2014 Group Project 7: Difference between revisions
| Line 339: | Line 339: | ||
* Macrocephaly is a syndrome of diverse etiologies rather than a disease and the most frequent cause is hydrocephalus.<ref><pubmed>21215908</pubmed></ref> | * Macrocephaly is a syndrome of diverse etiologies rather than a disease and the most frequent cause is hydrocephalus.<ref><pubmed>21215908</pubmed></ref> | ||
'''Hydrocephalus''' | |||
[[Image: Arachnoid_cyst_with_hydrocephalus.jpg|frame|right|middle|300x250px|Magnetic Resonance image showing Arachnoid Cyst with Hydrocephalus <ref><pubmed>22069421</pubmed></ref>]] | |||
* Progressive enlargement of head due to accumulation of cerebrospinal fluid in ventricles is known as hydrocephalus. | |||
* Excessive accumulation of cerebrospinal fluid is due to an imbalance between the formation and absorption of cerebrospinal fluid (communicating hydrocephalus) or obstruction of circulation of cerebrospinal fluid (non-communicating hydrocephalus). | |||
==References== | ==References== | ||
<references/> | <references/> | ||
Revision as of 13:16, 24 October 2014
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Neural - CNS
Introduction
The Central Nervous System (CNS) is a complex network of neurons which are responsible for the sending, recieving and integration of information from all parts of the body, serving as the processing center of the bodies nervous system. The CNS controls all bodily functions (sensory and motor), consisting of 2 main organs; The Brain and Spinal Cord.
The brain is the body's control center consisting of 3 main components; Forebrain, Midbrain and Hindbrain. The forebrain functions in receiving and processing sensory information, thinking, perception, and control of motor functions as well as containing essential structures; Hypothalamus and Thalamus, which are responsible in motor control, autonomic function control and the relaying of sensory information. The Midbrain along with the Hindbrain together form the brain-stem and are both important in auditory and visual responses
The spinal cord is a cylindrical shaped structure composes of nerve fiber bundles which is connected to the brain via the brain-stalk formed from the Midbrain and Hindbrain, running through the spinal canal in the vertebrae (in animals) from the neck to the lower back. The spinal cord plays the important role of transmitting information from bodily organs and external stimuli to the brain and acts as a channel to send important signals to other parts of the body. The nerve bundles in the Spinal cord are divided into ascending bundles, which transmits sensory information from the body to the brain, and descending bundle, which transmits motor function information from the brain to the body.
Before fetal period, neurulation occurs that ectoderm forms initial structure of the CNS and folds upon itself to form neural tube towards the end of week 3. The head portion becomes the brain, which further differentiates into forebrain, midbrain and hindbrain, while the middle portion becomes the brain stem. Around week 5, neural tube differentiates into the proencephalon (forebrain), the mesencephalon (midbrain) and the rhombencephalon (hindbrain). By week 7, the prosencephalon divides into the telencephalon and the diencephalon, while the rhombencephalon divides into the metencephalon and the myelencephalon. The formation of these 2 additional structures creates 5 primary units that will become the mature brain.
In this website, CNS development during the fetal period, including cellular processes and brain development will be discussed. Current research models and findings, and historic findings will be stated. The abnormalities associated to CNS during fetal period, and neural defects that occur during embryonic period and contribute in fetal development will be discussed.
Development during fetal period
Cellular process
In developing CNS, there are 4 major cellular processes, including cell proliferation, cell migration, cell differentiation and cell death. They are a cascade of events that the earlier occurring process may influence the subsequently occurring ones, but a late-occurring event cannot influence the earlier ones.
