Human Embryology and Morphology 1
Keith, A. Human Embryology And Morphology (1921) Longmans, Green & Co.:New York.
Human Embryology and Morphology: 1 Early Ovum and Embryo | 2 Connection between Foetus and Uterus | 3 Primitive Streak Notochord and Somites | 4 Age Changes | 5 Spinal Column and Back | 6 Body Segmentation | 7 Spinal Cord | 8 Mid- and Hind-Brains | 9 Fore-Brain | 10 Fore-Brain Cerebral Vesicles | 11 Cranium | 12 Face | 13 Teeth and Mastication | 14 Nasal and Olfactory | 15 Sense OF Sight | 16 Hearing | 17 Pharynx and Neck | 18 Tongue, Thyroid and Pharynx | 19 Organs of Digestion | 20 Circulatory System | 21 Circulatory System (continued) | 22 Respiratory System | 23 Urogenital System | 24 Urogenital System (Continued) | 25 Body Wall and Pelvic Floor | 26 Limb Buds | 27 Limbs | 28 Skin and Appendages | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chapter I. Early Changes in the Development of the Ovum and Embryo
The First Five Weeks of Development
In the first five weeks of human development changes take place very rapidly. In that short time the fertilized ovum passes from the condition of a single cell, with a diameter of 1/250 of an inch[1] to a fully formed human embryo about 1/5 of an inch in length (5 mm.), and contained within a spherical envelope of embryonal membranes which measures nearly an inch in diameter (see Figs. 1 and 2). By the end of the fifth week the beginnings of all the parts of the adult body are recognizable — the head, the trunk, the limb buds, the primitive segments, the eyes, the nose and mouth. A section across the abdominal cavity of an embryo at this stage (see Fig. 23) reveals the fact that the foundations of the genital glands are already laid, and that certain cells have been set aside for the reproduction of another generation. Thus by a cycle of developmental changes a new generation of reproductive cells has been produced from the fertilized ovum of a former generation all taking place within the short space of five weeks. In this chapter we are to follow the changes which lead from the fertilized egg or oocyte of the mother to the establishment of a new brood of genital cells in the embryo. In this way life is handed on from one generation to another.
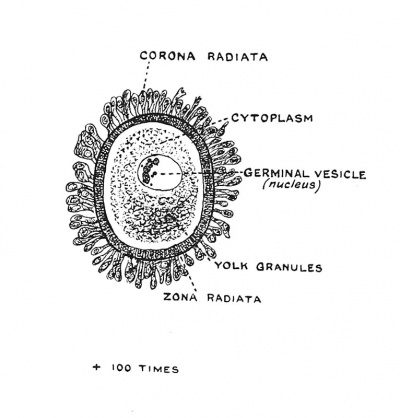
|
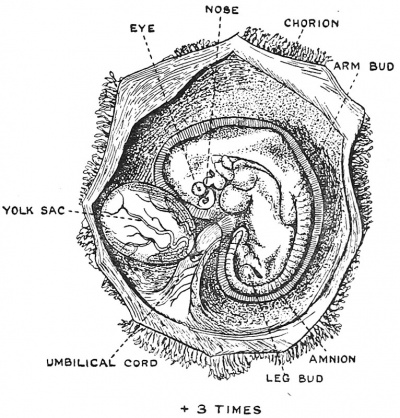
|
| Fig. 1. The parts of a Mature Human Ovum. (After Van der Stricht.) | Fig. 2. Human Embryo and its Membranes at the end of the fifth week of development. After Julius Kollmann (1834 - 1918) |
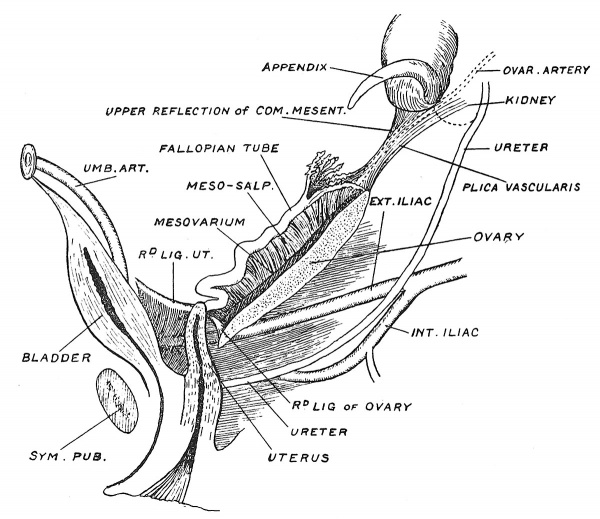
Fig. 3. The position of the Ovary and Fallopian Tube in the 5th month.
Descent of the Ovary
In tracing the cycle of changes which lead from the fertilized ovum of one generation to the production of a reproductive gland in the next generation, we may begin our study at any point, but for many reasons it is convenient to commence with the condition of the ovary in a fifth-month foetus. The ovary is descending or migrating from the region of the kidneys where it was formed, and has reached the iliac fossa. In all primitive vertebrate animals the genital glands are stationed above the kidneys, but in mammals, for reasons to be explained afterwards, they descend to the lower part of the trunk — a change which is especially well seen in the human subject. In the fifth month the ovary is long and narrow, with an upper or cranial and lower or caudal pole ; it is three-sided in section — the surfaces being medial, lateral and inferior or ventral (Fig. 3). The Fallopian tube or oviduct lies along the outer side of the ovary in the iliac fossa ; its upper fimbriated end terminates at, and is attached to, the upper or cranial pole of the ovary (Fig. 3). As the parts lie on the iliac fossa, the tube and the ovary are supported each by its own mesentery, the mesosalpinx and mesovarium. The two mesenteries have, however, a common origin or attachment to the posterior abdominal wall, and to the common attachment the name of common genital mesentery may be given (Fig. 4). The upper end of the common mesentery — the plica vascularis (Fig. 3), as it is reflected from the cranial pole of the ovary and fimbriated extremity of the tube, is continued up towards the diaphragm and in it the ovarian vessels and nerves pass to the ovary and tube. The caudal pole of the ovary is joined to the uterus by its round ligament. The round ligament of the uterus, corresponding to the gubernaculum testis of the male, passes from the brim of the pelvis, where it is attached to the horn of the uterus, almost straight to the internal inguinal opening and assists in the descent of the ovary and tube.
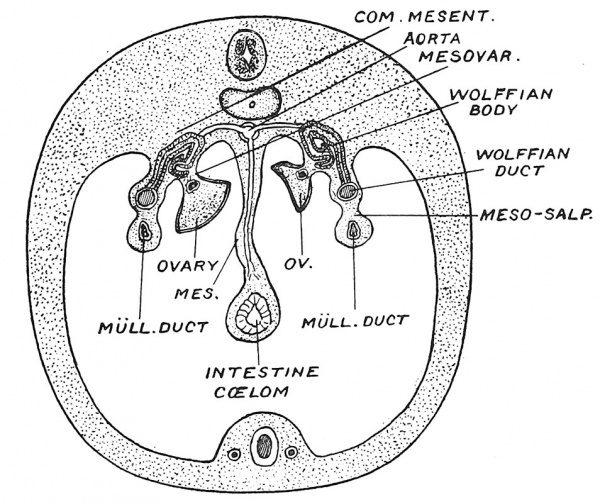
Fig. 4. Diagrammatic Section of a Foetus at the beginning of the 3rd month (30 mm. long), showing the attachments of the Ovary and Mullerian duct.
