2014 Group Project 3
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Gastrointestinal System
GIT system Overview
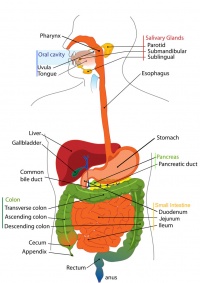
The GIT (gastrointestinal tract) system is a complicated system that extends from the mouth of the foregut to the anal canal of the hindgut. Its function is to ultimately turn food that is eaten into energy. The GIT system consist of the foregut, midgut and hindgut. Majority of the organs are located in the foregut. This includes the stomach, duodenum, Liver, pancreas and the spleen. Overall in fetal stage of the foregut development there is more rapid growth of already formed organs and important structures such as muscle layers emerge as well as bile production.[1] The midgut begins below the hepato-pancreatic ampulla and consist of the lower Duodenum, Jejunum, Ileum, Cecum, Appendix and the Ascending colon as well as the first two third of the transverse colon.In fetal development after the rotation and fixation of the midgut is complete, it starts to herniate at beginning of week 6 and continues to do so till week 10. [2] At week 11 midgut retracts back into the abdominal cavity and continues to grow. Hindgut begins from the left third of the transverse colon and ends to the cloaca (rectum). It consists of the left third of transverse colon, descending colon, sigmoid colon, rectum and anal canal. There is no rotation occurring in the hindgut instead it gets pushed to the left side by midgut during development.The colon matures and lengthens and the anal canal is formed.[3] The following page will explain the key fetal stage development of each segment of the Gastrointestinal tact (foregut, midgut and hindgut), some current research and models used to explain GIT system fetal development as well as the common fetal stage abnormalities and briefly list common abnormalities occurring in the embryonic stage.
Timeline
| GASTROINTESTINAL TRACT DEVELOPMENT | ||||
|---|---|---|---|---|
| Weeks | FOREGUT | MIDGUT | HINDGUT | |
| Embryonic Period | Week 4:
Week 5
Week 7: Week 8: |
Week 6: | Week 6: | |
| Fetal Period | Week 9-10:
Week 12: Week 13: Week 16 |
Week 9:
Week 10: Week 11: Week 16 Week 19: |
Week 9-10:
Week 8-12 Week 11 Week 12-15 Week 20 |
Current Research, Models and Historic Findings
Current ModelsFetal Intestine Transplant Injury Models 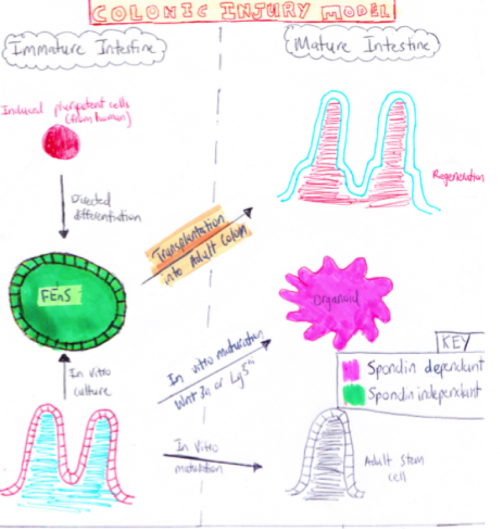 Diagram of Fetal Injury Colonic Transplantation Model[4] Using colonic injury models from both fetal humans and mouse it was found that the proliferative immature progenitor cells within the intestines could be expanded in vitro as Fetal Enterospheres (FEnS). Following transplantation after a in a colonic injury model, it was found that FEnS contribute to the regeneration of colonic epithelium by the formation of drypt-like epithelial structures that expressed region-specific differentiation markers. An overview of the process is shown in the digram on the right. This study revealed the existence of transitory populations of progenitor cells during the intestinal growth phase of both human and murine tissues[5].
