2014 Group Project 8
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Musculoskeletal
This webpage will be focusing on fetal muscular development.
Introduction
The musculoskeletal systems main purpose is to provide the body with structure, stability, support, protection, mineral storage, heat production and movement. It is made up of multiple structures; bone, cartilage, skeletal, tendon, ligaments and joints. This project will be focusing on the fetal development of the Muscular and Tendinous tissue. Both tissue have major embryonic contributions from the somites and during the fetal period undergo myogenesis and fibrillogenesis respectively to form mature tissue. Many molecules, particularly growth factors and proteoglycans regulate the growth of the muscle and tendons. Historical and current research and models in musculoskeletal development will be addressed, with addition of common muscular congenital fetal abnormalities.
Background Early Embryonic development
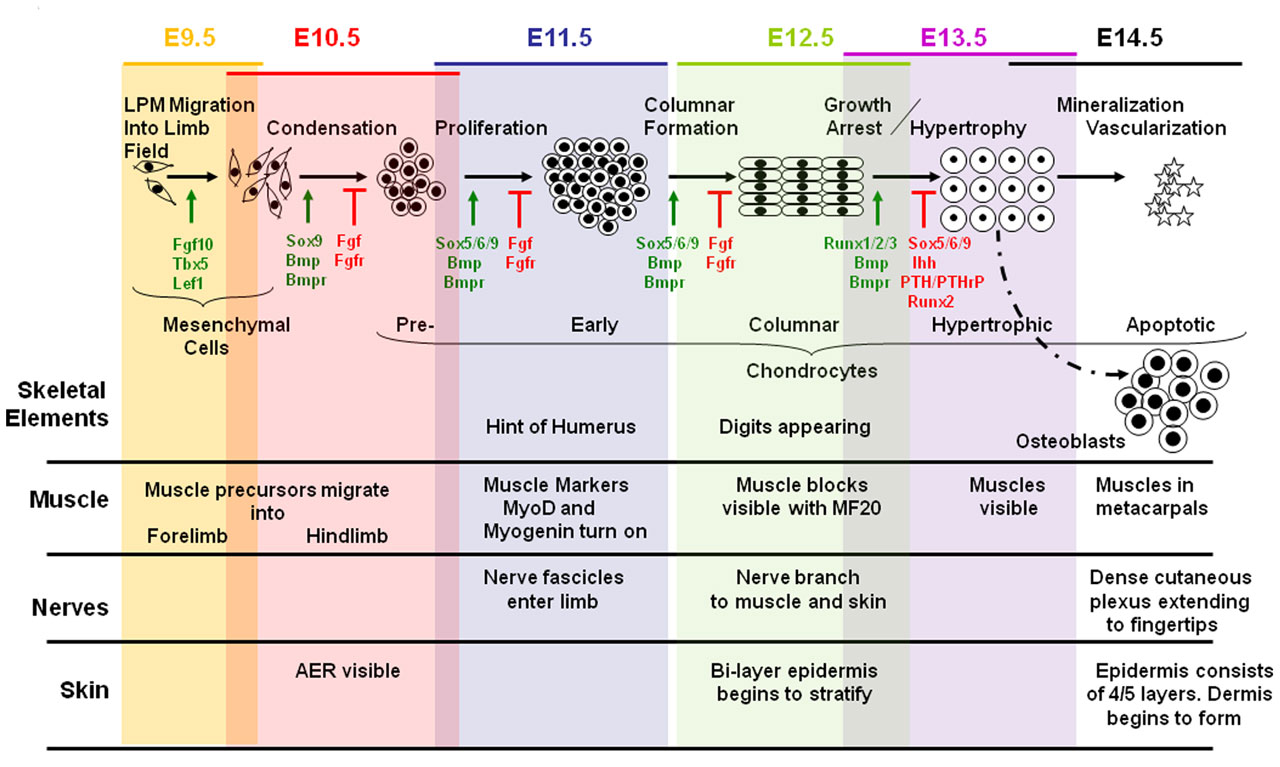
Skeletal Muscle develops from a process known as myogenesis. Mesenchymal cells which are embryonic connective tissue cels differentiates into embryonic muscle cells, myoblasts. Myoblasts which have single nuclei fuse and elongate to form myotubes which are multinucleated and cylindrical.
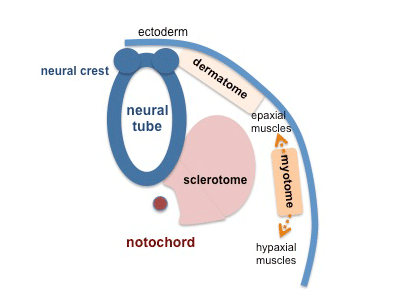
The neural tube and notochord release signaling molecules like Shh, Wnts and [BMP]-4. These signaling molecules act on transcription factors of the MyoD family and Pax [1].The MyoD or MrF family includes MyoD, Myf-5 , myogenin and Myrf4 [2]. The MyoD family of transcription factors, like MyoD are myogenic bHLH (basic helix loop helix) transcription factors. Pax-3 and the MyoD induce myogenesis, formation of myoblasts. Pax-3 also acts on c-met which is a migratory peptide.
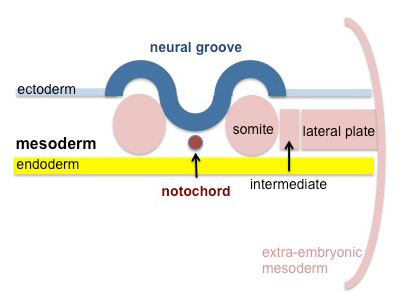
Skeletal muscle is derived from the somites. Paraxial mesoderm segments into somite structures on both sides of the notochord and neural tube.
