User talk:Z3389806
History
Huntington's disease has existed since at least the seventeenth century and several physicians provided earlier descriptions of hereditary chorea but without much detail. In 1872, Huntington’s disease was first documented with great details by George Huntington in “On Chorea” [1]. Huntington’s disease was initially known as chorea, derived from the Greek word khoreia which means dancing in unison. George Huntington described the disease as “an heirloom from generations away back in the dim past” as he realized that Huntington's disease was hereditary. This conclusion was reached when he observed that if one of the parents had the disease, the offspring will inevitably have the disease too. In his paper, “On Chorea”, he described:
| "Of its hereditary nature. When either or both the parents have shown manifestations of the disease ..., one or more of the offspring almost invariably suffer from the disease ... But if by any chance these children go through life without it, the thread is broken and the grandchildren and great-grandchildren of the original shakers may rest assured that they are free from the disease.".[2] |
Huntington thus was able to explain the precise pattern of inheritance of autosomal dominant disease years before the rediscovery by scientists of Mendelian inheritance.
TIMELINE OF HUNTINGTON'S DISEASE RESEARCH [3]
| Year | Milestones |
| 1842 | Huntington's disease was first described by Charles Oscar Waters in a letter in Robley Dunglinson's "Practice of Medicine." [4] |
| 1846 | Charles Gorman noticed that the symptoms associated with the disease seemed to affect many people in particular regions. Huntington's disease is observed as being localised.[4] |
| 1860 | Johan Christian Lund produced the first description of Huntington's disease while working at Jefferson Medical College.[5] |
| 1872 | George Huntington’s paper, "On Chorea" was published.[2] |
| 1888 | Hoffman describes juvenile Huntington's disease.[6] |
| 1900 | Mendel’s work on inheritance patterns of certain traits was rediscovered.[7] |
| 1908 | Punnett cites Huntington's disease as autosomal dominant.[8] |
| 1978 | Restriction fragment-length polymorphisms (RFLPs) were first described. This was used to locate the gene associated with Huntington's disease in 1983.[9] |
| 1981 | The US–Venezuela Huntington's Disease Collaborative Research Project was initiated. This project aims to find a cure by studying individuals in Venezeula which has the highest concentration of Huntington's disease.[10] |
| 1983 | The HD gene was mapped to the short arm of chromosome 4.[11] |
| 1989 | Linkage disequilibrium between HD gene and the loci D4S95 and D4S98 indicated a 2 Mb candidate region for localisation of HD gene near the loci.[12][13][14] |
| 1993 | The HD gene was isolated and a CAG repeat mutation was identified.[15] |
| 1994 | The Working Group on Huntington's disease of the WFN/IHA published guidelines on counseling for predictive testing of Huntington's disease.[16] |
| 1995 | A study done by Kremer et al. showed that sex of the transmitting parent is the major determinant for CAG intergenerational changes in the HD gene.[17] |
| 1996 | The first mouse model for Huntington's disease was described.[18] |
| 1997 | Aggregates were described in mouse [19] and patient brains with Huntington's disease.[20] |
| 2000 | An inducible mouse model of Huntington's disease was described.[21] |
| 2001 | The first phase-III clinical trials for Huntington's disease were published.[22] |
| 2001 | Huntington's disease-like 2 was first described. It is associated with a novel CAG repeat expansion.[23] |
| 2002 | The first high-throughput screen was published. High-throughput screening is useful in the discovery of HD therapeutics.[24][25] |
| 2004 | A study revealed that 40% of the variance remaining in onset age of Huntington's disease is attributable to genes other than the HD gene and 60% is environmental.[26] |
| 2004 | Langbehn et al. devised a formula based on CAG expansions that may predict whether an HD gene carrier of a given age is “close to” or “far from” onset.[27][28] |
| 2006 | The mitochondrial master gene, PGC1alpha, was found to be abnormally transcribed in Huntington's disease, thus resulting in mitochondrial dysfunction[29] |
| 2009 | Gene therapy stalls development of Huntington's disease in mice.[30] |
Epidemiology
There seems to be an increased prevalence of Huntington's disease among Europeans as compared to Africans and Asians. European populations exhibit a comparatively high prevalence with 4-8 per 100,000 individuals suffering from HD.[31] Two of the most well-known populations in which high prevalence of HD is found was notably in the state of Zulia, Venezuela[32] and Northern Ireland.[33] The overall prevalence of HD in Mexico was also expected to be comparable or even higher to that of European populations.[34].
