2016 Group Project 5
| 2016 Student Projects | ||||
|---|---|---|---|---|
| Signalling: 1 Wnt | 2 Notch | 3 FGF Receptor | 4 Hedgehog | 5 T-box | 6 TGF-Beta | ||||
| 2016 Group Project Topic - Signaling in Development
OK you are now in a group, add a topic with your student signature to the group page. | ||||
| This page is an undergraduate science embryology student project and may contain inaccuracies in either descriptions or acknowledgements. | ||||
| Group Assessment Criteria |
|---|
 Science Student Projects Science Student Projects
|
| More Information on Assessment Criteria | Science Student Projects |
| 2016 Student Projects | ||||
|---|---|---|---|---|
| Signalling: 1 Wnt | 2 Notch | 3 FGF Receptor | 4 Hedgehog | 5 T-box | 6 TGF-Beta | ||||
| 2016 Group Project Topic - Signaling in Development
OK you are now in a group, add a topic with your student signature to the group page. | ||||
| This page is an undergraduate science embryology student project and may contain inaccuracies in either descriptions or acknowledgements. | ||||
| Group Assessment Criteria |
|---|
 Science Student Projects Science Student Projects
|
| More Information on Assessment Criteria | Science Student Projects |
T-box genes and their signalling pathway
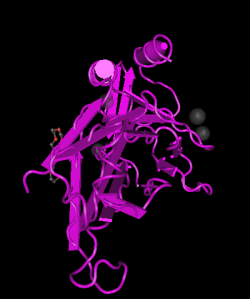
Introduction
The T-Box genes encode for a series of T-box proteins, a family of transcription factors that with more than 20 members identified in humans so far, and homologues in many other organisms including different vertebrates. These T-box proteins are termed as transcription factors because of their ability to regulate the expression and subsequent activity of other genes by binding to DNA. T-box proteins are characterised by a DNA-binding motif known as the T-box that binds DNA. Many researchers have identified that the T-box genes play an important roles of the development of the heart, respiratory system and limbs. Since these genes are involved in the development of certain important regions of human body, mutations of them leads into human gene disorders including Di-George Syndrome and Holt-Oram syndrome. [2] This page would give a board overview of how is the T-box signalling pathway worked and its importance, the discovering, abnormalities and animal models that used for researching. It is important to note that T-box genes have also found to regulate the patterning and cell fate, cell survival, and/or proliferation but not be included in this page. For a more comprehensive overview about the role of T-box genes, please click: PMID 25294936 [3] to read more.
T-Box gene and embryology
The T-box gene family, plays an essential role in controlling embryogenesis in a wide variety of organisms (invertebrates, amphibians, mammals) [4] . The box-gene family encodes related DNA-binding transcriptional regulators and the genes exhibit diverse patterns of both spatial and temporal expression in the developing embryo. Studies both in genetic and molecular embryology have shown the importance of these genes in regulating cell fate decisions which later during embryological development are important in establishing the early body plan and later are important during the organogenesis process. [4]
Mark's comments - what the signalling pathway does - clearly identify exactly what this page will talk about - eg. this pathway in reference to this system/embryonic development etc SEE ABOVE have done this - limitations of this page - what we won't discuss - put an image in straight away to break the copious amounts of text at the start of the page
PMID 9504043
Origins of the T-box name
The founding member of the T-box family is brachyura which comes from the greek and means short tail [3]. This gene was discovered following experimental studies with a short tailed mouse that harboured a mutation which affected tail length and embryonic development [5]. Brakhus means short in greek and oura meaning tail.
Nadezhda Alexandrovna Dobrovolskaïa-Zavadskaïa first described the brachyury mutation in 1927 as a mutation that affected tail length and sacral vertebrae in heterozygous mice [5]. Unfortunately, the original article had been lost and only the paraphrased version is subsisted.
 This image is a photograph of Nadezhda Alexandrovna Dobrovolskaïa-Zavadskaïa in 1948 taken from [6] |
The brachyury gene (which is also known as T) soon was studied in great detail due to its important role in the development of the notochord and posterior mesoderm. Mutations in T are shown to cause embryonic lethality in homozygote mice and short tails in heterozygote mice [3]. Now according to human and mouse genomes, the gene brachyura has the symbol T and gene name T. However the gene is described as brachyury.
Ancient origins and evolution of the T-box gene family
T-box gene family is ancient in origin and it is thought to be found in all metazoans [7]. Due to the increasing availability of sequenced genomes from a diverse group of animal taxa we now know that the origin of the T-box family has been pushed back to unicellular organisms and fungi, in which contain only one or two T-box genes ancestors , including T, have been identified. [8]
With the analysis of the genomes of bilaterian organisms and representatives of the different phyla, four basal metazoan phyla indicate that T is the most ancient member of the T-box family and that the family has expanded throughout metazoan evolution. [3] In Metazoan evolution it seems that genes were added progressively one by one by gene or genome duplication and some of these genes were lost and gained in specific lineages [3]. In present day or extant vertebrates we now know that the T-box family has radiated throughout the vast vertebrate linages and can be grouped into five subfamilies T, Tbx1, Tbx2, Tbx6 and Tbr1 [3]. In the common ancestor of vertebrates and sponges, four of these five subfamilies were already present [8].
Brachyury expression in 7.5dpc CD1 mouse embryos [9] Image from here
The discovery of T-box genes
The story of the T-box genes began in Paris at the Pasteur laboratory in the 1920s with the Russian scientist Nadine Dobrovolskaïa-Zavadskaïa, who embarked on a pioneering screen for X-ray-induced developmental mouse mutants. Her isolation of a mouse strain with a short tail, caused by a semidominant heterozygous mutation in a locus she called T, represented one of the first successful mammalian genetic screens, and provided one of the earliest links between gene activity and cell behaviour during embryogenesis[10]. The mid-gestational death of homozygous T embryos, with perturbed development of the posterior mesoderm and notochord, demonstrated an essential requirement for T during gastrulation, and led to the earliest insights into the inductive influences of notochord on neural tube and somite development. Over 60 years later T, now also known as brachyury, meaning ‘short tail’ in Greek, was cloned in one of the earliest positional cloning efforts in the mouse[11]. At the time, lack of homology in the T-gene product to any previously characterized protein gave no clues as to its biochemical role until, in 1993, it was revealed to be a novel sequence-specific DNA-binding protein[12]. Crystallographic determination of the structure of the DNA-binding domain, now called the Tbox, revealed a new way through which proteins recognize DNA[13].