Somites are mesodermal structures where the dorsal most end of the somite, which is known as the dermomyotome, becomes skeletal muscle and dermis [3]. A small anterior portion of the paraxial mesoderm remains un-segmented and eventually forms some muscles of the head [4].
The myotome lies in-between the scleratome which is ventrally located. The scleratome forms the cartilage and bone of the axial skeleton of the embryo. The dermomyotome is located dorsally and forms the first skeletal muscle in the embryo.
The medial part of the dermomyotome forms the dorsal and intecostal muscles whilst the lateral part of the dermomyotome forms the limb and ventral muscles [3].
As nearly all of the muscular system develops from the mesoderm. The Iris muscle comes from neuroectoderm. And Eosophagus skeletal muscle is derived from transdifferentiation of smooth muscle.
Myogenesis occurs in two phases; primary and secondary which occur in embryonic and fetal periods Primary myotubes express MHC slow myosin heavy chains [5]. The primary myotubes form the structure and scaffold upon which secondary myotubes form during secondary myogenesis which occurs in the fetal period [5].
Molecular and Cellular regulation of fetal myogenesis
Skeletal myofiber number is set at birth [6]. This was found using experiments on mice and pigs. Similar trends have been observed in humans as found by Widdowson et al (1972) where a huge increase and then levelling of gastrocnemius myofibers were found in the gestational period [7].
Multipotent mesenchymal cells (MSC) form myoblasts as well as adipocytes and fibroblasts [8]. Therefore specific the nutrients and growth are important in directing the growth of these MSC into either adipocytes or myoblasts [9]. Myoblasts differentiate to form the embryonic, fetal and adult skeletal muscle and in fact myoblasts differentiate to form all three early in development.
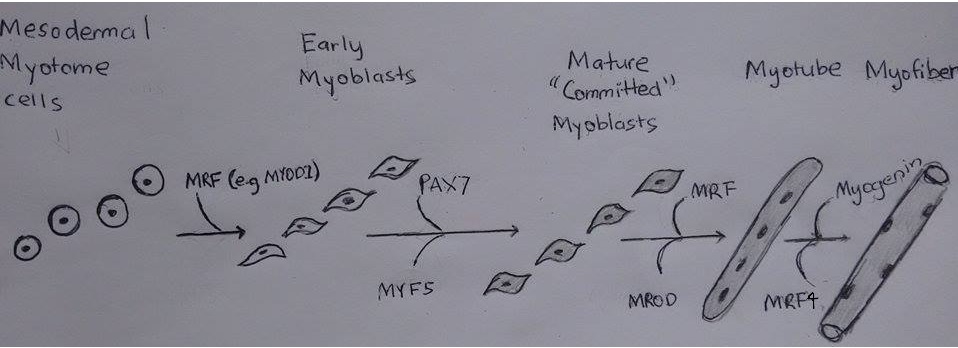
As discussed previously the mesoderm forms somites which divide into the dermomyotome, scleratome and myotome. The myotome becomes the skeletal muscle. Myotome cells migrate to different positions, for example limb buds. Once myotome cells travel to a target destination. Mrf like MYOD1 allow for the transdifferentiation of mesodermal myotome cells to the immature muscle cells, myoblasts.
Then the myoblasts proliferate under the influence of growth factors up until the neonatal period. Following this there is a decrease of growth factors which results in the myoblasts stop proliferating and align. The membranes of the myoblasts fuse together and become myoblasts.
Approximately 100 or more myotubes form a muscle fiber. Note that myoblasts still continually are fusing to this growing mass of muscle cells.Primary myotubes form the scaffolding for which the fetal myoblasts will differentiate into secondary myotubes and add too [10].
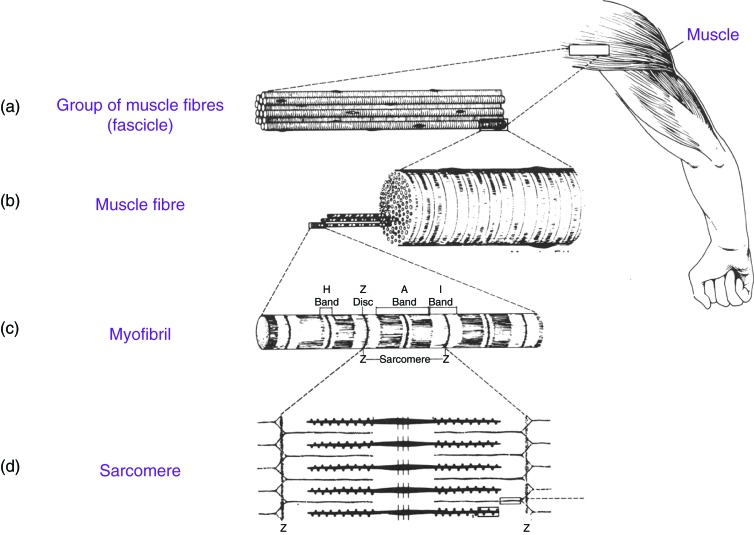
When myoblasts fuse and become myotubes there are number of changes that occur. In the cytoplasm of myotubes features of striated skeletal muscle develops like myofilaments and myofibrils. Myofibrils contain thick myosin and thin actin proteins which repeat along the myofibril to form repeating units known as sarcomeres. The sliding of myosin and actin allows for the contraction of muscle. In sarcomeres the nuclei are pushed to the side.