| Country/Region | Prevalence of HD (individuals per 100,000) |
| Tasmania (Australia) | 12.1 [35] |
| Northern Ireland | 6.4 [33] |
| South East Wales (UK) | 6.2 [36] |
| Olmsted County, Minnesota (US) | 6 - 6.6 (1960) & 1.8 - 2 (1990) [37] |
| Valencia Region (Spain) | 5.38 [38] |
| Slovenia | 5.16 [39] |
| Oxford Region (UK) | 4.0 [40][41] |
| New South Wales (Australia) | 0.65 [42] |
| Japan | 0.65 [43] |
| Finland | 0.5 [44] |
| Taiwan | 0.42 [45] |
| Hong Kong | 0.37 [46] |
| Table 1 Prevalence of Huntington's Disease in various parts of the world |
Table 1 shows the prevalence of HD in different parts of the world, with regions ranked according to how prevalent HD is.
Countries with the highest prevalence are from Europe with most appearing at the top of the table whereas Asian countries are found at the bottom half of this table. This indicates the lower prevalence of HD in Asia as compared to that in European populations. This observation is further supported by a study done by Shiwach and Lindenbaum (1990), it was found that the minimum prevalence of HD among immigrants from the Indian subcontinent was found to be almost half that found in the indigenous UK population.[47] For those areas where there are intermarriages with Europeans, there is a higher occurrence of the disease. This is related to the higher frequency of huntingtin alleles with 28–35 CAG repeats in Europeans and the fact the disease is autosomal dominant. [48]
In a paper by Warby et al. (2011), it was reported that HTT haplotypes contribute to the difference in prevalence of HD between European and East Asian populations. Haplotypes are sets of single-nucleotide polymorphisms (SNPs) on a single chromosome of a chromosome pair that are statistically associated. Different HD haplotypes have different mutation rates, resulting in expansion of CAG tract (marker for HD).[49] Hence, for HTT haplotypes with higher mutation risk such as A1 and A2 halotypes, individuals are more susceptible to HD due to CAG expansion and this corresponds to higher prevalence. This is supported by the findings that higher risk A1 and A2 HD halotypes composed the majority of HD chromosomes in Europe whereas it is absent in China and Japan.[50]
| HD Halotypes | General Population | HD Chromosomes | ||
| East Asia | Europe | East Asia | Europe | |
| A1 | 0.00 | 0.07 | 0.00 | 0.50 |
| A2 | 0.00 | 0.13 | 0.00 | 0.29 |
| B | 0.16 | 0.04 | 0.10 | 0.00 |
| C | 0.40 | 0.47 | 0.77 | 0.02 |
| Table 2 HTT haplotype frequency in East Asia and Europe [49] |
The incidence rate of HD increases with age. It was reported in Taiwan that the range of age at which most onset of HD occurs is between 40-49 years in males and between 50-59 years in females.[45] This trend is similar to that reflected in a Northern Ireland study, whereby the age group in which the highest number of HD onset occurs is 40-44 years.[33] Both of the above-mentioned studies concluded that there is no significant difference for the age of onset between males and females, indicating no sexual predominance for HD.
