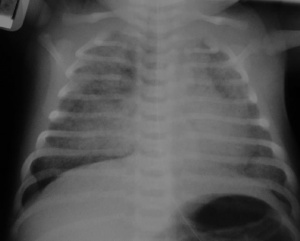
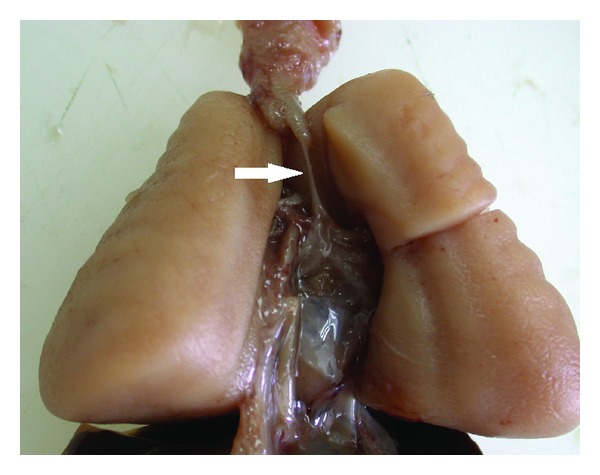
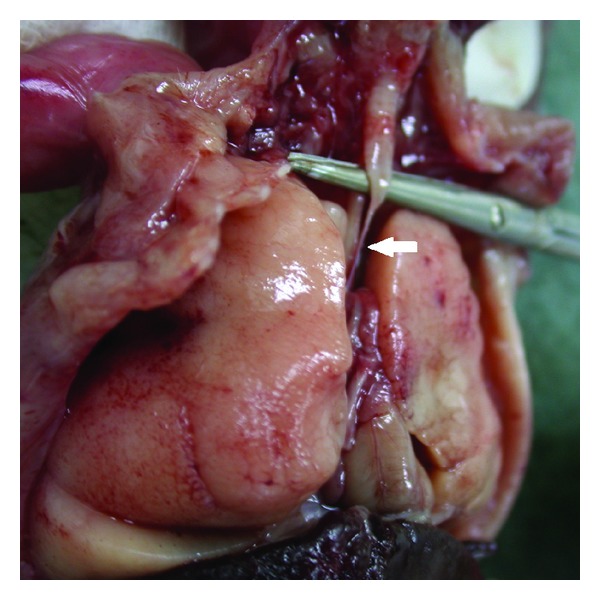
2014 Group Project 1
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Respiratory
This page focuses on the development of the respiratory system during the fetal stage, exploring the two significant zones and the major organ of the respiratory system, the lung. It places particular emphasis on the overview of fetal respiratory development. Discussion of current and historic findings during the fetal development of the respiratory system will also be elaborated on. Unfortunately, during the fetal development of the respiratory system, some things may go wrong leading to abnormalities in this important system. In respect to this, there will be a detailed explanation of abnormalities that we find relevant to this system to conclude.
Development of the Respiratory system Overview
Current knowledge of the development of the respiratory system portrays how understanding has advanced over time, from what was historically known about the system until what is known today. The respiratory system consists of organs and tissues that assist in breathing. Lungs are the most important organ for respiration. Humans have two lungs, a left and a right lung both located in the chest covered by many tissue, muscles and bones to protect them.
The purpose of respiratory system is for gas exchange to occur, gas exchange is the removal of carbon dioxide and intake of oxygen into the lungs. Gas exchange is imperative for the function of life as oxygen is needed to working muscles.[1]
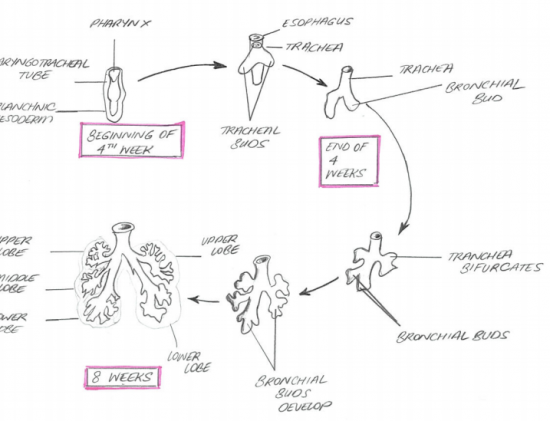
The respiratory system starts developing at week 4 of the embryo and a great deal of the development continues to take place in the fetal stage. A substantial portion of the respiratory system is formed by germ layer endoderm. The lung buds are lined by epithelium derived from the endodermal layer which later on differentiates into the respiratory epithelium. Nerves and neural innervations to the lungs are derived from ectoderm, on the other hand splanchnic mesoderm contributes to the pulmonary blood vessels, smooth muscle,cartilage and connective tissue.
