receptors occurs.
Experimental evidence in the cat supports the view that there is little spontaneous motor discharge in the absence of afferent impulses. In several hundred embryos and young fetuses de1ivered under good physiologic conditions and without using anesthesia. spontaneous movements have rarely been seen at the moment of delivery. They make their appearance within a few seconds or minutes. apparently because placental exchange has been jeopardized or becaIuse changes in the environment cause stimulation. One can not avoid manipulation entirely, and even though every effort is made to maintain the placental circulation intact, incision of the uterus disturbs the relationship between uterus and placenta. .The resulting anoxemia accounts partially for the movements.
If there were a true automaticity of embryonic motor neurons, spontaneous movements should be observed in many instances at the moment the specimens are brought into view. But there is very little motor discharge without afkerent stimulation. Rhythmical movements have been seen in sheep embryos 40 to 50 days old at the moment of delivery and it has been suggested that the responses are automatic« Some are- defmitely initiated by mechanical stimulation in the younger embryos. It was proposed that the automatic movements of the sheep fetus become inhibited as soon as descending secondary tracts grow down into the spinal cord from the brain stem. In the cat. no rhythmical movernents can be obtained until after tracts have. grown down; responses between 23 and 28 days of gestation are neither automatic nor rhythmic.
(c) Neural Threshold to Stimulation
Later in prenatal life all fetuses become less responsive to stimulation than they were at first. How much this may be due to inhibition over newly developed descending pathways from the brain we do not know. Guinea pig and cat fetuses near term do not respond activeIy to ordinary manual palpation through the intact abdomen but they can be aroused from their profound «slu1nber« by priclcing or prodding them with a needle thrust into the abdomen and uterus. It seems reasonable to conclude that the fetal nervous system has developed high thresholds. At any rate, fetal motor centers are less excitable than they were earlier in prenatal life and less excitable than those of newborn individuals.
But the excitability of fetuses in utero can be enhanced ex— perimentally. One way to do so is to reduce the oxygen available in the fetal brain without creating complete asphyxia Partial anoxemia at all times in prenatal life predisposes toward an in— crease in fetal movementsz but- most activities are not actually induced through the internal environment b"y chemical stimuli.
They follow mechanical stimuli which were subliminal before the anoxemia was set up. This was demonstrated in experiments like the following one.
At 63 days of gestation, fetuses of a decerebrated cat were observed to be very quiet in uter0. When a needle was passed through the abdominal wall and into the Uterus, the fetuses responded to proddingx they executed brief kicks on non-respira— tory jerks of the head which stopped almost immediately after stimulation ceased. With the needle still in place, the cat was allowed to rebreathe air in a rubber tube with wide bore. stimulation of the fetuses was repeatedz this time the responses were much more active, the fetuses kicked and squirmed for some time after the cessation of the stimulus and the movements were tonic and sustained. The rebreathing tube was then removed and as soon as the mother’s breathing had become normal again the fetuses were restimulated. Results were obtained like those before the anoxemia. One experiment of this type is illustrated in the parts of a continuous record reproduced in Fig. 62.
Fig. 62 Three Portions of a crystograph record showing maternal respirations (1arge was-es, 16 per minute) broken up by intrauterine fetal movementsh The cat (63 days gestation) had been decerebrated by the anemia method; no anesthesia was used during the experimentz the abdornen was not opened but a long needle had been passed through the abdominal wa11 into one amniotic sac. At the heavy solid lines, the fetus was stimulated with the needle and the irregular defiections on the record at these points are due to this mechanical eikech Fetal rnovenients were observed at the points indicated by the broken linesz these movements were siight in records i and z, during which the cat was breathing air. In reoord e, the cat was rebreathing to induce a partia1 anoxemiaz stimulation led to marked proionged fetal rnovements of a tonic squirming type (double broken 1ines) which caused considerable interference in the maternal respiratory record.
Certain very rapid rhythmica1 movements of respiratory muscles do appear to be elicited by endogenous chemical stimulation of the fetal respiratory center.37 They can be induced by raising the carbon dioxide level in the blood during the early part of active fetal life, but can not be called forth by this method in the late fetal period unless a rather marked oxygen deficiency is brought on too. These observations suggest a rising threshold in the fetal nervous system of the cat with advancing prenatal age.