Treatment
There is no cure for Huntington's disease. Similar to AIDS, only the symptoms of HD can be treated.[51]
Medications
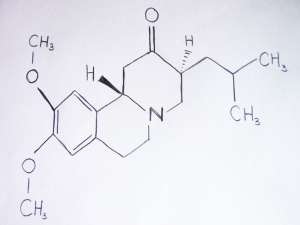
Tetrabenazine
Tetrabenazine was approved by the U.S. Food and Drug Administration in August 2008 to treat HD, making it the first drug approved for use in the United States to treat the disease.[52]
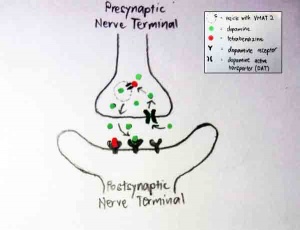
Tetrabenazine is a dopamine-depleting agent which helps to suppress chorea in HD and other hyperkinetic movement disorders such as Tourette's syndrome and tardive dyskinesia.[53] Its role as a dopamine-depleting agent is achieved by inhibiting the vesicular monoamine transporters (VMAT).
There are two types of VMAT: VMAT1 (located in pheripheral endocrine and paracrine cells) and VMAT2 (located predominantly in the brain and in sympathetic neurons).[54] Tetrabenazine binds selectively with a high affinity to VMAT2 and low affinity for VMAT1. VMAT2 is the only transporter that transports dopamine from the cytoplasm into synaptic vesicles for storage and eventual release.[55]
By inhibiting VMAT2, dopamine will not be packaged into vesicles and hence, unable to travel across the synaptic cleft. Tetrabenazine also binds and inhibit to dopamine receptors. This suppresses the amount of dopamine binding to the dopamine receptor located at the post synaptic nerve terminal. Thus, the neurones will not be stimulated and no cascade for the inducement of kinetic movements will be triggered.
Other drugs
| Types of symptoms | Types of medications | Active chemical ingredient | Drugs | Possible negative side-effects |
| Movement disorders | Antiseizure drugs
(anticonvulsants) |
Valproic acid [57] | Depakene Depakote | renal toxicity [58][59], hepatoxicity [60], hemorrhagic pancreatitis, coagulopathies, bone marrow suppression and encephalopathy [61] |
| Lorazepam | Ativan | sleepiness [62], withdrawal symptoms [63], memory impairment and confusion [64] | ||
| Lamotrigine | Lamictal | chorea [65], toxic epidermal necrolysis [66][67] and multiorgan failure [68] | ||
| Levetiracetam [69] | Keppra | somnolence, lethargy [70], rhinitis, asthenia [71], headache and dizziness [72] | ||
| Tetrabenazine [73] | Xenazine | drowsiness, depression, nausea, worsening of parkinsonism, xerostomia, hypotension [74] | ||
| Clonazepam | Klonopin Rivotril | drowsiness [75], depression, disinhibition and sexual dysfunction [76] | ||
| Antianxiety drugs | Diazepam | Valium Antenex | rebound anxiety after withdrawal [77] | |
| Psychiatric disorders | Antidepressants | Escitalopram | Lexapro Lexamil Lexam | insomnia, somnolence, dizziness, sweating, constipation, fatigue [78], sexual dysfunction [79], suicidal ideation [80] |
| Fluoxetine | Prozac Sarafem Fontex | mania [81], akathisia [82], asthenia, tremor, nausea [83] | ||
| Sertraline | Zoloft Lustral | similar effects to flouxetine and escitalopram: movement disorders and sexual dysfunction [84][85] | ||
| Nortriptyline | Aventyl Noritren Sensoval Pamelor | suicidal ideation [80], hepatic failure [86], dry mouth, sedation, constipation and increased appetite | ||
| Mirtazapine | Remeron Avanza Zispin | dry mouth, sedation, and increases in appetite and body weight [87], restless legs syndrome (RLS) [88] | ||
| Antipsychotic drugs | Haloperidol | Haldol Serenase Linton | acute dystonia, akathisia, parkinsonism [89], cognitive decline & brain damage [90] | |
| Clozapine | Clozaril Clopine Zaponex | agranulocytosis [91], myocarditis [92], gastrointestinal hypomotility [93] | ||
| Mood-stabilizing drugs | Lithium | Lithobid | renal failure [94], nystagmus [95], teratogenicity [96] | |
| Carbamazepine | Tegretol Carbatol Sirtal | congenital malformations [97], pitch perception deficit [98] |
Table 3 Symptomatic medications for Huntington's disease
Note: Some drugs have overlapping effects eg. valproic acid and lamotrigine can also used as mood-stabilising drugs.