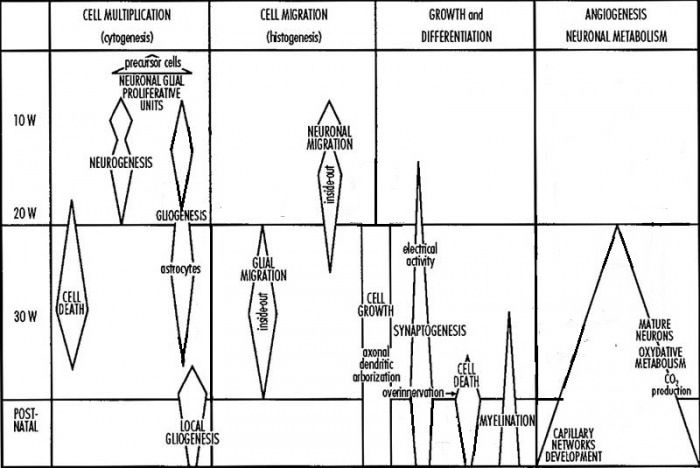
A simplified timeline of human neural development (modified) [1]
This simplified graph shows a timeline for major events of neural development that occur during fetal and postnatal periods. These events, which are summarized in the following table, are broadly classified by cell multiplication, cell migration, growth and differentiation and angiogenesis:
| Major Events | Descriptions |
|---|---|
| Cell multiplication |
|
| Cell migration |
|
| Growth and differentiation |
|
| Angiogenesis |
|
1. cell proliferation
- This process is responsible for the formation of neurons and glia.
- Begins around 40th embryonic day and is almost complete around the 6th month of gestation [3]
- Location: occurs in germinal matrix that comprised of ventricular and subventricular proliferative zones of cells.
- Ventricular zone: This is the proliferative zone that appears first which is a pseudostratified columnar epithelium [4]. In some part if the developing CNS, this is the only proliferative zone and therefore it is assume that ventricular zone produces all of the cell types. For example, in the hippocampus, all of the neurons of the major subdivisions (areas CA1, CA2 and CA3) are derived from the ventricular zone. There is substantial movement of the nuclei as they move through the cell cycle [5]. The nuclei move between the ventricular surface and the border of the ventricular zone with the subventricular zone [6].
- Subventricular zone: This is the second proliferative zone that appears in some parts of the developing CNS. Most of the glia for most of the brain are produced in this zone, therefore it is important to the adult brain [7]. In some parts of the brain, there is the production of a significant number of neurons in this zone [8]. For example, the subventricular zone contributes large number of cells to the neocortex, which is the youngest structure in the brain [9]. In contrast to ventricular zone, the proliferating cells do not move through the cell cycle.
2. cell migration
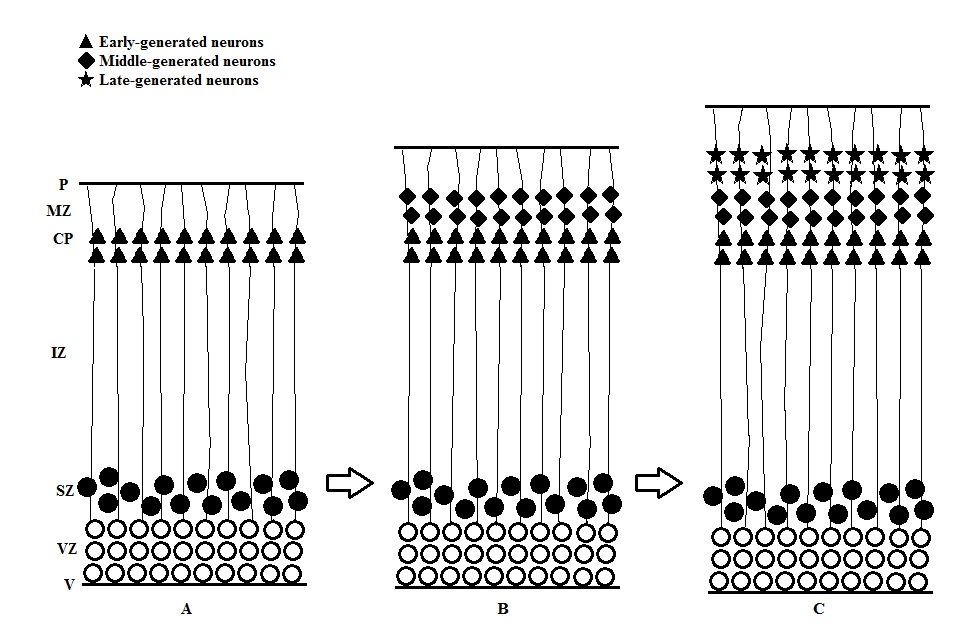
- This is the process that influences the final cell position by migrating the cells produced from the two ventricular zones.
- The postmitotic young neurons migrate from the proliferation site to their ultimate position in two different ways.
