2014 Group Project 2
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Renal
Introduction
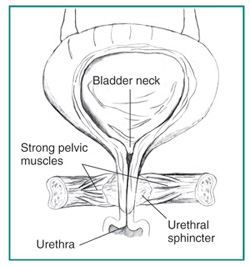
The renal system's main function is the production, storage and elimination of urine, and to maintain the balance of chemicals and water of the body. Kidneys are the primary organ of the renal system, and consist of smaller units known as nephrons - which filter the blood to remove urea and other wastes, and reabsorb or excrete excess water according to the needs of the body as directed by hormones released by the pituitary glands. Nephrons are made up of glomeruli to filter the blood, tubules to reabsorb any solutes or fluids, and more tubule networks to carry the urine to the bladder and outside the body. Small amounts of urine is released from the kidneys every 1 ~ 15 seconds into the ureter, which carry the urine to the bladder.[1] The bladder is a hollow organ which has the ability to change its epithelium according to how full the bladder is of urine. "The bladder's walls relax and expand to store urine, and contract and flatten to empty urine through the urethra. The typical healthy adult bladder can store up to two cups of urine for two to five hours.”[2] Two sphincter muscles are present at the base of the bladder, and two more at the end of the urethra (internal & external) to voluntarily control the excretion of urine.
Development of these components begin during the embryonic phase, and continue to develop and mature throughout the fetal stages. During the fetal stages, some abnormalities may form. During the embryonic period and fetal periods, the mother's placenta work to remove wastes from the fetus.
Abnormalities may arise during the embryonic and fetal stages of development of the renal system, such as Hereditary renal adysplasia, or polycystic kidney disease. Hereditary renal adysplasia is an inherited condition, where there is malformations in organs derived of the embryonic mesoderm [3]. Polycystic kidney disease is a fairly common genetic disorder in which fluid-filled cysts displace normal renal tubules[4].
Historic findings
| Year | Discovery |
| 1883[5] | The supra-renal bodies and the urinogenital organs are found to originate from the mesoblast |
| 1902[6] | It has been found that the pronephros exists first, before the mesonephros (Wolffian body).
The Wolffian duct is formed from the mesoblastic cells in the intermediate mass. These cells produce vesicles which form into tubular structures, opening into the duct. This has developed enough by the second month (Week 8) to be fully formed, but is almost completely degenerated by the end of that month. The permanent kidney forms in its place, arising from a stalked bud that extends forward from the Wolffian duct. The stalk portion of the bud becomes the ureter, which extends until it reaches a part of the cloaca that will form into the bladder later on in development. The common uro-genital mesentery connects the mesentery of the genitals and the mesentery of the Wolffian bodies together around Week 8. In females, the Müllerian duct forms the Fallopian tube, while the Wolffian body in males forms the epididymis, the seminal vesicles, the vas deferens and the common ejaculatory duct. |
| 1907[7] | The allantois is formed by a pouching-out of the ventral wall of the gut tract, and eventually goes on to form the urinary bladder and part of the urethra. The ureter is formed from the mesonephric ducts and opens into the urogenital sinus, later coming to open into the bladder. |
| 1912[8] | Before the pronephros begins to degenerate, both the mesonephros and pronephros function together.
The cloaca divides three times to form the rectum, bladder, urethra and the urogenital sinus. The bladder separates and becomes distinct from urethra by enlarging in size and by a differentiation in epithelium. This change begins to take play when the embryo is about 13mm in length. The first muscle layer of the bladder forms when the embryo is about 22.5mm in length due to a condensation of loose mesenchyme tissue that surrounds the bladder. This continues until the embryo is 80mm, when all muscle layers are distinct. |
| 1921[9] | When the tubules of the mesonephros are formed, a branch of the aorta enters the condensation of mesenchymal cells occurring at these areas, and divides and ramifies into smaller vessels, the entire structure of which forms a glomerulus. The tubule surrounding it flattens and becomes the two layers of epithelium around the glomeruli.