Xenopus.Laevis model system revealing molecular pathway of gut adaptation from embryo to adult The gastrointestinal tract is remodeled from embryo to adult to adapt to changing dietary environment. To study the mammalian gastrointestinal development as well as to determine genes and signalling programs which are important for gut development and maturation the Xenopus.Laevis metamorphosis system became a great model system for use. A change in gene expression during mammalian intestinal maturation and postembryonic development was observed by investigating similarities in gene regulation between X.Laevis and mouse. Both systems have increase T3 level which circulates in the blood stream during the postembryonic development therefore the study focused on genes whose expressions increased with more T3 levels. Genes were therefore ranked based upon their relative expression at stage 61 From this gene expression profiles, 6 major gene clusters which are involved in intestinal transformation from embryo to adult were identified. The two clusters of genes had peaks at around birth in mammals. [6]The X.laevis model helped contribute in identifying novel embryonic and adult specific genes providing a better insight of the molecular regulation of GI development as well as its function. 17 embryonic specific and 52 adult specific genes were identified and showed relationship to the development and physiology of the organs of the GIT. For example in mature intestine there is significant increase of PRSS2 and PRSS3 enzymes.[6] The pancreas was the only organ that showed synthesize of serine proteinases (trypsins) during maturation. The comparative analysis showed that during mammalian intestinal development genes that peaked at stages 61 were significantly up-regulated at birth in mouse suggesting molecular signatures are highly conserved in more than one species during GI development and that T3 can have potential role of regulating target genes in intestinal development. TGF-B signalling pathway was analysed used GenMAPP database and it was seen to have proliferating role in intestinal maturation and cell proliferation. [6] Overall the gene expression studying with the utilisation of X.Laevis system provided molecular description of maturation and remodelling of the postembryonic developing Gastro intestine improving understanding of intestinal organogenesis as well as the nature of molecular regulation during fetal development. |
Current ResearchFindings on Omphalocele: (Sonic hedgehog expression in the development of hindgut) In a recent research experiment, the Hh hedge-hog signalling pathway was believed to be a possible causative factor of omphalocele formation. The TM -inducible gene recombination system was used to show association of Hh signalling with omphalocele. Omphalocele was prominently observed in embryos from dams treated with the higher dose of TM (2 mg/40 g bw) but not with the lower dose (1 mg/40 g bw).[7]. The study observed ectopic Hh signal activity in the ventral wall region through del5-LacZ staining and a gain of function mutants of Hh signalling expressed defects in the body wall, therefore results suggested ectopically induced Hh signalling was a potential causative agent in a dose dependant formation of omphacele[7].
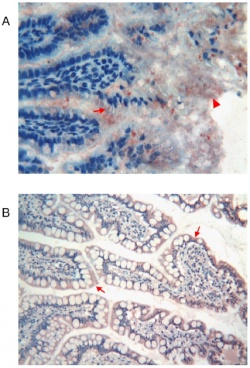
All the different intestinal segments including glands showed immunoreactivity for Tβ4 however each segments/sites had different levels of this. Highest reactivity for Tβ4 was seen in adult liver and pancreas while the lowest seen in the fetal developing liver. Interestingly granular reactivity for Tβ4 was seen in the epithelium that covered the ileal villi and even more in the cytoplasm of mucous cells (as can be seen on the right). Peptide reactivity was also observed in the mucous of the intestinal lumen at 21 week of gestation.[8]. The high level of Tβ4 its expression during gut development indicates its relevant role for development of the gut. As a whole, the data indicated a differentiation stage-specific regulation and expression pattern of Tβ4 in the gastrointestinal tract of human as well as annexed glands during fetal development. It was therefore suggested that Tβ4 could play variety of roles in development of organs in the GIT system during organogenesis.[8]. |
Historic Findings
| Year | Research and Findings |
|---|---|
| 1780 | The famous Dissertationi di fisica animale e vegetale by Lazzaro Spallanzani is published where the first interpreted process of digestion as a chemical mechanism was discovered through the study of gastric juices. known now as The Chemical Theory of Digestion |
| 1802-33 | contributions by Johann Friedrich Meckel to the study of the abnormalities occurring during the embryological development. Earning Meckel's diverticulum among other structures to be named after him [9] |
| 1806 | Oken believed the embryo was nourished through umbilical vesicle. In this year he published treatise, declaring proof for following ideas:
1. The intestine did not lie in the abdominal cavity originally but came from the vesicle (vesicular umbilicus) which was outside amnion. 2. Duodenum is prolongation of the stomach in which it splits into anterior and posterior intestine which go into abdominal cavity. 3. After intestine has no continuity and angular splicing with a valve, it begins to draw backwards to the umbilicus and enter abdominal cavity so the necessary umbilical hernia is present in embryos. [10] |
| 1810 | Kieser defended Oken’s propositions. He published a model of the intestine at 3 months showing his finding that coils of the intestine were lodged in umbilical cord rather than the abdominal cavity.[11] |
| 1900 | Choronschitzky presents findings on the emergence of spleen, liver, gallbladder, pancreas in vertebrate organisms [12] |
| 1900 | Berry J, M studied histological development of the small intestines where he noted that no evidence of villi by week 7 could be found. The villi could be observed in the duodenum by weeks 8-9 and along entire length of the small intestines by week 12 |
