2016 Group Project 5: Difference between revisions
| Line 158: | Line 158: | ||
T-box gene family is ancient in origin and it is thought to be found in all metazoans (Bollag et al., 1994). | T-box gene family is ancient in origin and it is thought to be found in all metazoans (Bollag et al., 1994). | ||
The origin of the T-box family has been pushed back to unicellular organisms and fungi, in which one or two T-box genes, including T, have been identified (Degnan et al., 2009; Sebe-Pedros et al., 2013). | The origin of the T-box family has been pushed back to unicellular organisms and fungi, in which one or two T-box genes, including T, have been identified (Degnan et al., 2009; Sebe-Pedros et al., 2013). | ||
PMID 25294936 | |||
===Glossary=== | ===Glossary=== | ||
Revision as of 07:20, 23 September 2016
| 2016 Student Projects | ||||
|---|---|---|---|---|
| Signalling: 1 Wnt | 2 Notch | 3 FGF Receptor | 4 Hedgehog | 5 T-box | 6 TGF-Beta | ||||
| 2016 Group Project Topic - Signaling in Development
OK you are now in a group, add a topic with your student signature to the group page. | ||||
| This page is an undergraduate science embryology student project and may contain inaccuracies in either descriptions or acknowledgements. | ||||
| Group Assessment Criteria |
|---|
 Science Student Projects Science Student Projects
|
| More Information on Assessment Criteria | Science Student Projects |
T-box genes and their signalling
Introduction
The T-box family of transcription factors exhibits widespread involvement throughout development in all metazoans. [1] PMID 9504043
From the limb development lecture, two major signalling pathways that involve Tbx: 1) FGF signaling (FGF10 and FGF8) 2) Wnt signaling pathway
THINGS TO STILL INCLUDE:
-Where the name comes from
The T-box gene and transcription factors of the T-box family play an important role in vertebrate development. The founding member of the T-box family is brachyura which comes from the greek and means short tail (reference). This gene was discovered following experimental studies with a short tailed mouse that harboured a mutation which affected tail length and embryonic development (Dobrovolskaïa-Zavadskaïa, 1927). Brakhus means short in greek and oura meaning tail.
Nadezhda Alexandrovna Dobrovolskaya-Zavadskaya first described the brachyury mutation in 1927 as a mutation that affected tail length and sacral vertebrae in heterozygous mice (reference).
The brachyury gene (which is also known as T) soon was studied in great detail due to its important role in the development of the notochord and posterior mesoderm. Mutations in T are shown to cause embryonic lethality in homozygotes and short tails in heterozygotes (Papaioannou, 2001). Now according to human and mouse genome the gene brachyura has the symbol T and gene name T, however the gene is described as brachyury.
Reference:
Dobrovolskaïa-Zavadskaïa, N. (1927). Sur la mortification spontanée de la queue che la souris nouveau-née et sur l'existence d'un caractère (facteur) héréditaire “non viable”. C. R. Seanc. Soc. Biol. 97, 114-116.
- The family of TBox genes -Where the gene is located with respect to other genes - using Omim, where the mutation occurs
Table of the main T-box genes
| T-box gene | Main expression sites during embryogenesis | Function | Abnormalities |
| Tbx1 | Pharyngeal endoderm, mesoderm core of the first pharyngeal arch, head mesoderm ventral to hindbrain, sclerotome | ||
| Tbx2 | Allantois, non-chanmber myocardium, optic and otic vesicles, naso-facial mesenchyme, limbs, lungs, genitalia |
Table adapted from PMID 16258075
Features of the T-box family
The defining feature of the T-box gene family is a conserved domain that was first uncovered in the sequence of the mouse T locus, or Brachyury gene. [2] This homology domain encodes a polypeptide region that we have named the T-box.[3]
STILL NEED TO INCLUDE: a table of the different Tbox genes, functions, location, which systems they are viable for etc.
Origins of the T-box genes
The story of the T-box genes began in Paris at the Pasteur laboratory in the 1920s with the Russian scientist Nadine Dobrovolskaïa-Zavadskaïa, who embarked on a pioneering screen for X-ray-induced developmental mouse mutants. Her isolation of a mouse strain with a short tail, caused by a semidominant heterozygous mutation in a locus she called T, represented one of the first successful mammalian genetic screens, and provided one of the earliest links between gene activity and cell behaviour during embryogenesis[4]. The mid-gestational death of homozygous T embryos, with perturbed development of the posterior mesoderm and notochord, demonstrated an essential requirement for T during gastrulation, and led to the earliest insights into the inductive influences of notochord on neural tube and somite development. Over 60 years later T, now also known as brachyury, meaning ‘short tail’ in Greek, was cloned in one of the earliest positional cloning efforts in the mouse[5]. At the time, lack of homology in the T-gene product to any previously characterized protein gave no clues as to its biochemical role until, in 1993, it was revealed to be a novel sequence-specific DNA-binding protein[6]. Crystallographic determination of the structure of the DNA-binding domain, now called the Tbox, revealed a new way through which proteins recognize DNA[7].