By full term the ovary lies at the brim of the pelvis or partly within it ; after birth the ovary, uterus and rectum come gradually to occupy their adult positions within the pelvis. This is due to a relatively great growth in the pelvis, which becomes marked as the child learns to walk, and especially in the female at the time of puberty. The ovary, as is more frequently the case with the testicle, may be arrested in its descent.
In Fig. 4 an earlier stage is shown ; it represents the condition about the beginning of the third month. The ovary and tube with the remnants of the Wolffian body — a primitive form of kidney — occupy the position in which they are developed. Both are suspended by mesenteries from the dorsal wall of the peritoneal cavity, at the side of the mesentery of the gut.
Normal Position of the adult Ovary
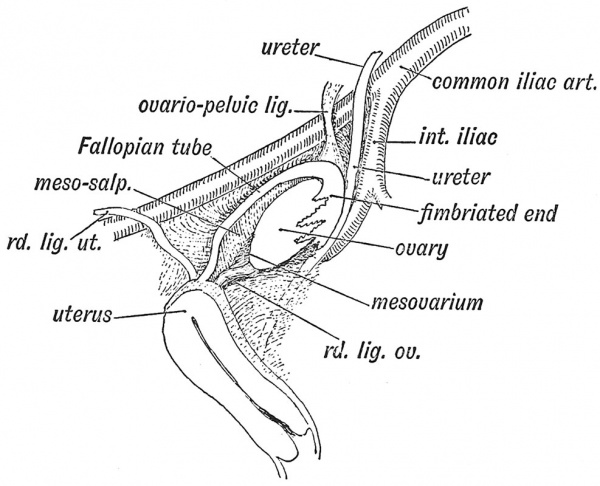
When the ovary descends within the pelvis it usually occupies a definite triangle — the ovarian triangle — on the lateral wall of the pelvic cavity (Fig. 5). The ovarian triangle is bounded above by the upper half of the external iliac artery, below and behind by the internal iliac artery, with the ureter lying on the artery ; in front by the reflection of the posterior layer of the broad ligament on the side of the pelvis. The peritoneum covering the triangle forms a depression, or occasionally a pouch, for the ovary. The fimbriated end of the Fallopian tube is applied to the ovary, ready to receive the ripe ova and transfer them to the uterus. One of the fimbriae — the ovarian fimbria — tethers the tube to the ovary. It will be seen that, with the descent of the ovary, the mesosalpinx, the mesovarium, and the common genital mesentery have come to form the major part of the broad ligament. The upper end of the common genital mesentery now forms the ovariopelvic ligament (Figs. 3 and 5). The ovary brings down with it, too, the ovarian arteries, veins, lymphatics and plexus of nerves. The nerves come through the aortic plexus from the 10th and 11th dorsal segments of the spinal cord, and the lymphatic vessels carry the ovarian lymph to a group of glands situated high up in the lumbar region.
Fig. 5. Showing the position of the Ovary on the lateral wall of the Pelvis and its relation to the Fallopian Tube.
An Ovum
As the infantile ovaries descend, each is laden with thousands of ova (over 10,000, T. G. Stevens ; 100,000, F. H. A. Marshall). It is estimated that not more than 200 in all become ripe and are shed.[2] The ova are embedded in the stroma of the ovary, each being surrounded by a special company or cluster of epithelial cells, which provide both a nest and nourishment for the ovum or oocyte (Figs. 6, 7). The cells which surround an ovum, with a condensed layer of the stroma cells outside them, form a Graafian follicle. As the ovary descends it is covered by a cubical epithelium, derived from the germinal epithelium which formed a stratum on the free surface of the ovary at its first appearance in the roof of the abdominal cavity. The ova and their accompanying follicular cells are derived from the surface stratum.[3] Amongst the columnar cells of the germinal epithelium and also in the stratum immediately beneath them are large peculiar cells. These are the primordial ova from which brood ova arise. The ova are thus carried within the ovary by ingrowths of the germinal epithelium. These tubular invasions into the ovary become broken up, the isolated masses of the germinal epithelium remaining to form the linings of the Graafian follicles. In the outer or cortical zone follicles continue to form in early foetal life, but after birth and even to the end of the fifth decade of a woman's life, follicles are being continually formed. With this new formation there is an equally constant process of degeneration or atresia of follicles. We shall see that another important constituent of the ovary, also arises from the tubular incursions of the germinal epithelium — namely the interstitial cells, which are glandular in nature, and supply an internal secretion which has much to do with the growth and regulation of the sexual structures of the body.
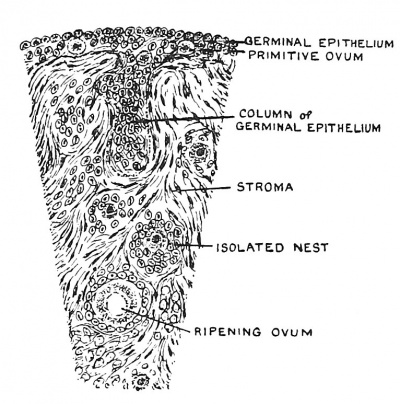
|
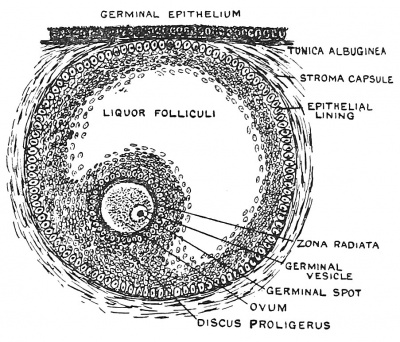
|
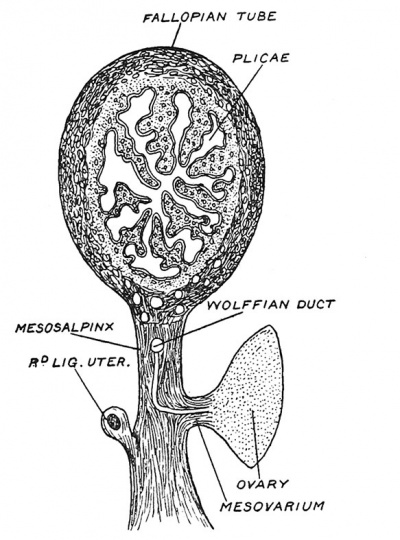
| Fig. 6. Diagrammatic Section of the Ovary of a fifth month Foetus, showing Nests of Germinal Epithelium and unripe Graafian Follicle. | Fig. 7. Ripe Graafian Follicle at Puberty. |
Discharge of the Ova
At puberty especially, also before it, and for 30 years after it, one egg after another ripens ; the ovum enlarges ; so does its Graafian follicle (Fig. 7). The cells of the epithelial lining proliferate and a cavity appears within the follicle, due to a collection of fluid — the liquor folliculi — amongst the cells. The ovum remains attached to the wall of the follicle by a group of epithelial cells, the discus proligerus or cumulus (Fig. 7). As the fluid collects, the follicle works its way to the surface of the ovary ; the tunica albuginea, which forms a capsule for the ovary, and the covering epithelium, gradually atrophy over it, and at last it bursts and discharges the ovum.[2] Ova may be shed at any point of a woman's menstrual cycle but the most usual time is during or just after the menstrual period. All the circumstantial evidence at our disposal points to the 10th or 11th day of a menstrual cycle, counting from the first day of the menstrual flow, as being the most common for conception. Whether ova are discharged from both ovaries at once, or from only one, and whether one or more than one in a month, are points not yet settled ; but the usual opinion is that one ovum is shed each month, and only from one ovary. An ovum shed from one ovary may occasionally pass down by the opposite Fallopian tube.