TO DO: Produce a timeline for the history of the TBox gene - type TBOx into PubMed and sort the findings by age - use this to see when the first journal articles where released related to TBox and limb development, how this research has improved and developed, where it is heading in the future
Timeline
1927 - Brachyury (T) locus was introduced to the world in a report describing the effects of a mutation at this locus on both embryonic viability in homozygotes and the development of the tail in heterozygotes. [5]
Over the following decades, the embryological defects caused by the T mutation were studied.
1990 - The T gene itself was cloned. [11]
1994 - Bollag and his colleagues showed the existence of a family of T-related genes in the mouse genome, which was christened the T-box gene family. [14]
1995 - The discovery of sequence homology between the mouse T gene and a newly cloned Drosophila gene called "omg". [15] The location of tbx-2 was found, 17q21-22 which means in the long arm (q) of chromosome 17, from region 2, band 1 to region 2, band 2. [16]
1997 - The mapping of the Holt-Oram Syndrome locus was refined to 12q24.1 by fluorescence in situ hybridization, which was tightly linked to Holt-Oram Syndrome.[17].
1998 - Alison Isaac and the team confirmed that in chicken embryo, Tbx-2 & Tbx-3 are related to both forelimb and hindlimb development, and Tbx-4 & Tbx-5 have limited expression domains in leg and wing respectively. [18]
2001 - It was proposed that TBX1 in humans is a key gene in the etiology of DiGeorge syndrome [19].
2003 - 3 mouse Tbx20 splice variants, were cloned and called Tbx20a, Tbx20b, and Tbx20c, and by database analysis they identified a fourth variant, Tbx20d.[20].
2004 - With collaboration of other signalling factor, tbx-3 is involved of mammary gland development in mouse model.[21]
2005 - A deletion of Tbx20 in mouse was found to be embryonic lethal. [22].
2006 - Tbx6 was found to be essential for Mesp2 expression during somitogenesis in mouse.[23].
2007 - A clinical human case shown tbx-5 is related to human pericardium agenesis and verified as one of the symptoms of Holt-Oram Syndrome. [24]
2009 - A Drosophila heart model involving mutation of pannier (pnr) was used to examine the function of GATA4 in adult heart physiology. [25].
2010 - Tbx3 showed to significantly improve the quality of induced pluripotent stem (iPS) cells [26].
2011 - TBX6-dependent regulation of SOX2 was demonstrated to determine the fate of axial stem cells [27].
MORE RECENT RESEARCH
PROTEIN DATABASE OF PUBMED THERE ARE 3D THINGS OF THE TBOX MEMBERS
Features of the T-box family
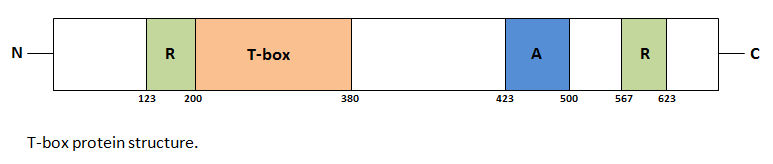
The defining feature of the T-box gene family is a conserved domain that was first uncovered in the sequence of the mouse T locus, or Brachyury gene[11]. This homology domain encodes a polypeptide region that has been named the T-box. [7]
Summary of the main T-box genes
The following table outlines the functions of some important T-box genes, including their location and associated human diseases.
| T-box gene | Main expression sites during embryogenesis | Function | Abnormalities |
| Tbx1 | Pharyngeal endoderm, mesoderm core of the first pharyngeal arch, head mesoderm ventral to hindbrain, sclerotome | Pharyngeal arch arteries development, governs the transition between stem cell quiescence and proliferation in hair follicles, associated with developmental abnormalities with the ear, facial and cardiac outflow | DiGeorge syndrome, Velocardiofacial syndrome, Conotruncal anomaly face syndrome, Tetralogy of Fallot |
| Tbx2 | Allantois, non-chanmber myocardium, optic and otic vesicles, naso-facial mesenchyme, limbs, lungs, genitalia | Potent immortalizing gene that acts by downregulating CDKN2A, regulates Anf expression in chamber myocardium development | None identified |
| Tbx3 | Non-chamber myocardium (sinoatrial region, AV canal and interventricular ring), expressed with TBX2, TBX3, and TBX5 in the embryonic neural retina, mammary gland | Provides positional information important for topographic mapping in differentiation of distinct cell types across the laminar axis of the retina, development of functional ectopic pacemakers, stimulates Nanog, Tbx3 specifies digit III and the combination of Tbx2 and Tbx3 specifies digit IV, acting together with the interdigital BMP signaling cascade. The fetal lung, kidney, heart, liver, and spleen in humans expresses this gene | Ulnar-mammary syndrome |
| Tbx4 | Hindlimb, mandibular and lung mesenchyme, atrium and body wall | Developmental pathways of the lower limbs and the pelvis in humans | Ischiocoxopodopatellar syndrome, Small patella syndrome |
| Tbx5 | Cardiac cresent, heart tube, sinus venous, common atrium, left ventricle (LV) and right ventricle (RV) forelimb, eye | Promotes cardiomyocyte differentiation, interaction with GATA4 cause of human cardiac septal defects | Holt-Oram syndrome |
| Tbx18 | Splanchnic mesoderm, septum traversum, epicardium | Maintain the separation of anterior and posterior somite compartments, specification of ureteral mesenchyme and SMC differentiation in the ureter | Congenital anomalies of kidney and urinary tract 2 |
| Tbx20 | Allantois, lateral plate mesoderm, cardiac crescent, heart tube, hindbrain, eye | Cardiac development and yolk sac vascular remodeling | Atrial septal defect 4 |
Table 1. adapted from: Table 1. Embryonic expression and mutant phenotypes of mouse cardiac T-box genes [28]
SEE IF WE CAN INSERT IMAGES FROM THE PROTEIN PART OF PUBMED - Mark said something about 3D models/images of these - Link to 3D structure: https://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?uid=81644
Functions of T-box in development
T-box genes are a growing family of transcription factors that are expressed in diverse patterns throughout vertebrate development. They have emerged as key players in embryonic patterning, tissue differentiation and morphogenesis, particularly in vertebrates and some specific examples have been described in the following sections.
Cardiac development
The heart is one of the first organs to develop and function in vertebrate embryos. Its formation is a complex process and requires contributions from multiple transcription factors, including GATA4, eHAND, dHAND, Irx4, and TBX genes [29].