This system has many airways that allows the movement of air from nose or mouth to the lungs. Some of the airways include;
- Nose (including the nasal cavity)
- Mouth
- Larynx
- Trachea
- Bronchi and their branches
During the embryonic and fetal stage the respiratory system is developing. The embryonic stage is the first 1-8 weeks and anything after that until about week 37 or birth . However the respiratory system does not carry out gas exchange until birth. Whilst the embryo or fetus is in the mother, gas exchange occurs through the placenta. Once born the lungs of the new born are drained and are filled up with air automatically. The lungs do not inflate completely until about 2 weeks of the new born. The surfactant in each alveoli assists in keeping the lungs open and prevents them from collapsing.[2]
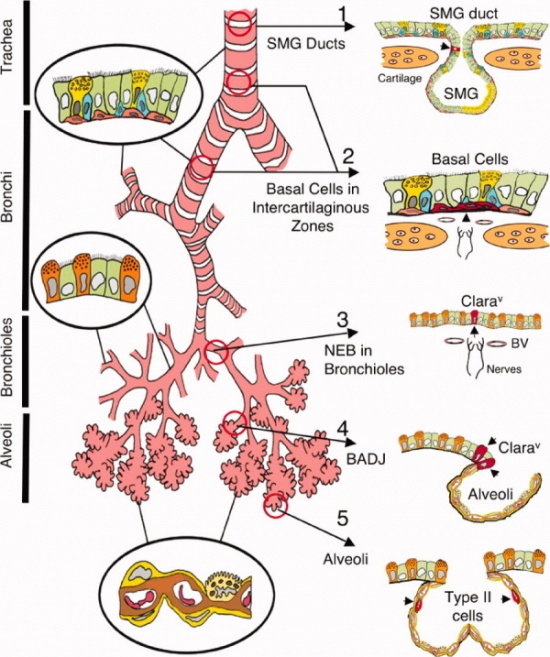
The respiratory tract is divided into two main parts; the conducting zone and the respiratory zone.
Development of the Conducting Zone
The conducting zone is made up nose to bronchioles, the main function is to filter, warm, and moisten air and conduct it into the lung. The conducting zone includes the nose, pharynx, larynx, trachea, bronchi and bronchioles. Nares are the opening into the nose and are where nasal cavities are lined with cilia, mucous membrane and consists of blood filled capillaries.