| 1914 | Johnson F, P made detailed descriptions on the early development of the anal canal and postulated that that an exact histological determination of the border between entoderm and ectoderm is impossible [13] |
| 1970 | Pace clears controversy on the age of appearance of the haustra within the human colon, showing Taeniae Coli and Huastral Clefts in week 11 of development[14]
|
Foregut
<html5media width="420" height="315">https://www.youtube.com/watch?v=uPBEgBIvRcI</html5media>
The foregut consists of the following organs [15] :
- Oesophagus
- Stomach
- Liver
- Gallbladder and Bile Duct
- Duodenum
- Pancreas
Nerve supply: Refer to specific structures of foregut
Blood supply: Celiac artery
Oesophagus
The primordium of the oesophagus is a portion between the respiratory diverticulum and stomach dilation. The oesophagus is a short tube initially but elongates significantly over time as the fetus grows. Oesophagus has an endoderm derived epithelial lining which proliferates. The epithelial lining also undergoes a series of transformations. Occluded lumen appears by week eight as well as vacuoles. With time the lumen is recanalized and the vacuoles combine. The epithelium of the oesophagus becomes stratified squamous during the fourth month. The development of muscle layers consisting of outer and inner layers, are recognised by eight weeks. The oesophagus contains smooth (splanchnic mesoderm derivative) and skeletal muscle fibres.[16]
Stomach
Dorsal and ventral mesenteries anchor the developing stomach to the body walls. In the 7th week, the stomach undergoes a 90 degrees clockwise rotation about a longitudinal axis. The rotation is such that the right side moves dorsally and left side moves ventrally. The vagus nerve follows this stomach rotation such that the anterior surface becomes left vagus nerve and the right vagus nerve becomes the posterior surface. Since the left vagus nerve is located on the anterior surface and right vagus nerve on the posterior surface, they are renamed as anterior vagal trunk and posterior vagal trunk respectively. The growth of the dorsal wall of the stomach is faster than the ventral wall. [17] The greater and lesser curvatures of the stomach are established via the different growth on the left and right sides. Pylorus is tipped superiorly by cranio-caudal rotation. Pyloric sphincter is formed by the proliferation of the mesoderm derived smooth muscle. This smooth muscle lies in the caudal end of the stomach. The stomach is pulled up by the rotation of the stomach and duodenum about a ventrodorsal (A-P) axis. In the 8th week, due to these rotations, the duodenum is pulled into a C-shaped position. Hence the postnatal position for the stomach and duodenum is achieved.
The greater and lesser omenta are formed by the dorsal and ventral mesenteries of the stomach respectively. Also the ventral mesentery is attached to the developing liver. Distinct spaces of the peritoneal cavity is produced by the rotations of the forgut structures and the development of the omenta. The space posterior to the stomach is called the lesser sac or omental bursa. The space anterior to the stomach is called the greater sac. The greater sac is anteriorly inferior to the stomach. A small opening is located near the liver’s hilum which is called the epiploic foramen. The greater and lesser sacs communicate through the epiploic foramen. From 4 layers of peritoneum, a thick sheet is forms via the anterior and posterior folds of the greater omentum fusing.[18]
Liver, Gallbladder and Bile Duct
Early in the fourth week of the foregut development, the liver, biliary duct system and gallbladder are seen as ventral outgrowth (hepatic diverticulum). This ventral outgrowth is from the caudal/distal part of the foregut. Hepatic diverticulum is formed via the interaction between bipotential cells and FGF’s which is secreted by the developing heart. There is a mass of splanchnic mesoderm between the midgut and the developing heart. This mass is called septum transversum which is an extension of diverticulum. The ventral mesentry for this region is then formed by the septum transversum.
As the hepatic diverticulum grows in between the layers of ventral mesogastrium, it divides into two parts. Primordium of the liver is the larger cranial part of the hepatic diverticulum. The kupffer cells, fibrous and hematopoietic tissues of the liver are all derived by mesenchyme in the septum transversum.[19]
From the 5th to 10th weeks, the liver grows rapidly and fills the upper abdominal cavity largely. The development and segmentation of the liver is determined by the amount of oxygenated blood flowing from the umbilical vein and into the liver itself. At first, both right and left lobe are of the same size but eventually the right lobe becomes larger. During the 6th week, hematopoiesis begins which gives liver a bright reddish appearance. The liver will account for 10% of the total fetus weight by the ninth week. The bile formation begins during the 12th week. Bile is formed by hepatic cells.
The gall bladder is formed by the small caudal part of the hepatic diverticulum. The cystic duct is formed by the stalk of the diverticulum. The connection between the hepatic and cystic ducts via stalk to the duodenum becomes the bile duct. Initially the bile duct is to the ventral aspect of the duodenum but as the duodenum grows and rotates, the bile duct entrance is carried to the dorsal aspect of the duodenum. After the 13th week, the bile entering the duodenum via bile duct gives the meconium (intestinal contents) a dark green colour.[20]
Ventral mesentery is a thin double layered membrane which gives rise to:
- The lesser omentum passing from the liver to the lesser curvature of the stomach (hepatogastric ligament) and from liver to duodenum (hepatoduodenal ligament)
- Extending from the liver to the ventral abdominal wall is the falciform ligament.