In the developing heart, Tbx5 expression can be first detected at stage 12 along the entire rostrocaudal length of the fused heart tube[30]. Although it is expressed very early in cardiac embryogenesis, Tbx5 is not essential for cardiac crescent formation or for development of the early heart tube. The growth of the posterior segment (atria and left ventricle) of the heart is what requires Tbx5, and does not occur in embryos that do not have Tbx5. In contrast, right ventricle and outflow tract development have been shown to be Tbx5-independent[29]. It is interesting to note that the role of TBX5 in the growth and maturation of posterior heart is evolutionarily conserved from amphibia (Xenopus) to mammals[31].
The signalling mechanism of TBX5 involves interaction with the cardiac homeobox protein NKX2-5 which synergistically promotes cardiomyocyte differentiation. Both these molecules bind directly to the promoter of the gene encoding cardiac-specific natriuretic peptide precursor type A (NPPA) alongside each other, and the 2 transcription factors induce activation. Hiroi et al. (2001) proved this by showing that cell lines over-expressing wild-type Tbx5 gene started to expressed more cardiac-specific genes and started to contract earlier. However, cell lines that expressed a mutation in Tbx5 gene did not differentiate into beating cardiomyocytes which indicates that Tbx5 is crucial in cardiomyocyte differentiation.[32].
GATA4 is a transcription factor essential for heart formation has been known to interact with TBX5 to induce normal cardiac septation. An article by Misra et al. (2014), showed that Gata4 and Tbx5 are co-expressed in the embryonic atria and ventricle and that a disruption of myocardial Gata4 and Tbx5 results in defects in cardiomyocyte proliferation and atrioventricular septation. [33] A mutation of GATA4 can result in human congenital heart defects where functional separation of the four cardiac chambers is perturbed[34]. Therefore Tbx5 is involved in directing gene expression in morphogenetic processes associated with specific chamber formation and any mutations can cause heart septal defects as seen in Holt–Oram syndrome[31] (See 'Abnormalities' section below for further info).
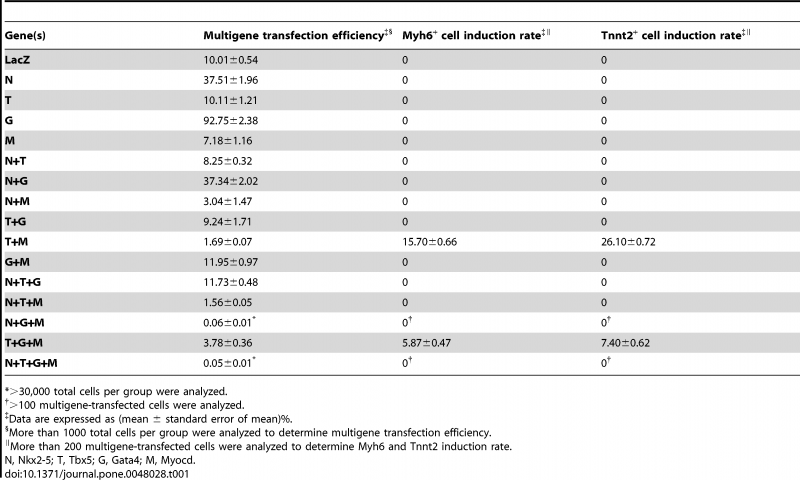
Moreover, cardiac genes Myh6+ and Tnnt2+ cell induction rate was investigated by Zhou et al. (2012) using combining the expression of various genes. The results of the article are summarised below:
Table 2. Multigene transfection efficiency and cardiac genes Myh6+ and Tnnt2+ cell induction rate.
The results from this table demonstrates that cardiac marker protein expression of Myh6 and Tnnt2 was only induced by Tbx5+Myocd (T+M) and Tbx5+Gata4+Myocd (T+G+M) combinations in 10T1/2 non-myoblastic cells . This further supports previous findings that Tbx5 and Gata4 interactions are vital activators of cardiac genes and activated the fewest genes associated with non-cardiac processes[35].
In embryonic development, arteries form within pharangeal arches, and subsequently undergo extensive remodeling that ensures proper outflow connection between the heart and systemic and pulmonary circulation. Tbx1 has been implicated in regulating formation and growth of the pharyngeal arch arteries, growth and septation of the outflow tract of the heart, interventricular septation, and conal alignment. Vitelli et al. (2002) showed that homozygous loss of Tbx1 gene can cause severe vascular and heart defects[36] . This is evidenced through the study done by Lindsey et al. (2001) demonstrated that homozygous Tbx1 mutant embryos do not form pharyngeal arch arteries 3, 4, and 6 [37]. Another study by Jerome and Papaioannou (2001) revealed that mice with a heterozygous Tbx1 mutation had a high incidence of cardiac outflow tract anomalies. They noticed that this modelled one of the major abnormalities of the human DiGeorge Syndrome/Velocardiofacial syndrome and proposed that TBX1 in humans is a key gene in the etiology of this human disorder (See 'Abnormalities' section below for further info) [19].
Limb Development
The initiation of limb outgrowth involves T-box genes as well as Homeobox (Hox) genes. Specific Hox genes are upregulated by retinoic acid in limb fields which then initiates downstream signaling cascades that ensures correct limb growth along its three axes: anterio-posterior, dorso-ventral and proximo-distal [38] [39]. An interesting study conducted by Mohanty et al. (1992) amputated the tails of tadpoles to demonstrate that when their tail stumps were treated with retinoic acid they regenerated legs instead of a tails at the site of amputation [40]. The role of β-catenin in forelimb initiation, however, has not been studied in detail[41].
The initiation of limb outgrowth other transcription factors are expressed to control specific areas of patterning, i.e. forelimb versus hindlimb. Various vertebrate limb models have identified three genes that determine the identity of the developing limb i.e. forelimb or hindlimb. Tbx5 and Tbx4 are T-box family transcription factors specifically expressed in the forelimb and hindlimb, respectively[42][43]. Pitx1, another transcription factor, is expressed in the developing hindlimb, but not the forelimb[44].