ORAL CAVITY
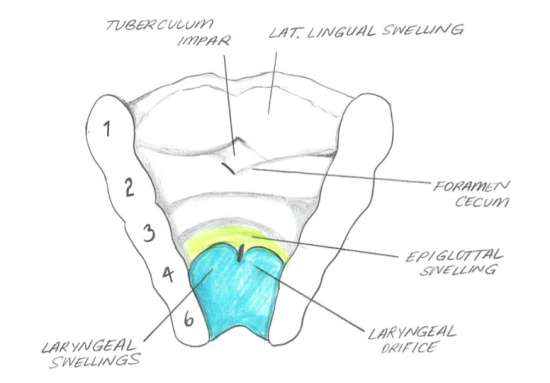
The oral cavity is formed by the stomodeum, which is the depression in the embryo located between the brain and the pericardium. This depression is known as the precursor of the mouth. The stomodeum is ectoderm-lined depression and separates the primitive pharynx by the buccopharyngeal (oropharyngeal) membrane. The membrane later breaks down and stomodeum opens into the pharynx which then forms the vestibule of oral cavity. [3]
LARYNX
The larynx is developed from endoderm of laryngotracheal tube. The splanchnic mesoderm contributes to the development of connective tissue and muscle as well as the laryngeal cartilages, they develop from the 4th to the 6th pharyngeal arch mesenchyme. [4]
TRACHEA
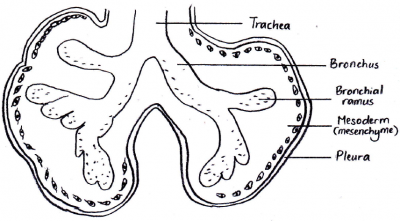
The laryngotracheal tube develops in the 4th week. The oseophagotracheal ridge separating the diverticulum forms the trachea.The epithelial cells from the foregut endoderm invade the surrounding mesenchyme to form the trachea. The trachea then divides into 2 bonchial buds, giving rise to the main bronchi, left main and right main.[5]
BRONCHI
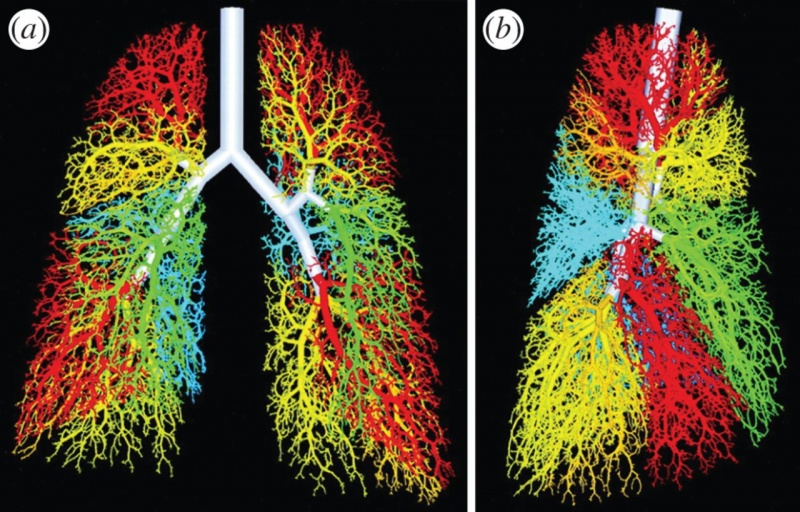
The bronchi is formed in week 4 and the lung buds develop and further divide into more divisions making up 2 divisions for the left and the 3 for the right. These secondary bronchi (3 branches on the right and 2 on the left), then again divides into tertiary bronchi which occurs in week 7. The surrounding mesenchyme then develops into bronchopulmonary segments.[6]
BRONCHIOLES
By the 24th week there would have formed approximately 17 subdivisions. After the birth of the baby the bronchiole tree further divides another 6 more divisions.[7]
Development of the Respiratory Zone
The respiratory zone is where the oxygen and carbon dioxide exchange with the blood. The respiratory zone includes the terminal bronchioles, alveolar ducts and alveoli.The alveolar ducts and the bronchioles cause the 10% of gas exchange. The rest of the 90% is due to the alveoli. [8]
TERMINAL BRONCHIOLES
Terminal bronchioles is a passageway where air passes through from the bronchioles to the alveoli (air sacs) of the lungs, they are lined with simple columnar epithelium. This first begins to develop between week 12 and 13 of the fetus. They develop from thin squamous epithelium, and then differentiate into alveolar cells type 1 and alveolar cells type 2.[9]
ALVEOLAR DUCTS
The alveolar ducts permits oxygen and the carbon dioxide to move between the lungs and bloodstream. Alveolar ducts begin to develop during the late fetal period until about 8 years postnatally. They develop with thin walls along with many capillaries that are in close association with the alveolar epithelial cells. [10]
ALVEOLI
The alveoli is where the carbon dioxide and the oxygen exchange. Each alveolar consists of alveolar cells; type 1 and type 2. Type 1 is a membranous pneumocyte and it serves for gas exchange, on the other hand type 2 is a granular pneumocyte that produces surfactant and it reduces surface tension and prevents the alveoli from collapsing. There would be a remodelling of the alveolar wall that results in a single capillary network, that concludes in the maturation however not a full-sized lung. [11]
Lung Development Stages
|
Historic findings
Historic knowledge of the shifts in understanding of the respiratory development during the fetal stage is essential for robust appreciation of current accepted ideas of how this system comes to be in the human body. Historical knowledge predating modern imaging techniques has most often been confirmed by contemporary studies that provided evidence for the claims of early respiratory development. At times, theories put forward for fetal respiratory development were enhanced with further detail, whereas elsewhere paradigms were shifted and challenged due to the availability of proof otherwise [19]. The understanding of the development of the upper and lower respiratory system during the fetal period from week 8 onwards, as well as their respective functions, have been around since before 19th Century [20].