TO DO: Produce a timeline for the history of the TBox gene - type TBOx into PubMed and sort the findings by age - use this to see when the first journal articles where released related to TBox and limb development, how this research has improved and developed, where it is heading in the future
Functions of T-box in development
T-box genes are a growing family of transcription factors that are expressed in diverse patterns throughout vertebrate development. They have emerged as key players in embryonic patterning, tissue differentiation and morphogenesis, particularly in vertebrates and some specific examples have been described in the following sections.
Cardiac development
The heart is one of the first organs to develop and function in vertebrate embryos. Its formation is a complex process and requires contributions from multiple transcription factors, including GATA-4, eHAND, dHAND, Irx4, and Tbx5 [8]. In the developing heart, Tbx5 expression can be first detected at stage 12 along the entire rostrocaudal length of the fused heart tube[9]. Although it is expressed very early in cardiac embryogenesis, Tbx5 is not essential for cardiac crescent formation or for development of the early heart tube. Subsequent growth of the posterior segment (atria and LV) of the heart requires Tbx5, and does not occur in Tbx5 del/del embryos. In contrast, RV and outflow tract development appears to be Tbx5 independent[10]. What is interesting is that the role of Tbx5 in the growth and maturation of posterior heart is evolutionarily conserved from amphibia (Xenopus) to mammals[11]. Thus Tbx5 transcription factor is involved in directing gene expression in morphogenetic processes associated with specific chamber formation and any mutations can cause heart septal defects in Holt–Oram syndrome[12] (discussed below in Abnormalities).
PMID 11572777 PMID 15580613 PMID 16258075 PMID 1851989
Limb Development
Four members of the T-box family of transcription factors (Tbx2-Tbx5) are expressed in developing limb buds, and that expression of two of these genes, Tbx4 and Tbx5, is primarily restricted to the developing hindlimbs and forelimbs, respectively[13]. Tbx4 and Tbx5 are first expressed in lateral plate mesoderm within clearly defined territories at the time the prospective limb fields are being specified by Hox genes. Hox genes may therefore be responsible for regulating expression of these Tbox genes within the limb fields. Fgf-10 expression is also initiated in lateral plate mesoderm around this time, and FGF10 is a good candidate for the mesodermal factor that initiates limb outgrowth and signals the adjacent ectoderm to express FGF8.
PMID 9609833 PMID 9655805 PMID 9550719 PMID 8798150 PMID 11782414 PMID 12490567 PMID 22872086 PMID 12736212 PMID 26212321 PMID 21932311
http://www.ncbi.nlm.nih.gov/books/NBK10003/ http://dev.biologists.org/content/130/3/623/F1
Respiratory Development
In a chicken model, all Tbx2 subfamily genes, Tbx2, Tbx3, Tbx4 and Tbx5 are expressed in the developing lung buds and trachea between stages 15–21 [14]. In the mouse however, Tbx1 is expressed in lung epithelium at E12.5, Tbx2 and Tbx3 are expressed in lung mesenchyme at E11.5, and Tbx4 and Tbx5 are expressed in both lung and trachea mesenchyme at E12.5 and later[15].
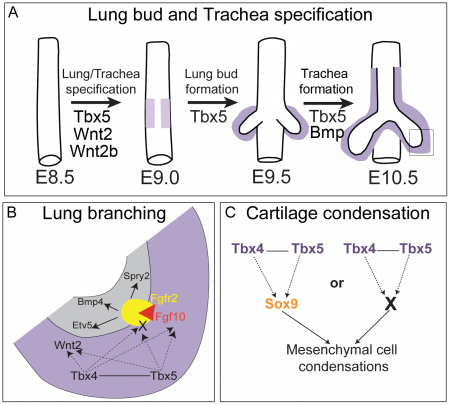
In lung branching morphogenesis, Tbx4 and Tbx5 genetically interact with one another. In an article by Ripla et al. (2012), they demonstrated that both Tbx4 and Tbx5 are expressed throughout the mesenchyme of the developing lung and trachea[16]. They showed that a loss of Tbx5 leads to a unilateral loss of lung bud specification and absence of tracheal specification in organ culture. Concordant with this defect, the expression of mesenchymal markers Wnt2 and Fgf10, as well as Fgf10 target genes Bmp4 and Spry2, in the epithelium is downregulated. This suggests that Fgf10 signaling pathway is activated downstream of Tbx4 and Tbx5 in the developing lung and that Fgf10 genetically interacts with Tbx4 and Tbx5. This is consistent with finding from Sakiyama et al. (2003) that found out that the Tbx4-Fgf10 system controls lung bud formation during chicken embryonic development. Tbx4 was found to trigger Fgf10 expression in the lung primordium mesoderm which then acquires an inductive capability for the initial budding morphogenesis of primary lung buds[17]. Of significance, lung-specific Tbx4 heterozygous;Tbx5 conditional null mice died soon after birth due to respiratory distress. These pups have small lungs and show severe disruptions in tracheal/bronchial cartilage rings, highlighting the important role of Tbx4 and Tbx5 in respiratory development.