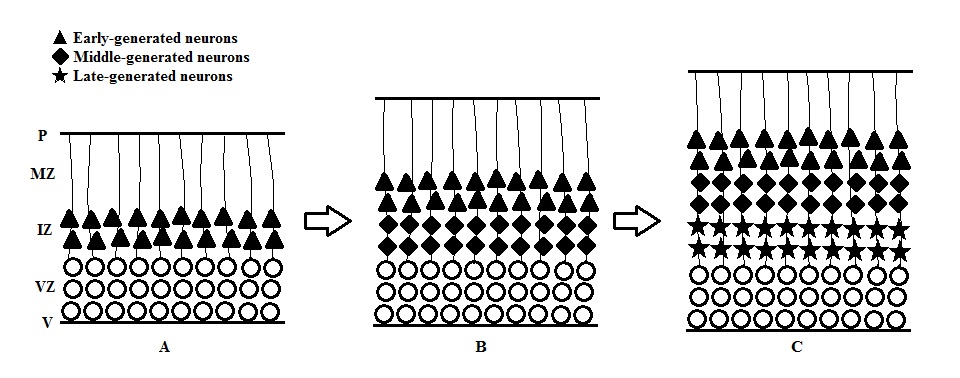
- Passive cell displacement: Moving of cells in this way does not require active locomotor activity. In some parts of the developing CNS, the postmitotic neurons that only move a very short distance from the border of proliferative zone are displaced outward by newly produced cells (figure). This results in an “outside-to-inside” spatiotemporal gradient that the earliest generated neurons are located farthest away from the proliferative zone, where the youngest generated ones are located closest to the zone. This pattern can be found in the thalamus [11], hypothalamus [12], spinal cord [13], dentate gyrus of the hippocampal formation [14] and many regions of the brainstem.
- Active migration: This requires the active participation of the moving cell for its displacement which neurons move at a greater distance and the migrating young neuron bypasses the previously generated cell (figure). This results in an “inside-to-outside” spatiotemporal gradient. This pattern can be found in well-laminated structure including cerebral cortex and several subcortical areas [14].
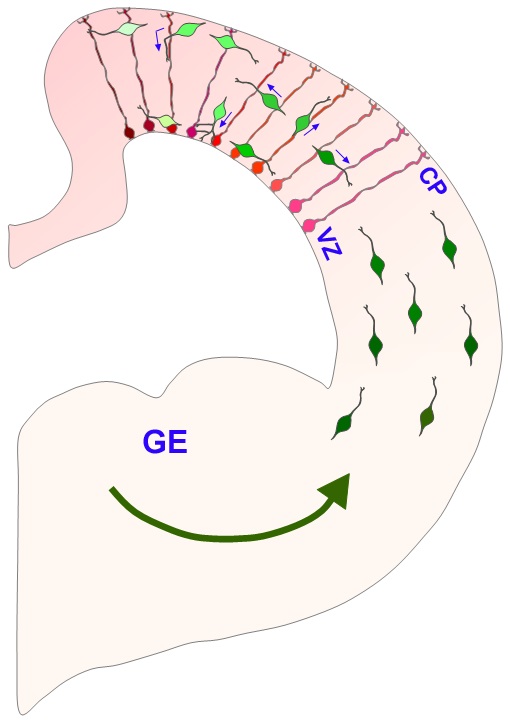 Image of interneurons migration and interactions with radial glia in the developing cerebral cortex [15] |
3. cell differentiation[10]
- This is the process begins after the migration of neuronal and glial cells to the final positions and it is responsible for the generation of a wide variety of cells in the adult CNS. During the differentiation, each neuron grows out its axon and dendrites.
- Time: starts about the 25th month of gestation until adolescence
- The axons do not grow directly to their final targets, but they transiently innervate areas and cells in two ways that the connections cannot be found in adult brain. These two types of transient connections are not mutually exclusive and can be found within a single population of cells.
- Divergent transient connections: one neuron innervates more cells than normal which will be eliminated by the reduction of projection area.
- Convergent transient connections: several neurons innervate one target neuron where only one of these neuronal connections is found in adult.
4. cell death (apoptosis)
- This is the process where elimination of transient connections occurs and two mechanisms, axonal retraction and neuronal pruning [10], are involved.
- Axonal retraction: The transient connections are removed by the recession of the collaterals of the neuron’s axon or by the shrinking of the terminal arborisation of the axon.
- Neuronal pruning: The transient connections are removed through a selective cell death that neurons die due to the failing to establish projections.