What is known as primary myofibre forms when after growth factors attract the nerve and it attaches to the bed of muscle. The nerve gives of branches to secondary myofibres that form later.
| Muscle Myogenesis Movie |
|---|
| <html5media height="400" width="400">File:MYOGENESIS_video.mp4</html5media> |
External laminae develops for the myotubes and separates the myotubes from surrounding mesenchymal tissue. Sheaths containing the myofibers develop; endomysium, epimysium and perimysium. External laminae and reticular fibers form the endomysium. Perimysium and epimysium layers are the formed by fibroblasts.
There are a greater deal of secondary myofibers than primary and are receptive to nutrients and growth factors. Muscle regulatory factors (MRFs) control the proliferation of secondary myoblasts. MRF’s are helix-loop-helix transcription factors.
| Cell type/Stage | Associated Molecules | Action |
|---|---|---|
| Mesodermal Myotome cells | MYOD1 | Allow for transdifferentiation into myoblast |
| Early Myoblasts | PAX7 , MYF5 | Act on CDK4 and Cyclin D1 to dephosphorylate Rb and induce cell proliferation [11] [12] [13] |
| Mature ‘Committed’ Myoblasts | MRF, MROD | MYOD acts on myostatin to take myoblast out of cell cycle and ready for differentiation [11] |
| Myotubes | Myogenin, MRF4 | Inhibit cell cycle and proliferation by acting on regulatory proteins like P21 to stop the cell cycle allowing for the conversion of myotube to myofiber. [11] [14] |
Following the formation of myofibers, growth factors, amino acids and stretch/load activity act on myofibers to affect subsequent hypertrophy maturation of myofibers [11]
Myofiber hypertrophy is occurs when protein synthesis is greater than protein degradation. The resulting accumulation of protein results in hypertrophy. Therefore maintaining protein levels is important in the hypertrophy of myofibers. This is regulated by nutrients and growth factors (GFs). Growth factors There are a large number of growth factors that affect fetal myogenesis. Some of the main GFs as Brown (2014) describes in her article “IGF1, insulin, basic fibroblast growth factor (bFGF), transforming growth factor-β (TGF- β)” [11] [15] [16] Experiments have shown that if IGF1 is removed reduced muscle mass and hypoplasia has resulted. And increased IFG1 expression has resulted in hyperplasia and increased skeletal muscle. IFG1 has also been found to enhance protein synthesis. [11][17][18] Insulin similarly has been found to promote fetal muscle growth and protein synthesis. Basic fibroblast growth factor (bFGF) and transforming growth factor-β (TGF- β) induce proliferation and myogenesis by upregulating cyclin D.
Nutrients Being the building block of proteins, amino acids are important in muscle protein synthesis. But they are more important in adult muscle protein synthesis than fetal muscle protein synthesis. Experiments have shown for example an amino acid infusion did not always result in a fetal muscle growth, only when there was a rise in the insulin levels. And little is known of the interaction of amino acids and growth factors in the context of fetal myogenesis. Research has shown that fetal or secondary myofibers are more prone to suffer from nutrient deficiency than primary myofibers in pigs and sheep. [19][20] [21]
Fetal myofiber number have been observed to decrease with detal nutrient deficiency. This is concerning as myofiber numbers are set at birth. Nutrients are also important for fetal myofiber hypertrophy. Stretch and loading Stretch and loading also affect hypertrophy. [22]
Tendon Development
Tendons are connective tissue which join muscle and bone allowing the transmission of force. They organised fibrils which form fibers, which along with fibroblasts are surrounded by connective tissue to form fascicles[23]. Tendons primary embryonic structure originate from mesenchymal progenitor somite cells with further contributions of Neural Crest and Lateral plate mesoderm.
Appearance of tendons begins in the 20th Carnegie stage and marks the beginning of fibrillogenesis. This process is initiated by fibroblasts in series of extracellular compartments; they enlarge the cells domain into extracellular space. Channels deep in the cytoplasm drive the process of elongation, these channels location is associated with Golgi bodies. [24]
First type of compartments are formed by collagen containing secretory vacuoles which fuse with surrounding cell membranes. Initial fibrillogenesis is mediated my macromolecular interactions based on vacuole content, with a lesser input from receptor membrane interactions. Fibril groups as fibres close to the cell surface and Secondary extracellular compartments form; at this stage fibroblast are arranged adjacently[24] . Third level of compartmentalization forms later when fibroblast are adjacent with 2 or more other fibroblasts. As the tendon matures fibres coalesce invading each other with interdigitating processes. Secretory vacuole persist, laterally aggregating with further growth preserving sites for fibril deposit. [25] Collagen fiber assembly branch creating fibre networks along fascicles. Fibres branch within tendon fascicle
The main regulatory factors of tendon fibrillogenesis are Leucine-rich repeat proteoglycans.