The outgrowth of the mesonephric duct forms the epithelium for the ureter, renal pelvis, and the collecting tubules, while the mesenchyme gives rise to the convoluted tubules and the glomeruli. |
| 1943[10] | The maturity of the fetus can be determined by the state of glomerular production, which ceases at Week 35 and/or when the fetus is 2.1-2.5kg in weight. |
| 1962[11] | Glomeruli begin to mature at about Week 6 until roughly about Week 35 of gestation. It is believe that at five months of gestation, the kidney has formed structurally enough to fully function. |
| 1973[12] | Luciano Barajas was able to demonstrate that when renal sympathetic nerve terminals are in contact with the basement cell membrane of the renal tubules, they are able to innervate all segments of this area. It was previously thought these nerves served little function. |
| 1976[12] | Sodium reabsorption increased due to renal sympathetic nerve stimulation and occurred in the proximal convoluted tubule, the thick ascending limb of Henle’s loop, the distal convoluted tubule, and the collecting duct. |
Developmental Timeline
| Week | Development |
| 3 | Nephrogenesis begins; pronephri formation. |
| 4 | Embryonic development of ureter begins from the ureteric bud. |
| 5 | Metanephros formation occurs. |
| 8 | Mature kidney is formed. |
| 9 | Urethral sphincter complex histologically identifiable within the urogenital sinus
The kidneys complete their ascension and achieve their correct anatomical position |
| 10 | Kidneys begin to produce urine.
The epithelial lining of the ureter begins to differentiate. External urethral sphincters develop |
| 11 | Degeneration of the mesonephri.
The bladder wall begins to change and develop. |
| 12 | The urinary bladder is developed from the urogenital sinus and the surrounding splanchnic mesenchyme. |
| 15 | Allantois regresses and becomes known as the urachus. Vesicourethral canal develops into the pelvic segment of the urethra in the male fetus, and the entire urethra in the female fetus.
Inner layer of smooth muscle fibres and outer layer of striated muscle layers of urethral sphincter complex developed. |
| 21 | The epithelium of the bladder is mostly developed; it is 3-4 layers thick. |
| 36 | Nephrogenesis is complete, however can occur as early as week 32. |
| Postnatal | Maturation of neonatal glomerular filtration. |
Current research models
Research is constantly being done on the development of the renal system; it is important to know what events happen at different stages of development and what initiates the different stages. The main events of development have been discovered so researchers now are looking at the development in more detail; for example looking at the origins of the structures, the impacts mothers have on their unborn child and ways to cure abnormalities. Animals are good models and are often used for research as there are fewer ethical issues surrounding them compared with humans and their generation time is much shorter so mutations can be identified faster. Below highlights a few of the recent studies that have been carried out to look at specific parts of renal development.
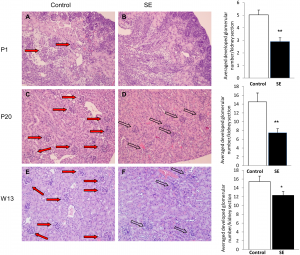
The impact of maternal cigarette smoke exposure in a rodent model on renal development in the offspring[13]
- Mice have been used to determine the impact of maternal cigarette smoke exposure on the development of the renal system
- It was predicted that smoke exposure would lead to a change in the expression of growth and transcription factors which would lead to kidney disease later on in life
- An experiment found that there was up-regulation of some fibroblast growth factors and down-regulation of others which led to elayed nephron development and fewer nephrons present at birth
- The image on the left shows left glomerular numbers in the control offspring are higher compared with smoke exposed offspring at birth, early postnatal period and adulthood
- Glomerular in smoke exposed offspring were not mature and were not fully vascularised
- Overall it was discovered that cigarette smoke exposure during pregnancy and lactation period leads to underdeveloped renal system which can result in chronic kidney disorders in adulthood
- As well as the impact of smoke exposure the effect of nicotine on development has been researched[14]
Development of an animal model to study congenital urinary obstruction[15]
- Lambs were used to look at the possible cures for Lower Urinary Tract Obstruction
- It’s important to find a cure for Lower Urinary Tract Obstruction as it affects many newborns and is one of the most common causes of renal failure in young children
- Bladder obstructions were created in lambs by ligating the urachus
- 3 or 4 weeks after the obstruction was created shunts were placed in the lambs so the urine could bypass the obstruction
- Bypasses with and without valves were created
- Shunts without valves led to small, shrunken and thick-walled bladders with poor function developing
- Shunts with valves had much better bladder function
- It was found that the cycle of filling and emptying the bladder with urine during foetal development is very important for the formation of the bladder.