The Graafian follicle, after rupture, fills up with blood ; a cellular tissue is soon developed within its cavity from the lining cells of the follicle but particularly from cells of the inner sheath of the follicle. The inner sheath cells break into the follicle and carry blood vessels with them. These cells come to contain lutein, which gives them a yellowish colour when seen in the mass. If pregnancy does not occur, a false corpus luteum is formed, a formation which begins to disappear before the next menstrual period. If pregnancy occurs, however, the cellular mass continues to increase in size until it forms a glandular body as large as a pigeon's egg and is known as a true corpus luteum. It reaches its maximum size about the fourth or fifth month of pregnancy ; it is much reduced in size by the end of that period. Experiments have been made by Marshall and Jolly and by Blair Bell which show that the secretion of the corpus luteum acts on the decidual or lining membrane of the uterus, sensitizing it so that it responds by growth when the fertilized ovum comes in contact with the decidua. If the corpus luteum is excised pregnancy is prevented, or if begun, is arrested. Both forms of corpus luteum lead to the formation of cicatrices which are to be seen on the surface of the ovary. The ovary of an old person is commonly covered with such scars. The Graafian follicles may become cystic and give rise to enormous ovarian tumours.
The Fallopian Tube
When the ovum or oocyte drops from the ovary it cannot easily escape the ciliated fimbriae of the Fallopian tube which surround and clutch the ovary. In Fig. 5 the relationship of the Fallopian tube to the ovary is shown. The tube may be demarcated into three parts : (a) the isthmus or arm directed outwards to the wall of the pelvis (I to 1 inch) ; (6) the forearm or ampullary part, directed backwards on the lateral pelvic wall above the ovary ; (c) the hand, infundibular, or fimbriated part, folded backwards and grasping the free border and exposed surface of the ovary. The tube is fastened by one of its fimbriae to the cranial pole of the ovary.
Course of the Ovum in the Tube
The cilia on the fimbriae work towards the ostium abdominale, the abdominal mouth of the Fallopian tube, which is situated at the bases of the fimbriae, and carry the discharged ovum through the ostium within the tube. The ostium abdominale is shut when the tube is examined after excision ; the closure is probably due to reflex contraction of the tube muscle, caused by handling and cutting. In the infundibular and ampullary segments of the tube, the mucous membrane is thrown into long plicated folds shown in section in Fig. 8. They are covered with ciliated epithelium, which urge the ovum towards the uterus. Between these folds, in the upper reach of the tube, the ovum, if it is to be fertilized, usually meets the male cell or spermatozoon, for we know that spermatozoa can remain alive in the tube for at least seven days after connection. The passage of the fertilized ovum along the tube takes place slowly for it undergoes its first developmental changes during this journey which is supposed to extend over a period of four or five days. If the passage of the fertilized ovum is obstructed, which may result from an inflammation or cicatrization of the epithelial lining of the tube, development may proceed at the point of obstruction.[4] When tubular pregnancy occurs, the growing ovum expands and ultimately perforates the tube — usually in the second month — an accident which is always attended by a grave haemorrhage.
The History of the Ovum within the Fallopian Tube
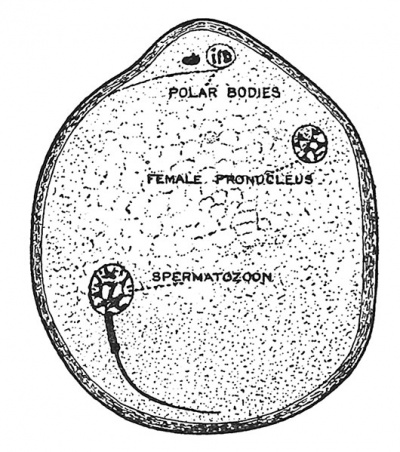
When the ovum enters the Fallopian tube, it is a cell of very considerable size (100/ji) with a cell wall — the zona radiata (Fig. 1), a nucleus — the germinal vesicle, and a nucleolus — the germinal spot. Then, or before then,[5] the ovum prepares for fertilization by the extrusion from its nucleus of first one, then a second polar body, and, with the extrusion, the germinal vesicle becomes the female pronucleus (Fig. 9). The polar bodies or polocytes, for they really represent cells, lie outside the protoplasm of the ovum, but within the zona radiata ; they are parts of the germinal vesicle which are extruded with all the display of karyokinesis — the peculiar changes manifested by the nucleus when a cell is about to divide. We shall see that the three polar bodies really represent three aborted ova — which have left their cell bodies to enrich the principal ovum.
Karyokinesis
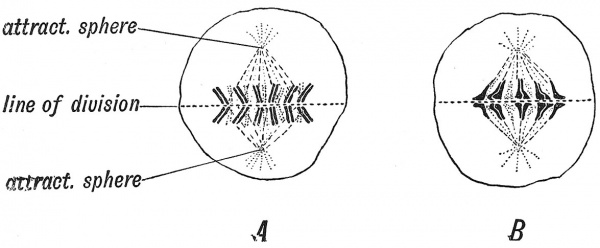
The preparatory or maturation changes which take place in the nucleus of the ovum and also in the nucleus of the male germinal cell are of the greatest interest to us, for we have good grounds for suspecting that the mechanism which regulates the shaping of the adult body is represented in the substance of the nucleus of the germ cell. The nucleus appears to be the chief vehicle of heredity — the medium by which the features of the parent are handed on to the child. Hence the importance attached by embryologists to the elaborate changes undergone by the nucleus of a maturing male or female germ-cell. When an ordinary cell of the body is about to divide, the nucleus undergoes certain changes before cleavage takes place. The nuclear division precedes that of the whole cell. This mode of cell division or cell propagation is known as Karyokinesis or Mitosis.[6] Two elements within the nucleus play a part in the process — the chromatin, which readily combines with certain staining reagents, and the acuromatin, which does not absorb dyes. In the resting phase, the chromatin is scattered as minute particles in the substance of the nucleus, but when mitosis is to take place the particles unite into filaments ; the filaments break up into segments or rods, each rod being known as a curomosome (Fig. 10). The number of chromosomes appearing in each somatic cell is approximately constant for each species of animal ; in man twenty-four is the usual number (Broman) . As the chromosomes form, an achromatin formation appears in the substance of the cell body just outside the nucleus — the centrosome, which appears to yield a commanding influence on the division of the nucleus. The centrosome divides ; the two halves move apart until they lie at opposite poles of the nucleus where each forms an attraction sphere (Fig. 10). The attraction spheres become joined by a spindle of achromatin threads, the chromosomes of the nucleus then appearing as if they were supported by the spindle between the attraction or centrospheres. The chromosomes move towards the equatorial plane of the nucleus — midway between the attraction spheres ; during the movement each chromosome divides longitudinally, so that each is split into two, the two halves lying side by side, often bent into V-shaped forms (Fig. 10, A) . As the nucleus divides in the equatorial plane 24 chromosomes pass into one half and 24 into the other. The attraction spheres fade away ; the division of the cell body is completed, each half having now its own nucleus ; the chromosomes break up in the network of the daughter nuclei and the two cells enter a resting phase. By this means an equitable distribution of the chromatin material of the parent nucleus is made to the two daughter cells.