Tbx in lung and trachea development[18]
Other developmental events
Tbx signalling also involved in palate development,
In humans, Tbx1 mutation is responsible for the major phenotypes of 22q11.2 deletion syndrome (Velo-cardio-facial/DiGeorge syndrome, discussed in Abnormalities section) as well as non-syndromic submucous cleft palate, suggesting that Tbx1 is a regulator of palatogenesis. A study by Funato et al. (2012) revealed that Tbx1 regulates oral epithelial adhesion and palatal development and showed that Tbx1 -/- mice exhibit various forms of cleft palate phenotypes, including submucosal cleft palate and soft palate cleft.
Dorsoventral Patterning of the Mouse Coat by Tbx15 PMID 14737183 Palate Development PMID 22371266
Abnormalities
Z5039628 (talk) 19:37, 1 September 2016 (AEST)
TBX1/DIGEORGE SYNDROME; TBX3/ULNAR-MAMMARY SYNDROME; TBX5/HOLT– ORAM SYNDROME; TBX19/ISOLATED ACTH DEFICIENCY; TBX22/CLEFT PALATE
Z5039628 (talk) 23:16, 8 September 2016 (AEST)
PMID 10235264 PMID 18505863 PMID 15066124
Holt-Oram Syndrome primary article PMID 11161571
OMIM 142900
Animal models
The gene brachyury is important in all bilateral organisms (vertebrates- chordates and invertebrates such as mollusca). The brachyury gene is believed to have a conserved role in defining the midline of a bilateral organism [19] and are also fundamental in the establishment of the anterior-posterior axis [20]
Its ancestral role, or at least the role it plays in the Cnidaria, appears to be in defining the blastopore.[5] It also defines the mesoderm during gastrulation.[9] Tissue-culture based techniques have demonstrated one of its roles may be in controlling the velocity of cells as they leave the primitive streak.[10][11]
Transcription of genes required for mesoderm formation and cellular differentiation.[clarification needed]
Brachyury has also been shown to help establish the cervical vertebral blueprint during fetal development. The number of cervical vertebrae is highly conserved among all mammals; however a spontaneous vertebral and spinal dysplasia (VSD) mutation in this gene has been associated with the development of six or fewer cervical vertebrae instead of the usual seven.[12]
PMID 25294936
PMID 15034714
PMID 11880350
Marsupial forelimb development PMID 21098569
PMID 24299415
PMID 11427155
PMID 22235805
This study [21]published in 2012 is the first which describes the HOX gene expression in a marsupial (Tammar wallaby Macropus eugenii) and how these genes are also responsible for limb and digit formation.
The genes responsible for digit patterning are the HOXA13 and HOXD13 which are also present and highly conserved in other vertebrates such as the chicken and the mouse. In the tammar wallaby the forelimbs develop much faster than the hind limbs. In the tammar wallaby the hindlimb autopod has only four digits and the fourth digit is greatly elongated while the digits two and three are syndactylous. The authors found that the differences in the gene structure in the tammar and the changes in the expression and timing may be driving the difference in the development of the syndactylous hind limb. They suggest that the polyserine region may be responsible for marsupial syndactyly. This also needs to be studied in other marsupials with syndactyl hind limbs such as bandicoots and bibles (Peramelemorphia). Our findings support the hypothesis that changes to the structure and function of HOXA13 and HOXD13 affect regulation of digit identity in this marsupial.
Ancient origins and evolution of the T-box gene family
T-box gene family is ancient in origin and it is thought to be found in all metazoans (Bollag et al., 1994). The origin of the T-box family has been pushed back to unicellular organisms and fungi, in which one or two T-box genes, including T, have been identified (Degnan et al., 2009; Sebe-Pedros et al., 2013).
PMID 25294936
Glossary
Good places to look
PubMed Biomed Central OMIM PNAS PLOS
References
- ↑ <pubmed>25294936</pubmed>
- ↑ <pubmed>2154694</pubmed>
- ↑ <pubmed>792065</pubmed>
- ↑ <pubmed>11268043</pubmed>
- ↑ <pubmed>2154694</pubmed>
- ↑ <pubmed>8344258</pubmed>
- ↑ <pubmed>9349824</pubmed>
- ↑ <pubmed>11572777</pubmed>
- ↑ <pubmed>9651516</pubmed>
- ↑ <pubmed>11572777</pubmed>
- ↑ <pubmed>10079235</pubmed>
- ↑ <pubmed>10079235</pubmed>
- ↑ <pubmed>9609833</pubmed>
- ↑ <pubmed>9651516</pubmed>
- ↑ <pubmed>8853987</pubmed>
- ↑ <pubmed>22876201</pubmed>
- ↑ <pubmed>12588840</pubmed>
- ↑ <pubmed>22876201</pubmed>
- ↑ <pubmed>15034714</pubmed>
- ↑ <pubmed>11880350</pubmed>
- ↑ <pubmed>22235805</pubmed>