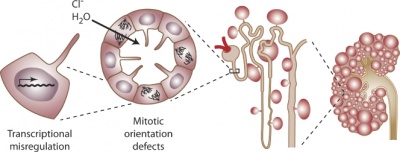
Fate mapping using Cited1-CreERT2 mice demonstrates that the cap mesenchyme contains self-renewing progenitor cells and gives rise exclusively to nephronic epithelia [16]
- The fate of cap mesenchyme and pre-tubular aggregates that comprise the metanephric mesenchyme were investigated
- Previous experiments have shown that metanephric mesenchyme gives rise to nephronic epithelial structures but it is not clear from which of the sub-populations, cap mesenchyme and pre-tubular aggregates, the structures arise.
- Transgenic mice were used with a mutation that allowed the fate of the transcriptional regulator, Cited1, only expressed in the cap mesenchyme to be followed.
- It was found that collecting duct epithelium does not contain any cells originating from cap mesenchyme
- The cap mesenchyme was also found to have a population of epithelial progenitor cells that could self-renew.
Kidney
Early Development
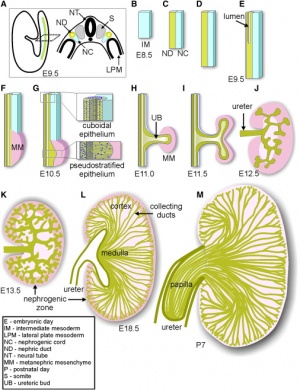
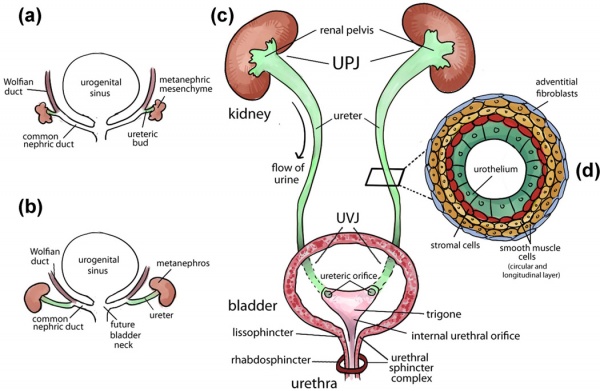
The kidneys first develop in the embryo by a process called nephrogenesis, in which self-renewing mesenchymal renal stem cells produce nephrons, the main functional unit, and form a simple embryonic kidney called the pronephros [17]. This process of nephron formation is stimulated by the signaling between the ureteric buds and these stem cells, named progenitor cells and located at the tips of the ureteric buds, causing nephrons to develop and the ureteric buds to branch [18]. The following events occur:[19]
- During week 3 two pronephri are produced and nephrotomes, a series of tubules, begin to fuse together with the pronepheric duct.
- The pronephri elongate and induce the nearby mesoderm to form mesonephri, and the pronepheric duct to become the mesonephric (Woffian) duct.
- The ureteric bud and metanephric mesoderm (blastema) react together to form the metanephros which goes on to form the mature kidney.
- The cells of the ureteric bud differentiate to form the major and minor calyces as well as the collecting tubules, while the cells of the metanephrogenic blastema develop into the renal tubules and glomeruli.
- This process ends by week 8 of gestation, while the development of the nephrons continue through to week 32-36.
Fetal Development
There are a number of factors that occur in regards to the kidneys during the fetal period of development, the most important of which is the continued generation of nephrons. There are specific genes expressed for the continued nephrogenesis, with vasculature created to supply the newly formed kidneys. There are certain events that occur for the kidneys to achieve their correct anatomical position before they are fully formed, as well as further maturation of the glomeruli post-natally before the kidneys are fully functional.
Nephrogenesis
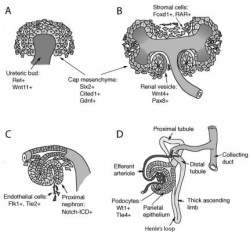
The formation of nephrons continues well after the embryonic period in concert with the branching of the ureteric bud [18], ending between week 32-36 of gestation [20]. It is during this period that the self-renewing process of the progenitor cells ceases and they differentiate into nephrons for the final time he ureteric bud [18]. The process of nephron formation is shown in the first diagram to the right [21]. Although nephrogenesis is terminated by week 36, the resulting kidney is not yet fully functional. At birth, although the infant’s kidneys are developed enough to maintain homeostasis and are sufficient for growth and development, their functional capabilities are decreased. This is a result of the transition from depending on the placenta to maintain homeostasis of fluid and electrolyte balance while in-utero, to maturation of the neonatal glomeruli once born [22]. This maturation phase due to postnatal adaption to extrauterine life occurs during early infancy [23].