Fig. 10, A. Diagram of Karyokinesis in a somatic cell (homotypical division). B. Diagram of Karyokinesis during the production of matured ova and spermatozoa (heterotypical division). (After J. B. Farmer.)
The two karyokinetic divisions undergone by the ovum before fertilization differ in three particulars from the process as seen in a somatic cell : (1) Only 12 chromosomes are formed — each being really double ; (2) the chromosomes are peculiar in shape and in manner of division (Fig. 10, B) ; (3) the cell body divides very unequally — only a very minor part accompanying that half of the nucleus which is separated at the first and second divisions of the ovum and which form, when thus separated, the first and second polar bodies or polocytes. A division of the first polar body accompanies the separation of the second polar body from the ovum, there being thus three polocytes formed during the maturation of the ovum. Thus the two divisions undergone by the ovum result in the formation of one matured ovum and three polocytes. Three-fourths of the chromatin in the nucleus of the original ovum are extruded in the polar bodies. The number of chromosomes in the ripe ovum has been reduced from 24 to 12.
The cells of a malignant tumour frequently show in their divisions the peculiar mitotic changes which are seen in the preparation of the female pronucleus. Prof. J. B. Farmer regards such cells as essentially germinal in character. It has also been found that the heterotypical or reducing form of mitosis may occur in leucocytes and in inflamed tissues.
Formation of Spermatozoa
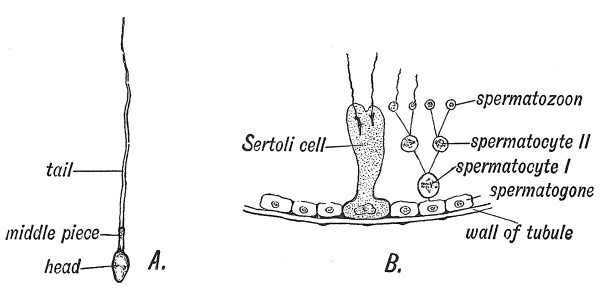
Having thus described the maturation of the ovum, and followed it within the Fallopian tube, it is necessary to trace the history of its counterpart in the male — the spermatozoon. The manner in which a spermatozoon is produced by a primary and secondary division from a spermatocyte is very similar to the production of a mature from an immature ovum. The form of mitosis is the same (heterotypical), the chromosomes being reduced to 12 in number and to a peculiar shape. The two divisions take place within the seminiferous tubules of the testis, and result in the production of four spermatozoa — corresponding to the matured ovum and three polar bodies (Fig. 11, B). The seminiferous tubules correspond to the ingrowths of germinal epithelium which carry the primordial ova within the ovary. Lining the tubules are two kinds of cells — those of Sertoli (Fig. 11, B), large cells for nourishing the spermatozoa — representing those of the stratum granulosum in the Graafian follicles — and other cells known as spermatogonia, corresponding to primordial ova. Spermatogonia divide and give rise to primary spermatocytes which correspond to immature ova. A primary spermatocyte divides into two secondary spermatocytes, each of which again divides and thus four cells are produced, which become modified into spermatozoa (Fig. 11, jB). The nucleus forms the head, the junctional part represents the centrosome,[7] while the tail is all that remains of the substance of the cell body (Fig. 11, A). While the ripe ovum has a diameter of 100µ (1/250th in.) the total length of a spermatozoon is only 50µ. While the ovum represents a large passive cell, laden with nourishment or yolk, its male counterpart becomes highly modified within the Sertoli or nurse cell, has its cell substance reduced to a minimum and is given a power of active motion. While ova are ripened singly, spermatozoa ripen by the million. Gemmill has shown that spermatozoa can be kept alive for many days in nutritive media ; probably the secretions of the epididymis, vesiculae seminales, prostate and Cowper's glands are for this purpose.
Fig. 11, A. Diagram of a Spermatozoon. B. Diagram showing the origin of Spermatozoa from the lining cells (spermatogonia) of the tubules of the testicle.
Fertilization
In the course of fecundation thousands of spermatozoa are lodged in the genital passage ; many stem the adverse current of the uterine cilia, reach and live for days within the interlaminar grooves in the wider parts of the tube.[8] In the course of its descent within one of the grooves the egg may be fertilized. The spermatozoon, attracted to the ovum by a force we do not yet understand, bursts through the zona radiata, loses its tail, its head enlarges, and forms the male pronucleus. The male and female pronuclei unite, and from their union springs the nucleus of the fertilized ovum. This is the centre from which all future developmental changes start. In the pronuclei, it will be remembered that the chromosomes were reduced to half the usual number ; by their union the full complement of twenty-four is again restored in the fertilized ovum. By the process of fertilization the characters of two human individuals are mingled. The mixed chromosomes of the nucleus of a fertilized ovum are laden with an assortment of the virtues and vices of both father and mother in a latent form. They transmit the characters of the race from one generation to another. The ovum may be, but rarely is, fertilized in the ovary, or between the ovary and ostium abdominale, the result being a pelvic gestation. The length of time the fertilized ovum takes to reach the uterus is not known exactly, but probably it spends from four to five days within the Fallopian tube. The musculature of the tube, as well as the action of the cilia, assist the fertilized and developing ovum in its progress to the cavity of the uterus.
Formation of the Embryo
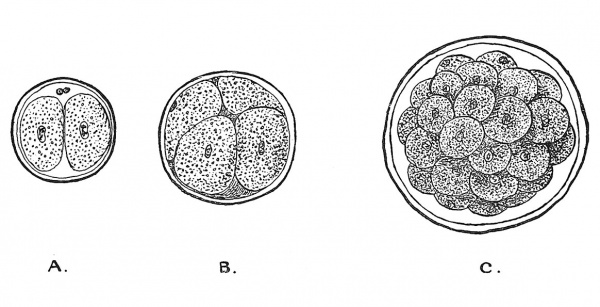
We are now to follow, step upon step, the changes which are to transform the fertilized ovum into a human embryo.[9] With the fusion of the male with the female pronucleus the ovum begins to divide, thus giving rise to the first brood of cells, two in number ; these in turn give rise to a second brood, four in number, and so on through successive stages, until a minute mass of cells replaces the ovum (Figs. 12 and 13) and thus a blastula or morula is formed — the first stage in the production of an embryo.
Fig. 12. Showing the production of the Blastula or Morula from the Ovum. The oolemma (zona radiata or egg-membrane) persists up to the morula stage, even later. A. The Ovum after the first division. B. After the second. C. The Blastula stage.
The production of the blastula takes place as the egg passes towards the cavity of the uterus, but before it has come into actual contact with the prepared lining membrane or decidua, it has entered a second and very important stage. A space or cavity appears within the blastula (Fig. 14) so that its cells become arranged in a definite manner. The cells which are going to give rise to the structures by which the embryo is to be nourished become arranged around the central cavity as a covering layer, while the cells which are to build up the embryo are enclosed within the covering layer (Fig. 14). In this manner the blastocyst is produced. At this stage, when the developing ovum is probably only half a millimetre in diameter (1/50 inch), it reaches the uterus. Its enveloping layer or trophoblast comes in contact with the decidua. In the blastocyst we recognize an embryogenic and a vegetative or yolk pole (Figs. 13 and 14). In Vertebrates with huge stores of yolk in their ova, such as birds have, the vesicle is filled by yolk-bearing cells, continuous with the enveloping layer at the vegetative pole, opposite to the inner cell mass.
