Paper - Description of a 4 mm human embryo (1906)
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bremer JL. Description of a 4-mm human embryo. (1906) Amer. J Anat. 5: 459-480.
| Online Editor |
|---|
| This historic 1906 paper is of course before the Carnegie embryo staging system was established, but represents an embryo probably at Carnegie stage 12 during Week 4 of development. |
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Description of a 4-mm Human Embryo
J.L. Bremer
with 16 text figures. (1906)
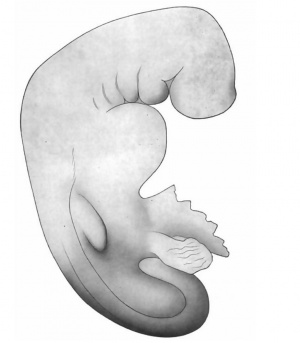
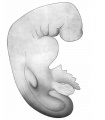
This embryo, series No. 714 of the Harvard Embryological Collection, noted as about three weeks old, is an excellent subject for study because of its good preservation and successful sectioning. Unfortunately the drawings of the whole embryo are inadequate, so that the sketch given here has been altered slightly to conform more accurately with the shape as we find it in the serial sections. The necessity for a part of this alteration may, of course, be due to shrinkage, but the form, as given in Fig. 7, is certainly at least approximately correct. This shows a large head, flexed sharply on the body, a curving back ending in a curled tail, twisted spirally to the right; a marked protuberance below the head for the heart, and an outgrowth for the fore limb. There is no trace of posterior limb. Appended to the ventral surface below the heart is the yolk-sac, represented here as cut irregularly, and art the right side of this, posteriorly, the body-stalk, cut near the chorion. Four pocket-like depressions, the gill clefts, lie behind a larger depression, the mouth. There is no surface marking to indicate the eye. Protuberances corresponding to the primitive segments are not shown in the original drawings, though plainly visible in a model of the rump region (Figs. 15, 16)- perhaps the irregularities of surface have increased with shrinkage. There is no sign of distortion or injury.
The embryo was preserved in 10 per cent formalin, imbedded in paraffin, and cut in a transverse plane. On microscopic examination the tissues are found to be excellently preserved; even the frequent mitotic figures in multiplying cells show clearly. In fact, by the histological condition, as well as by the external appearances, we are led to believe that the specimen is normal, and yet the state of growth of some parts and the form of others do not agree with the descriptions of human embryos of the same size given by His and other investigators.
The most notable difference is in the stage of growth of the nervous system. His describes the thickened medullary plates as having already, in an embryo of this size and general stage of development, been changed throughout their entire extent from a groove to a closed tube; but in this embryo the closure is incomplete in the anterior part of the head, and also for a considerable distance from the end of the tail, giving both an anterior and a posterior neuropore. This is the more interesting when we consider that in both the pig embryo and the rabbit embryo, as described in Reibel’s Normentafeln, the closure of the medullary tube is completed only after the formation of the head and neck bends, as in this human embryo.
Description of Models
Model of Brain
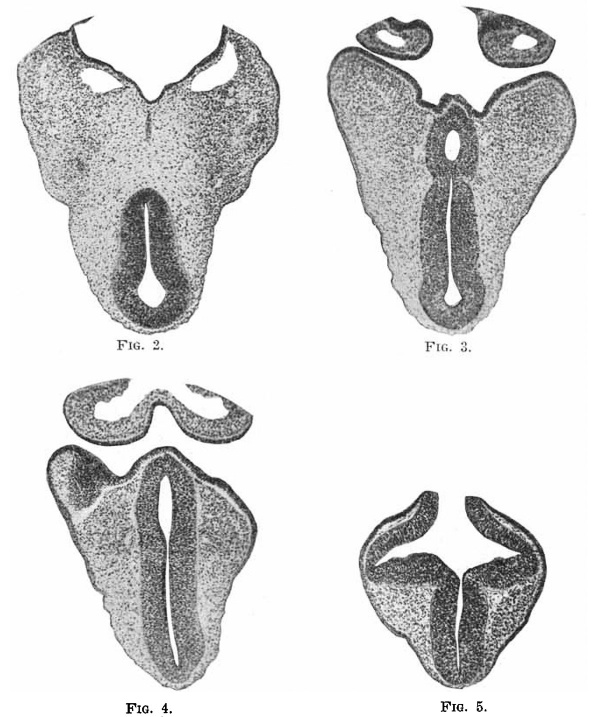
This model is shown in side view in Fig. 1, and again in Fig. 6, there turned so as to be seen nearly from below. The fore brain is divided into two parts, one of which includes the anterior neuropore (marked by the rolled ectoderm, shown only where that of the brain joins that of the skin), the lateral pocket of the optic vesicle, and further from the anterior end on the ventral side the enlargement for the hypophysis, which fits between the two lobes of this gland arising from the pharynx. The relations of the pharyngeal and medullary portions of the hypophysis are shown in Fig. 3, the photograph of a section of this part of the embryo at this level. Of the other photographs of the brain, Fig. 4 represents a section of the anterior part of the hypophysis, showing its continuity with the cavity of the brain, and Fig. 5, a section passing through the optic vesicle and the anterior neuropore. The plane of section is shown by the line between the two pieces of the model.