|
Current Understandings and Areas of Research
The development of the respiratory system is one of the most crucial for the survival of the neonate, and hence it is a system that is highly studied. Current research looks at the molecular processes that underpin two important developmental stages of the lung. The lung can anatomically be divided into two parts; an upper respiratory tract and a lower respiratory tract [30]. However, physiologically, the organ can be divided into two parts that occur subsequently:
- The Conducting system- consisting of all the tubular structures such as the larynx, trachea, and bronchi.
- The Functional unit- An alveolus. Alveoli (Plural). Specialised epithelial cell, the at which gas exchange of carbon dioxide and oxygen takes.
Much research has been undertaken to understand how each of these processes occurs individually. However, a study conducted last year shows evidence that during later stages of fetal development, when the expands, these two important processes involve co-ordinated cellular interactions and take place at a precise time within development and at a specific location [31].
By week 8, the respiratory system of the fetus is well underway and the development of the lung is at the pseudoglandular stage (see table above for more information about the properties of each stage of lung development). The three germ layers (ectoderm, mesoderm and endoderm) have each contributed to the development of the lung and their involvement is crucial for regulating a cascade of sequential of events including bronchial branching (see 1. The conducting system) and alveolar differentiation (see section 2. Functional Unit).
1. The Conducting system - The respiratory network
|
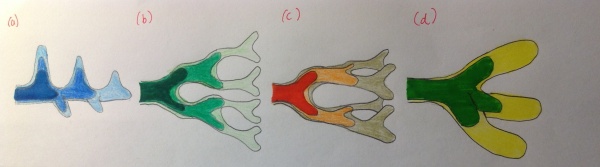
Branching morphogenesis is the growth and branching formation to build a treelike tubular network ending with specialized air bubbles (alveoli) as sites for gas exchange. • In 2013, a review study conceptualised how we now currently understand the model of branching morphogenesis. There are currently three geometrically models proposed for the way in which the primary bronchial buds branch[32]:
a) Domain branching: In this type of mode, the respiratory network develops and continues to grow in a direction perpendicular to the future trachea. New lung bud formations become apparent appear on either side of the stalk. The most recent lung buds that are formed are shown in lighter colors, typically where outgrowths are observed. b) Planar bifurcation: these types of bifurcations form the thin edges of the lobes c)Orthogonal bifurcation: this type of bifurcation creates the lobe surfaces and fill the interior part of the respiratory system with the diaphragm, lies beneath. Note both b) and c)as the name suggests, these branching models are responsible for bifurcating the airways in consecutive rounds of tubular divisions d) Trifucation: Researches have recently identified that this mode of branching is responsible for the backbone of the respiratory tree[33] |
|
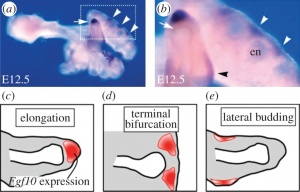
• Another recent study conducted in 2013 [34], suggests that there is a correlative interaction between the lung epithelium and the surrounding plural mesenchyme. The mesenchyme secretes fibroblast growth factor (FGF10) secreted by the mesenchyme, which in turn activates its membrane receptor co-worker (FGFR2). The epithelium, sequentially then generates a small amount of GTPase (KRAS). Both these contributions are involved in a cascade of signaling pathways essential for normal branching morphogenesis of the lung. • The high concentrations of FGF10 at the distal tip of the lung bud would initiate growth in that direction and thus elongate the tube in that direction.