- Critical for appropriate brain development
Brain Development
- The human brain development begins in the 3rd gestational week with the start marked by differentiation of the neural progenitor cells. By the end of the embryonic period in gestation week 9 (GW9), the basic structures of the brain and CNS are established as well as the major parts of the Central and Peripheral nervous systems being defined [16]
- The early fetal period (mid-gestation) is a critical period in the development of the neocortex, as well as the formation of vital cortical neurons which are vital in the brain processing information
During Fetal Period
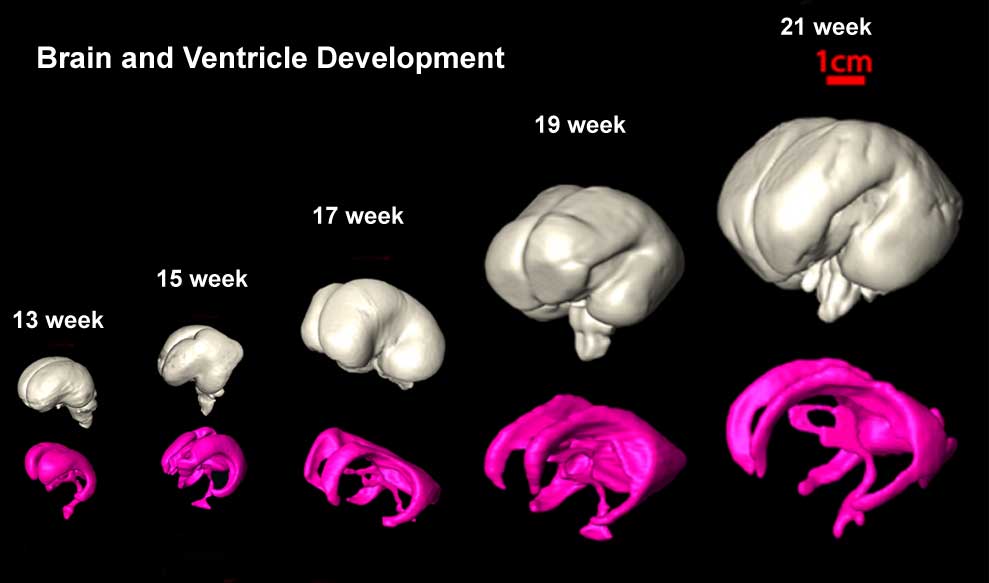
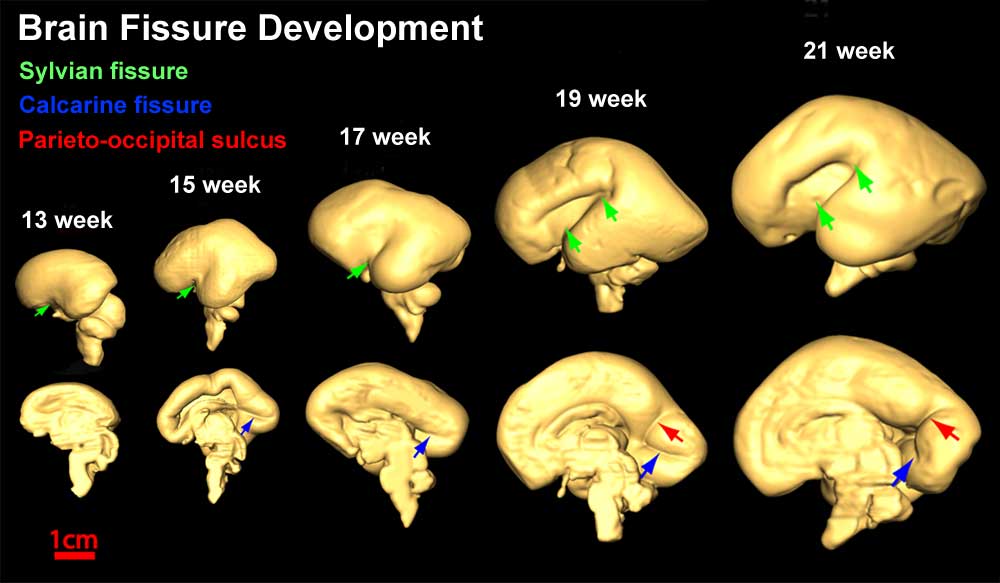
- The gross morphology of the developing brain undergoes striking change during this time, beginning as a smooth structure and gradually developing the characteristic mature pattern of gyral and sulcal folding.
