User:Z3332337
Lab Attendance
Lab 1 --Z3332337 11:49, 25 July 2012 (EST)
Lab 2 --Z3332337 10:15, 1 August 2012 (EST)
Lab 3 --Z3332337 10:17, 8 August 2012 (EST)
ANAT2341 – LAB 1 25/07/12
- The type of cells in the zona radiata are the ‘granulosa’ cells.
- Zona pellucida is a specific extracellular matrix to the (development) of the oocyte. Consists of 3-4 thick glycoproteins made by the oocyte itself and the granulosa cells are attached to the outside.
- The fusion of the spermatozoa to the zona pellucida stimulates the oocyte to continue into the final stages of meiosis.
- The production of the final polar body (which contains half the chromosomes of the original germ cell) is also stimulated.
Occasionally, the 3rd polar body is made by the 1st polar body which also undergoes meiosis. NB: The polar bodies are “trash bags” of the oocytes.
- In meiosis the abnormality trisomy 21 (down syndrome) “leaves behind chromosome 21” (ie: isn’t separated properly).
Two other trisomy’s exist, trisomy’s 18 & 13 (occur in this order). Major genetic abnormalities are spontaneously aborted in the first 2 weeks of development because they are incompatible with proper development.
- The proliferating spermatagonia (make MORE spermatogonia which) are the ones who continue on to complete meiosis, which create spermatids (haploid cells).
- These daughter cells are initially cross-linked until maturation of spermatid into spermatozoa.
- FINAL (functional) MATURATION, doesn’t occur in the epididymis (although altered here).
It actually occurs after ejaculation into the vagina where it undergoes capacitation. The pH in the vaginal canal causes this change.
Capacitation
Sperm undergo morphological, physiological and biochemical changes during the journey through the female reproductive tract; a process called Capacitation.
Semen contains factors that do not allow the sperm to penetrate the ovum and these are removed in the female reproductive tract.
Capacitation needs to occur before the sperm are capable of penetrating and fertilising an ovum.
Many sperm are required to dissolve the zona pellucida of the ovum, but only one gets the chance to fertilize.
Oogenesis
- NB: the ovary is located in the PERITONEAL CAVITY.
- Ovarian follicle atresia: atresia refers to the degeneration and subsequent resorption of one or more immature ovarian follicles.
NB: this can happen at any time in the cyle.
- The medullary region of the ovary is highly vascularized because hormones (FSH and LH) are being brought in to regulate menstruation and pregnancy (negative feedback loop).
Supported by the granulosa cells.
Follicle Classification The above images show the histological changes that occur with follicle development (folliculogenesis). In humans, this entire process occurs over the timecourse of at least 3 menstrual cycles. This means that within the ovary during each cycle (at any point in time) many follicles can be either developing (folliculogenesis), regressing (atresis) and only a single follicle will be selected as ready for release. The selected follicle readied for release, generally one of the largest antral follicle, and can be classifed or described as: an antral preovulatory follicle or Graafian follicle or type 8 follicle (depending upon the classification used).
Classification systems - There are several different nomenclatures for the stages of follicle maturation (shown below) all of which makes the literature very confusing. The simplest is primordial, preantral, antral, Preovulatory (Graffian). You can also use the 5 step follicle classification: Primordial, Primary, Secondary, Tertiary, Preovulatory. Note that some classifications refer to the antral follicle as a secondary follicle and do not use the term tertiary follicle.
- Primordial Follicle - Alternative nomenclature: small follicle or type 1, 2, 3 (25 cells) less than 50 micron diameter.
- Preantral Follicle - Alternative nomenclature: preantral follicle or type 4 (26-100 cells), type 5 (101-300 cells) up to 200 micron diameter.
- Antral Follicle - Alternative nomenclature: small antral type 6 (301-500 cells), large antral type 7 (501-1000 cells) small antral 500 micron diameter, large antral 1000-6000 micron diameter.