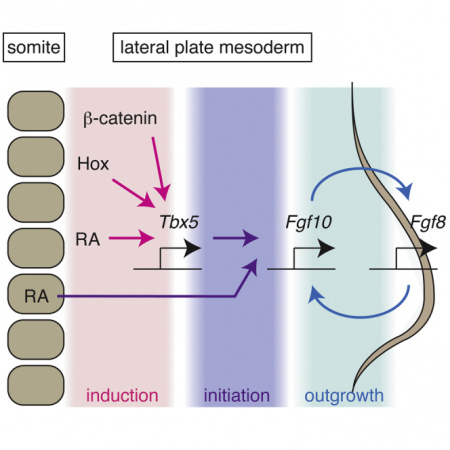
Tbx4 and Tbx5 are essential regulators of limb outgrowth whose roles seem to be tightly linked to the Fibroblast Growth Factor (FGF) and Wnt signaling pathways (more information on these two signalling pathways are described in 2016 Group Project 3 and 2016 Group Project 1 respectively). Initial activation of fibroblast growth factor-10 (Fgf10) in the lateral plate mesoderm of the forelimb and hindlimb is regulated by Tbx5 and Tbx4 respectively [45]. It has also been found that Tbx5 binding sites have been identified in the Fgf10 promoter sequence in mice and humans [46]. The Fgf10 signals the overlying distal ectoderm to induce Fgf8, which is crucial for the formation of the apical ectodermal ridge (AER) at the tip of the developing limb bud. A positive feedback loop, regulated by the Wnt signalling pathway, is then established between Fgf8 and Fgf10, such that Fgf10 promotes Fgf8 expression and Fgf8 promotes Fgf10 expression. If Fgf10 is flanked in mice the resultant embryos develop without limbs, indicating the importance of this fibroblast growth factor. A deletion of either Tbx5 or Tbx4 will also cause outgrowth defects of limb buds and the the FGF and Wnt regulatory loops required for limb bud outgrowth are not established, including initiation of Fgf10 expression [47][48].
The important role of Tbx trancription factors is highlighted in experiments where Tbx5 is knocked out or inactivated. In these studies the embryos that develop do so with complete failure of formation of any elements of the forelimb. These studies further support that Tbx5 interacts with Fgf and Wnt to initiate outgrowth of the limb bud.
A schematic representation of T-box involvement in limb development is outlined in the image below:
Close up of Tbx5 role in the Initiation of Limb Bud Formation[49]
Respiratory Development
Members of two T-box gene family (Tbx2/Tbx3 and Tbx4/Tbx5) are expressed in embryonic lung mesenchyme [50] and have been shown to play important roles in: 1) lung bud and trachea specification, 2) lung branching morphogenesis, and 3) tracheal/bronchial cartilage formation [51].
In lung branching morphogenesis, Tbx4 and Tbx5 genetically interact with one another. In an article by Ripla et al. (2012), they demonstrated that both Tbx4 and Tbx5 are expressed throughout the mesenchyme of the developing lung and trachea[51]. They showed that a loss of Tbx5 leads to a unilateral loss of lung bud specification and absence of tracheal specification in organ culture. Concordant with this defect, the expression of mesenchymal markers Wnt2 and Fgf10, as well as Fgf10 target genes Bmp4 and Spry2, in the epithelium is downregulated. This suggests that Fgf10 signaling pathway is activated downstream of Tbx4 and Tbx5 in the developing lung and that Fgf10 genetically interacts with Tbx4 and Tbx5. This is consistent with finding from Sakiyama et al. (2003) that found out that the Tbx4-Fgf10 system controls lung bud formation during chicken embryonic development. Tbx4 was found to trigger Fgf10 expression in the lung primordium mesoderm which then acquires an inductive capability for the initial budding morphogenesis of primary lung buds[52]. Of significance, lung-specific Tbx4 heterozygous;Tbx5 conditional null mice died soon after birth due to respiratory distress. These pups have small lungs and show severe disruptions in tracheal/bronchial cartilage rings, highlighting the important role of Tbx4 and Tbx5 in respiratory development.
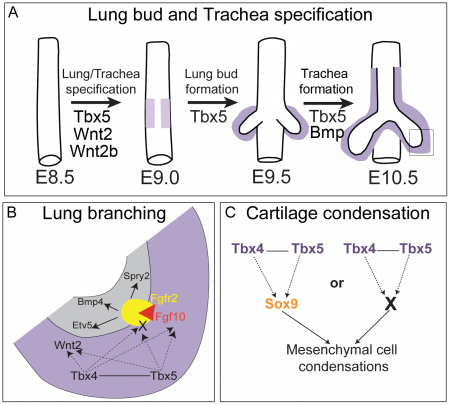
Other developmental events
TBX signalling also involved in palate development. In humans, TBX1 mutation is responsible for the major phenotypes of 22q11.2 deletion syndrome (Velo-cardio-facial/DiGeorge syndrome, discussed in Abnormalities section) as well as non-syndromic submucous cleft palate, suggesting that Tbx1 is a regulator of palatogenesis. A study by Funato et al. (2012) revealed that Tbx1 regulates oral epithelial adhesion and palatal development and showed that Tbx1 -/- mice exhibit various forms of cleft palate phenotypes, including submucosal cleft palate and soft palate cleft.
Dorsoventral Patterning of the Mouse Coat by Tbx15 PMID 14737183 Palate Development PMID 22371266
Tbx6 interacting with Ripply for the formation of somite boundaries (in zebrafish) PMID 25725067 Tbx6 is required for the expression of mesp-b and ripply1 in the paraxial mesoderm during somite formation, and for the specification of the central Pax3+/Pax7+ dermomyotome. Mesp-b is necessary and sufficient for central dermomyotome formation, it inhibits myogenic differentiation and promotes dermomyotome development. Ripply1 function is required for maturation and fast muscle fiber differentiation.
Results show that Tbx6 protein has to be removed for the expression of pax3/7 and myoD in the lateral paraxial mesoderm, indicating that Tbx6 and/or Tbx6-dependent genes inhibit maturation of myogenic cells. Downstream of Tbx6, Mesp-ba promotes dermomyotome development.
Abnormalities
A number of human disorders have been linked to mutations in T-box genes, confirming their medical importance. They include Holt– Oram syndrome/TBX5, Ulnar-Mammary syndrome/TBX3, and more recently DiGeorge syndrome/TBX1, ACTH deficiency/TBX19 and cleft palate with ankyloglossia/TBX22 and it is trusted that more disease would be found. [54] [55] [56]
TBX1/DiGeorge Syndrome
The TBX1 gene can be mapped on chromosome 21 within the DiGeorge syndrome region. Studies using mice, have shown that the TBX1 gene is haploinsufficient.[57] This means that while although mice are diploid organisms, only one functional copy of the gene exists, while the other copy has undergone a mutation, making it inactivated. This single gene is unable to produce sufficient amounts of the protein needed for TBX1 to carry out its function- the development of pouches and pharyngeal arches in these mice. In this study of mice, a deficiency of the TBx1 gene resulted in heart defects, hence suggesting the close association between the TBx1 gene and cardiovascular development.