| Non-fibrillar components | Molecular Characteristics | Function |
|---|---|---|
| Fibromodulin | Keratin sulphate, proteoglycan | Fibromodulin modulates the site-specific cross-linking ultrastructure of collagen, ensuring mechanical strength. Control the pattern of lysyl oxidase-mediated collagen cross-linking by reducing access of the enzyme to telopeptides, by binding to the collagen. [26] |
| Decorin | Chondoirin sulphate, proteoglycan | Regulates expression of multiple leucine-rich proteoglycansins(SLRP) during tendon fibrillogenesis, via Class I and II Small(SLRP). Competes with Biglycan for binding sights on collagen types I-VI . Concentration increases as fetal development continues. [27] |
| Biglycan | Chrondroitin sulphate and Dermatan sulphate, proteoglycan | Process of regulation closely mimic Decorin though concentration is maximal expression at day 16-18 during embryonic development, reducing after this point. Competes with Decorin for bonding sights on collagen I-VI. [28] |
Historical findings
Predating 1970 the adult musculoskeletal system was heavily researched which provided some overlap with prenatal development, though few studies existed which focused on the fetal stages. During the 70's a majority of research in the field focused on the histologist differences and development of differing fiber types. Research on morphological development was very limited with eh exception of two Czechoslovakian studies in the late 1980's which displayed muscle formation for multiple primordia and sexual differentiation.
The differing developments of alpha and beta fibers was revealed in a study by University of California published in 1972 using lamb fetuses as an experimental model. Beta muscle fibers are formed during the first stages of fusion, the individual Beta fibers create a network for the alpha fibers to develop on. Red muscle fasciculi are formed by merging of small fiber bundles. White muscles are formed by ongoing addition of alpha fibers. Additionally it was concluded that fetal muscle contraction didn't significantly effect number of fibers present. Most fibers had already been formed by the 20th week, during this period the limit muscle contractions result in little mechanical tension.
Gluteus Maximus muscle Morphogenesis
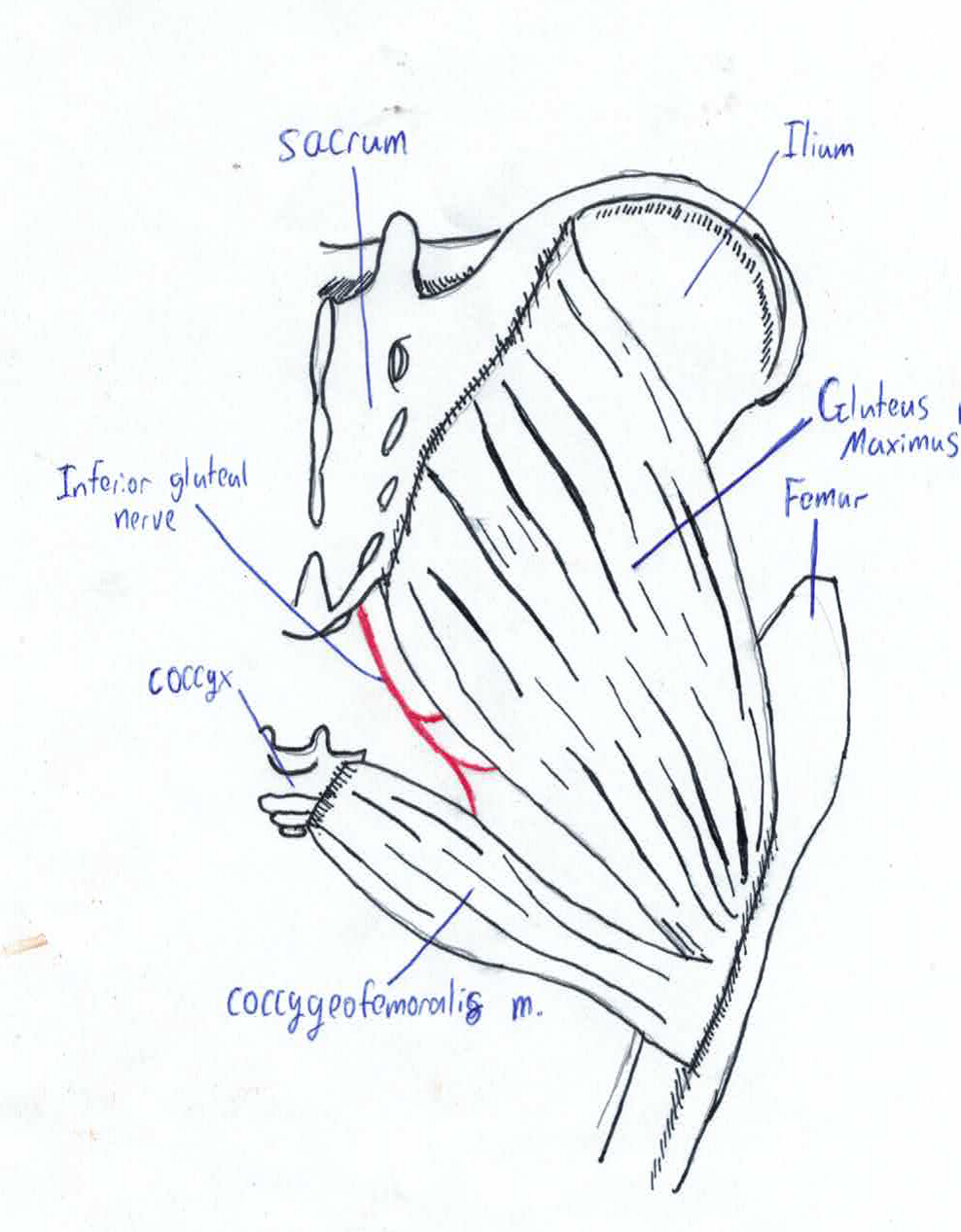
A 1985 Czechoslovakian study investigated the development of Gluteus Maximus during the embryonic and fetal periods. Pelvic micro-dissection of human embryos and foetuses with crown length varying from 22-215mm were compared with newborns and adults. The presence of a muscle not present in post-natal adults[29] was discovered called the coccygeofemoralis or pars coccygea; this muscle originates from sides of coccyx and inserts onto the gluteal tuberosity. In the adult human this muscle fuses with the larger pars sacroiliac or fetal gluteus maximus to create the adult gluteus maximus. Pars sacroiliac originates from ilium and sacrum and inserts onto the gluteal tuberosity[30].