Fig.2-5. Sections through head.
In this first portion of the fore brain there are, beside the anterior neuropore, two other areas, shown in the model, where the ectoderm of brain and skin are continuous ; one in the mouth region, and one .behind (following the curve of the brain) the neuropore. Moreover just behind this last area there is a distinct prominence of the brain tube, not attached to the skin, which simulates a neural crest, as though for a pair of cranial ganglia. This of course must soon disappear, as no such pair of ganglia exists at a later period, but its presence is interesting as showing a tendency of this anterior portion of the brain to develop as do the more posterior portions.
Between this anterior part of the fore brain and the mid brain, not marked distinctly from either, is found a narrow strip of less caliber than the anterior portion, with no special prominence and no connection with the ectoderm of the skin. A cross section of this is shown in Fig. 2.
The mid brain, occupying the head bend, is also without special interest, except that it is of smaller caliber than might be expected. It merges without line of demarcation into the hind brain. The hind brain is again divisible into two parts; one posterior, smooth, of nearly even caliber, merging gradually at the neck bend into the spinal cord; and one anterior, showing a further development. The roof of this anterior part is thin, has become shrunken and crumpled. As yet, however, there is no sign of a Varolian bend. Attached to the side, ventrally, are two ganglia, one for the Trigeminal nerve, and one for the Acoustico-facial complex, in which there is only a faint indication of separation into distinct nerves. Behind this second ganglion, applied closely to it, liea the otocyst, a rounded hollow vesicle, close to the brain, but not touching it, and !till attached externally to the ectoderm, a patch of which can be seen on its outer surface. Of the other cranial ganglia nothing is shown in the model, as they are represented only by diffuse groups of cells on each side of the hind brain, not yet divided into separate ganglia, and not attached to the brain wall. There is a peculiar notch in the floor Of this part of the hind brain, shown in Fig. 1, between the two ganglia, bounded by two rounded prominences seen better in Fig. 6, the significance of which I do not know.
Fig. 6. Model of brain, seen from below. X 60.
On taking dff the top part of the model, we find that the cavity of the hind brain is marked by a median ventral furrow. The sides are smooth, except in the anterior portion, where there are distinct traces of neuromeres. Toward the cord the cavity is compressed laterally, but there is no sign of a groove between the zones of His. Fig. 6 shows the wide open anterior neuropore, through which one can look into the hollow optic vesicles, and into the median cavity of the fore brain, which is seen to be a narrow slit, compressed laterally. The edges of the opening are broadly curved or rolling, with no demarcation to indicate where the future line of closure is to be. This is shown in section in Fig. 5.
Model of Pharynx and Aortic Arches
The pharynx, of which the model is a cast, the shape of the cavity being represented, is shown in Fig. 1, in its proper relation to the brain. It consists of a broad body opening anteriorly into the mouth, the lateral extension of which, between the maxillary and mandibular processes, is represented as a cut surface. On the dorsal surface of this body, near the fore brain, are irregularities, chiefly two rounded ridges, one on each side of the median line, representing the out-poclietings for the pharyngeal lobes of the hypophysis, as shown in Fig. 3, and explained above. Behind these, at the angle made by the roof of the pharynx corresponding to the curve made by the brain at the head bend, is a median ridge or point which is continuous with the notochord, and marks the anterior end of the latter. Toward the Esophagus, into which the body of the pharynx merges, the cavity becomes more and more compressed laterally, until it is an antero-posterior slit. From the sides of this main cavity three lateral out-pocketings project, the most dorsal one being divided into two smaller projections near the end. These represent the gill pouches, and diminish in size from before backward. The first two rapidly become compressed antero-posteriorly and end in blunt, more or less vertical ridges, while the third is more tapering, and the fourth ends in two distinct pointed branches.