- The brain development begins rostral in GW8, proceeding caudally until it is complete at GW22 [18]
- The formation of Secondary Sulci is between GW30-35, while the formation of of Tertiary Sulci begins during GW36 and into the postnatal period.
- Different population of neurons form grey matter structures in many regions of the brain including hindbrain and spinal column, cerebellum, midbrain structure and the neocortex
- Neurons, after production, migrate away from the proliferative regions of the VZ, the neurons that will form the neocortex migrate in an orderly fashion forming the 6 layered neocortical mantle
- The major fibre pathways make up the brain white matter
- AS development proceeds, the brain becomes larger and the primary mode of neuronal migration from the VZ changes.
Neuron Production
- The human brain contains billions of neurons which are produced by mid-gestation; during fetal development [19]
- Processes of Neuronal production is initiated by first increasing the size of the Neural progenitor cell population within the body, these cells are mitotic in nature and are capable of forming new cells.
- Initially (from the end of gastrulation through to embryonic day 42), the neural progenitor cell population is increased greatly when the progenitor cells divide through 'symmetrical' mode of cell division. Multiple repeats of the cell division occurs throughout this period. This 'symmetrical' division method brings about the formation of 2 new identical neural progenitor cells
- Mode of cell division changes from a symmetrical cell division type to an 'asymmetrical' cell division from the beginning of E42. This asymmetrical cell division forms 2 different cell types; one Neural progenitor & one Neuron [20]
- Newly formed Neural progenitor cells remains to undergo more processes of cell division whereas the newly formed neuron moves into position in the developing neocortex
Neuron Migration [21]
Outlined above was a broad summary on neuron migration. Neuron migration in the cortex will be covered below in more detail.
- The ventricular zone (VZ) is where the majority of neurons migrate radially out and into the neocortex. The type of migration utilised here is referred to as somal translocation where the neuron cell body translocates out of the VZ and into the outer brain region with the nucleus moving across the cytoplasm and into the outer brain region.
- With increasing brain size, somal translocation is no longer efficient for neuronal migration out of VZ. Since greater distances are evident, radial glial guides are adopted to allow the migration of neurons to continue. Similar to somal translocation, these cell populations establish a scaffolding system where neurons can attach to (basal process occurs) and then move into the developing cortical plate. These cell populations are comprised of neural progenitor cells and are therefore able to support mass neuron migration.
- Another mode of transport utilised by neurons situated within a second zone of the ventral telencephalon is referred to as tangential migration. The neurons here migrate tangentially to the cortical mantle whilst adopting different signalling pathways (guidance molecules) as opposed to radial migration. As a result of neuronal migration, a 6-layered structure forms within the developing neocortex.
- Once completion of the pre-plate has occurred, two regions are formed and which are the subplate (SP) and th marginal zone (MZ).In between these two regions, the cortical plate arises as well. Earliest neuron arrival forms the deepest layer of the cortex; cortical layer 6. More superficial layers of the cortex will develop with continuous cell migration. It is important to note that a specific cell class is located within the MZ in which are the Cajal-Retzius cells (CR). CR's are important for correct neuron positioning within the cortical layers as well as having an inhibitory effect on neuron migration. Rheelin, a molecular signal produced by the CR's is important here as it stops neurons from migrating and signals them to up their corresponding positions.
Folding: sulcation and gyration
- During the fifth and sixth month of gestation, the smooth cortex begins to fold by sulcation and gyration. [22]
- Sulcation: This is the development of sulci, including primary and secondary. The formation of primary sulci involve the appearance of shallow grooves on the surface of the brain, which then become more deeply infolded, while the formation of secondary sulci is due to the development of side branches of the primary ones [23].