In this study, a mouse model was used where the relevant region on chromosome 16 of the mice species, corresponding to chromosome 22q11.2, or del22q11, in humans, was deleted. [58] These 'mutated' mice exhibited a range of cardiovascular abnormalities and defects, as well as behavioural changes. They also revealed incomplete or mutated pharangeal arch and pouch development. Tbx 1 (7-9) was identified as the gene responsible for these cardiovascular malformations. The normal development of the cochlear and vestibular organs was also observed in these mice. Thus, Tbx1 was also identified as crucial for the development of otic epithelial cells, which then later contribute to the development of these inner ear organs. These defects are also exhibited in the birth defects in humans, hence making this mouse model highly effective and suitable. [57]
For more info: https://omim.org/entry/188400?search=TBX1&highlight=tbx1
TBX3/Ulnar-Mammary Syndrome
| Mutations of the TBX3 gene leads to ulnar-mammary syndrome, caused by a reduce in the levels of the functional proteins needed for normal development of limbs, mammary glands and other structures. Like DiGeorge Syndrome, this syndrome is a result of the haploinsufficiency of TBX3. This disorder is expressed in abnormalities of the limbs, teeth, genitals and mammary glands. [59] Again, animal models of mice have shown abnormalities in mammary glands, limbs and genitalia, often dying before birth. These abnormalities are often characterised by short, stunted growth of the hindlimbs of mice, as well as missing elements to the forelimb. Images of these can be seen on the right.
On the other hand, when this gene is abundant and over-expressed, cancers in the breast, liver and skin have been seen to develop, as high levels of this gene assist in the development of tumours. Lung cancers, breast cancers, ovarian cancers, bladder cancers and liver tumours have been shown to have high levels of the TBX3 gene. [59] For more info: https://omim.org/entry/181450?search=TBX3&highlight=tbx3 |
File:Ums.jpg a-c: ums patient and d-f: mother of patient, normal [60] |
TBX5/Holt– Oram Syndrome
| Mutations of the TBX5 gene has been shown to cause defects in cardiac septation and the production of isomers in humans affected with holt-oran syndrome. [61] The TBX5 gene is mutated in affected individuals, on chromosome 12q24. [62] Holt-Oram syndrome is an autosomal-dominant disorder, and in humans, is expressed by deformities of the upper limb, the shoulder girdle as well as defects in the septa of the heart. Animal models such as chicks have been used, where the overexpression of this gene in chicken embryos has resulted in incomplete growth of the myocardium and the trabeculae and septa of the heart. In humans, the overexpression and mutation of this gene has also been shown to inhibit normal cardiac development. This is because TBX5 is responsible for the normal proliferation of cells during cardiac development. [63]Thumb anomaly is another expression of holt-oran syndrome, where the thumb may be completely absent, or may develop as another finger-like digit, not different from the other digits of the finger. Other indicators of this disease are heart and skeletal lesions, patent ductus arteriosus (PDA), malformation of the ventricles in the heart, mitral valve prolapse and superior vena cava anomaly. [62] These thumb abnormalities can be seen in the images on the left.
For more info: https://omim.org/entry/142900?search=TBX5&highlight=tbx5 |
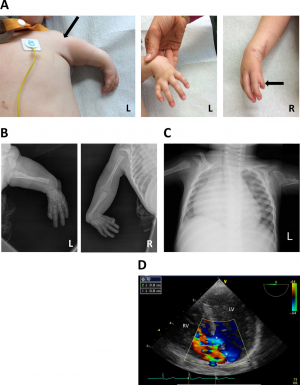 shortened thumb (Fig. 1A). radial flexion (Fig. 1B). enlarged heart (Fig. 1C). [64] |
TBX19/Isolated Adrenocorticotropic Hormone (ACTH) Deficiency
The TBx19 gene initiates the transcription process of the Proopiomelanocortin (POMC) gene. [57] This gene contains the instructions for the synthesis of the proopiomelanocortin protein. This protein is further transformed into smaller peptides which bind to proteins in the body, initiating various signalling pathways throughout the body. When this gene is mutated, congenital isolated adrenocorticotropic hormone deficiency develops, caused by the reduction of the secretion of Isolated adrenocorticotropic hormone (ACTH) from the anterior pituitary gland. This is because ACTH-producing cells of the pituitary gland are damaged and cannot secrete sufficient amounts of adrenocorticotropic hormone. [65] This results in severe hypoglycaemia and seizures in neonates. Cholestatic Liver disease also arises from this mutation, blocking or reducing the normal flow of bile through the liver. [65]This disease has also been seen to cause a range of other clinical symptoms, including urinary incontinence, gait disturbance and dementia in older patients. Ventricular enlargement in the brain, loss of appetite and vomiting are other symptoms associated with isolated ACTH deficiency. The main treatment for this disease is hormone replacement therapy. [57]
For more info: https://omim.org/entry/201400?search=TBX19&highlight=tbx19
TBX22/Cleft Palate
| Studies on mice with cleft palate have shown that the mutations of the gene encoding TBX22, causing the gene to no longer function. Tbx22 is involved in the development of the intramembranous bone formation of the posterior hard palate, rather than palate closure, and hence a mutation of this gene results in cleft palate. These mutations of the TBX22 gene included frame shifts of the gene, splices, nonsense and missense changes to the gene sequence. TBX22 is regulated by the Mn1 transcription factor, working together to achieve normal palate development. Mutations of Tbx1 and Tbx10 are also responsible for cleft palate. The mutation of this gene also causes ankyloglossia, the development of a short and thick lingual frenulum under the tongue, limiting tongue movement. This can be corrected by surgery. Choanal atresia, a blockage of the nasal airway, was also seen in affected mice. Other developmental mutations can be seen in the incomplete formation of the vomer bone in the skull. [66] [67] [68]
For more info: https://omim.org/entry/303400?search=tbx22&highlight=tbx22 |
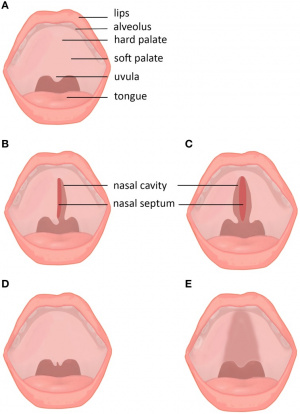 Subtypes and subclinical forms of cleft palate. (A) Normal lip and palate. (B) Unilateral cleft palate. (C) Bilateral cleft palate. (D) Cleft uvula. (E) Submucous cleft palate. [69] |
Animal models
The gene brachyury is important in all bilateral organisms (vertebrates- chordates and invertebrates such as mollusca). The brachyury gene is believed to have a conserved role in defining the midline of a bilateral organism [70] and is also fundamental in the establishment of the anterior-posterior axis [71]
It is thought to play a role in the development of organisms in the Phylum Cnidaria, and appears to be in defining the blastopore during early development[72]. It is also important during gastrulation where it defines the mesoderm [73], and experiments using tissue culture have demonstrated that the gene brachyury is also important in controlling the velocity of cells as they leave the primitive streak. [74]
Another important role that brachyury has also been shown to have is to help establish the cervical vertebral blueprint during (human) fetal development. In mammals the number of cervical vertebrae is highly conserved; however a spontaneous vertebral and spinal dysplasia (VSD) mutation in this gene has been associated with the development of six or fewer cervical vertebrae instead of the usual seven. [75]
The T-box gene family have been identified in organisms ranging from hydra to humans and due to extensive research by many investigators , we now know that T-box is important in metazoan development including transcriptional activity, genetic targets, developmental regulatory functions, and associated disease mechanisms (reviewed in Papaioannou, 2001).