While it is not well known the mechanisms by which nephron number is determined, the causes of several disorders and diseases, such as renal disease and hypertension, have been attributed to a low nephron count [18]. It has been determined that a decrease in the number of progenitor cells, a possible result of genetic abnormalities, toxic insults, and nutritional deficiencies [20], can result in fewer branching of the ureteric buds, leading to impaired kidney growth [18]. Therefore, nephron number is important as it can show the success/extent of nephrogenesis, and thus be used to determine if any and what genes and environmental factors may aid this process [24].
Genes
While there are many genes and transcription factors that regulate the formation of early kidney development, the main factor that continues to be expressed as the fetal kidney develops is the Gremlin gene. The gene, coded GREM1, has been found to play a key role in the formation of the kidneys and nephrogenesis in general. When the kidney is fully formed, the expression of this gene is relatively low in an adult. However, it is thought that many renal diseases and their progressions are linked to an overexpression of this gremlin gene [25].
Anatomical Position
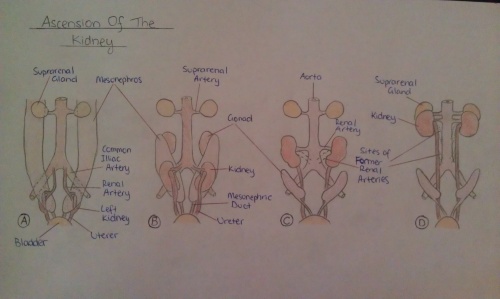
The kidneys initially begin to develop proximally to the pelvis, located at the level of the upper sacral segments. In order to attain the correct anatomical position, as the fetus develops, they continuously become more cranial in position until they reach the dorsal sides of the body at around the T12~L3 levels. This process is usually completed by week 9 of fetal development and occurs as a result of the kidneys coming into contact with the supra-adrenal glands, as well as due to the growth of the embryo’s body and abdominal cavity. These organs are medially rotated by up to ninety degrees during their ‘ascension’, so that the renal pelvis will lie on their medial aspect [26][27].
During the entire process of anatomical positioning, the kidneys receive a bloody supply from arteries branching off at various points of the abdominal aorta. While they are initially vascularized by the lateral sacral branches of the aorta, during the ascent, they continue to receive blood at progressively higher levels. When the kidneys have achieved their correct position, they are supplied by the renal artery at the level of the 2nd lumbar vertebrae. The simultaneous rise of the vasculature helps to contribute to the ascending of the fetal kidneys [26].
Renin Production
While the kidneys develop, particularly the tubules, the production of renin can be found within these areas. As the tubular activity increases and this component proliferates in number and size, it is noted that growth is proportional to the amount of renin produced; as growth and functional state of the tubules increase, so does the concentration of renin created [28].
Urethra
The urethra is a structure that allows for excretion of urine, and presents as a tube from the urinary bladder to the external urethral orifice. The urethra originally develops from the cloaca during fetal development. The cloaca can be divided into the anorectal canal (dorsally) and the urogential sinus (ventrally). The bladder develops from the superior portion of this urogenital sinus (endoderm), and the inferior portion develops into the urethra. The endoderm of the urogenital sinus derives the urethral epithelium, and the splanchnic mesenchyme develop into the connective tissue and smooth muscle components of the urethra.[29]
The anterior part of the urogenital sinus (develops into the bladder) has an opening at the apex which is connected to the allantois during fetal development. The function of this opening at the apex of the fetal bladder is to drain the developing bladder to the allantois via the umbilical cord. At around week 15 of fetal life, the allantois regresses and becomes known as the urachus; at which time the vesicourethral canal develops into the pelvic segment of the urethra in the male fetus, and the entire urethra in the female fetus. The remaining parts of the urogenital sinus (posterior portion) "later develops into the phallic urethra in the male and the lower portion of the vagina and vaginal vestibule with perineal urethra orifice in the female." (Hila Milo Rasouly, Weining Lu. 2013)[30]
The urethral sphincter complex is important in controlling urination frequency and timing, and is histologically identifiable within the urogenital sinus from week 9 of development as a mesenchymal condensation after the cloacal division. It develops into an inner layer of smooth muscle fibres and outer layer of striated muscle layers by week 15 of development, which further develop into an omega shaped muscle apparatus surrounding the urethra. The urethral sphincter complex muscles are innervated by autonomic and somatic nervous systems, and correct development of the muscle nerve innervation and the urethral sphincters are essential in the maintenance of normal urinary continence postnatally [30]. External urethral sphincters are thought to develop within week 10 of development. [31]
Ureter
The ureter is a muscular tube structure responsible for the transportation of urine produced in the kidney to the bladder. The development of the ureter typically begins during week 4 of gestation in infants. The process begins from the ureteric bud, which arises from the caudal region of the mesonephric ducts (also known as Wolffian ducts) that run along the edge of the intermediate mesoderm [32]. Following the migration of the ureteric bud into the metanephric mesenchyme, the reciprocal interaction that occurs between the tip of the ureteric bud and the metanephric mesenchyme results in the ureteric bud branching morphogenesis for the formation of the renal collecting system [30]. This occurs simultaneously with the elongation of the trunk of the ureteric bud, the portion remaining outside of the metanephric mesenchyme, forming the ureter [33][32].