From the umbilical cord to the liver, the umbilical vein passes on the border of falciform ligament. Also the visceral peritoneum of the liver is derived from ventral mesentery which is derived from mesogastrum. The whole liver is covered with peritoneum except the bare area which is in direct contact with the diaphragm.
Duodenum
The proximal duodenum if formed by the caudal part of the foregut. Proximal duodenum is supplied by the anterior and posterior branches of the superior pancreaticoduodenal artery. This is a branch of the celiac artery. The duodenum and the pancreas are pushed up against the posterior abdominal wall as the rotation of the stomach takes place. The mesothelium which is covering the duodenum contracts and eventually fuses with the mesothelium covering the posterior abdomen wall. Due to this the duodenum becomes located behind the peritoneum which is known as retroperitoneal. However since the duodenum has not yet began its development in a retroperitoneal position, it is said to be secondary retroperitoneal organ.
During 5th and 6th week of development, the lumen of the duodenum becomes progressively small and occludes. The lumen recanalizes by the end of week 8. If recanalization fails, this results in duodenal stenosis or atresia. Most of the ventral mesentery of the duodenum has disappeared by this time.
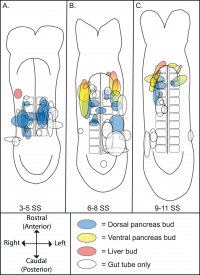
Pancreas
The ventral pancreatic bud and the dorsal pancreatic bud are two caudal outgrowths from the forming liver. The endoderm develops into branched tubules which are attached to the secretory acini (exocrine pancreas) within each bud. Islet of Langerhans (endocrine pancreas)[22] is formed by stem cells at the duct branch points. This then develops into discrete islands of vascular endocrine tissue[23]. This tissue is within the parenchyma of the exocrine glandular tissue.[24]
The ventral and dorsal buds fuse together via the primary rotation of the gut tube which gives a single organ in the adult. The head of the pancreas consist of an uncinated process which is derived from the ventral pancreatic bud. The rest of the head, body and tail of the pancreas are derived from the dorsal pancreatic bud. The distal duct systems of both ventral and dorsal buds join together to form the main pancreatic duct, which drains into the common bile duct. At this site, duodenal papilla is present. Failure for the fusion of buds can lead to annular pancreas.
During the 5th month of development, insulin secretion begins. Along with duodenum, the pancreas is also pressed against the posterior abdominal wall hence becomes a retroperitoneal structure. [25]
For this section some references were from the Moore, KL, Persuad, TVN & Trochia MG. (2011) The Developing Human: Clinically Oriented Embryology (9th edition). Philadelphia: Saunders
Midgut
Structures of Midgut:
-Ileum
-Appendix
-Ascending colon
-Hepatic flexure of colon
-Cecum
-Jejunum
-Duodenum (distal half of 2nd part, 3rd and 4th parts)
-Transverse colon (proximal two-thirds)
Nerve supply: Superior mesenteric plexus
Blood Supply: superior mesenteric artery[26].
Midgut Rotations (Embryonic)
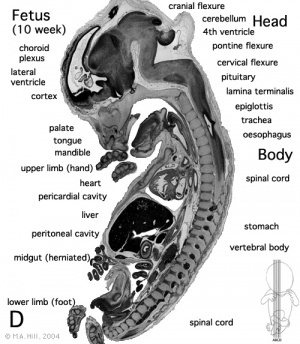
Development of the midgut in the embryonic period is characterized by rapid elongation of the gut and its mesentery. The loops of the week 10 intestine position themselves as shown in the hand drawn illustration below is a result of embryonic rotations.
Midgut Herniation
Herniation of the midgut usually begins around week 5 of the embryonic period. Connected by dorsal mesentery the herniation occurs as a result of the intestine (particularly the ileum) growing faster than the abdominal cavity during this embryonic period. As seen in the table below it is thought that all midgut herniation’s of the fetus should occur by weeks 9-10 [2].
| Week of Gestation | Percentage of Foetuses Herniated |
|---|---|
| 8 | 64% (note: embryo) |
| 9 | 100% |
| 10 | 100% |
| 11 | 25% |
| 12 | 0% |
Table 1.1: Percentage of Herniated midguts during weeks 8-12[27].
Week 10: The herniation occurs because of the lack of space in the intra-abdominal cavity mainly due to the large liver and kidneys. As a result the rapidly growing intestinal loops of the midgut are herniated at the umbilicus to accommodate for this lack of space.