Organisms used in animal models for T-Box
Animal model studies have been performed in a number of organisms including: Drosophila (the fruit fly), Xenopus (a genus of aquatic clawed frog native to sub-Saharan Africa) zebrafish, avians, and mice. These different animals are used to examine T-box gene regulation in the developmental and disease process [2].
T-box genes are involved in the development and patterning of many organ systems and embryonic structures including the heart, limb, eye, central axis, and face. In addition, T-box genes are subject to regulation by singling molecules and also induce the expression of developmentally important signaling molecules, such as retinoic acid (RA), bone morphogenetic proteins (BMPs), fibroblast growth factors (FGFs), and Wnts, in different organ systems [2]. T-box proteins can act as transcriptional activators or repressors with a variety of cofactors to regulate expression of genes involved in cell lineage determination, differentiation, and maturation [76]. Overall, T-box genes are integrated into regulatory networks that control patterning, growth, and maturation of many cell types and tissues in the developing embryo.
File:Evolution of T box gene Family.jpg This figure is modified from Papaioannou (2014) [3]and shows the subfamilies or classes of genes that have been identified in the different animal groups that are indicated in green in the boxes. What can be demonstrated is that there has been a remarkable conservation of transcription factors between lineages that have been evolving independently since the last common ancestor to metazoans, and many of the T-box gene families have their origin at the base of the tree. However, diversification at the onset of metazoan evolution is evident. T, which is the most ancient T-box gene, is represented in unicellular organisms, as is Tbx7/8, a class not present in Bilateria. Sponges have a diverse set of T-box genes including several that have not been retained in Bilateria, whereas Tbx6 subfamily genes apparently arose in Bilateria. Note that this diagram represents one possible order of divergence of phyla. |
T-box in the Mouse
Brachyury was the first T-box gene to be discovered, and is the most studied gene so far in the T-box gene family. As mentioned above it was discovered in the mouse through a semi-dominant mutation in which heterozygotes have short tails and the homozygotes are distinguished by embryonic lethality. That short tailed heterozygotes have the hall mark short tail and this is why the whole family of T-Box gene family bears the T for tail. The brachyury gene is responsible for the development of the posterior mesoderm during the gastrulation phase. In homozygote mice, the anterior part of the embryo develops normally, however the axial and posterior mesoderm structures develop abnormally. There is no connection with the allantois and chorion and without a vascular connection between the embryo and the placental and the embryo does not survive [77].
T-box in the zebra fish
T-box orthologs have been recognised in different species. Brachyura expression using genetic analysis was found to be confined to tissues that showed developmental defects in mutants. In the Zebrafish ortholog the zf-T is expressed as a nuclear protein in a pattern that resembles mouse T. A mutant in this gene in the zebrafish is called "no tail" and has a similar phenotype that seen in mouse mutations.
T-box in Drosophilia
The Drosophila ortholog of Brachyury is dm-Trg and mutations in the Drosophila ortholog shows effects in the posterior structures of the fly, which are possibly analogous to the posterior structures found in mammals.
T-Box in Amphibia: Xenopus
Using molecular developmental techniques the role of Brachyury was further examined in the frog Xenopus laevis The Xenopus homologue of Brachyury, Xbra, was cloned by Smith and coworkers in 1991 [78] . This study was based on sequence homology between the frog and mouse sequences, and its expression has shown to represent a true orthologue of the mouse gene with a high degree of similarity in both sequence and expression pattern. In the unfertilised egg, low levels of maternal Xbra are found, but the gene is expressed predominantly at the mid-blastula to neurula stages[78]. Studies in Xenopus have contributed significantly in understanding the mechanism of action of Xbra and the Brachyury gene and have shown that the activation of Xbra in response to mesoderm inducing factors.
T-Box in the chick
In the avian model (the chick) the following T-box genes have been isolated Tbx-2, Tbx-3, Tbx-4, and Tbx-5 and, like the mouse homologues, are expressed in the limb regions [79]. Other Tbox genes cTbx2, cTbx3, and cTbx5 have been found to also be involved in the chick embryo heart development.
File:T box in chick.jpg This image above demonstrates Tbx4 and Tbx5 genes Expression (marked by arrows) of Tbx4 (a, b) and Tbx5 (c, d) in the developing chick embryo at early (a, c) and late (b, d) limb-bud stages. Note that Tbx4 is expressed in the hindlimb and Tbx5 in the forelimb. Image taken from[80] |
T-box gene in Marsupial forelimb development
A study [81] published in 2012 describes for the first time the T Box gene expression in marsupials in the Tammar wallaby (Macropus eugenii). This study describes how these genes are also responsible for limb and digit formation in marsupials. Marsupials mammals differ from placental mammals because at birth, neonates are born highly altricial and not very developed. They lack fur, their eyes and ears are not developed and most of the skeleton is still cartilaginous. Another interesting feature that is observed in all marsupial neonates is that the forelimbs are more "developed" than the hindlimb. This is believed to be an adaptation to aid the tiny neonate after birth to climb from the urogenital opening to the pouch or the mammary area, and thus the neonate can attach to the teat where it completes its development- using its forelimbs to help move towards the pouch.