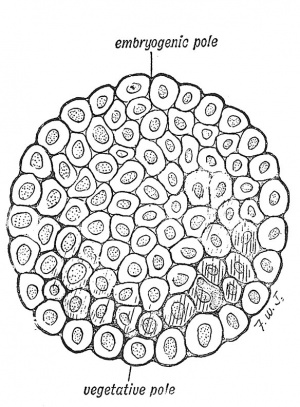
|
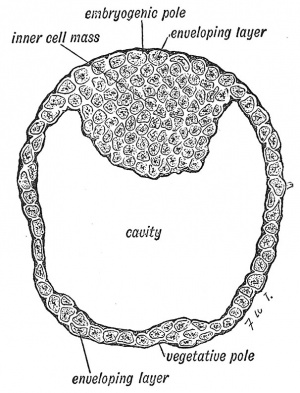
|
| Fig. 13. Stage I. The Blastula. | Fig. 14. Stage II. The Blastocyst. (After Van Beneden.) |
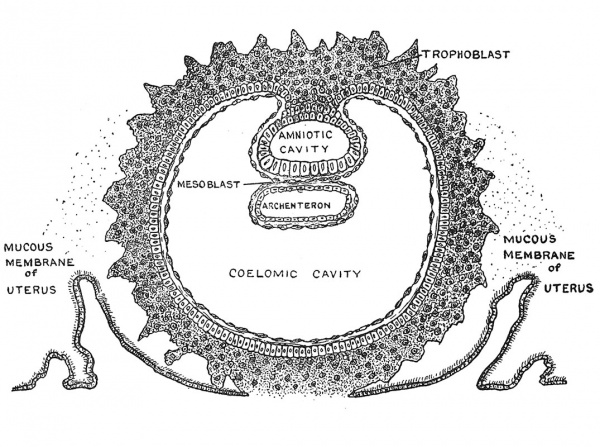
We now pass on to a further or third stage, concerning which our knowledge is as yet imperfect. By virtue of the phagocytic power oi its outer or trophoblastic layer, the blastocyst embeds itself in the decidual membrane of the uterus towards the end of the first week of development. In its earlier stages all developmental efforts are concentrated on the growth of the outer or trophoblastic layer which is to provide the embryo with nourishment ; hence the rapid expansion of the blastocyst and the multiplication and spread of the trophoblastic cells. Early in the second week a vesicular structure, measuring little more than a millimetre in diameter, has been produced (Fig. 15). The inner cell-mass shown in Fig. 14 has now become differentiated into three sets or systems (Fig. 15) : (1) a hypoblastic or entodermal set, grouped so as to form the wall of a minute vesicle — the fore shadow of the alimentary or archenteric system of the embryo ; (2) an epiblastic or ectodermal set, enclosing another minute fluid space — the cavity of the Amnion. We shall see that the ectodermal cells in the floor of this cavity, the side abutting on the archenteric vesicle, will go to the formation of the embryo, while the cells of the side and roof will form merely the lining of the amniotic cavity, within which the embryo will become developed ; (3) a third system — the mesoblastic or mesodermal — of cells has made a precocious appearance, surrounding the archenteric and mesodermic vesicles, lining the inner surface of the trophoblast and filling the space between the vesicles — the trophoblastic wall — with exceedingly fine fibrils (Fig. 15). These mesodermal cells are heralds of the great system out of which are to arise the blood and the vessels, muscle, bone, ligaments and all the connective tissue structures of the body. Thus in the second week, embedded within the decidua, the developing human blastocyst reaches a third stage — one in which the embryo is represented by two vesicular structures — the bivesicular blastocyst. The youngest human embryo of which we have accurate knowledge represents the terminal phase of this stage of development. This embryo was investigated and described by Teacher and Bryce in 1908.[10] The inner vesicles are still excessively small, the amniotic measuring only 0.5 mm. in diameter, while the archenteric is still less. On the other hand, the containing or trophoblastic vesicle is relatively large, measuring almost 2 mm. in its longest diameter and the trophoblastic cells are pressing outwards into the decidua by a process of most active growth. It is estimated that the Teacher and Bryce blastocyst is at the end of the second week of development. Towards the end of the second week or commencement of the third a very important change, reproduced in Fig. 16, carries the blastocyst on to a further or fourth stage of development. The coelomic cavity or space, the primitive representative of the peritoneal, pleural and pericardial cavities is produced by the cleavage or separation of the mesoderm into two layers. One layer — the inner — covers the archenteron, its wall being now made up of two strata — an inner of entoderm and an outer of mesoblast or mesoderm ; this double-layered wall is known as the splanchnopleure. The other layer of mesoderm — the outer, covers the outer surface of the amniotic cavity (Fig. 16) and the inner aspect of the trophoblastic wall. We shall see that the double-layered amniotic wall really represents the wall which encloses the abdominal and thoracic cavities ; the double stratum made up of epiblast or ectoderm and mesoderm or mesoblast is known as the somatopleure. The addition of mesoderm to all of these epithelial walls endows them with the power of forming blood vessels and blood. So far the growing blastocyst has depended for its sustenance on what the trophoblastic layer could absorb from the decidua but with the addition of mesoderm to the trophoblastic layer we have the outer or enveloping layer endowed with new and important properties. We shall see that the mesoderm on the archenteron and also that which lines the trophoblastic wall are the first to produce blood vessels and blood. With the addition of mesoderm to the trophoblastic wall we apply a new name to the outer or enveloping wall — namely the chorion. Through the chorion the embryo is to draw its oxygen and nourishment from the mother and get rid of its carbon dioxide and waste products.
Fig. 15. The Blasto-dermic Stage.
Fig. 16. Showing the Origin of the Primitive Coelom, the Mesoblast and Cavity of the Amnion during the Development of the Human Ovum. (After T. H. Bryce.)
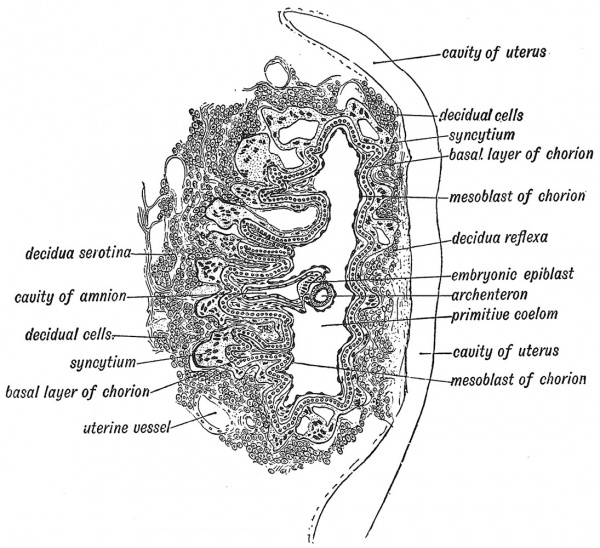
In 1899 Dr. Peters gave a full and clear description of an embryo at this stage of development. The whole blastocyst was as yet of small size — only 1.6 mm. (1/16 inch) on its longest diameter (Fig. 17). The enveloping epiblast and its lining of mesoblast now form a distinct but non- vascular chorion. The archenteric vesicle is still of minute dimensions (Fig. 17). The amniotic cavity, formed within the enclosed ectoderm is larger, and the cells lining it have become differentiated into two kinds (Fig. 17). An area of columnar cells, forming the floor plate of the cavity, produces ultimately the epithelial covering of the body, and all the cells and fibres of the nervous system. The flatter cells which line the dome of the cavity will form the epithelial lining of the Amnion ; the outer layer of mesoderm afiords a covering to the amniotic ectoderm (Fig. 17). Fluid collects within the cavity of the amnion ; floating in the fluid, the human embryo will develop. Thus the delicate embryonic tissues, being equally supported on all sides by the amniotic fluid, may pursue their developmental courses, unhindered by the influence of gravity, and uninjured by the pressure, to which the uterus within the abdomen is subjected by the movements of respiration or bending of the trunk. If the fluid is deficient or absent then many forms of malformation may result.