Disclaimer: The table above is not a comprehensive reference. Please consult your doctor for further information.
Therapies
Psychotherapy: Aims to help a person manage behavioural problems, develop coping strategies, manage expectations during progression of the disease and facilitate effective communication among family members.
Speech therapy: HD significantly impairs control of muscles of the mouth and throat that are essential for speech, eating and swallowing Hence, this therapy addresses difficulties with muscles used in eating and swallowing.
Physical Therapy: It helps to enhance strength, flexibility, balance and coordination. These exercises can help maintain mobility as long as possible and may reduce the risk of falls. Patients may need to use a walker or wheelchair to assist them.
Occupational Therapy: This therapy requires the use of assistive devices that improve functional abilities.
- Handrails at home
- Assistive devices for activities such as bathing and dressing
- Eating and drinking utensils adapted for people with limited capabilities
Glossary
Agranulocytosis: Failure of the bone marrow to make enough white blood cells (neutrophils).
Akathisia: Also known as the restless legs syndrome (RLS), it is a disorder in which there is an urge or need to move the legs to stop unpleasant sensations.
Asthenia: The lack of strength or energy.
Asymptomatic: Showing no evidence of disease
Atrophy: A wasting away of the body or of an organ or part, as from defective nutrition or nerve damage.
Autosomal dominant: An inheritance pattern in which a gene on one of the non-sex chromosomes that is always expressed, even if only one copy is present.
Cerebral Cortex: Grey, neural tissue (1.5mm to 5mm) that covers the outermost layer of the brain. It is involved in important functions of the brain such as language, motor function, planning and organisation, attention, personality, memory, touch and consciousness.
Chorea: a disorder characterised by an abnormal involuntary jerky dance-like movement. Chorea is derived from the Greek word khoreia which means dance.
Coagulopathies: a disease or condition affecting the blood's ability to coagulate.
Cognitive: of or pertaining to the mental processes of perception, memory, judgment, and reasoning, as contrasted with emotional and volitional processes.
Computed tomography (CT): A technique for producing 2-D and 3-D cross-sectional images of an object from flat X-ray images
Degeneration:A process by which a tissue deteriorates, loses functional activity, and may become converted into or replaced by other kinds of tissue.
Dopamine: A catecholamine neurotransmitter
Dystonia: A movement disorder which causes involuntary repetitive contractions of muscles.
Encephalopathy: diseases of the brain.
Gastrointestinal hypomotility: A condition resulted from the lack of gastrointestinal movement, giving rise to severe constipation, fecal impaction, paralytic ileus, bowel obstruction, acute megacolon, ischemia or necrosis.
Haplotypes: Sets of single-nucleotide polymorphisms (SNPs) on a single chromosome of a chromosome pair that are statistically associated.
Hemorrhagic pancreatitis: Inflammation of the pancreas accompanied by the formation of necrotic areas on the surface of the pancreas and in the omentum and, frequently, also accompanied by hemorrhages into the substance of the gland.
Hepatoxicity: Poisoning of the liver.
Hypotension: Low blood pressure.
Linkage Analysis: Study aimed at establishing linkage between genes
Magnetic Resonance Images (MRI): a medical imaging technique used in radiology to visualize detailed internal structures. MRI makes use of the property of nuclear magnetic resonance (NMR) to image nuclei of atoms inside the body.
Mania: A state of abnormally elevated or irritable mood, arousal or energy levels.
Myocarditis: Inflammation of the heart muscle.
Neuronal: A specialized, impulse-conducting cell that is the functional unit of the nervous system, consisting of the cell body and its processes, the axon and dendrites.