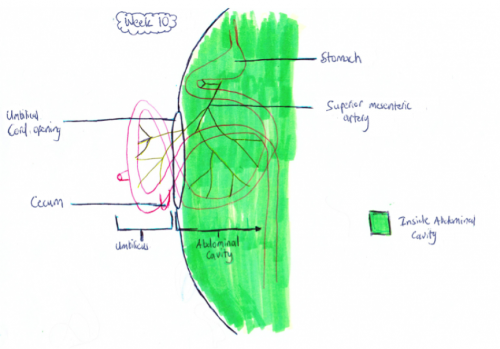
Retraction of Midgut
Week 11: The intestinal loops that have migrated to the umbilicus usually return to the abdominal cavity between 8 and 12 weeks of gestation[29]. This is shown in figure 1.b as now all the intestine is back in the abdominal cavity in comparison to the week 10 fetus whose midgut was still herniated. Although it is not exactly known why the midgut goes back into the abdominal cavity it is thought that the following factors play a major role:
-growth and expansion of the abdominal cavity to cover the herniated midgut
-regression of mesonephric kidney for more space
-reduced growth of the liver for more space[30]
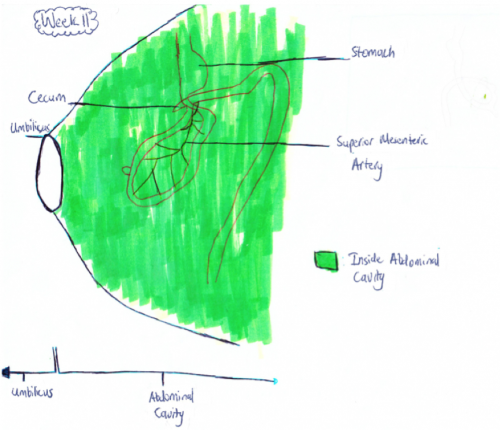
Features of Midgut
Peyer's Patches: Peyer's patches are organised lymphoid nodules. By week 30 of gestation the fetal small intestine contains on average 60 peyer's patches[31]. During weeks 15-16 there is a rapid spurt in which the development and maturation of lymphoid follicles of T and B cells allows the continual development of the foci of peyers patches at a continual rate within the small intestine[32]. At week 19 these aggregations mature into recognisable peyer's patches. In week 24 of gestation the patches become macroscopically visible[31].
Interstitial cells of cajal: Interstitial cells of Cajal (ICC) are specialised network-forming cells that play important roles in the control of digestive motility. In the small intestine c-kit immunoreactive (c-kit IR) cells identifiable as interstitial cells of Cajal appear in week 9. ICC cells then appear in between weeks 10-12 in the large intestine[33]. The ICC cells are only present in the proximal duodenum at the end of the embryonic period in the form of a wide belt of tightly packed cells around the inception of the myenteric plexus ganglia. The ICC cells emerge in the distal duodenum at the beginning of the fetal period in the arrangement of thin rows of pleomorphic cells at the level of the myenteric plexus[34].
Villi Formation: At week nine in fetal development the small intestine contains mesenchyme and pseudo stratified columnar epithelium. Around week 10 small Lumina develop near the base of the epithelium[35]. Ridges are formed by an elongation of individual epithelial cells. By approximately week 11 these ridges form a longitudinal zig-zag pattern after folding in a concertina fashion. The main lumen is not in balance with the small lumina formed at the base and as a result there is an extension of the small lumina to the main luminal surface and exofoliation of the redundant cells. This leads to a division of the zig-zag folds into primary villi. Therefore the villi can be seen by week 11 in the small intestine[36]. The primary villi trun into secondary villi following the development of cyst like structures within the epithelium to extend the lumen. By week 16-20 the villi appear throughout the entire intestinal tract. [35]
Hindgut
The hindgut consists of the following organ/structures:
- Distal transverse colon
- Descending colon
- Sigmoid Colon
- Rectum (Cloaca before urogenital partitioning)
Innervation: inferior mesenteric plexus
Blood supply: inferior mesenteric artery
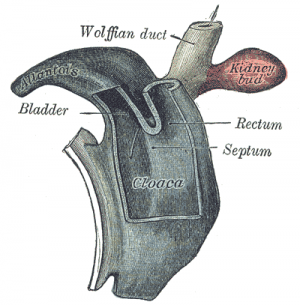
Cloaca partitioning
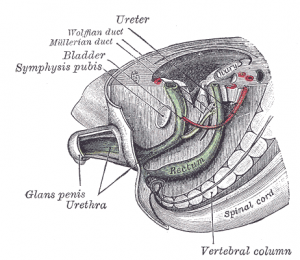
During embryological development, human gastrointestinal and urinary tract share a common vessel known as the cloaca.[37] By the end of week seven, the urorectal septum, a coronal ridge of mesenchyme, will have formed down the angle between the allantois and hindgut and fuses with the cloacal membrane. This forms an anterior urogenital sinus and a posterior anorectal canal. The anterior portion of the membrane that forms the urogenital membrane is larger than its posterior anal membrane. In Week 9, there is proliferation of the the mesenchyme around the anal membrane raising the surrounding ectoderm, forming the proctodeum (a shallow pit). There are swelling around this pit that are referred to as anal folds. The rectal membrane at the base of the anal pit, soon ruptures to create the anal canal which connects the GIT from the rectum to the amniotic cavity.[3]