In the tissue between the opening of the mouth and the first gill pouch, and also between the first and second, and second and third gill pouches, run the first, second, and third aortic arches on each side, joined dorsally by the dorsal aork, which make an impression on either side of the median line of the body of the pharynx, and then continue downward, beside and behind the swphagus. On the model, the aortic bulb is represented as cut off just above the standard; a cup-shaped chamber leads from this forward, and gives off on each side two vessels; one pair of vessels forms the first aortic arches, a second pair divides into the second and third arches of each side. The connection between these last two pairs of arches and the aortic stem is very slender, compressed laterally, as though the arches had grown from the dorsal aortae and had only just united with the ventral aortae. Of the fourth and fifth aortic arches the only trace is the rim of the cup-shaped part of the ventral aorta, shown in the figure below the bend-of the third aortic arch. No branches come from the dorsal aortae in the position of these fourth and fifth arches. The dorsal aorts are joined in a single median vessel for a considerable distance along the back; there is no trace of carotid branches from the first arches toward the head.
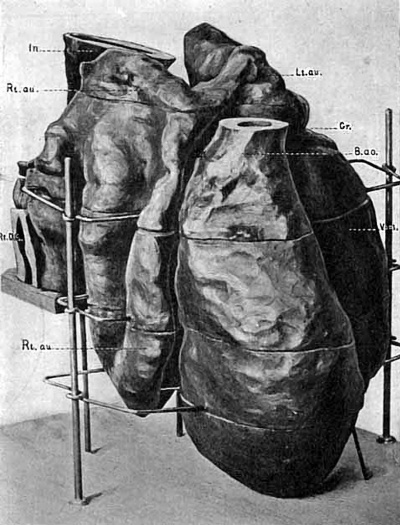
Model of Heart
An anterior view of the model of the heart is given in Fig. 9, the model is seen slightly from the right side. The most anterior portion is the large rounded aortic bulb (B.no.), cut off just below its division into aortic arches as shown in the model of the pharynx. Continuous with this is the ventricle (Vent.) a large single chamber, beginning at a constriction marked by a groove on the outside (Gr.), extending downward on the left side, turning on itself to forni the apex of the heart, and then tapering upward to merge with the aortic bulb. The walls of the ventricle are thick, smooth externally, but internally very irregular, with deep pockets lined by endothelium leading from the main chamber, representing the sinusoids of the heart. In the aortic bulb the walls become smooth internally; there is no sign of division into right and left ventricles. The left auricle (Lt. au.) is placed above the ventricle, marked from it externally by the groove. Its walls are thin and folded, probably by pressure, and internally the endothelium is smooth, with no pockets nor trabeculae. The left and right auricles are joined near their upper ends anteriorly by a prominent ridge, enclosing a cavity, which extends downward, compressed antero-posteriorly, and opens by a narrow channel into the ventricle to the left of the median line. Above this connection the right auricle rises dorsally to a rounded peak, not quite so high as the top of the left auricle, from which it is separated by the intestine (In.), but, unlike the left auricle, the right extends far below the auriculo-ventricular canal as a spacious pouch, reaching almost to the level of the apex of the ventricle, and occupying a position behind and to the right of the aortic bulb and the lower part of the ventricle. Here, as in the left auricle, the walls are thin, smooth on both external and internal surfaces, and thrown into great folds. If this auricle were distended it could certainly contain a larger volume of blood than the ventricle. This enormous right auricle is not, to my knowledge, figured in any model or drawing of human or other mammalian hearts of any stage, but conforms almost exactly to the Selachian heart.