Neuropathological: The pathology of the nervous system.
Nystagmus: Involuntary eye movements.
Parkinsonism: A neurological syndrome characterized by tremor, hypokinesia, rigidity, and postural instability.
Polymorphisms: The existence of two or more clearly different phenotypes in the same population of a species.
Positron Emission Tomography (PET): a nuclear medicine imaging technique that produces a three-dimensional image or picture of functional processes in the body.
Renal toxicity: Poisoning of the kidney.
Rhinitis: Irritation and inflammation of some internal areas of the nose, resulting in ‘stuffy nose’.
Single-photon emission computed tomography (SPECT): a nuclear medicine tomographic[1] imaging technique using gamma rays.
Somnolence: Better known as drowsiness, it is a state of near-sleep, a strong desire for sleep, or sleeping for unusually long periods.
Striatum: A subcortical part that is situated in the centre of the brain and is part of a larger system called the basal ganglia. It receives input from the cerebral cortex.
Teratogenicity: The capability of inducing fetal malformations.
Toxic epidermal necrolysis: A life-threatening dermatological condition in which the epidermis is deattached from the dermis all over the body.
Visuospatial: pertaining to perception of the spatial relationships among objects within the field of vision.
Xerostomia: Dry mouth resulting from reduced or absent saliva flow.
References
- ↑ Huntington G (1872). "On Chorea". Medical and Surgical Reporter of Philadelphia (The Hague: Nijhoff) 26 (15): 317–321. ISBN 9061860113. [1]
- ↑ 2.0 2.1 <pubmed>11232352</pubmed>
- ↑ <pubmed>16136077</pubmed>
- ↑ 4.0 4.1 Harper P (2002). "Huntington's disease: a historical background". In Bates G, Harper P, and Jones L. Huntington's Disease – Third Edition. Oxford: Oxford University Press. pp. 3–24. ISBN 0-19-851060-8.
- ↑ Wexler A, Wexler N (2008). The Woman Who Walked Into the Sea: Huntington's and the Making of a Genetic Disease. Yale University Press. p. 288. ISBN 978-0-300-10502-5.
- ↑ Hoffmann, J. Über Chorea chronica progressiva (Huntingtonsche Chorea, Chorea hereditaria). Virchows Arch. A 111, 513–548 (1888) (in German)
- ↑ On the Origin of Mendelian Genetics Amer. Zool. (1986) 26 (3): 753-768. [2]
- ↑ Punnett, R. C. Mendelian inheritance in man. Proc. R. Soc. Med. 1, 135–168 (1908)
- ↑ <pubmed>281713</pubmed>
- ↑ <pubmed>2881213</pubmed>
- ↑ <pubmed>6316146</pubmed>
- ↑ <pubmed>2531223</pubmed>
- ↑ <pubmed>2531224</pubmed>
- ↑ <pubmed>1680285</pubmed>
- ↑ <pubmed>8504314</pubmed>
- ↑ <pubmed>7966192</pubmed>
- ↑ <pubmed>7668260</pubmed>
- ↑ <pubmed>9267033</pubmed>
- ↑ <pubmed>8898202</pubmed>
- ↑ <pubmed>9302293</pubmed>
- ↑ <pubmed>10778856</pubmed>
- ↑ <pubmed>11502903</pubmed>
- ↑ <pubmed>11761463</pubmed>
- ↑ <pubmed>12200548</pubmed>
- ↑ <pubmed>18336216</pubmed>
- ↑ <pubmed>14993615</pubmed>
- ↑ <pubmed>15025718</pubmed>
- ↑ <pubmed>21626556</pubmed>
- ↑ <pubmed>17055784</pubmed>
- ↑ <pubmed>19864571</pubmed>
- ↑ <pubmed>1535611</pubmed>
- ↑ <pubmed>2139171</pubmed>
- ↑ 33.0 33.1 33.2 <pubmed>7562964</pubmed>
- ↑ <pubmed>19672992</pubmed>
- ↑ <pubmed>2142982</pubmed>
- ↑ <pubmed>9231935</pubmed>
- ↑ <pubmed>8018043</pubmed>
- ↑ <pubmed>9528016</pubmed>
- ↑ Peterlin B, Kobal J, Teran N, Flisar D, Lovrecić L.Epidemiology of Huntington’s disease in Slovenia. Acta Neurol Scand.: 2009 PMID:18976322 [3]
- ↑ <pubmed>21088431</pubmed>
- ↑ <pubmed>8108531</pubmed>
- ↑ <pubmed>11008591</pubmed>
- ↑ <pubmed>8752454</pubmed>
- ↑ <pubmed>2889026</pubmed>
- ↑ 45.0 45.1 <pubmed>20881427</pubmed>
- ↑ <pubmed>7586664</pubmed>
- ↑ <pubmed>2151860</pubmed>
- ↑ DC Rubinsztein, Molecular biology of Huntington's disease (HD) and HD-like disorders. In: S Pulst, Editor, Genetics of movement disorders, Academic Press, California (2003), pp. 365–377.