Respiratory rhythms are quite independent of most other somatic movements in the cat. At the time they are first obtain— able, they involve only the muscles which are normally used by the adult for breathing, i. e» the diaphrag·m, intercostals and abdominals A little later in fetal life, they can be made to involve neclc and trunlc muscles if the degree of anoxemia used to elicit them is increase-d. Respiratory rhythms often set off other somatic movements in cat fetuses but are themselves less frequently started by some non-respiratory twitch. Indeed, the cat fetus when stimulated in the intact uterus can be induced to lcick or move its head vigorously without any movement of a respiratory nature following.
(d) Muscle Tonus and Mass Movement
Not only does anoxemia facilitate the efkectiveness of subliminal mechanical stimulation and induce automatic rhythmical respirationdilce movements but it also brings about changes in the character of motor responses when its severity is increased beyond the point of facilitation. Early in the gestation period of the cat, local reflexes of the limbs are abolished more readily than those of the trunlc and neclc under anoxemia. Throughout the middle of prenatal life, stimulation of anoxemic fetuses leads to responses which resemble mass reactions, any adequate stimulus eliciting not local movements but generalized activities such as squirming. 38
Movements of anoxemic cat fetuses laclced the "jerky" quality they had shown beforehand and became more sustained and tonic. A single stimulus often results in repeated movements suggestive of considerable aftepdischarge in motor centers. Under marked anoxemia, such as that following occlusion of the umbilical cord, fetal muscles sometimes become so hypertonic that the fetus resembles a decerebrate animaL similar postures have been observed in lcittens and rabbits (see Fig. 64) decerebrated by sectioning the brain at the rostral border of the mesencephalon.39 The increase in muscle tonus under asphyxial conditions may be interpreted as a protective mechanism. It acts to insure the expansion of the chest which is necessakyfor air breathing.40
To what extent anoxemia is normal and physiologic in prenatal life is not Known.* 0bservations in early stages of several species of animals suggest that it may follow experimental procedures more readily in some than in others. This may be due in part to species variation in the placenta as an organ for oxygen exchange in early fetal life. It is possible that embryos of animals like the sheep, which have rather primitive syndesmochorial p1acentas, may tolerate operative procedures involving sdme manipulation of the uterus less well than other forms such as the cat or guinea pig, which have their maternal and fetal blood streams in more intimate contact (see Chapter I) . Theoretically, an equal amount of trauma would be more disastrous in the former than in the latter. The fact that it was more diilicult to observe the very first reliexes in sheep than in cat and guinea pig embryos and that there is more of a tendency for movements, sekjningly automatic, to manifest themselves in sheep than in cat embryos, would iit into such a conception.
- see J. Barcrofh et a1., 194o, J. Physiol» 97: 338, 347.
(e) Susceptibility Gradients to Asphyxia
The exact nature of changes which depress neural thresholds under oxygen deliciency is unknown. They may be chemical or physical. some investigators have suggested that certain fetal movements result from stimulation of the nervous system by accumulating metabolic end-products, principally carbon dioxide.41-44 Although it is true that automatiq rhythmicaL respiratory movements can be initiated by increasing the carbon dioxide content of fetal blood experimentally, those induced by asphyxiation may be related less to an increase in the chemical stimulus than to a depression of thresholds (increase of neuron excitability).
It has been postulated45, 46 that endogenous (chemical) stimulation afkects the motor centers directly and acts first on the most recently developed units, that the new neurons have the highest physiologic gradient and are consequently stimulated first by accumulating metabolites in the blood. The evidence is open to question because experimental conditions were not well controlled. The embryos were studied in unbalanced saline solutions. It has been demonstrated more recently« that the waving of embryonic limbs and tail, which suggested the theory, occurs only in embryos placed in solutions delicient in calcium and potassium and is not necessarily related to accumulating metabolites.