In fetus with crown length of less than 45 mm the two muscle primordia are separated by small amount of loose connective tissue. This point onwards the muscles become fused by a small furrow which persists until 215mm crown length. By the time of birth the furrow is absence and the muscles are entirely fused [31]. The coccygeofemoralis in long tailed mammals remains separate from the gluteus maximus, known as the caudofemeralis muscle [32]. In these animals its function is lateral flexion of the tail [33]. This is the sole morphogenesis study on any of the large muscles; it is likely other muscles with multiple origins and insertion have separate fetal muscle primordia.
Morphogenesis of human sphincter urethrae muscle
This study was completed in 1989 at the same Czechoslovakian institute of that of Gluteus maximus morphogenesis. . Study displayed three developmental phasess differentiating them by morphogenesis, histology and sexual dimorphism. External urethral sphincter in embryos and fetuses with crown length varying 18-320mm, neonates, children and adults was fixed in formaldehyde and embedded in paraplast. Then cut in series and stained with hematoxylin and eosin for histological analysis. Morphogenesis and Sexual dimorphism specimens were micro-dissected using a stereomicroscope. [34]
| Phase | Time Period | Characteristics |
|---|---|---|
| Indifferent Phase | Before 10th week | Muscle primordia discernible from neighbouring muscle by week 8. Grow to forms a shallow arch; connecting the urethra and urogenital diaphragm. Consists of condensation of myoblasts up to 9.5 weeks, past this point myotubes and muscle fibres appear. |
| Sexual dimorphic Phase | 10th week to Birth | Associated with the development of prostate and vagina. Primordia spread along urethra wall posteriorly.
In Males: Spreads to create the infraprostatic part of external urethral sphincter. Arches anteriorly to join prostate and urethra In Female: Spreads to create upper part of external urethral sphincter. Lower sixth of sphincter connects anterior and lateral urethral walls, additionally projects to lateral vaginal walls. |
| Definite Structuring Phase | After birth | Position of urethral sphincter does not alter in relation with prostate and inferior part of vagina. Infraprostatic region in males and upper part in females grow to form a complete ring. |
Current Research and Findings
Research Models
Current research models used to explore fetal myogensis include both human and animal models. Below is a list of a few different models;
- Rats and Mice as Langlois et al (2014) use in their experiment to do with Pannexin 1 and Pannexin 3 Channels and Skeletal muscle. [35]
- Pigs as Yang et al (2014) use in their experiment in comparing lean and obese pigs’ genes and muscle development[36]
- Sheep as Duckett et al (2014) use in their experiment on the effects of ergot alkaloids on fetal growth in sheep. [37]
- Humans as Langlois et al (2014) use in as Langlois et al (2014) use in their experiment to do with Pannexin 1 and Pannexin 3 Channels and Skeletal muscle. [38]
Intermediate tendon of Human Digastricus and Omohyoideus
Katori et al (2011) did a study to understand the development of the intermediate tendon within digastricus and omohyoideus muscles and their differences and similarities.
| Digastricus | Omohyoideus |
|
|
Stapedius
Rodri´guez-va´ zquez et al (2010) in order to understand the isolated case of a unilateral agenesia of the stapedius tendonduring week 14 of post-conception development (PCd), have tried to understand the way the stapedius muscle develops. The stapedius is essentially formed by two anlagen; one anlagen forms the tendon of the stapedius and the other forms the muscle belly of stapedius. The anlagen forming the tendon are derived from the internal segment of interhyale. The anlagen forming the muscle belly are derived from the 2nd pharyngeal arch near the interhyale, medial to the facial nerve. Interhyale is the internal part of the second branchial arch and it develops into stapedius’ tendon. The observed unilateral agenesia of the tendon of stapedius was found by Rodriguez-va et al (2010) to be due to the internal segment of the interhyale regression. Instead the belly of stapedius was accompanied with a pseudo tendon formed by the external segment of the interhyale. To come to this conclusion the formation of the stapedius and pyramidal eminence was tracked.[40]
Interhyale was observed as a mesenchymal condensation formed at O’Rahilly stage 16 at cranial component of the second branchial arch. The stapes and reichert cartilage are eventually differentiated from the interhyale. At Stage 18 and 19 a mesenchymal bridge forms by the interhyale which bridges stapes and cranial part laterohyale (Reichert’s cartilage). And by stage O’Rahilly stages 20 and 21 the interhyale fully develops. Rodriguez-va et al (2010) found the belly of stapedius to derive from a blastema which develops adjacent the interhyale to form its own anlage. In O’Rahilly stage 22 Rodriguez-va et al (2010) found that the interhyale began to take on an angular shape and the anlage of the stapedius belly connected with the vertex of the now angular interhyale.[40]
Interhyale developed two segments; internal thick segment and an external thin segment. The thick internal segment as discussed contributes to the formation of the tendon of the stapedius muscle. The external segment begins to regress in the embryonic period and by the fetal period, week 9, the external segment was observed to be fully regressed. In weeks 10-11 a conical belly continuous with the stapedius tendon was observed and the stapedius tendon attached at the back of the stapes head.The anlage of the pyramidal eminence formed around week 12-14 and around the stapedius muscle belly. The mesenchymal condensation forming the pyramidal eminence grew until it was inhibited around week 15-17. After week 9 there was not much morphological change of the stapedius. From this study we can see how the stapedius muscle develops its shape and attachments in the embryonic and fetal periods.[40]
Two studies on Triceps Brachii and Biceps Brachii and their parameters
Grzonkowska et al (2014) studied 30 fetuses from the age of 12-29 weeks and observed that with an increase in fetal age there was an increase in fetal triceps brachii parameters. [41] Similarly Szpinda et al (2013) studied the anatomical parameters of biceps brachii of 30 fetuses aged 17-30 weeks. [42]
| Triceps Brachii | Biceps Brachii |
|
|
The importance of these studies is that they measured the parameters of these developing muscles which were not done before. And they found that the individual components of these muscles developed proportionately as they are seen when they are fully developed.