It is in this stage (Stage IV.) that it becomes possible to detect the foundation or Anlage of the embryo. It is represented by the plate or lamina of tissues which separates the cavity of the archenteron from the cavity of the amnion (Fig. 17). The growth of the embryo remains in abeyance ; all the developmental energy is thrown into the upbuilding and expansion of the enveloping epiblast or Trophoblast as Hubrecht named it in 1889, for he recognized that its chief function was to provide the embryo with the means of nourishment (τροφδς, a feeder). Thus in the earlier stages of development the actual embryo remains in abeyance, while the tissues which protect it and nourish it grow and develop with exceeding rapidity. Already, in Stage IV., it is seen that the epithelium forming the trophoblast has become differentiated into (a) a Basal Layer (Langhan's cells), (b) masses of cells, which have undergone multiplication without separation ; this formation is known as Syncytium (Fig. 17). The syncytium is chiefly developed on that aspect of the developing ovum which is directly in contact with the wall of the uterus. It is remarkable not only for the imperfect -separation of its cells, due probably to the rapidity of its growth, but also for the extraordinary phagocytic power it exercises on the mucous membrane of the uterus. Processes of the syncytium burrow within the thickened and vascular mucous membrane in a systematic and regulated manner ; they absorb the tissue with which they come in contact, and lay open blood vessels of the mucous membrane. The maternal blood escapes into spaces enclosed by the syncytial processes or into lacunae formed by the vacuolation of processes. In certain circumstances syncytial cells escape into the general circulation and form malignant growths. If the developing ovum be arrested in the Fallopian tube the syncytium, owing to the extreme thinness of the lining membrane, quickly eats its way into and through the wall of the tube.
Fig. 17. Stage IV. Section through the bivesicular blastocyst embedded in the wall of the Uterus. (Modified by F. W. Jones from figures given by Peters and Selenka.)
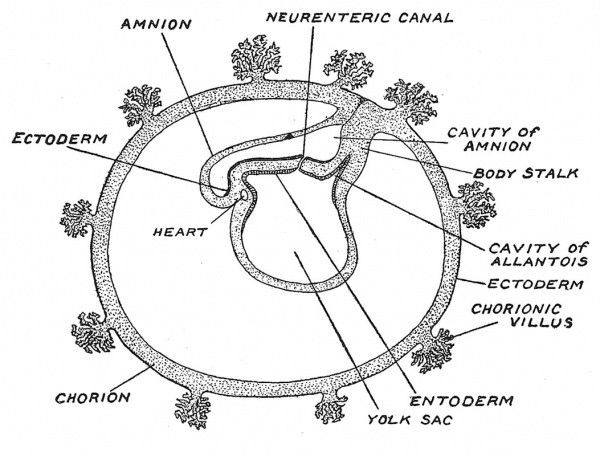
Fig. 18. Stage V. Diagrammatic Section of a human pregnancy towards the end of the 3rd week of development, showing its demarcation into embryo and membranes. (After Graf Spee.)
In Fig. 18 there is given, in a diagrammatic form, the stage of development reached about the end of the third week. A very rapid growth sets in during this week ; the chorionic vesicle which at Stage IV. measured only 2 mm. in its longest diameter, has become five times that length — an object easily visible to the naked eye. Villi grow out from it, at first simple and then branched ; blood spaces filled by maternal blood are formed between the villi. In the villi, blood vessels and blood are being formed but a circulation is not yet established. The embryo is now definitely represented by a plate or shield — the embryonic plate, composed of three layers of tissues — an upper or ectodermal derived from the floor of the amniotic vesicle ; a lower or endodermal formed by the roof of the archenteric vesicle and an intermediate formed by mesoderm or mesoblast. On the upper surface of the flat embryonic plate, which has a total length of about 1-5 mm., appears on its hinder or caudal half, the primitive streak ; at the anterior end of the streak a perforation is formed — the neurenteric canal which places the amniotic cavity in communication with the archenteric vesicle (Fig. 18). The nature of the primitive streak and of the neurenteric canal we shall discuss later (see p. 38). The archenteric vesicle has also undergone a rapid growth, now measuring 2 mm. in diameter and we can recognize in it (see Fig. 18) the beginning of a division into two parts, the yolk sac — which contains a stock of nourishment and will come to lie outside the embryo and a part which remains applied to the embryonic plate and will form the alimentary canal system. The part which will come to lie within the embryo already shows a division into three parts — a forward diverticulum — the rudiment of the foregut, a posterior diverticulum — the rudiment of the hind gut and an outgrowth from the hind gut — which represents the structure known as the allantois. The embryonic plate, with the amniotic and archenteric vesicles, is bound to the chorion by the body-stalk (Fig. 18) — the rudiment of the umbilical cord. Thus towards the close of the third week human pregnancy is represented by (1) an embryonic plate, (2) a yolk sac, (3) amnion, (4) body-stalk, (5) chorion.[11]
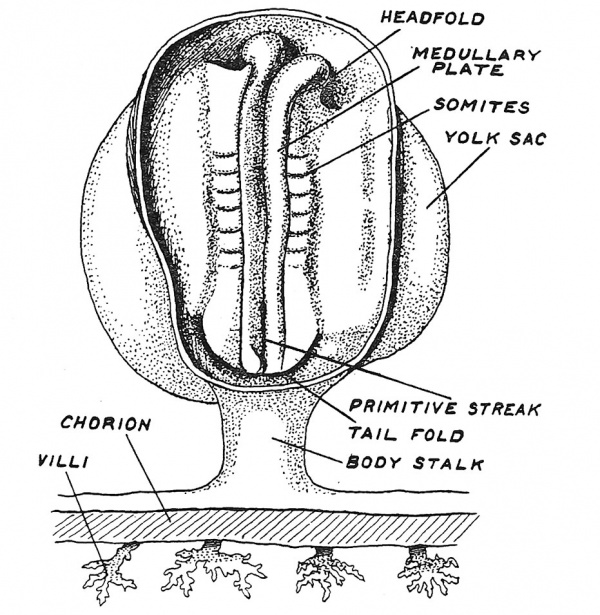
We shall now concentrate our attention on the growth of the embryo which from the end of the third week to the end of the fifth undergoes a very rapid transformation. The changes to be described follow very rapidly and constitute a sixth stage. About the end of the third week two folds — the medullary folds — begin to rise up along the head end of the embryonic plate (Fig. 19), thus enclosing the neural plates from which the brain and spinal cord are to be developed. The rising up of the medullary folds is accompanied by the appearance of another very important developmental process — the cleavage or segmentation of the mesoderm on each side of the medullary folds into segments or somites. Segmentation commences in the posterior region of the head and spreads backwards. Thus the head region of the embryo is the first to be differentiated and we have every reason to suppose that the segments at the cranial end are the oldest in an evolutionary sense. With the appearance of somites the human embryo manifests its vertebrate character.
Fig. 19. The formation of the medullary folds and somites on the embryonic plate. From Prof. Pfannenstiel's model of an embryo measuring 1.95 mm. in length.