- Preovulatory Follicle - Alternative nomenclature: largest antral follicle or Graafian follicle or type 8 (>1000 cells) greater than 6000 micron diameter.
NB: IT TAKES MORE THAN 1 CYCLE TO MATURE TO BE SELECTED TO BE RELEASED IN OVULATION.
Spermatogenesis
- Microtubule organisational centre radiates for motility.
- Acrosome (‘acrosomal head’) is a modified golgi apparatus.
Acrosome is a large vesicle, containing enzymes and proteins that enable sperm to penetrate oocyte by dissolving the zona pellucida.
- Mitochondria are tightly packed within the mid-tail of the spermatozoa to generate the ATP required to drive the whip-like movements of the tail.
- There are 3 types of cells: germ, support and hormonal.
- Only 2 cells (sertoli and spermatogonia).
Lab 1 Assessment
Article Summary
Effect of the method of conception and embryo transfer procedure on mid-gestation placenta and fetal development in an IVF mouse model.
This article discusses the effects of using In Vitro Fertilisation (IVF) against "In Vivo" mouse models on the fetal development, using birthweight as the a quantifiable variable.
This article showed that IVF fetuses had a lower birthweight than fetuses from In vivo pregnancies.
They implanted sites for all IVF mice had similar implantion sites, however this study shows that IVF mice have higher abortion rates along with a lower birthweight when compared with in vivo embryos. It shows here that IVF embryos are more likely to be delayed in reaching the blastocyst stage when compared to "in vivo". They also show delayed development after the blastocyst stage which may be related to the low birthweight of IVF embryos.
Reference: Delle Piane, L., Lin, W., Liu, X., Donjacour, A., Minasi, P., Revelli, A., Maltepe, E. and Rinaudo, P.F. (2010). Effect of the method of conception and embryo transfer procedure on mid-gestation placenta and fetal development in an IVF mouse model. European Society of Human Reproduction and Embryology , 25 (8), 2039-2046.
IVF Nobel Prize
Professor Robert Edwards won the Nobel Prize in Medicine in 2010.
http://www.abc.net.au/news/2010-10-04/father-of-ivf-wins-nobel-prize/2285078
Lab 2 Assessment
Detection of methylation by zygote staining
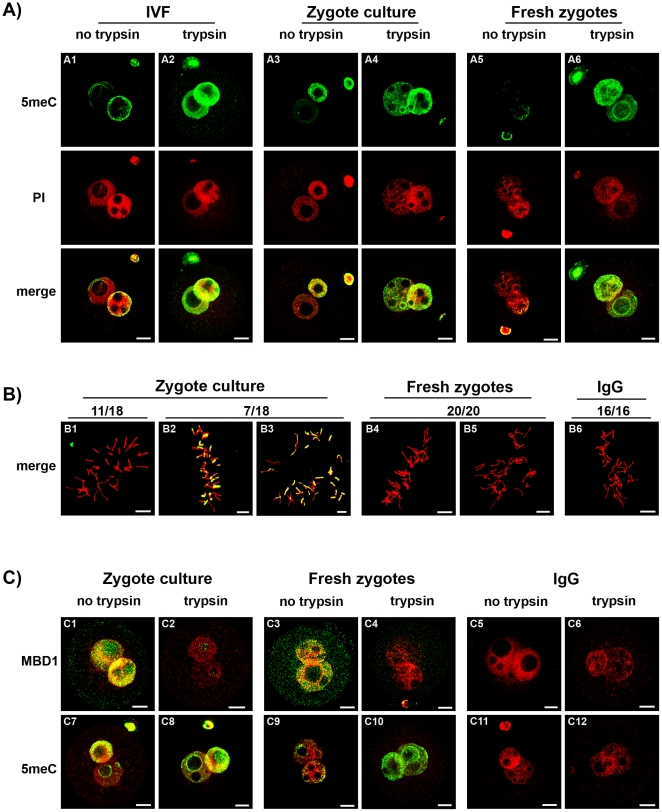
Asymmetric anti-5meC staining of the male and female pronucleus after acid-pretreatment has been reported [5], [6], [20], [21] yet was not confirmed by this study. The zygotes used in past studies were commonly generated by in vitro fertilization or subjected to culture in vitro (which provides logistic advantages for the feasibility of such studies). After antigenic unmasking with acid, the smaller (female) pronucleus in zygotes produced by in vitro fertilization (Fig 7A1) and or cultured in vitro (Fig 7A3) showed more anti-5meC staining compared to those collected directly from the oviduct (Fig 7A5). After antigenic unmasking by acid and trypsin, however, high levels of anti-5meC staining were consistently observed in both pronuclei of IVF (Fig 7A2), cultured (Fig 7A4) and fresh PN5 zygotes (Fig 7A6). Analysis of metaphase zygotes showed that culture from the early zygote