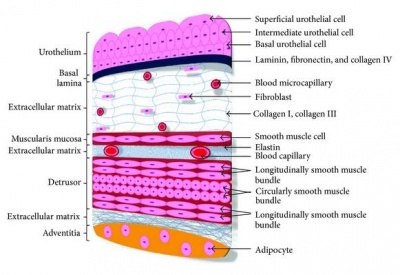
By week 10, the differentiation of the ureter lining occurs in response to the molecular signals from the ureteral epithelial and mesenchymal cells. This then initiates differentiation of the early simple cuboidal ureteral epithelial cells to form the multilayered urothelium. The urothelium is covered by urothelial plaques that express uroplakin proteins and is impermeable to urine. Concurrently, the stromal cells, smooth muscle cells and the adventitial fibroblasts (which are later maturated to form the serosa) all give rise from the differentiated mesenchymal cells [30].
The smooth muscle cells go on to be further arranged into layers with inner circular and outer longitudinal orientation and are characterized with strong expression of α-smooth muscle actin. Together, the mechanism involved give rise to the four layers of lining present in the mature ureter; urothelium, stromal cells, smooth muscle cells, and adventitial fibroblasts [30].
Bladder
The role of the urinary balder in the renal system is to store urine produced by the kidneys before it is excreted via the urethra [34]. A flexible epithelium is essential for the bladder as it changes volume by contracting and relaxing depending on the volume of urine in the body. A fusion event occurs between the common urogenital sinus and the mesonephric duct, this divides the rectal components from the urine components and it allows the bladder to develop.
The urinary bladder develops in the first 12 weeks of gestation from the urogenital sinus and the surrounding splanchnic mesenchyme, these development events are controlled by complex epithelial–mesenchymal signals. The vesical part of the urogenital sinus is attached to the allantois and goes on to form the bladder [35]. The lamina propria, the muscle coat and the adventitia all develop from the splanchmic mesoderm whilst the epithelial lining is derived from the endoderm of the urogenital sinus [36].
Initially the epithelium lining of the urinary bladder is made from two distinct cell layers, the superficial layer and the basal layer. Up to week 11, the rest of the bladder wall consists of mesenchyme that gradually matures into lose connective tissue. At the 13th week collagen begins to appear, by the 14th week it is abundant in the lamina propria and by week 15 it has extended into the superficial muscle bundles. Smooth muscle cells begin to appear in the connective tissue during week 12, they initially appear in the proximal part of the organ but they spread distally over time. At 21 weeks the epithelium is 3-4 layers thick, the superficial layer, the intermediate layer, lamina propria and the basal lamina. This epithelium is specialised and contains features that are characteristic of urothelial differentiation so that urine is unable to pass through the bladder wall [36].
The muscular coat of the bladder does not develop until after the kidneys have begun to produce urine this is not a problem for the embryo as urine is released directly into the amniotic cavity [36]. The muscles in the bladder however are very important for normal urine output once the foetus have been delivered; as development continues the walls of the bladder muscle thicken and there is a decrease in collagen content, this increases the compliance of the bladder [36].
During development the bladder only produces immature reflexes rather than the voluntary bladder control that is only seen once the infant is toilet trained. It is suggested that the switch between involuntary reflexes and voluntary contractions is due to the development of the central and peripheral neural pathways that control the contraction of the bladder [37]. It also could be due to the increasing number of nerves in the detrusor muscle during fetal development, this would lead to an increase control over the bladder wall due to the increase in nerves present [34].