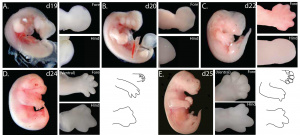 This image above demonstrates the development of tammar fetal limbs at selected stages before birth. (A) day 19, (B) day 20, (C) day 22, (D) day 24 and (E) day 25 (one day before birth). High magnification of the fore- and hindlimb are from samples stored in methanol whilst wholemounts were stored in 70% ethanol. A diagrammatic representation of the fore and hindlimb at day 24 and day 25 is provided showing dorsal and ventral views. All limbs are viewed from the dorsal aspect unless indicated. NB: images not to scale. [81] |
At birth marsupials are born with a more developed forelimb than hindlimb. The more developed forelimb can be observed in the tammar wallaby (Macropus eugenii) which clearly demonstrates that the hindlimb development clearly lags behind the forelimb development. However some other marsupials such as the South American didelphid Monodelphis domestica also known as grey short-tailed opossum, has significantly less difference between forelimb and hindlimb development at birth, however there is still more development in the forelimbs than the hindlimb. This may be because Mondelphis domestica does not have to climb such a distance to the pouch as the tammar wallaby, and also because gestation is less for didelphid marsupials than for macropods. This recent study [81] has demonstrated that the key patterning T box genes TBX4, TBX5, PITX1, FGF8, and SHH are also involved in the developing limb buds in the tammar wallaby. The results show that all the T box genes examined were highly conserved in the tammar wallaby with orthologues from opossum and mouse. TBX4 expression appeared earlier in development in the tammar wallaby than in the mouse, but appeared later in the tammar wallaby than in the opossum. Other results demonstrate that SHH expression is restricted to the zone of polarising activity, while TBX5 (forelimb) and PITX1 (hindlimb) showed diffuse mRNA expression. FGF8 is specifically localised to the apical ectodermal ridge, which is more prominent than in the opossum. The conclusions of this study demonstrate that in kangaroos and wallabies there is a very marked difference in limb size when the forelimb is compared to the hindlimb. The faster development of the fore limb compared to that of the hind limb correlates with the early timing of the expression of the key patterning genes in these limbs.
T-Box in Amphioxus
In amphioxus two brachyury like genes have been found and are referred to as paralogous genes which are orthologous to vertebrate Brachyury [77]. See below for more information.
Evolution of the T-box family: Insights from the living chordate Amphioxus
The lancelets ( also known as amphioxus -singular or amphioxi- plural) consist of 32 species of fish-like marine chordates and all are placed in the order Amphioxiformes. They have a distribution in shallow temperate and tropical seas. The amphioxus is a bilaterian cephalochordate and is considered a close relative of vertebrates. They are important in the study of zoology as they provide indications of the evolutionary origin of vertebrates and how vertebrate organisms have evolved. Lancelets have split from vertebrate more than 520 million years ago, however their genomes give us insite on how vertebrates evolved and how vertebrates have employed old genes for new functions.They are regarded as similar to the archetypal vertebrate form.
Phylogenetic analyses indicate that two genome duplications have occurred in the vertebrate linage after cephalochordates diverged so that each amphioxus gene corresponds to two or three vertebrate genes [3]
For example the amphioxus gene AmphiTbx1/10 corresponds to two vertebrate T box genes Tbx1 and Tbx10 so these have arose presumably during genome duplications.
AmphiTbx1/10 is expressed in amphioxus during gastrulation in the ventral somites and branchial arches [82] and this corresponds to the mammalian mouse T box gene Tbx1 and the expression of this gene in the ventromedial somites and pharyngeal arches. While the mouse Tbx10 is only expressed in the developing hindbrain [83]
Therefore the function of Tbx1/10 in chordates might originally have been involved in branchial arch patterning and ventral somite specification. These functions are retained by the Tbx1 gene while the Tbx10 has lost its role in pharyngeal arch patterning and instead have gained a novel new role in hindbrain development.
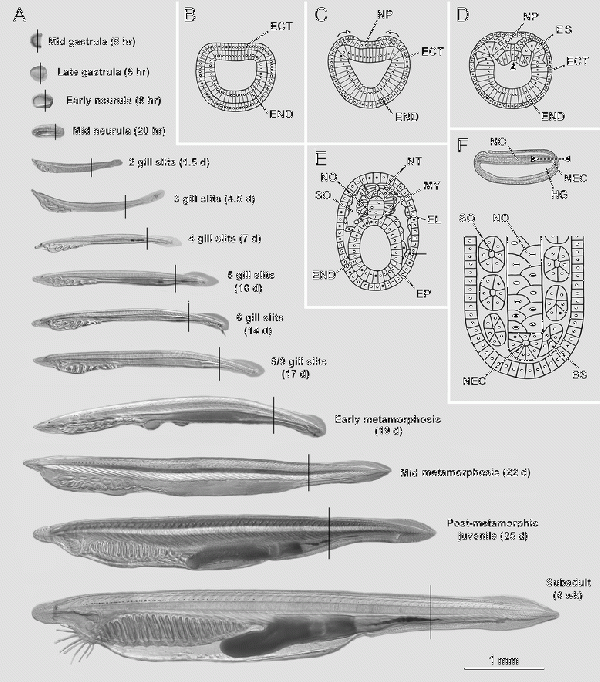 This image above demonstrates an overview of amphioxus development and the stages examined in the study by [84] Amphioxus are classified in the Subphylum Cephalochordate and are approximately 22 mm long and are chordates, and considered to be one of the closest living relatives to all vertebrates. |
Glossary
Apical ectodermal ridge (AER): a thickened area of pseudo-stratified columnar epithelium at the tip of the developing limb bud. It is a major signaling centre for the developing limb.
Congenital: a condition attributable to events prior to birth, could be related to a baby born with disease.
Homologous: existence of shared ancestry between a pair of structures, or genes, in different taxa.
Homologue: something homologous.
Limb fields: areas where the limb buds will develop.
Motif : a pattern or design
Nkx2-5: a cardiac homeobox protein essential in cardiac development, and mutations in Csx (which encodes Nkx2-5) cause various congenital heart diseases.