|
|
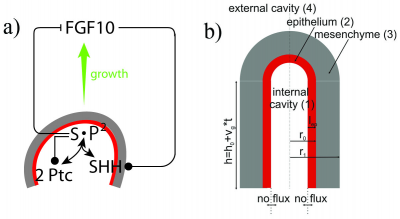 FGF10 is transcribed at high levels in the distal mesenchyme (grey) and experiments suggest that FGF10 promotes both the proliferation of the endoderm and its outward movement (green arrow). FGF10 stimulates the expression of SHH in the epithelium (red). SHH reversibly binds its receptor Ptc1 which is expressed in the mesenchyme (grey). SHH-Ptc binding results in the repression of FGF10 expression A research group in 2011, identified two key signalling factors; fibroblast growth factor (FGF10) and sonic hedgehog (SHH) [35][36]. Other signalling factors such as sonic hedgehog (SHH) receptor patched (Ptc), Bone morphogen protein (BMP4) were also identified from experiments and developed a model to explain the branching network. From their research they also conclude that the sequence of branching events may be the result of different growth speeds:
|
2. The Functional Unit-Alveolus
At the end of the conducting system or at the end of the tertiary bronchial, lie the sites of gas exchange- alveolar air sacs. This process of differentiation from building on to the branched duct to specialised alveolar cells is a process named alveolar differentiation. There are two types of epithelial cells that typically line this tract and both play important role. These two types are described below and provide important background information for the modern research today.
• Although the first breath the newborn takes is after existing the birth canal, a recent article suggests that the two types of alveolar cells only appear to be mature 1 day prior to birth, when the distal tube dilates. [37]. This is a major concern for preterm infants, who many not have complete developed airways by the time that the new infant is born. This can lead to serious diseases including Idiopathic Pulmonary Fibrosis and Respiratory Distress Syndrome (For more information about this visit Newborn Respiratory Distress Syndrome under the "Abnormalities" section of this page)
• It was previously thought alveolar type I arise from type II cells but recent studies propose otherwise. By using molecular markers on the mouse model, this research [38] concludes that during development Type I and II cells arise directly from a bipotent progenitor, whereas after birth new Type I derive from rare, self-renewing, long-lived, mature Type II cells that produce slowly expanding clonal foci of alveolar renewal. Mapping alveolar cell locations is important for cancer treatment for patients.
• This article [39] looks at the current findings of different physiological factors that affect normal neonatal, functioning lungs upon during fetal development. The size of the paired organ to be able to exchange carbon dioxide with oxygen for the very first time at birth, is crucial to be able to withstand that pressure. As we know surfactant, is a lipid-protein composite that aids in this process. Both these epithelial cells that lines this tract a play important role and crucial to the function of the lung prior to birth and especially post-natally (Type 2alveolar cells) because [40]:
- A) Type I alveolar cells are flat and cover more than 90% of the alveolar surface, across which gases diffuse. The exchange of carbon dioxide, CO2 for 02. However, whilst the fetus is still growing inside the uterus this gas exchange does not occur. The first breath is just after delivery and hence the first time in which the alveolar serve their purpose is after birth.
- B) Type II alveolar cells are cuboidal and play and crucial role in the respiratory development of the fetus post-natally. They synthesize pulmonary surfactants, lipoprotein complexes that hydrate the alveolar surface and prevent alveolar collapsing by reducing surface tension.
However, current research suggests that the production of surfactant which is reliant on hormonal factors, have little influence on fetal lung growth. In contrast, the following physiological lung growth factors were found to permit the lungs to express their inherent growth potential.
Current Models
When considering models of research for analysing human development, animals such as the zebrafish, rabbit and mouse are most popular. These models have been used as part of research for scientists to study how different animals can be used to mimics the way the lung is developed in humans. These models have been chosen for various reasons, their genomic patterns, however, are the main reason. For example, at around E16.5 in the mouse, lung development switches from branching morphogenesis to the canalicular and saccular stages [41] . These, in turn, lead to the final process of alveologenesis that generates the functional units for gas exchange. The timing of alveolar development varies between species. In mice it occurs postnatally (∼P5–30), but in humans few alveoli have formed before birth and the process continues for many months- years afterwards.