- Gyration: This is the development of gyrus that occurs during late during fetal development until the end of the pregnancy or after birth [23]. This process is the formation of tertiary sulci, which is the formation of other side branches of the secondary sulci [23].
| Weeks of gestation | Visible anatomical details |
|---|---|
| 20-21 | smooth, with the impression of "lissencephalic" brain; wide Sylvian fissures; visible interhemispheric fissure and parieto-occipital fissure |
| 22-23 | beginning of calcarine and hippocampal fissures and of callosal sulci |
| 24-25 | smooth cerebral cortex surface; visible shallow grooves in the central sulci, interparietal sulci and superior temporal sulci; start of opercularization of Sylvian fissures; presence of calcarine fissures and cingular sulci |
| 26 | presence of central and collateral sulci |
| 27 | presence of marginal and precentral sulci |
| 28 | presence of postcentral and intraparietal sulci |
| 29 | presence of superior and inferior frontal sulci; narrower Sylvian fissure; clear corpus callosum; bright white matter; dark cortical ribbon |
| 30-31 | beginning of infolding of cortex which is first apparent in the occipital lobe; narrower ventricular system and subarachnoid spaces |
| 32 | presence of superior and inferior temporal sulci |
| 33 | presence of external occipitotemporal sulci |
| 34-35 | close to final shape of gyration; compactly and extensively folded cortex |
| 36-37 | completed opercularization of Sylvian fissures; narrow pericerebral fluid spaces; dark subcortical fibres and corona radiata |
| 38-40 | dark posterior limbs of internal capsules |
table of the process of sulcation and gyration [24]
Historical Research and Findings
Historical knowledge, predating when modern research techniques were made available, in understanding and studying the Central Nervous structure of humans and other animals were gathered by various investigations by Pathologists, Anatomists, Physiologists from the early 1800’s.
| Year | Research and Findings |
|---|---|
| 1824 | Luigi Rolando first discovered a method to study Central Nervous system structures via cutting chemically hardened pieces of brain tissue into thin sections for microscopical observations [25] |
| 1833 | Robert Remak discovers that the brain tissue is cellular. Ehrenberg discovers that it is also fibrillar |
| 1842 | Rolando’s method of observing CNS structures was perfected by Benedikt Stilling by cutting series of consecutive slices of the same tissue, this allowed the ability to trace nerve tracts and establish spacial relations |
| 1858 | Joseph von Gerlach brings forth a new process to differentiate between the different microstructures in the brain by treating the sample to a solution of ‘Carmine’.
This solution made the sample no longer appear homogenous under the lens but able to be differentiable to its components |
| 1889 | Camille Golgi comes forth with the procedure of impregnating hardened brain tissues with silver nitrate solution which resulted in the staining of nerve cells.
Possibility to trace cellular prolongations definitely to their termini now present. Ramon y Cajal announces his discoveries. Old theory of union of nerve cells into an endless mesh-work is discarded altogether, the new theory of isolated nerve elements ‘theory of neurons’ is fully established in its place [26] |
HOW DO WE REFERENCE BOOKS? A History of Science by Henry Smith Williams, M.D., LL.D. assisted by Edward H. Williams, M.D. (1904)
Current research models and findings
Current Research
Most previous studies describe overall growth of brain based upon in utero imaging studies with the use of magnetic resonance imaging (MRI) and ultrasound; however, complicated folding of the cortex in adult brain is due to different rates of regional tissue growth. In the following study, maps of local variation in tissue expansion are created for the first time in the living fetal human brain, in order to examine how structural complexity emerges in fetal brain.