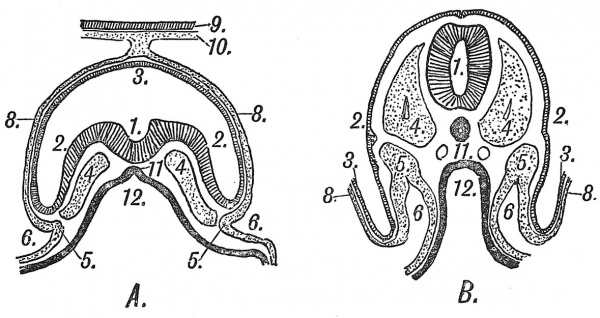
The human embryo reconstructed from serial sections by Professor Thompson in 1907 shows the great advance made during the fourth week[12] (Fig. 20). The medullary folds have fused along their crests and enclosed the neutral plates to form a canal or tube, the process of segmentation is spreading rapidly backwards, the head and gill arches can now be recognized and although the embryo measures as yet less than 3 mm. in length, the main parts of the adult body, saving the limbs, are clearly foreshadowed. We have reached what may be termed a seventh stage in the development of the human embryo. We must now turn to some of the chief internal changes which have been taking place, and this can best be done by comparing sections across the flat embryonic plate of a pregnancy in the third week of development (Stage V.) with a section of one such as made by Professor Thompson which has reached Stage VII. (see Fig. 21, A, B). When such sections are compared the following changes will be noted : (1) A narrow plate of modified ectoderm or epiblast, stretching along what will be the median dorsal line of the body, becomes depressed, thus forming the floor of a groove ; the lateral margins of the groove rise up, meet together and fuse along the middle line. Out of the neural tube thus enclosed are developed the brain and spinal cord. (2) In a somewhat similar manner a strip of cells along the median dorsal wall of the archenteron is separated as a tube to form the notochord (Fig. 21, A, B). Round the notochord are developed the spinal column and the greater part of the base of the skull. (3) Indications are to be seen of a separation of the archenteron into an intra-embryonic part, which will form the alimentary tract, and an extra-embryonic part, which becomes the yolk sac. (4) The mesoderm on each side of the embryo shows a division into four parts : (a) paraxial mesoderm (4, Fig. 21) from which the voluntary musculature, as well as other parts of the body system arise ; (6) intermediate cell mass (5, Fig. 21), in which the renal and genital organs are developed ; (c) somatic mesoderm, this layer with the ectoderm over it forms the somatopleure, the outer wall of the coelom ; from the coelom are developed the pericardium, pleura and peritoneum ; [d) the splanchnic mesoderm, which covers the intestine and yolk sac ; the splanchnic mesoderm and entoderm together form the splanchnopleure. (5) Indications can be seen of the division of the coelom into intra- and extra-embryonic parts (6, Fig. 21).
Fig. 20. Human Embryo 2.5 mm long towards the end of the fourth week of development. (Professor Peter Thompson.)
Fig. 21. Schematic Transverse Sections of two Human Embryos. A, In the 3rd. week of development. B, In the 4th week of development. The numbers are placed on corresponding points : Epiblast, shaded ; hypoblast, black ; mesoblast, stippled.
| 1. Neural groove and canal.
2. Epiblast of embryo. 3. Epiblast lining amnion. Only the attachment of the amnion is represented in B. 4. Paraxial mesoblast. 5. Intermediate cell mass. |
6. Coelom, bounded by the somatopleure externally and splanchnopleure internally.
8. Mesoblast on amnion. 9, 10. Chorion. 11. Notochord. 12. Archenteron. |
When the umbilicus contracts and closes, these two parts of the coelom are finally separated. It is also during the fourth week that the paraxial mesoderm becomes separated into primitive segments, or somites, formerly known as protovertebrae. In the embryo shown in Fig. 20 twenty-three pairs are already separated.
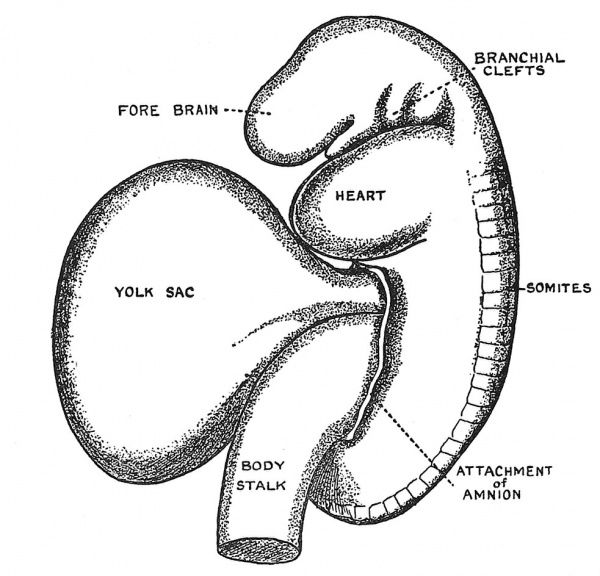
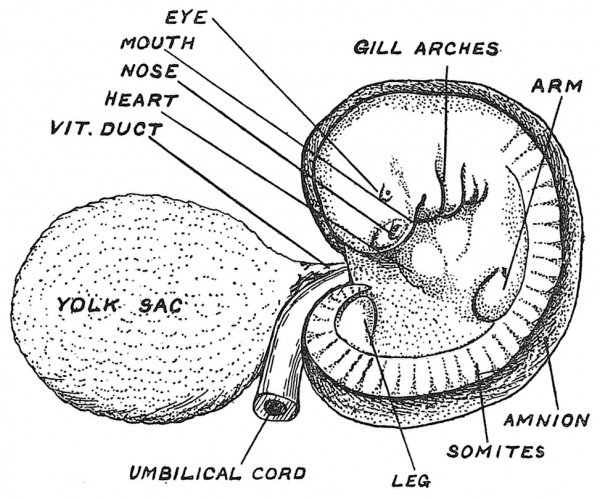
In Fig. 22 is represented the fully differentiated human embryo — a stage of development — which we may call the eighth stage, reached about the end of the fifth week. From crown to rump the embryo about this time measures 5 mm. — one-fifth of an inch. The buds of the upper and lower extremities have now appeared ; segmentation has reached almost to the tip of the tail, there being three occipital and 35 body somites, the last representing the sixth coccygeal or caudal. The mouth is becoming apparent ; so are the eye and nose ; the gill arches, four of which are apparent in the neck, have reached their highest development ; a blood circulation is now fully established, the body stalk having been transformed into the umbilical cord. The yolk sac is now joined to the embryonic gut by a long narrow duct — the vitello-intestinal duct (Fig. 22).