Pannexin 1 and Pannexin 3 and Myoblast differentiation and proliferation
Langlois et al (2014) investigate the role of these Panx1 and Panx3 in the skeletal muscle of rodents and humans and their expression in the skeletal muscle of fetal and adult life.[44]
Panx1 and Panx3 are part of the pannexin channel protein family. Panx1 is known to play many roles, a few of which are vasodilation and inflammatory responses.[45] Panx3 is known for fewer functions such as carcinogenesis and osteoprogenitor cell proliferation. [46] [47] However recent studies have shown the potential role of Panx1 and Panx3 in skeletal muscle development.
Pannexins are known to be present within skeletal muscle. Pannexins were found in mRNA of skeletal muscle [48]. And Panx1 in sarcolemma of rodent skeletal muscle [49] Pannexins have also been observed to play certain roles in contraction as studies showed that Panx channel blockers reduced ATP release from electrical stimulation of myotubes [49][50] The role however of pannexins in cell differentiation and proliferation is unknown and what Langlois et al (2014) investigate in this experiment. [45] HSMM (primary Human Skeletal Muscle Myoblasts) was obtained from post quadriceps and psoas major. And SkMC (primary Human Skeletal Muscle cells) was got from upper arm or leg. [45] Western Blot analysis confirmed the presence of Panx1, Panx2 and Panx3 in the human, rat and mouse skeletal muscle tissue samples. Pan1 and Panx3 was detected in the tissue samples but Panx2 wasn’t. Panx1 and Panx2 stained differently within skeletal muscle and hinted that they had different functions. [45] It is known that in the fetal stage there is lots of proliferation whilst in the adult stages there is decreased proliferation. Decreased proliferation was indicated by decreased proliferating cell nuclear antigen (PCNA). Whilst increased differentiation resulted in increased myosin heavy chains (MHC). [45] Panx1 of higher molecular weight is the main pannexin found in skeletal muscle, more so in fetal than adult cells. Lower molecular forms of both Panx1 and Panx3 are more increased in adult forms and the higher weight forms are more increased in fetal forms. This shift possibly suggests that Panx1 and Panx3 plays a role in skeletal muscle development. [45] Langlois et al (2014) also found that Panx1 and Panx3 had a role to play in the sophisticated process of myogenesis, in particular myoblast proliferation and differentiation. Panx1 was low in in undifferentiated skeletal muscle cells and myoblasts but promoted myoblast differentiation and were thereafter more abundant. Panx3 of low molecular weight (~ 43 kDa) acted differently to higher weight species (~ 70 kDa). Expression of low molecular weight Panx3 promoted myoblast differentiation and repressed proliferation. Whilst expression of high molecular weight Panx3 coincided with proliferation and subsided with differentiation of myoblasts. Low molecular weight Panx3 were low in differentiated and undifferentiated HSMM but was expressed in skeletal muscle tissue which may indicate that lower molecular weight Panx3 plays a role more later on in differentiation. [45] Langlois et al (2014) used 2 Panx3 shRNAs to reduce Panx3 expression and they observed an inhibition of Proliferation whilst not initiating differentiation. Other impacts on Panx were studied such as their glycosylation, phosphorylation and being sialylated are important in their functioning as membrane channels. Post-transcriptional modifications and molecular interactions of Panx are thus important in regulating Panx channel function and thus important in myoblast differentiation and proliferation. [45] All evidences led to the conclusion that both Panx1 and Panx3 are expressed in skeletal muscle cells and participate in proliferation and differentiation of myoblast muscle cells.