The reason why these animal models are currently used in research is for a number of reasons. Below is a list of reasons why the mouse model is widely used as part of research within the scientific community:
A Comparison of lung development stages in the human and rabbit with their relationship towards gestational length can be found in Figure 1 of this article [1]
Abnormalities
As mentioned earlier, the development of the respiratory system in the fetus is one that has been known for many years dating back into history, and one that is still currently studied today. However, despite knowledge of respiratory system development being well understood, unfortunately, there still occurs a myriad of abnormalities in the neonate due to complications in development during the fetal stages of the developing human. In this section, we will discuss some of the primary abnormalities that is found in respect to the development of the respiratory system in the fetus.
Newborn Respiratory Distress Syndrome (Hyaline Membrane Disease)
Newborn Respiratory Distress Syndrome (NRDS), also known as Hyaline Membrane Disease (HMD) is characterised by the lack of or inability to synthesise surfactant in the premature lung of neonates.
The incidence of NRDS occurs in babies suffering form immature lung development, usually from premature birth with increased severity and incidence in correlation to decreased gestational age [45]. Preterm births do not allow for full lung maturation of the preterm infant due to process in which the respiratory system forms (from upper respiratory tree to lower). Type II Pneumocytes secrete surfactant into the alveoli, reducing surface tension and thus preventing the collapse of the alveolus – they are the last respiratory cells to differentiate. Preterm infants usually lack Type II Pneumocytes in their lung tissue causing the instability of their alveoli, oedema from immature alveolar capillaries and hyaline membrane formation[46].
NRDS mostly occurs in preterm neonates but can occur in post-term and term babies for a variety of reasons including:[47]
- Intrauterine Asphyxia – commonly caused by wrapping umbilical cord around the neck of the neonate, impairing development[45]
- Maternal diabetes – high levels of insulin can delay surfactant synthesis[48]
- Multiple pregnancy (twins, triplets etc) – associated with high rates of preterm births and resulting lung immaturity [48]
- Rapid labour, fetal distress, placenta previa, preeclampsia, placental abruption – that impair lung maturation in final stages of pregnancy [48]
- Preterm Caesarean delivery – not allowing for lung maturation[49]
- Genetic abnormalities that impair surfactant synthesis (ABCA3)[50]
- Meconium Aspiration Syndrome (MAS) - causes damage to the lower respiratory tract after aspiration of Meconium in amniotic fluid[51].
Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia (BPD) is a common complication in the treatment of Newborn Respiratory Distress Syndrome (NRDS) in infants born more than 10 weeks premature and of low weight. Efforts to treat breathing difficulties associated with NRDS can cause damage to the vulnerable lungs of the infant[52]. The complications can occur from a number of reasons following treatment[53]:
- Oxygen therapy causing inflammation to the lung epithelium due to the higher amounts of oxygen administered
- Used in more critical cases because of the complications associated with this form of treatment, air pressure from ventilation machines can further damage the premature lungs.
- There is some growing evidence that genetics may play a role in the predisposition of BPD [54].
- Infections from treatments involving ventilation can also occur leading to inflammation of the upper respiratory tract.
Meconium Aspiration Syndrome (MAS)
Meconium Aspiration Syndrome (MAS) affects newborn infants in response to some form of fetal stress during the third trimester and/or parturition, often due to: acute hypoxia, intrauterine hypoxia (often caused by the wrapping of the umbilical cord around the neck of the baby) and other physiological maturational events. [51][55].
Stress on the baby before or during labor can cause relaxation of the anal sphincter leading to expulsion of Meconium by the foetus into the surrounding amniotic fluid which can then be aspirated by the fetus, damaging the upper respiratory tract and possibly the lower respiratory tract. [56].
Problems associated with Meconium aspiration include[51]:
- Pulmonary gas exchange deficiency - caused by damage to the lower respiratory tract epithelium.
- Pneumitis and pneumonia - due to chemical damage and irritation from Meconium interaction with the airways.