|
Local tissue growth patterns underlying normal fetal human brain gyrification quantified in utero[27] Recent development in fetal MRI motion correction and computational image analysis techniques were employed in this study to help with the understanding of the patterns of local tissue growth. These techniques were applied to 40 normal fetal human brains in the period of primary sulcal formation (20–28 gestational weeks). This time period covers a developmental stage from the point at which only few primary sulci have developed until the time at which most of the primary sulci have formed, but before the emergence of secondary sulci on MRI. This developmental period is also important clinically, since the clinical MRI scans are also performed at this gestational age. Therefore it is important to describe the normal growth patterns in this period in order to be able to recognise abnormalities in the formation of sulci and gyri. Techniques mentioned previously were utilised to quantify tissue locations in order to map the tissues that were expanding with a higher or lower growth rate than the overall cerebral growth rate. It was found that relatively higher growth rates were detected in the formation of precentral and postcentral gyri, right superior temporal gyrus, and opercula whereas slower growth rates were found in the germinal matrix and ventricles. Additionally, analysis of the cortex illustrated greater volume increases in parietal and occipital regions compared to the frontal lobe. It was also found that gyrification was more active after 24 gestational weeks. These maps of the fetal brain were used to create a three-dimensional model of developmental biomarkers with which abnormal development in human brain can be compared. |
| The following recent studies use a similar model to what was described above:
Mapping Longitudinal Hemispheric Structural Asymmetries of the Human Cerebral Cortex From Birth to 2 Years of Age [28]
|
|
Asymmetry of White Matter Pathways in Developing Human Brains [29]
|
|
In the following studies new models and imaging techniques have been employed to gain a better understanding of the changes in brains of patients with schizophrenia Sexually Dimorphic White Matter Geometry Abnormalities in Adolescent Onset Schizophrenia [30]
Moreover, in the following study, a neuroimaging technique known as Proton Magnetic Resonance Spectroscopy (1H-MRS) is used to examine the brain of patients with schizophrenia. Multimodal neuroimaging of frontal white matter microstructure in early phase schizophrenia: the impact of early adolescent cannabis use [31] 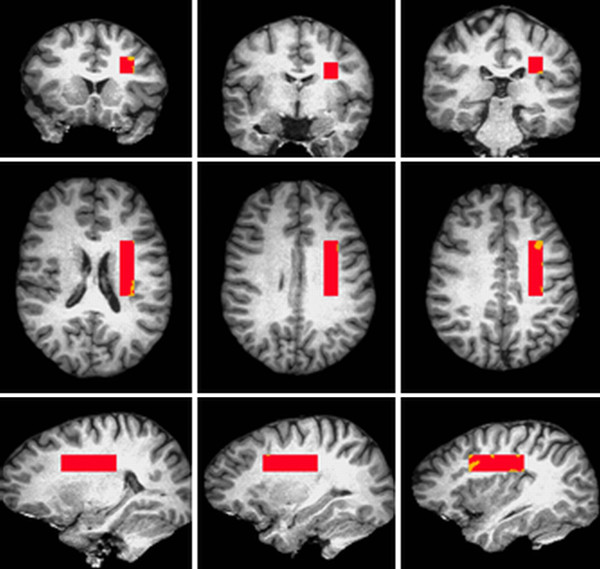 Proton magnetic resonance spectroscopy (1H-MRS) is used to examine the white matter tissue in brain of patients with schizophrenia. The image shows online placement of the 1H-MRS volume of interest.The 1H-MRS volume of interest is placed parallel to the AC-PC line (AC =anterior commissure, PC = posterior commissure). [32]
|
Abnormalities
Microcephaly, Macrocephaly and Hydrocephalus
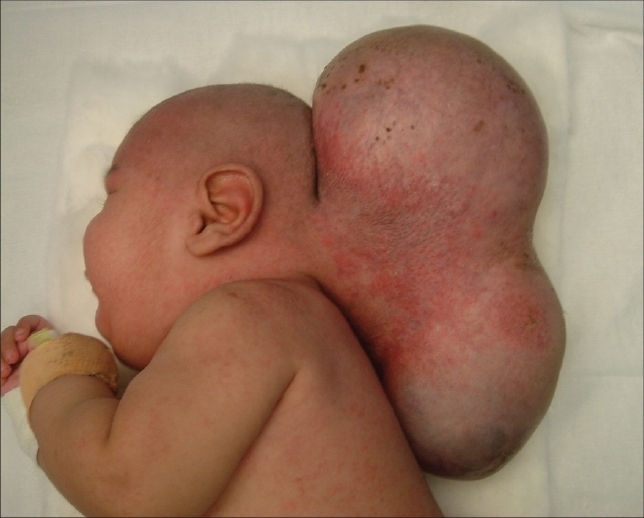
Microcephaly and macrocephaly refer to abnormal head size. These abnormalities are seen in less than 2% of all newborns. Learning abnormalities and neurophysiological malfunctioning associated with these abnormalities are dependent on etiology, severity and patient’s age.
Microcephaly
- Noticeable reduction in the size of brain is observed due to factors that kill the dividing cells in the ventricular germinal zone. These dividing cells give rise to brain cells (both neurons and glia).
- Microcephaly is specifically defined as a head size more than two standard deviations below the mean for age, gender and race.There are two diagnostic types of primary and secondary microcephaly.
- Primary Microcephaly: Abnormal development is observed in the first seven months of gestation.
- Secondary Microcephaly: Abnormal development occurs during the last 2 months of gestation (prenatal period).