stage caused variable levels of anti-5meC staining to persist in acid-only treated zygotes (Fig 7B). The level of methylation was assessed further by comparing staining with anti-MBD1 and anti-5meC in fresh and cultured zygotes (Fig 7C). This analysis showed that a similarly high level of MBD1 staining was observed in PN5 cultured (Fig 7C1) and fresh (Fig 7C3) zygotes, yet 5meC staining persisted in an asymmetrical fashion in cultured (Fig 7C7) but not fresh (Fig 7C9) zygotes. After acid and trypsin unmasking the MBD1 staining was lost from both treatments (Fig 7C2,4) and resulted in a similarly high level of staining with anti-5meC in both cultured and fresh zygotes (Fig 7C8,10). No staining was detected with non-immune control antisera for either antibody (Fig 7C5,6 and C11,12). The current results show that manipulation of the early embryo interferes with the maturational changes in zygotic chromatin that results in acid-resistant antigenic masking of 5meC, and this reduced level of masking was greatest in the female pronucleus giving an artifactual appearance of asymmetric demethylation.
Reference: Li, Y., & O'Neill, C. (2012). Persistence of cytosine methylation of DNA following fertilisation in the mouse. PLoS One., 7(1), e30687. Epub 32012 Jan 30626. Copyright Li, O'Neill. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Identify a Protein Associated with Implantation
A protein associated implantation is Proprotein 6 Convertase (PC6), a serine protease. It is involved in endometrial receptivity and is integral for cellular remodelling by cleavage of scaffolding protein ezrin-radixin-moesin binding phosphoprotein 50 (EBP50). Cleavage event allows for the ezrin protein to integrate the actin cytoskeleton and the plasma membrane, increasing chances of binding and therefore implantation. Knockout mice were used to down-regulate PC6 and results in failure of cleavage of EBP50 and therefore a decrease of healthy embryo implantation when compared to the wild type mice.
LAB 3 Assessment
1. Identify the difference between "gestational age" and "post-fertilisation age" and explain why clinically "gestational age" is used in describing human development.
The "gestational age" is any time between conception and birth, typically measured from the woman's last menstrual period until the current time. Whereas, "post-fertilisational age" is specifically the time after the ovum is fertilised.
"Gestational age" is used to in describing human development because it is measures pregnancy in weeks, as a pregnancy typically lasts 38-42 weeks.
http://www.parentsconnect.com/pregnancy/trying-to-conceive/Gestational_Fetal_Age.html
2. Identify using histological descriptions at least 3 different types of tissues formed from somites.
Somites which develop from the paraxial mesoderm, give rise to 3 different types of tissues: dermatome, myotome and sclerotome tissues.
Dorsolaterally there is the: Dermatome which forms the dermis and and contributing fibroblasts, & Myotome which forms the myoblasts (primordial muscle cells).
Ventromedially there is the: Sclerotome which forms the vertebrae and the ribs.