- Blockage of the airways
Laryngeal Atresia
Laryngeal Atresia (LA) is incredibly rare and occurs as a failure of the laryngo-tracheal tube to recanalise, obstructing the upper respiratory tract leading to a larynx with no lumen[57]. This can cause Congenital High Airway Obstruction Syndrome (CHAOS) [58]. Genetic abnormalities have been identified as having an association with AL [59].
Congenital High Airway Obstruction Syndrome (CHAOS)
Congenital High Airway Obstruction Syndrome (CHAOS) is extremely rare and is the result of an obstruction to the fetal airways. This obstruction can be caused by atresia of the larynx or trachea, laryngeal cysts, laryngeal webs, subglottic stenosis, inversion of the diaphragm and hyperechoism of the lungs[60]. Reviews have revealed that most cases are fatal[61] but ex-utero partum treatments (EXIT) have been successful in treating this condition[62]
Congenital Laryngeal Webs
Similarly to Laryngeal Atresia, Congenial Laryngeal Webs (CLW) are caused by failure of the laryngo-tracheal tube to recanalise, usually at the level of the vocal chords. The lumen and vocal chords of the larynx is usually developed after the epithelium is reabsorbed but in the case of CLW, this reabsorption is incomplete leaving ‘web-like’ formations in the larynx that obstruct normal development and airflow. [63]
Congenital Pulmonary Airway Malformation
Congenital Pulmonary Airway Malformation (CPAM) occurs at varying degrees and is defined by its location in and the level of differentiation of alveoli[64]. In the cases of type I and II, CPAM involves the presence of cysts affecting the terminal bronchioles and lung parenchyma. CPAM is thought to be caused by an abnormal development of the lung bud in week 4-5 of development and leads to the malformation of the pulmonary airways via the formation of lung abscesses, pulmonary infections and the sequestration of areas of the lung[65]. Recent reviews have also suggest that thyroid transcription factor 1 (TTF1) may have a role in CPAM as it is involved in the differentiation of lung epithelium and overall pulmonary development. [66].
- Type I - is defined by large multilocular cysts occurring in one of the pulmonary lobes
- Type II – define by the presence of smaller more uniform cysts.
- Type III – is defined by larger lesions that affect the lung parenchyma of en entire lobe.
Azygos Lobe
Azygos lobe (also known as Adam's lobe) occurs due to the aberrant formation of the azygos vein as it veers from its normal course over the apex of the right lung to penetrate the upper lobe. An accessory fissure is formed in the upper lobe and the pulmonary parenchyma located in the medial portion is identified as the Azygos Lobe. There have been three observed types of azygos lobe that are relatively harmless and present little clinical significance (except during surgery due to variations in the course of the phrenic nerve): [67].
- Upper Azygos Lobe
- Lower Azygos Lobe
- the Lobe of the Azygos Vein
Cystic Fibrosis
Cystic fibrosis (CF) is caused by a mutations of the cystic fibrosis transmembrane conductance regulator (CFTR)[68]. The defect associated with this mutation results in the excretory glands of the body producing a thick sticky mucus as well as salty sweat. The disease affects several organs in the body but mainly affects the respiratory system allowing impairing the response to bacterial infection and causing inflammation in the airways[69][70]. This aberrant production of mucus can lead to the mucus stasis in the pulmonary epithelium, airway plugging, inflammation and chronic bacterial infection causing the decrease in lung function.