The blood enters the heart by way of the right auricle, from the sinus venosus, as is shown in Fig. 10. In this figure the heart is viewed from behind and slightly from the left side, and a portion of the model, the dorsal wall of the sinus venosus, has been removed. The jugular veins from the head region and the umbilical veins from the body join on either side, on the left more anteriorly than on the right, and from this junction on each side an irregular sinus extends across the embryo, interrupted in places by bands of tissue (the cut end of one of which is seen in the figure) into which open dorsally the two vitelline veins. From this sinus venosus the blood passes to the right auricle by two separate openings (z and y). Here again this embryo differs from those previously described or modelled, in all of which a single channel leads from sinus venosus to right auricle. Dr. Minot has recorded separate openings into the heart for the cardinal and omphalo-mesaraic veins in the chick (Textbook, p. 282), but the double openings just described do not appear to be correlated with those in the bird.
The heart lies in the body cavity or ccelum, and is attached to the body wall by the sinus venosus, and to the intestinal tract by a short fold of mesenchyma. Fig. 10 shows also a portion of this intestinal tract; the mesodermal part has been partially dissected away from the entoderm in order to show the outgrowth destined to become the lung (Lu.).This outgrowth, situated at the level of the left auricle on the ventral side of the intestine, where the pharynx is becoming narrowed to form the esophagus, is rounded, irregular, and extends to right and left and caudally. The right side of the outgrowth is the larger and extends further tailward, leaving the esophagus at a more acute angle than the left, a reversal of the adult condition. There is no distinct trachea, as the cavity of the esophagus is continued directly into the cavities of the right and left lobes of this outgrowth.
Model Of Veins Near Heart
This model represents the venous channels as solid, and includes the junction in the sinus venosus of the three large pairs of veins, the jugular and the umbilical veins in the body wall, and the vitelline veins in the intestinal wall. The sinus venosus is shown as a cavity in the model of the heart, so that these two models overlap to a certain extent. Fig. 11 shows the model as seen from the dormm of the embryo ; Fig. 12 as seen from the left side; and Fig. 13 is a view taken from the caudal end of the embryo, and a little from the right side.
The umbilical veins, entering the embryo by the body stalk, run in the body wall forward, at a level ventral to the intestine, varying only slightly in diameter. At points differing slightly on the two sides, more anteriorly and dorsally on the left, they join the two jugular veins and turn inward (on the left side also ventrally) as the sinus venosus. From each jugular vein, nearly at its junction with the umbilical vein, arises a small bud, the future posterior cardinal vein (P . curd.). The Duct of Cuvier (D.C.), later a well marked trunk extending from the junction of jugular and cardinal veins to the entrance of the umbilical veins, can in this embryo, then, scarcely be said to exist, so short and ill-defined is it.
The sinus venoms is seen, as in the model of the heart, extending across the embryo, an irregular cavity subdivided by bands of mesenchyma (represented in this cast as holes), and emptying anteriorly into the right auricle by two channels (Bt. au.). Into this sinus empty also the two vitelline veins (Vit.w.) one on each side of the intestine, at a level dorsal to the umbilical and jugular veins (Fig. 12). The vitelline veins, unlike the two other pairs modelled, vary greatly in caliber; they are small and rounded at their entrance into the sinus venosus, but more caudally spread into large channels, each with a mesial wall straight, vertical, fitting closely the sides of the entodermic intestine, which lies between them (Figs. 11 and 13). The lateral wall of each vitelline vein is very irregular, giving off many branches, which join to form new longitudinal channels, the whole making an irregular plexus. The plexus from the right vitelline vein is more developed than that from the left, and has even acquired an opening of its own into the sinus venosus (Figs. 11 and 13). From the plexus on each side branches extend ventrally, those on the right ending blindly, but one on the left side joining by a narrow channel (Figs. 12 and 13, x) with another venous plexus ( V . pl.) extending across the median line, ventral to the intestine, and posterior or caudal to the sinus venosus, from which it is entirely free. This ventral plexus is in intimate relation with the liver, and will be spoken of more fully in the next section.
Posterior to the outgrowth of these plexuses the vitelline veins divide into many branches which spread out over the yolk sac, into which the intestine soon opens. Cut ends of some of these branches may be seen in Fig. 13.