The junction between the anal pit ectoderm and hindgut endoderm Is designated by the anatomic anorectal or pectinate (dentate) line, the former site of the anal membrane. This area is where the epithelium changes from columnar to stratified squamous epithelium. This squamous tissue begins to keratinise and becomes continuous with the perineum. Within the hindgut portion, the anal canal is supplied by the inferior mesenteric artery, whereas the anal pit is supplied by the internal pudenal branch of the internal iliac artery.[13]
Colon Development
During fetal stages, the Colon undergoes lengthening and maturation. After the abdominal cavity has enlarged enough to accommodate all of the Gastrointestinal tract, the return of the herniated small intestines forces the distal end of the colon into its final position in the left side of the peritoneal cavity. the proximal end of the colon returns after the Small intestines, with the cecal end swinging to the right and downwards
- Week 8-12 the intramural ganglia develop[38]
- Week 11: Taeniae Coli and Huastral Clefts begin to appear[14]
- Circular Muscle in Large intestines form in Week 8 and Longitudinal muscle formed in Week 12-15[39]
- Week 20 intestinal villi formation is completed. incorporates all of the colon as well as the Small intestines[40]
Abnormalities
| BEFORE FETAL STAGE | |||
|---|---|---|---|
| Abnormality name | Affected region | phase of occurrence | Definition |
| Esophageal atresia | Foregut (Esophagus) | occurs in 8th week | 1 in 3000-4500 birth- results from deviation of trachoesophageal septum and there is incomplete separation of the eosphagus from laryngotracheal tube. [41] |
| Eosphageal stenosis | Foregut (Esophagus) | during week 8 | Due to incomplete recanalisation of Esophagus [42] |
| Congenital hypertrophic pyloric stenosis | Foregut (stomach) | onset at 3rd week and can present neonatally | 1 in 150 male and 1-750 female- muscular thickening of the pylorus Cite error: Invalid <ref> tag; invalid names, e.g. too many
|
| Anal agenesis | Hindgut | week 7 | Anal canal end blindly or ectopic anus, or anoperineal fistula. Anal canal may open into vagina in female or urethra in male.
|
DURING FETAL STAGE
Cleft Palate
Cleft palate is one of the most common congenital birth defects affecting at rates of 75.9 per 100000 births in 2003.[43]. Females are affected with this abnormality more than male at ratio of 2:1 according to studies. It occurs between the 6th and 11th week of fetal development. There is strong data which links to maternal smoking being possible cause of cleft palate in the offspring.[44] Alcohol consumption, solvents and agricultural chemicals have also been seen as possible causes however there are many genetic errors (such as aberrant gene variants) that can possibly lead into cleft palate abnormality making its etiology mostly unknown.[44]
Cleft palate can occur with or without cleft lip. It is a defect of the normal development of soft and hard palate (part of the oral cavity). Cleft of the palate occurs as a result from disruptions that affect cellular migration, proliferation, apoptosis, extracellular matrix deposition and morphogenetic movements.[45] These events are all important in the formation of lip and palate.
Secondary palate form from the pair of palatal shelves (vertically orientated outgrowths of the maxillary process). The shelves go into a horizontal position just above the tongue by elevating and expanding and then fuse at the midline with each other. When there is disruption/deregulation in any of these processes the shelves are unable to fuse and this is called cleft palate. [45]. Mesenchyme derived from cranial neural crest cells and pharyngeal ectoderms normally interact so that the palate growth and fusion occur. Interruption in the interaction of these two tissues therefore also leads to cleft palate.
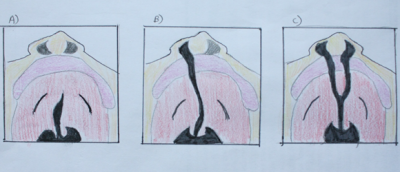
Cleft of the palate can either be incomplete, unilateral complete or bilateral complete. Complete cleft means the maximum degree of cleft of any of the types. The image on the right shows the different forms of cleft of the palate (with or without cleft of the lip). The end result of this abnormality leads the roof of the mouth directly connected with nasal cavity.
NB: The picture is showing the roof of the mouth with the nose on top, and the lips just below it in pink and oral cavity in red.