References
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ Cite this page: (2014) National Heart, Lung, and Blood Institute Health. Retrieved 20 September, 2014, from http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0063039/
- ↑ <pubmed>11936451</pubmed>
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ <pubmed>11992723</pubmed>
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ <pubmed>12107102</pubmed>
- ↑ 8.0 8.1 8.2 8.3 <pubmed>20692626</pubmed>
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ <pubmed>8815817</pubmed>
- ↑ Cite this page: Rothstein, P (2014) Lung Development. Retrieved September 10, 2014, from http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf/ Lung Development
- ↑ Cite this page: Rothstein, P (2014) Lung Development. Retrieved September 10, 2014, from http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf/ Lung Development
- ↑ Cite this page: Mazurová, Y. Hrebíková, H. Embryology: Respiratory System. Retrieved 26 September, 2014, from http://web.lfhk.cuni.cz/histologie/Histols_web/Vyuka/en/tuition/general/doc/histology_II/G_II_lect_11_E_respir_syst.pdf
- ↑ Cite this page: Rothstein, P (2014) Lung Development. Retrieved September 10, 2014, from http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf/ Lung Development
- ↑ Cite this page: Rothstein, P (2014) Lung Development. Retrieved September 10, 2014, from http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf/ Lung Development
- ↑ Cite this page: Rothstein, P (2014) Lung Development. Retrieved September 10, 2014, from http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf/ Lung Development
- ↑ <pubmed>24058167</pubmed>
- ↑ <pubmed>23431607</pubmed>
- ↑ <pubmed>16601307</pubmed>
- ↑ <pubmed>5118050</pubmed>
- ↑ 22.0 22.1 <pubmed>1399659</pubmed>
- ↑ <pubmed>23377345</pubmed>
- ↑ <pubmed>19972530</pubmed>
- ↑ Keith, A. (1902) Human Embryology and Morphology. London: Edward Arnold.
- ↑ <pubmed>18446178</pubmed>
- ↑ <pubmed>15985753</pubmed>
- ↑ <pubmed>14509914</pubmed>
- ↑ 29.0 29.1 <pubmed>24160653</pubmed>
- ↑ Cite this page: Hill, M.A. (2014) Embryology Lecture - Respiratory Development. Retrieved September 10, 2014, from https://php.med.unsw.edu.au/embryology/index.php?title=Lecture_-_Respiratory_Development
- ↑ <pubmed></pubmed>24058167
- ↑ <pubmed>24004663</pubmed>
- ↑ <pubmed>22844507</pubmed>
- ↑ <pubmed>24004663</pubmed>
- ↑ <pubmed>22359491</pubmed>
- ↑ ,<pubmed>24004663</pubmed>
- ↑ <pubmed>24499815</pubmed>
- ↑ <pubmed>24499815</pubmed>
- ↑ <pubmed>24429276</pubmed>
- ↑ <pubmed>24058167</pubmed>
- ↑ <pubmed>18654673</pubmed>
- ↑ https://embryology.med.unsw.edu.au/embryology/index.php/Mouse_Development
- ↑ https://embryology.med.unsw.edu.au/embryology/index.php/Mouse_Development
- ↑ http://www.oneofus.eu/wp-content/uploads/2014/06/One-of-Us.pdf
- ↑ 45.0 45.1 <pubmed>20468585</pubmed>
- ↑ <pubmed>6071188</pubmed>
- ↑ <pubmed>10829971</pubmed>
- ↑ 48.0 48.1 48.2 <pubmed>20848797</pubmed> Cite error: Invalid
<ref>tag; name 'PMID20848797' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID20848797' defined multiple times with different content - ↑ <pubmed>14629318</pubmed>
- ↑ <pubmed>15044640</pubmed>
- ↑ 51.0 51.1 51.2 <pubmed>10612363</pubmed>
- ↑ <pubmed>22785261</pubmed>
- ↑ <pubmed>19712501</pubmed>
- ↑ <pubmed>25031518</pubmed>
- ↑ <pubmed>16651329</pubmed>
- ↑ <pubmed>19399004</pubmed>
- ↑ <pubmed>14325849</pubmed>
- ↑ <pubmed>2342705</pubmed>
- ↑ <pubmed>3566610</pubmed>
- ↑ <pubmed>22167132</pubmed>
- ↑ <pubmed>12778398</pubmed>
- ↑ <pubmed>9802816</pubmed>
- ↑ <pubmed>16798587</pubmed>
- ↑ <pubmed>24672262</pubmed>
- ↑ <pubmed>21355683</pubmed>
- ↑ <pubmed>21762550</pubmed>
- ↑ <pubmed>16333920</pubmed>
- ↑ <pubmed>24685676</pubmed>
- ↑ <pubmed>16928707</pubmed>
- ↑ <pubmed>22763554</pubmed>