Model of the Liver
This model is seen from the caudal end, and a little from the right side in Fig. 14. The intestine is a large tube much compressed laterally, and the vertical right side is shown. From the ventral border of this tube hangs a large outgrowth, extending first ventrally, then laterally, then dorsally, composed of an irregular mass of entodermal cells, which tends to break up into cords, often anastomosing with each other. Only the right side of this mass has been modelled. The cavity of the intestine passes for a short distance into this mass, but without subdivision, so that all the cords and irregularities are solid. In the model a part of the left wall of the intestine has been cut away to allow a view of this cavity.
It has been stated by other writers that the liver arises by cords of cells which grow into the territory occupied by a large vein, .pushing before them, and thus becoming invested by the endothelium, and forcing the blood to run in small, anastomosing channels, the sinusoids. This statement would not be true of this embryo, for the liver cords are found growing into mesenchyma, at a level ventral to the vitelline veins; in this same mesenchyma, however, we find the branches of the vitelline veins ramifying, and forming plexuses, and in certain places these plexuses come into intimate relation with the liver cords. These points of contact, wh’ere veins and liver actually touch, are marked in the model of the liver (Fig. 14, V,), and are seen to correspond with the anterior surfaces of the portion of the plexuses marked in the model of the veins (Fig. 13, li.). These two models might be fitted together, in which case we should find the intestine lying between the vertical, mesial walls of the two vitelline veins, the liver spreading in the same mesenchyma that contains the lateral and ventral venous plexuses, between the sinus venosus in front and the ventral venous plexus behind; but the two models would come into actual contact only at the points marked and already noted. As both the liver and the venous plexuses continue to grow, they will come into contact over more and more of their extent, until finally the result of small, anastomosing channels, the endothelium of which is in close contact with the liver cords, will be produced. The method by which this result is attained is, however, in this embryo different from that usually described; in the one case, the cords of liver cells push against the walls of a pre-existing large venous channel, and by invaginating these walls, make finger-like trabecula: within this large space, yet separated from the cavity by the endothelium which invests them. These cords then anastomose until the cavity of the vein is subdivided into many smaller channels, the sinusoids, all lined by the original endothelium, and having for walls, beside this endothelium, the cords of liver cells. In this embryo, on the other hand, the cords of liver cells and the small branches from the vitelline veins are both pushing into the same mesenchyma, both growing rapidly, until, the mesenchyma remaining small in amount, the whole enlarged mass will be made up practically of the veins and the liver cells, which by their continued growth must come into contact, the veins filling practically all the spaces between the cords, and the thin endothelium becoming wrapped around the cords ;and thus the adult condition will be reached.
In this model is also shown the anlage of the pancreas. (Fig. 14, Pa.) It is a small, knob-like mass of cells, with no cavity, growing from the ventral border of the intestine just caudal to the liver outgrowth, but distinct from the latter. Further down the intestine (nine sections in the specimen) where it has begun to expand to the yolk sac, is found another, smaller mass of cells, growing also from the ventral border, which probably represents a second anlage of the pancreas; there is no sign of a dorsal anlage. The pancreas also, then, differs from those usually described, as it has probably two outgrowths, neither from the liver stalk, for the duct of Wirsung, and as yet no outgrowth to represent the duct of Santorini. There is no enlargement of the intestine above the liver
for the stomach.
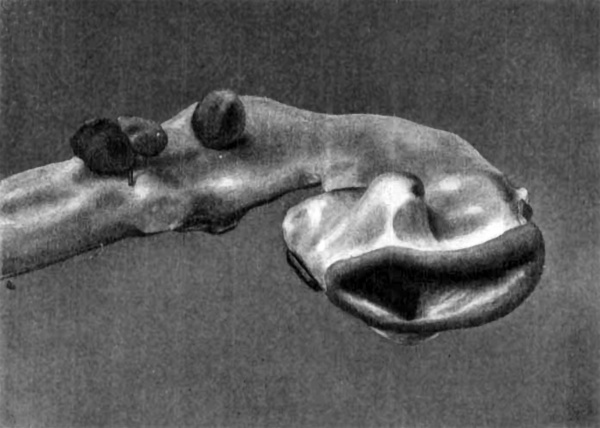
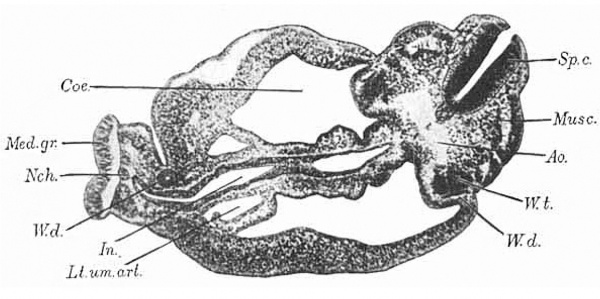
Model of the Tail End of the Embryo
Fig.8. Section through body of embryo, near the cloaca.