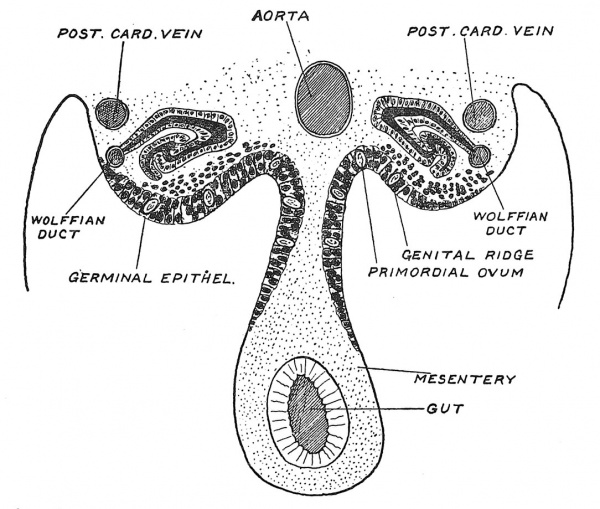
Origin of Ova and Spermatozoa. — Towards the end of the stage just described, the genital ridges arise from the intermediate cell mass and project into the coelom, one at each side of the root of the mesentery (Fig. 23). The mesothelial cells which line the coelom assume a columnar form at the root of the mesentery and over the genital ridges ; between these cells appear primitive germ-cells (primordial ova) characterized by their large size and reaction to certain stains. Hitherto it has been assumed that the germ-cells arose from the mesothelial columnar cells which cover the ridge. Beard, during a prolonged and accurate investigation of the development of fishes, especially of the skate, discovered that the germinal cells were not formed in the genital ridges but appeared at a very early stage corresponding to that described here as Stage I. When the coelom is formed they migrate towards the genital ridges. There is nothing strange in such a migration for it is a daily occurrence in the adult body for leucocytes to be drawn in crowds to a site of infection by an obscure force which is named chemotactic. It is, then, far from unlikely that the primitive germ-cells are separated at an early stage in the division of the ovum and then subsequently seek a nidus in the genital ridge. We shall see that nerve cells migrate under similar influences. We may suspect tiaat the germ-cells which fail to reach the suitable nidus, which the genital ridges afford, are absorbed, or, as Beard has suggested, they may give rise to those curious tumours known as teratomata. The manner in which the primitive germ-cells are carried within the genital ridge by tubular incursions of the mesothelium covering the ridge has been already described, but we are ignorant of the circumstances which determine the production of spermatozoa and a testicle in one individual, and the formation of ova and an ovary in another. It is not until the embryo has attained a length of 15 mm. in the seventh week of development that it is possible to distinguish testicle from ovary.
Fig. 22. Showing a human embryo, 5 mm. in length, at the end of the 5th week of development. (After Keibel and Mall.)
Fig. 23. Diagrammatic Section of the roof of the Coelomic cavity of a human embryo in the fifth week of development, showing the position of the Genital Ridges in which the Ovary or Testicle is formed.
Thus in the space of five weeks the cycle which produces new human seed from the old is accomplished and all the parts of a new human body are laid down in outline. In these five weeks the fertilized ovum has given rise to (1) germ-cells which are endowed with the combined properties of the ovum and spermatozoon from which they were produced ; (2) an embryo in which these cells are nourished and protected ; (3) membranes by which the embryo is protected and nourished during intrauterine life.
Having thus followed the chief developmental changes of the ovum, and seen how the embryo, the membranes and the reproductive cells are differentiated, we shall review in the next chapter the manner in which the ovum establishes itself in the cavity of the uterus and, for the space of nine months, passes a parasitic life there.
- ↑ Measurements are given at first according to our English standard, but throughout this book the more convenient metric system vnW be employed. One inch=25.4 mm. One millimetre = 1000µ, or micromillimetres, or mikrons.
- ↑ 2.0 2.1 For fuller details and literature see Francis H. A. Marshall, The Physiology of Reproduction, London, 1910. For more recent investigations on the maturation of Graafian follicles in Man see Prof. Arthur Thomson, Journ. of Anat. 1919, vol. 53, p. 172, vol. 54, p. 1. See also Prof. Arthur Robinson, Trans. Roy. Soc. Edin. 1918, vol. 52, p. 303.
- ↑ Online editor - We now know that this is incorrect and the germinal epithelium is a misnomer, as it is not the source of the ova, these migrate into the ovary as primordial germ cells.,
- ↑ See F. P. Mall, Surg. Gynaec. and Obstet. 1915, vol. 21, p. 289.
- ↑ Prof. Arthur Thomson has shown that ova are to be seen in the human ovary with both polar bodies already extruded and that maturation changes can be seen to take place before the ova are shed, Journ. Anat. 1919, vol. 53, p. 172.
- ↑ For literature and significance of Mitosis see C. E. Walker, Essentials of Cytology, London, 1907 ; Bashford and Murray, " Significance of Mitosis," Proc. Roy. Soc. 1904, vol. 73, p. 66 ; R. Fick, Ergebnisse der Anat. 1906, vol. 16, p. 1 ; S. Tschassownikow, Anat. Hefte, 1911, vol. 45, p. 197 ; Prof. W. E. Agar, Cytology, 1920 ; Prof. F. R. Lillie, Fertilization of the Ovum, 1919.
- ↑ In the sheath of the middle piece is also included an element scattered through the substance of the cell body of the parent germinal cell — the element known as mitochondria or chondriosomes (see J. Duesberg, Biol. Bulletin, 1919, vol. 36, p. 71 ; E. V. Cowdry, Contributions to Embryology, 1918, vol. 8, p. 41.
- ↑ For literature on fate of spermatozoa in the uterus see J. H. F. Kohlbrugge, Boux's Archives, 1912, vol. 35, p. 1.
- ↑ For literature on early stages in the formation of the mammalian blastula see J. P. Hill, Quart. Journ. Mic. 8c. 1911, vol. 56, p. 1 ; 1918, vol. 63, p. 91.
- ↑ For literature on very early human embryos see T. H. Bryce and J. H. Teacher, Contributions to the Study of the Early Development and Embedding of the Human Ovum, Glasgow, 1908 ; the more recent literature and data are given by Dr. Geo. L. Streeter, Contributions to Embryology, 1920, vol. 9, p. 389.
- ↑ For recent literature on embryos at Stasje V. see Geo. L. Streeter, Contributions to Embryology, 1920, vol. 9, p. 389; N. W. Ingalls, ditto, 1918, vol. 7, p. Ill ; H. Triepel, Anat. Hefte, 1916, vol. 54, p. 149.
- ↑ For description of embryos in the 4th week of development, and for references to other descriptions see P. Thompson, Journ. Anat. and Physiol. 1907, vol. 41, p. 159 ; F. Wood Jones, Proc. Anat. Soc. Grt. Brit. June, 1903 ; A. Low, Journ. Anat. and Physiol. 1908, vol. 42, p. 237 ; W. E. Dandy, Amer. Journ. Anat. 1910, vol. 10, p. 85 ; Sussana P. Gage, Amer. Journ. Anat. 1905, vol. 4, p. 409. For the most recent descriptions : see Prof. D. Waterston, Journ. of Anat. 1915, vol. 49, p. 92 ; J. Crawford Watt, Contributions to Embryology, 1915, vol. 2, p. 5.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Human Embryology and Morphology: 1 Early Ovum and Embryo | 2 Connection between Foetus and Uterus | 3 Primitive Streak Notochord and Somites | 4 Age Changes | 5 Spinal Column and Back | 6 Body Segmentation | 7 Spinal Cord | 8 Mid- and Hind-Brains | 9 Fore-Brain | 10 Fore-Brain Cerebral Vesicles | 11 Cranium | 12 Face | 13 Teeth and Mastication | 14 Nasal and Olfactory | 15 Sense OF Sight | 16 Hearing | 17 Pharynx and Neck | 18 Tongue, Thyroid and Pharynx | 19 Organs of Digestion | 20 Circulatory System | 21 Circulatory System (continued) | 22 Respiratory System | 23 Urogenital System | 24 Urogenital System (Continued) | 25 Body Wall and Pelvic Floor | 26 Limb Buds | 27 Limbs | 28 Skin and Appendages | Figures