- ↑ 49.0 49.1 <pubmed>19249009</pubmed>
- ↑ <pubmed>21248742</pubmed>
- ↑ <pubmed>19588393</pubmed>
- ↑ <pubmed>20957126</pubmed>
- ↑ <pubmed>16466307</pubmed>
- ↑ <pubmed>8643547</pubmed>
- ↑ <pubmed>2751365</pubmed>
- ↑ <pubmed>PMH0000677</pubmed>
- ↑ <pubmed>16507108</pubmed>
- ↑ <pubmed>8108303</pubmed>
- ↑ <pubmed>21868496</pubmed>
- ↑ <pubmed>21492891</pubmed>
- ↑ <pubmed>18201150</pubmed>
- ↑ <pubmed>4341107</pubmed>
- ↑ <pubmed>6131080</pubmed>
- ↑ <pubmed>3960963</pubmed>
- ↑ <pubmed>16900938</pubmed>
- ↑ <pubmed>9061826</pubmed>
- ↑ <pubmed>12892003</pubmed>
- ↑ <pubmed>19380079</pubmed>
- ↑ <pubmed>16340384</pubmed>
- ↑ <pubmed>18728811</pubmed>
- ↑ <pubmed>10908898</pubmed>
- ↑ <pubmed>10999557</pubmed>
- ↑ <pubmed>16476934</pubmed>
- ↑ <pubmed>19050408</pubmed>
- ↑ <pubmed>12390050</pubmed>
- ↑ <pubmed>2889724</pubmed>
- ↑ <pubmed>6145363</pubmed>
- ↑ US Food and Drug Administration Review and Evaluation of Clinical Data: Escitalopram Oxalate
- ↑ <pubmed>16430968</pubmed>
- ↑ 80.0 80.1 <pubmed>19832967</pubmed>
- ↑ <pubmed>3485926</pubmed>
- ↑ <pubmed>2549018</pubmed>
- ↑ Eli Lilly and Company Prescribing Information of Prozac(June 15, 2011)
- ↑ <pubmed>8909330</pubmed>
- ↑ <pubmed>11229450</pubmed>
- ↑ <pubmed>8848811</pubmed>
- ↑ <pubmed>11607047</pubmed>
- ↑ <pubmed>18756499</pubmed>
- ↑ <pubmed>17054159</pubmed>
- ↑ <pubmed>18228005</pubmed>
- ↑ <pubmed>8515788</pubmed>
- ↑ <pubmed>17194170</pubmed>
- ↑ <pubmed>18452342</pubmed>
- ↑ <pubmed>19940841</pubmed>
- ↑ <pubmed>12552061</pubmed>
- ↑ <pubmed>11948561</pubmed>
- ↑ <pubmed>21127116</pubmed>
- ↑ <pubmed>12581810</pubmed>