Dll1 signaling and craniofacial myogenesis
Craniofacial skeletal muscle is derived from cranial mesoderm and trunk skeletal muscle is derived from somatic mesoderm. Hence craniofacial muscles are developmentally contrasting from trunk muscle. It is known that Notch signalling plays a role in myogenesis in trunk muscles. However Notch signalling role in myogenesis in craniofacial muscles has not been investigated that much, and is the focus of Czajkowski et al (2014) experiment. [51]
The normal myogenic regulatory factors (MyoD, Myf5, Mrf4) act in myogenesis and all three are necessary for myogenesis to occur. However there are other regulatory factors which act upstream to affect this standard myogenic pathway. This is another area where trunk and cranial muscle differs in regulation as Pax3/7 act upstream of the normal myogenic regulatory factors. However in craniofacial muscle development Pitx2 and Tbx1 play this role.[52]
The role of Notch signalling is not well known for craniofacial muscles. In mice Notch signalling is normally the result of a ligand such as DII1 or DII3 binding to a Notch receptor1-4. Subsequently this is followed by translocation into the nucleus and activation of target genes. In the context of myogenesis Notch signalling was observed repress MyoD and induce MyoR expression in C2C12 cells. [52]
Fetal Skeletal muscle contains a renewing pool of progenitor cells. Notch signalling results in the regulation of myogenic differentiation. Therefore when notch signalling is lost or mutated uncontrolled myogenic differentiation results and the formation of muscle. Things that repress notch signalling like DII1 and Rbpj impair muscle growth. Therefore mutations in DII1 and Rbpj results in muscle growth. Therefore Notch signalling plays role in removing MyoD and compensating for mutations in DII1 or Rbpj, and maintaining progenitor muscle pool. Notch signalling therefore suppresses myogenic differentiation in trunk muscles. But not much is known of its role in craniofacial muscles. [52]
Czajkowski et al (2014) found that in myogenic progenitor cells and muscle growth are deficient during early fetal development. Czajkowski et al (2014) observed that in the absence of these myogenic progenitor cells supernumerary myoblasts temporarily come and express MyoD. The overexpression of MyoD is in this case detrimental to the limited craniofacial progenitor cell in the mutant mice. Therefore the mutation and repression of MyoD which Czajkowski et al (2014) propose that Notch facilitates, allows for the craniofacial myogenesis. [52]
Czajkowski et al (2014) finds out that Notch signalling acts in a similar as it does in the trunk muscles in the craniofacial muscles. It does this by regulating progenitor cell differentiation by controlling MyoD expression. The difference however between Notch signalling between craniofacial and trunk muscles is in its other functions such as homng of satellite cells and the independence of Pax7 expression from DII1 signalling in craniofacial muscles. [52]
Abnormalities
Duchenne Muscular Dystrophy(DMD)
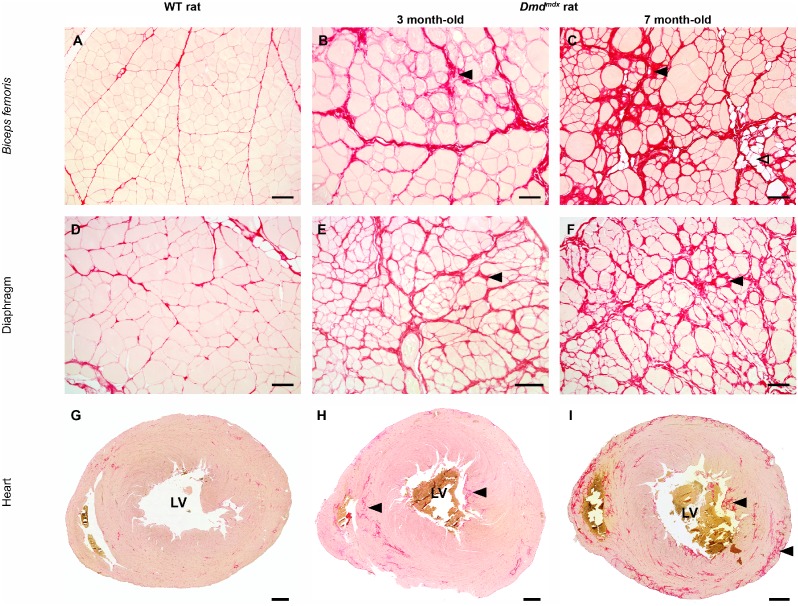
Caused by a mutation of the dystrophin protein on locus Xp21; this protein complex connects the cytoskeleton of muscle fibres to the extracellular matrix. Abnormal dystrophin results in a degradation of cellular integrity, excessive penetration of sarcolemma by calcium and water entering mitochondria increasing pressure and bursting. The lack of significant load during fetal development results in minimal wasting, it is detected in postnatal babies at 3-5 years old with muscles resisting gravity being the first to waste. Patients are restricted to wheelchairs by their early teens and have a life expectancy of 25 years. The incidence in male infants is 1 in 36,000. Typically males are affected while females are carriers. In the offspring of a carrier mother and unaffected father; sons have 50% chance of affected and daughter’s 50% chance of becoming carriers. Since the disease is a terminal illness killing in mid-twenties; affected fathers are not considered in the situation. Absence of affected fathers means it is very unlikely for daughters to be affected.