- Microcephaly is caused by various factors that prevent normal proliferation and migration of cells during CNS development. These factors are divided into physical (irradiation, raised maternal temperature), chemical (anticancer drugs) and biological (infection of uterus due to rubella, cytomegalovirus and herpes simplex virus).
- Genetic defects and chromosomal disorders can also play a role.All of these factors result in destruction of the brain tissue (encephalopathy) with multiple areas of scarring and cyst formation. [35]
Macrocephaly
- In patients with macrocephaly the head is enlarged.
- Macrocephaly is specifically defined as a head size more than two standard deviations above the mean for age, gender and race.
- Macrocephaly is a syndrome of diverse etiologies rather than a disease and the most frequent cause is hydrocephalus.[36]
Hydrocephalus
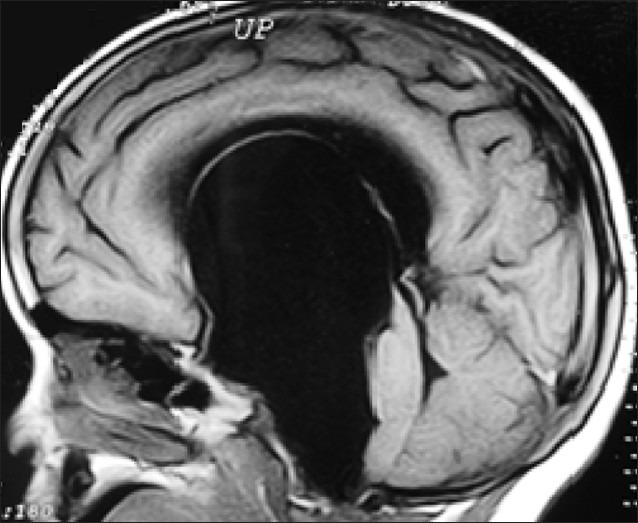
- Progressive enlargement of head due to accumulation of cerebrospinal fluid in ventricles is known as hydrocephalus.
- Excessive accumulation of cerebrospinal fluid is due to an imbalance between the formation and absorption of cerebrospinal fluid (communicating hydrocephalus) or obstruction of circulation of cerebrospinal fluid (non-communicating hydrocephalus).
References
- ↑ Report of the Workshop on Acute Perinatal Asphyxia in Term Infants, U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Child Health and Human Development, NIH Publication No. 96-3823, March 1996.
- ↑ <pubmed>22272298</pubmed>
- ↑ <pubmed>4203033</pubmed>
- ↑ <pubmed>5414696</pubmed>
- ↑ <pubmed>7204662</pubmed>
- ↑ <pubmed>12764033</pubmed>
- ↑ <pubmed>8523077</pubmed>
- ↑ <pubmed>9712307</pubmed>
- ↑ <pubmed>1713238</pubmed>
- ↑ 10.0 10.1 10.2 10.3 <pubmed>10532616</pubmed>
- ↑ <pubmed>489804</pubmed>
- ↑ <pubmed>5029133</pubmed>
- ↑ <pubmed>4407392</pubmed>
- ↑ 14.0 14.1 <pubmed>17533671</pubmed>
- ↑ <pubmed>17726524</pubmed>
- ↑ <pubmed>21042938</pubmed>
- ↑ 17.0 17.1 <pubmed>19339620</pubmed>
- ↑ <pubmed>560818</pubmed>
- ↑ <pubmed>8361683</pubmed>
- ↑ <pubmed>12764028 </pubmed>
- ↑ <pubmed>21042938</pubmed>
- ↑ <pubmed>21571694</pubmed>
- ↑ 23.0 23.1 23.2 <pubmed>11158907</pubmed>
- ↑ <pubmed>20608424</pubmed>
- ↑ <pubmed>7472570</pubmed>
- ↑ <Pubmed>17490748</pubmed>
- ↑ <pubmed>21414909</pubmed>
- ↑ <pubmed> 23307634 </pubmed>
- ↑ <pubmed> 24812082 </pubmed>
- ↑ <pubmed> 23307635 </pubmed>
- ↑ <pubmed> 3852698 </pubmed>
- ↑ <pubmed> 3852698 </pubmed>
- ↑ <pubmed>17275978</pubmed>
- ↑ <pubmed>3271622</pubmed>
- ↑ <pubmed>15806441</pubmed>
- ↑ <pubmed>21215908</pubmed>
- ↑ <pubmed>22069421</pubmed>