Gastroschisis
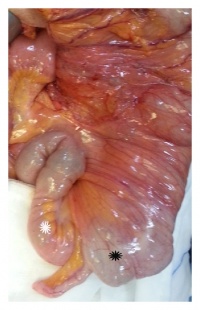
Gastroschisis is a congenital birth defect which can be diagnosed early in fetal development. It is the most common neonatal abdominal wall defect which occurs equally in males and females. Studies suggest white population is more affected by this abnormality in comparison to Hispanics and dark skinned people.[46] It is a defect of the anterior abdominal wall, usually to the right of the umbilical cord and the peritoneal membrane that normally covers the bowel is absent. The defect involves all layers of the abdominal wall and in this abnormality, an infant’s intestine (small and large intestine) protrude out and into the amniotic fluid. [47]Other organs such as the stomach and liver can also bulge out from the hole and no membrane covers these organs in this abnormality.
Possible cause:
The cause of Gastroschisis is not completely clear however a potential mechanism include tearing at the base of umbilical cord before the umbilical ring closes or the failure of one or more folds in the abdominal wall to fuse completely and in the correct manner. [46]
Due to the failure of the closing as stated above, organs protrude out and the eviscerated bowel is exposed to amniotic fluid for a long period of time, causing mucosal and muscular injury. The organs that remain out of the body wall are also exposed and vulnerable to infection in the open air as an infant is born. In more uncommon cases, chromosomal syndromes such as trisomy 18, 13, or 21 or sex chromosome anomalies have been found in association with Gastroschisis. [48] In recent findings Gastroschisis has also been seen to be a result from vascular events causing disruption of fetal abdominal wall. For example Maternal vascular under perfusion is a possible underlying cause of intermittent ischemia to the abdominal wall.
Omphalocele
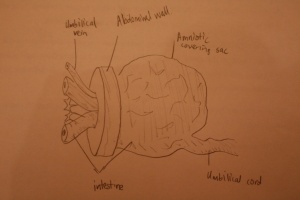
Omphalocele is a common midline abdominal wall defect of variable size affecting 2-3 infants per 10,000 births worldwide. [47] It is a feature of the below abnormality (Cloacal Extrophy). It is similar to Gastroschisis however, it is characterised with the absence of fascia, muscle, and skin and occurs due to a defect in the development of the muscles in the abdominal wall.[47] A fetus with omphalocele tends to have herniation of abdominal contents into the base of the umbilical chord. A membranous sac that has amnion and peritoneum covers these contents lying within the umbilical chord. Omphalocele in the later stage of fetal development (approximately week 11) occurs after normal infolding of the embryo therefore having formed an abdominal cavity. When the umbilical ring does not close around the umbilical cord then this small defect occurs containing only the intestine.[47]
Reverse rotation
Congenital abnormality of the Midgut seen in one of 5000 live births. Normally after the midgut has undergone retraction into the abdominal cavity it undergoes a 180 degree counterclockwise rotation at week 10-11. Reverse rotation is when this midgut loop rotates in clockwise direction instead. Doudenum lies anterior to the superior messentric artery ( normally it should lie posteriorly) and transverse colon lies posterior instead of anterior to it ( artery). [47] It is crucial that an early diagnosis is done as there are life threatening complications associated with it.
Anorectal deformities
There are 3 major types of anorectal deformities that can be found clinically
- imperforate anus (abscent): the rectal membrane doesn’t rupture leaving Gastrintestinal tract to be completely or incompletely walled off from the outside. All varieties of this deformity require immediate surgical intervention at birth
- Insufficient anus leads to problems of meconium evacuation and should be treated without delay
- Ectopic sinus: does allow some intestinal transport, but is usually functionally insufficient
Cloacal Extrophy
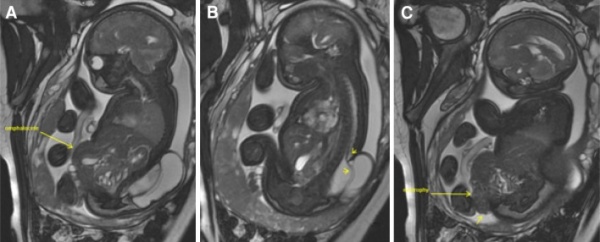