Duchenne Muscular Dystrophy has no present cure, treatment is aimed at altering onset of symptoms and maximising quality of life. [54] The most common approach is Corticosteroid based medication, which significantly improves muscle strength and function over a short term. Patients using the drugs long term gradually have reduced doses to avoid severe side effects which can include; weight gain, behavioural disorders and osteoporosis. Most effective corticosteroids are prednisolone, and deflazacort. Theories on how the steroids work include; include activation of T-Cell pathways, directly reducing muscle regeneration, modulating cell inflammation and enhancement of myogenic precursors. [55] Additionally assisted ventilation, supportive equipment, proton-pump inhibitors, increased excercise, beta-blockers and diuretics culminate to reduce symptoms and increase sufferers quality of life. [56] [57] [58] [59]
Amyoplasia
Amyoplasia is characterised by replacement of newborn muscle tissue with fat and dense fibrous tissue. Is the most common of Arthrogryposis multiplex congetia disorders; these result in multiple joint contractures. Affected limbs have significantly altered positioning, typically clubfoot is present and elbows are extended[60]. Primary cause is limited fetal movement, muscle is replaced by dense fibrous tissue. Conditions which limit fetal movement include abnormal uterus morphology and reduced amniotic fluid. Presently no genes have been linked to the deformity. [61] 85-90% of newborns undergo surgery within days of birth primarily on the legs and hips, releasing tendons from contractures. Motion can be improved by casting and splinting, lower limbs are typically cast and upper limbs are typically splinted. [60]
| Involvement subtype | Percentage of Case |
|---|---|
| Four-limb symmetrical | 55% |
| Three limb | 5% |
| Upper limb only | 17% |
| Lower limb only | 16% |
Nemaline Rod Myopathy
Nemaline rod myopathy is congenital non-dystrophic muscle disease. Sufferers experience general muscle weakness, weakness is most severe in face, neck and limbs[62]. Further clinical manifestations include feeding problems, scoliosis of spine, foot deformities and respiration difficulties. The disease is an inherited both autosomal dominant and recessive, 30% autosomal dominant, 20% autosomal recessive and 50% simplex. In total 6 genes have been associated with the formation of nemaline myopathy with the NEB and ACTA1 genes being the most prominent[63] [64]. Over 60 NEB mutations have been discovered which result in nemaline myopathy, it results in the decreased production and decreased length of the nebulin protein, half of all NM cases are associated with NEB mutations[63]. About 140 mutations to ACTA1 can lead to formation of nemaline myopathy, this results in either aggregation of α-actin fibres preventing functional muscle contraction or total absence of α-actin production[64].
References
- ↑ <pubmed>10809386</pubmed>
- ↑ <pubmed>7748174</pubmed>
- ↑ 3.0 3.1 <pubmed> 9094722</pubmed>
- ↑ <pubmed> 12587921</pubmed>
- ↑ 5.0 5.1 <pubmed> 21204650</pubmed>
- ↑ <pubmed>5804561</pubmed>
- ↑ <pubmed>5046781</pubmed>
- ↑ <pubmed>10102814</pubmed>
- ↑ <pubmed>23100595</pubmed>
- ↑ <pubmed>640968 </pubmed>
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 <pubmed>24532817</pubmed>
- ↑ <pubmed>22445545</pubmed>
- ↑ 13.0 13.1 <pubmed>12242286</pubmed>
- ↑ <pubmed>10733231</pubmed>
- ↑ <pubmed>2190237</pubmed>
- ↑ <pubmed>22682632</pubmed>
- ↑ <pubmed>3546571</pubmed>
- ↑ <pubmed> 7744859</pubmed>
- ↑ <pubmed> 2041547</pubmed>
- ↑ <pubmed>8014156</pubmed>
- ↑ <pubmed>15317692</pubmed>
- ↑ <pubmed>23629510</pubmed>
- ↑ <pubmed>2337297</pubmed>
- ↑ 24.0 24.1 <pubmed>8115369</pubmed>
- ↑ <pubmed>7780173</pubmed>
- ↑ <pubmed>24849606</pubmed>
- ↑ <pubmed>16518859</pubmed>
- ↑ <pubmed>16810681</pubmed>
- ↑ <pubmed>5043313</pubmed>
- ↑ <pubmed>15788867</pubmed>
- ↑ <pubmed>4083527</pubmed>
- ↑ <pubmed>1255730</pubmed>
- ↑ <pubmed>8843689</pubmed>
- ↑ <pubmed>2610390</pubmed>
- ↑ <pubmed> 25239622</pubmed>
- ↑ <pubmed> 25229314</pubmed>
- ↑ <pubmed> 25191653</pubmed>
- ↑ <pubmed>25239622</pubmed>
- ↑ 39.0 39.1 39.2 39.3 39.4 39.5 39.6 39.7 39.8 39.9 <pubmed>21538565 </pubmed>
- ↑ 40.0 40.1 40.2 <pubmed>19899117</pubmed>
- ↑ <pubmed>24913107</pubmed>
- ↑ 42.0 42.1 42.2 42.3 42.4 42.5 42.6 42.7 <pubmed> 23468258</pubmed>
- ↑ 43.0 43.1 43.2 43.3 43.4 43.5 <pubmed>24913107</pubmed>
- ↑ <pubmed>25239622</pubmed>
- ↑ 45.0 45.1 45.2 45.3 45.4 45.5 45.6 45.7 <pubmed>25239622</pubmed>
- ↑ <pubmed>22947051</pubmed>
- ↑ <pubmed>24338011</pubmed>
- ↑ <pubmed>15028292</pubmed>
- ↑ 49.0 49.1 <pubmed>23321639</pubmed>
- ↑ <pubmed>23583931</pubmed>
- ↑ <pubmed> 25220152</pubmed>
- ↑ 52.0 52.1 52.2 52.3 52.4 <pubmed>25220152</pubmed>
- ↑ <pubmed> 25310701</pubmed>
- ↑ <pubmed> 25187493</pubmed>
- ↑ <pubmed> 17541998</pubmed>
- ↑ <pubmed> 8143083</pubmed>
- ↑ <pubmed> 12467747</pubmed>
- ↑ <pubmed> 19945914</pubmed>
- ↑ <pubmed> 19945913</pubmed>
- ↑ 60.0 60.1 <pubmed>24459070</pubmed>
- ↑ <pubmed>9260643</pubmed>
- ↑ <pubmed> 14090530</pubmed>
- ↑ 63.0 63.1 <pubmed> 10051637</pubmed>
- ↑ 64.0 64.1 <pubmed> 15236405</pubmed>