Under asphyxia, the behavior of fetal sheep tends to revert to a type characterizing younger specimens but a clear cut recapitulation of development of reilex movements was not found« simple reflexes most recently acquired apparently were not called forth during asphyxia. In the catss it has been observed that asphyxia abolishes activities in a rather orderly manner. It has a more destructive action upon the appendicular motor mechanism than upon that of the neclc ånd trunlc. The last activity to disappear under asphyxia is a respiratory movement of the chest. This conlirms a previous observation in bird fetuses-S; it was found that deep rhythmical gasping could be induced repeatedly by tying and untying the allantoic vessels. It seems probable that motor centers show gradients of susceptibility to asphyxia but there is no evidence that the oldest reflexes, i. e» those of the neclc and forelimb, are the last to be afkected by the asphyxia.
(f) Inhibition of Motor Mechanisms by Higher Centers
It has been suggested that the changes ·in behavior of sheep fetuses under asphyxia may result from the removal of inhibitory inliuences of new descending nerve tracts of higher order upon lower motor neurons« This view conforms to the conception of a gradient of susceptibility to destruction by asphyxia, the new higher order neurons being thrown out of function before the older motor neurons. It also assumes some sort of automaticity in the lower motor centers.
Transection of the fetal brain stem and spinal cord at various levels below the mesencephalon was performed in ten fetuses zo to 76 days old without removing them from the uterusås 0ne to 13 days later the abdomens of the ewes were opened again and the fetuses delivered by Caesarean Section. Fetal movements at that time resembled those characterizing unoperated specimens at 40 to 50 days gestation. It was suggested that the operations had released lower motor mechanisms from inhibitory influences of nervous centers above the transection level in the region of the red nucleus. 0n the other hand one investigator49 encountered no qualitative difference in behavior of newborn rats whose cerebrum (alone or with other parts of the brain above the medulla oblongata) had been destroyed in utero and their normal litter mate controls.
It is probably true that sheep as well as other mammalian fetuses are endowed with relatively lower thresholds in early fetal life than later on toward term, and that structural growth within the brain plays an important part in determining the nature of thresholds The theory that motor centers are held in checlc by the descending tracts is a very attractive one and deserves careful study, but more experiments in other species of animals must be performed before it can be proved.
0ther studies have been made with rssults suggestive of the phenomenon in question. Minlcowslciso observed a reversal of the type of response in the human plantar reflezc During the course of its development plantar flexion preceded dorsal flexion from stimulating the sole, and after the latter had become well established it commonly changed baclc to plantar ilexion under narcosis as well as progressive asphyxia. 0thers have disagreed with his interpretationsJU and Personal experience has shown that the Babinslci reflex (dorsal ilexion) of the human fetus is remarlcably resistant to asphyxia. When it does succumb, the plantar flexion which remains appears to result from direct stimulation of the muscles in the sole of the foot.
The earliest head movemen of cat embryos are contralateral. With further development theyschange to homolateral responses, but under asphyxia they sometimes become contralateral again. Extension of the head of the early sheep fetus usually accompanies the respiratory-lilce rhythms of movement, but after a time, during which a partial anoxemia builds up, extension changes to ilexionR These and other examples show how progressive asphyxia exerts selective action upon the central nervous System. similar results have been observed in adult animals. 53
References Cited
. Davenport, I-I. A» W. F. Windle s: R. I-I. Beech. 1934. stain Tech., g: Z. . Windle, W. F. 1931. J. comp. Neur., Zzx 71.
. Windle, W. F. 1932 Ibid., 55: gg.
. Winde, W. F. 1932 Ibi·d., 55: gis.
. Windla W. F. 1g33. Ibid., 58: 643.
. Windle, W. F. 1934. Ibid., 59: 487.
. Windle, W. F. 1g35. 1bid., 63: 139.
. Windle, W. F. s« R. F» Baxten 1936. Ibids Eis: 173.
ON! CAN-Poe v s—172 . Windle, W. F. sc R. E. Baxten
. Tello, J. F.
. Trillo, F. i938.
. Lanyworthy, O. R. . Langworthy, O. R. i933. contn Ernb» e4: Z.
. Angulo y Gonzalen A. W. i9e9. J. Cornp. Neur., 48: 459. . Windle, W. F., M. W. Fish sc J. E. O’Donnell.