Phylogenetic: is the study of the evolutionary history and relationships among individuals or groups of organisms.
Transcription factor: a transcription factor is a protein that binds to specific DNA sequences, thereby controlling the rate of transcription of genetic information from DNA to messenger RNA.
Wild-type: refers to having the gene that encodes the phenotype most common in a particular natural population.
Zone of Polarising Activity (ZPA): a collection of cells at the posterior border of the limb close to the AER, adjacent to the body wall.
Glossary doesn't have to be referenced
Good places to look
PubMed Biomed Central OMIM PNAS PLOS
DO A QUIZ AT THE BOTTOM OF THE PAGE - maybe collapse it in a tab to make the page look cleaner etc.
to make sure its a comprehensive quiz useful for the student - explain why the correct option is correct and why the other options are wrong
References
- ↑ Pubmed structure (2010) Crystal Structure of Human Tbx5 in the DNA-free Form, https://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?uid=81644, accessed 21/10/16
- ↑ 2.0 2.1 2.2 <pubmed>16285859</pubmed>
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 <pubmed>25294936</pubmed>
- ↑ 4.0 4.1 <pubmed>14699590</pubmed>
- ↑ 5.0 5.1 5.2 Dobrovolskaïa-Zavadskaïa, N. (1927). Sur la mortification spontanée de la queue che la souris nouveau-née et sur l'existence d'un caractère (facteur) héréditaire “non viable”. C. R. Seanc. Soc. Biol. 97, 114-116.
- ↑ <pubmed>11268043 </pubmed>
- ↑ 7.0 7.1 <pubmed>7920656</pubmed>
- ↑ 8.0 8.1 <pubmed>24043797</pubmed>
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedZD1927 - ↑ <pubmed>11268043</pubmed>
- ↑ 11.0 11.1 11.2 <pubmed>2154694</pubmed> Cite error: Invalid
<ref>tag; name 'PMID2154694' defined multiple times with different content - ↑ <pubmed>8344258</pubmed>
- ↑ <pubmed>9349824</pubmed>
- ↑ <pubmed>7774921</pubmed>
- ↑ <pubmed>8530034 </pubmed>
- ↑ <pubmed>8597636</pubmed>
- ↑ <pubmed>8988165</pubmed>
- ↑ <pubmed>9550719</pubmed>
- ↑ 19.0 19.1 <pubmed>11242110 </pubmed>
- ↑ <pubmed>14550786</pubmed>
- ↑ <pubmed>15255957 </pubmed>
- ↑ <pubmed>15843414</pubmed>
- ↑ <pubmed>16505380</pubmed>
- ↑ <pubmed>16376438</pubmed>
- ↑ <pubmed>19494035</pubmed>
- ↑ <pubmed>20139965</pubmed>
- ↑ <pubmed>21331042</pubmed>
- ↑ <pubmed>16258075</pubmed>
- ↑ 29.0 29.1 <pubmed>11572777</pubmed>
- ↑ <pubmed>9651516</pubmed>
- ↑ 31.0 31.1 <pubmed>10079235</pubmed>
- ↑ <pubmed>11431700</pubmed>
- ↑ <pubmed>24858909</pubmed>
- ↑ <pubmed>12845333</pubmed>
- ↑ <pubmed>23144723</pubmed>
- ↑ <pubmed>11971873</pubmed>
- ↑ <pubmed>11242049</pubmed>
- ↑ <pubmed>9655805</pubmed>
- ↑ <pubmed>8625833</pubmed>
- ↑ <pubmed>1731249 </pubmed>
- ↑ <pubmed>26212321</pubmed>
- ↑ <pubmed>9609833</pubmed>
- ↑ <pubmed>10235263</pubmed>
- ↑ <pubmed>22071103</pubmed>
- ↑ <pubmed>9187149</pubmed>
- ↑ <pubmed>12490567</pubmed>
- ↑ <pubmed>7889567</pubmed>
- ↑ <pubmed>9435295</pubmed>
- ↑ <pubmed>26212321</pubmed>
- ↑ <pubmed>8853987</pubmed>
- ↑ 51.0 51.1 51.2 <pubmed>22876201</pubmed>
- ↑ <pubmed>12588840</pubmed>
- ↑ <pubmed>9916808</pubmed>
- ↑ <pubmed>10235264</pubmed>
- ↑ <pubmed>18505863</pubmed>
- ↑ <pubmed>15066124</pubmed>
- ↑ 57.0 57.1 57.2 57.3 <pubmed>1197183</pubmed> Cite error: Invalid
<ref>tag; name 'PMID1197183' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID1197183' defined multiple times with different content - ↑ McKusick, V. (1997). T-BOX 1; TBX1, OMIM, accessed 3rd October 2016
- ↑ 59.0 59.1 <pubmed>3374754</pubmed>
- ↑ <pubmed>19938096</pubmed>
- ↑ <pubmed>11161571</pubmed>
- ↑ 62.0 62.1 McKusick, V. (1986). HOLT-ORAM SYNDROME, OMIM, accessed 3rd October 2016
- ↑ <pubmed>17534187 </pubmed>
- ↑ <pubmed>27652283</pubmed>
- ↑ 65.0 65.1 Genetics Home Reference (2016). POMC Gene. https://ghr.nlm.nih.gov/gene/POMC. accessed 3rd October 2016
- ↑ <pubmed>19648291</pubmed>
- ↑ <pubmed>18948418</pubmed>
- ↑ <pubmed>17846996</pubmed>
- ↑ <pubmed>26973535 </pubmed>
- ↑ <pubmed>15034714</pubmed>
- ↑ <pubmed>11880350</pubmed>
- ↑ <pubmed>12536320</pubmed>
- ↑ <pubmed>12921737</pubmed>
- ↑ <pubmed>3327671</pubmed>
- ↑ <pubmed>25614605</pubmed>
- ↑ <pubmed>11148447</pubmed>
- ↑ 77.0 77.1 <pubmed>9504043</pubmed>
- ↑ 78.0 78.1 <pubmed>1717160</pubmed>
- ↑ <pubmed>9550719</pubmed>
- ↑ <pubmed>10203826</pubmed>
- ↑ 81.0 81.1 81.2 <pubmed>22235805</pubmed>
- ↑ <pubmed>15372236</pubmed>
- ↑ <pubmed>12915323</pubmed>
- ↑ <pubmed>26052418</pubmed>