This model has been photographed in two positions, once (Fig. 15) squarely from the right side, and again (Fig. 16) still from the right side, but also a little from behind. The left side of the model shows the surface of the embryo with the umbilical cord attached to it. A portion of the right side, that at the tip of the rump, shows the outer surface also; but for the remainder of this side, the ectoderm of the skin and the inesenchyma have been dissected away to the median line, which in this part of the embryo is distinctly curved on account of the spiral twist of the tail. The curve can be clearly understood by referring to the photograph of a section (Fig. 8) at the level of the two brass handles for moving the model, shown in Fig. 16. The plane of section is shown by the bottom line of the dissected part of t,he model. The outline of this section will explain the irregularities of the surface at the rump end as seen in the model. Posteriorly, from the median line forward extends a rounded, smooth area overlying the spinal cord; next this anteriorly, another rounded area, broken by transverse grooves into irregular mounds, representing the primitive segments ; and still more anteriorly a broad, smooth area, separated from the last by a longitudinal groove, covering the body wall, within which lies the coelom. These three itreas are curved upon themselves at the rump end, the protuberant body wall making a distinct ledge-like projection, as seen in the photographs (Figs. 15, 16, B. w.).
Features
Spinal cord
The spinal cord is shown in both drawings, appearing at the dorsal side of the embryo, directly under the ectoderm of the skin. The shape in cross section can be seen in Fig. 8 ; the opening of the canal to the outside is artificial, a tear in the section. The spinal cord follows the curve of the rump, passing nearly out of sight in the undissected part, but reappearing nearer the tail. Here a change has taken place. The ectoderm of the spinal cord and that of the skin have been gradually becoming continuous, and just after rounding the curve of the rump the medullary tube is replaced by an open medullary groove. This opening of the spinal cord to the outside is accompanied, as in the case of the anterior neuropore, by a rolling of the edges, as shown in Fig. 16, and in section in Fig. 8. The groove becomes wider, until ihe sides lie in the same plane, but the medullary plate is distinguishable from the ectoderm of the skin, almost to the end of the tail, by its thickness. At the tip of the tail, which has been cut off in the model, all ihe tissues become merged into an undifferentiated mass.
Notochord
The notochord, shown in the back region in Fig. 15, and in the tail region in Fig. 16, lies just ventral to the spinal cord to which a t times i t seems attached (Fig. 8 ) . I n cross section it is a t first rounded, but at the curve of the rump becomes oval, with long axis dorso-ventral, and considerably larger. It follows the curve of the spinal cord, and is lost not far from the end of the tail by merging with the spinal cord (Fig. 16).
Aorta
The aorta lies ventral to the notochord, separated from it by a narrow strip of mesenchyma. At first, i. e., in the back region, it sends off small buds, not always directed laterally, apparently not segmentally arranged, but presumably representing intersegmental arteries. At the apex of the rump curve the aorta, now entirely separated from its fellow on the opposite side of the body, leaves the notochord and curves sharply forward. At this point a branch is found, extending laterally and toward the spinal cord, mesial to the Wolfian duct; this is the iliac artery (Fig. 16, II. ur.). Beyond Ohis branch the aorta passes, as the right umbilical artery, lateral to the cloaca and the allantois, to the beginning of the body stalk, but here becomes obliterated on the right side (Rt.urn. ar.). On the left side, however, the left umbilical artery (Lt. urn. ar.) becomes a much larger channel, and continues along the body stalk. The mesenchyma of the body stalk has been partially disseated away above a certain level to show the left umbilical artery and allantois. Throughout the first part of its course the umbilical artery is of irregular caliber, but soon it becomes smooth.