. Beclcen R. F., W. F. Windle, E. E. Barth sc M. D. Schule.
iskiYsionocY or· THE. Fisrus
i936. Ibid.- As: i89. Windle, W. F. i937. Proc.· soc. Expen Biol. sc Med., 36: 64o.
. Windle, W. F. sc J. E. Fitzgeralch ·i937. J. Comp. Neur., 67: 493.
scharpenbergx L. G. sc W. F. Windle. i938. J. Anat» 7e: 344. Tello, J. F. i934. Ztschn inilc.—anat. For-seh» 36: See.
Tello, J. F. i934. Trav. Lab. Rech. Biol., Univ. Madrid, e9: 339. i935. Ibid» so: 447.
i936. Ibid» Si: 77.
Ibid., se: i. .
Bok, s. T. i9e8. in W. von Möllendorkks Handbuch mik. Anat. Mensch.,
4 (I)- 478— shanen R. F. i93e. J. cotnp. Neun, H: 493. shanen R. F. i934. Ibid» 6o: z.
Tello, J. F.
. shanen R. F. i934. Anat» 68: 3i4. . Eos-g, l. D. Cited by D. Hooken
i936. Yale J. Biol. sc Med., 8: 579.
Jaspen H. H» C. s. Bridgman sc L. catmichaeL i937. Expen Psychol» ei: 63.
Langworthy, O. R. i9e7. 'contr. Emb» i9: i77.
Flechsig, P. i876. Die Leitungsbahnen im Gehirn und Rüclcenmarlc des Menschen auk Grund entwiclcelungsgeschichtlicher Untersuchungen. W. Engelmann, Leipzig.
Tilney, F. sc L. casainajon i9e4. Arch. Neun sc Psychiat» ie- i.
Lang-worthy, O. R. i9e6. Contn Kind» i7: ie5.
Langworthy, O. R. i9e8. J. Comp. Neun, 46: eoi.
Lang-worthy, O. R. i9e9. contn Emb., so: ie7. Use. Arch. Neurol. sc Psychiat» es: i365.
i934. Ibid., 59: iZ9. Watson, J. B. i9o3. Animal Education. Univ. chicago Press. i94o. surg»
Gyn. sc Obst., 7o: sog. Barcrokn J. sc D. H. Barron. i937. J. Physiol» 9i: 3e9.
Windle, W. F» M. Monnier sc A. G. steeles i93s. Physiol. Zool» it: 4e5.
. Windle, W. F. sc R. F. Becken i94o. Arch. Neun sc Psychiat, 43: 9o. . Windle, W. F. . Hendersom Y. i937. science, 85: 89.
. Zuntn N. i877. Pklügens Arch., i4: sog. . Brown, T. G.
i9e9. Cornp. Neun, 48: 2e7.
i9i5. Physiol» 49: 2o8.
Grahain, E. A. i9i3-i9i5. Trans. Chicago Path. soc» g: ie3.
. Walz, W. i9ee. Monatschn Geburt. Gyn., 6o: zzn . Angulo y Gonzalen A. W. . Angulo y Gonzalen A. W. i934. Anat. Rec., 58: suppL 45.
i930. Proc. soc. Exp. Biol. sc Med., e7: 579.
Windle, W. F. i939. Physiol. Zool» re: 39.
. Windle, W. F. sc J. Barcrokn i938. Am. J. Physiol» iei: 684. . corey, E. L. . Minlcowskh M. i9e3. schweizer Arch. Neun Psychiat» i z: 475.
. Bolaklio, M. sc G. Artom. i9e6. Zeitschr. Neun Psychiat., io3: 3eo. . Barcrokn J. sc D. H. Barron. . Kaban H. sc c. Dennis.
i934. Proc. soc. Exp. Biol. sc Med., Zi- 95i.
ig36. J. Physiol» 88: 56. Proc. soc. Exp. Biol. sc Med., 38: 864.
Cite this page: Hill, M.A. (2024, April 28) Embryology Book - Physiology of the Fetus 11. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Physiology_of_the_Fetus_11
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G