If the cloaca ruptures before completely partitioning, it can lead to extrophy. Although extremely rare with only occuring in one in 200 000-400 000 births, it requires immediate surgical attention postnatal [49]. It arises from the failure of the caudal fold to close. Cloacal extrophy results in a child being born with with many inner-abdominal structures exposed. A portion of the large intestine lies outside of the body, and on either side of it are the two halves of the bladder. To diagnose this, MRI Imaging is usually performed during development to detect the 'elephant trunk sign' of cloacal extrophy[50]
Superficial anorectal deformities;' [51]
• Anal agenesis or insufficient anus (with or without fistula): • Membranous atresia or covered anus (with or without fistula • Anorectal agenesis with or without fistula
Deep deformities:
• Pure rectal atresia: complete failure of the formation of the inferior part of the rectum and anal canal • Rectal atresia with fistula: always insufficient. The length and degree of anastomosis differentiate the various types of anomaly
'Mixed deformities ' include all forms of ectopic anus
• May involve abnormal anastomoses of the anus to the perineum and reflect both perineal and cloacal abnormalities
Hirschsprung disease
Hirschsprung disease is congenital disease that affects 1 in 5000 live births and has a male dominance over female with a ratio of 4:1.[52]
The gastrointestinal tract derives from endoderm extending from mouth to anus. The function of the GIT depends on the enteric nervous system. Neurons and Glia of enteric nervous system are organised into ganglia. This abnormality is characterised when there is a congenital absence of these neurons in the distal colon (part of Hindgut) of the GIT. The absence of neuron can be due to disrupt in normal migration, proliferation, differentiation, survival or apoptosis of neural crest cells which occurs between the 5th and 12th week of gestation. [53]
Certain mutation in genes such as GFRα1, NRTN, EDNRB,PHOX2b, SOX10, and SHH have been thought to play a part in the pathogenesis however it is not fully understood. [47] Due to this disease the normal function of the Hindgut does not proceed this includes movement such as peristalsis.[54]
References
- ↑ <pubmed>9551687</pubmed>
- ↑ 2.0 2.1 <pubmed>14745932</pubmed>
- ↑ 3.0 3.1 <pubmed>17853405</pubmed>
- ↑ <pubmed>3858813</pubmed>
- ↑ 5.0 5.1 <pubmed>24139758</pubmed>
- ↑ 6.0 6.1 6.2 <pubmed>20482879</pubmed>
- ↑ 7.0 7.1 <pubmed>16369776</pubmed>
- ↑ 8.0 8.1 8.2 <pubmed>20161756 </pubmed>
- ↑ J. F. Meckel: Über die Divertikel am Darmkanal. Archiv für die Physiologie, Halle, 1809, 9: 421-453.
- ↑ Keibel,F. and Mall, F.P. J. B. Lippincott Company, Philadelphia (1912)
- ↑ Keibel,F. and Mall, F.P. J. B. Lippincott Company, Philadelphia (1912)
- ↑ CHORONSCHITZKY: Die Entstehung der Milz, Leber, Gallenblase, Bauchspeicheldruse und des Pfortadersyssems bei den verschiedenen Abteilungen der Wirbeltiere. Anat. Hefte, Bd. XIII, 1900.
- ↑ 13.0 13.1 <pubmed>20563874</pubmed>
- ↑ 14.0 14.1 <pubmed>5556677</pubmed>
- ↑ <pubmed>19575677</pubmed>
- ↑ <pubmed>22750256</pubmed>
- ↑ <pubmed>9664826</pubmed>
- ↑ <pubmed>16052677</pubmed>
- ↑ <pubmed>23720330</pubmed>
- ↑ <pubmed>23799566</pubmed>
- ↑ <pubmed>22815796</pubmed>
- ↑ <pubmed>23630303</pubmed>
- ↑ <pubmed>22743232</pubmed>
- ↑ <pubmed>15072563</pubmed>
- ↑ <pubmed>23597482</pubmed>
- ↑ <pubmed>24891783</pubmed>
- ↑ <pubmed>2528908</pubmed>
- ↑ 28.0 28.1 <pubmed>2908440</pubmed>
- ↑ <pubmed>9203209</pubmed>
- ↑ <pubmed>8345550</pubmed>
- ↑ 31.0 31.1 <pubmed>18668776</pubmed>
- ↑ <pubmed>2276071</pubmed>
- ↑ <pubmed> 24414177</pubmed>
- ↑ <pubmed> 21352475</pubmed>
- ↑ 35.0 35.1 <pubmed> 7158824</pubmed>
- ↑ <pubmed> 507402</pubmed>
- ↑ <pubmed>12740945</pubmed>
- ↑ <pubmed>1232090</pubmed>
- ↑ <pubmed>12982018</pubmed>
- ↑ <pubmed>1214115</pubmed>
- ↑ <pubmed>25286746</pubmed>
- ↑ <pubmed>25305782</pubmed>
- ↑ <pubmed>24689077</pubmed>
- ↑ 44.0 44.1 <pubmed>19884687 </pubmed>
- ↑ 45.0 45.1 <pubmed>23516636</pubmed>
- ↑ 46.0 46.1 <pubmed>11778986</pubmed>
- ↑ 47.0 47.1 47.2 47.3 47.4 47.5 8590188<pubmed>8590188</pubmed> Cite error: Invalid
<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content - ↑ <pubmed>23554304</pubmed>
- ↑ <pubmed>12651743</pubmed>
- ↑ <pubmed>22878705</pubmed>
- ↑ <pubmed>23073994</pubmed>
- ↑ <pubmed>23528997</pubmed>
- ↑ <pubmed>PMC3691347</pubmed>
- ↑ <pubmed>PMC1683643</pubmed>