Intestine
The entodermic intestine (Figs. 15, 16, In.) appears as an oial tube, with long axis antero-posterior. It is situated in front of the aorta, but separated from it by the mesoderm of the mesentery, which has been dissected away to the median line. Ventral to the entodermic intestine, also, the mesoderm of the intestinal wall has been cut away to the median line. The entodermic tube follows the general curve of the embryo, growing gradually larger, and empties into a large rounded pouch, also compressed laterally, the cloaca. I n Fig. 8 the section passes nearly through the junction of the intestine and the cloaca, and the compressed shape can be more easily seen. The mesentery becomes gradually shorter and finally disappears entirely. The cloaca, along its greater curvature, lies close to the notochord, and passes into the tail as the tail gut, which soon loses its lumen and ends near the tip of the tail by fusing with the ectoderm in an undifferentiated mass. At the root of the tail the cloaca comes into close contact w-ith the ectoderm of the skin at the anal plate (An.pZ.). From the upper, posterior corner of the cloaca, just above the entrance of the intestine, passes off the allantois, which, as a slender rounded tube, curves out into the body stalk close to the wall of the ccelom, between and posterior to the two umbilical arteries, and ends blindly. A slight, irregular swelling occurs in its course, marking perhaps the position of the future bladder.
Wolffian duct
The Wolffian duct on the right side (Figs. 15, 16, W . d.) is shown in the model standing out at the side, because freed from the mesoderm. It follows the general curve of the body, just ventral to the plane of the notochord, gradually approaches the median line on its way toward the tail, and ends before reaching the level of the anal plate by joining the cloaca (W.d.’) . As can be seen in Fig. 8, its position corresponds with the external groove between the area of primitive segments and the body wall. On the left side the Wolffian duct does not extend so far as the cloaca, but ertds blindly, and so is cut only once in the section (Fig. 8, W . d.). Dorsally the duct is oval in cross section, with long axis parallel with the lateral surface of the Wolffian ridge, which is seen in Rg 8, projecting into the ccelom dorsally; but nearer its union with the cloaca, the duct assumes a rounded form.
The Wolfian tubules, seen in Fig. 8, just mesial to the duct, are not shown in the model. They are short, sometimes with an S-shaped curve, frequently overlapping, as in the section given, where portions of two tubules are shown on bhe left side ( W . t.). I n all there are twenty- eight tubules, arranged in two groups; one caudal group, consisting of twenty-three tubules, close together, but not definitely arranged in relation to the primitive segments or muscle plates (Fig. 8, musc.). This represents the true Wolffian body, and the tubules are all closed at their distal ends. Beyond this the Wolffian duct can be traced anteriorly for some distance (through twenty sections), and still further anteriorly is found a second group of tubules, consisting of three rather indistinct, scattered units, not connected by a duct. The most anterior tubule lies at the level of the liver and opens by a funnel-shaped mouth into the ccelom. This smaller group of tubules belongs to a rudimentary pronephros.
In conclusion, I wish to point out the chief differences between this embryo and those of the same general stage of growth already described. The most important is the presence of anterior and posterior neuropores, which were supposed to have closed in human embryos of this size, though known to be open in comparable embryos of the pig and rabbit. Next in importance, perhaps, is the large right auricle of the heart, and the double passage into it from the sinus venosus. Next, the growth of liver cells, not into territory occupied by a large vein, but into the mesenchpma surrounding the intestine, where also are growing branches of the vitelline veins; from the intercrescence of cords of liver cells and venous channels, sinusoids would inevitably result. Finally the presence of a few pronephric tubules, and the tardy growth pf the left Wolffian duct, which has not yet acquired an opening into the cloaca. Let me add once more that embyro is, histologically and anatomically, in excellent condition, and as far as can be seen normal.
Abbreviations Used
|
|
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
- Human Embryo 4 mm: Fig. 1. Brain and Pharynx Models | Fig. 2-5. Head Sections | Fig. 6. Brain Model | Fig. 7. Embryo Drawing | Fig. 8. Body Section | Fig. 9. Heart Model
Reference
Bremer JL. Description of a 4-mm human embryo. (1906) Amer. J Anat. 5: 459-480.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - Description of a 4 mm human embryo (1906). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Description_of_a_4_mm_human_embryo_(1906)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G