Abnormalities
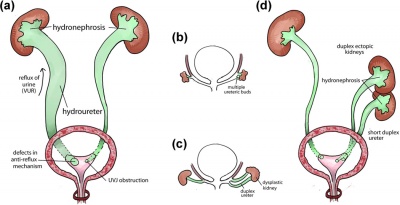
Renal agenesis
Renal agenesis is a congenital abnormality referring to the failure of the development of the kidneys and ureter produced by a lack of interaction between the ureteric bud and the metanephric mesenchyme. Renal agenesis can occur in two forms, infants can be born with either bilateral or unilateral renal agenesis. Infants born with bilateral renal agenesis are incompatible with life and are born usually stillborn, or die within a few days after birth[38]. Unilateral renal agenesis has a frequency of 1 in 5,000 newborns, in contrast to bilateral renal agenesis the rarer of the two, occurring in 1 in 30,000 infants [39].
Induction of the ureteric bud from the nephric duct is mediated by glia cell-derived neurotrophic factor (GDNF), which is secreted by the metanephric mesenchyme and interacts with the tyrosine kinase c-Ret receptor expressed in the ureteric bud in order to induce branching of the nephric duct [38]. In animal models, renal agenesis is linked to signalling failures failure of the GDNF–RET signalling [40]. A complex molecular network that includes regulation by EYA1, the sine oculis homeobox homologues SIX1 and SIX4, and PAX2, controls the activation of GDNF in the metanephric mesenchyme. Furthermore, members of the HOX11 paralogous genes13 and the signalling molecule growth and differentiation factor 11 (GDF11) are necessary for GDNF signals that are derived from sources other than the metanephric mesenchyme (for example, the developing gut) are not interpreted in more rostral parts of the embryo [39].
Although Mutations in GDNF or RET have yet to be linked with mutations in patients with kidney defects such as renal agenesis. A possible explanation for this could be because human kidney development is not susceptible to the slight reduction in protein level that results from heterozygous inactivation [39].
Renal agenesis is typically associated with other congenital defects, including:
- Oligohydraminos
- Facial abnormalities including: wide set eyes, a recessed chin and low-set ears
Polycystic Kidney Disease
Polycystic kidney disease (PKD) is a common genetic disorder characterized by the formation of fluid filled cysts in the kidneys, which displace normal renal tubules. There are two types of PKD, autosomal dominant polycystic kidney disease (ADPKD) and autosomal recessive polycystic kidney disease (ARPKD).
PKD affects approximately 1 in 1000 individuals, with ADPKD accounting for the majority of the cases reported [4][38]. ADPKD, also known as adult-onset polycystic kidney disease is commonly reported to present in adulthood in association with hypertension and renal failure [41]. The disease is linked to the mutations in the genes encoding polycystin-1 (PC1) and polycystin-2 (PC2), which is characterized by perturbations of renal epithelial cell growth control, fluid transport, and morphogenesis [4]. These mutations ultimately affect multiple signaling pathways, which cause aberrant gene transcription, cell proliferation, and ion secretion, which in turn result in the formation of benign fluid-filled cysts. As cysts balloon out from individual nephrons, their collective effect leads to the displacement of the normal renal parenchyma and the formation of a cyst-filled kidney with reduced functional capacity [4]. In ADPKD, the growth of renal cysts produces a progressive increase in renal volume and destruction of the parenchyma, leading to terminal chronic renal failure in adulthood [38].
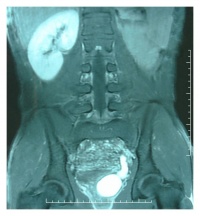
ARPKD, however is the rarer form of PKD, affecting approximately 15% of cases or 1 in 20'000-40'000 live births [42] [43]. It commonly presents during the second or third trimester of fetal development. Ultrasound images show kidneys that are usually 'bright' or echogenic and are often associated with progressive oligohydramnios [41].
Horseshoe Kidney
Horseshoe kidney is a common congenital abnormality causing a fusion of the kidneys. The abnormality occurs when the poles of the lower kidney fuse, and the kidneys develop into one structure, forming an L or U shape, instead of the distinct two separate structures [44] [42]. Horseshoe kidney is the most common abnormality associated with fusion, occurring in 1 in 400 infants, with males being twice as likely to develop the disease [45][46].
Horseshoe kidneys are commonly positioned lower than normal kidneys, being located at the lower lumber vertebrae in between L3-L5, in front of the aorta and inferior venae cavae, and posterior to the inferior mesenteric artery, which usually crosses the isthmus. This is caused by the inferior mesenteric artery, as it prevents the ascent of the kidneys to its normal anatomical position, and it thus trapped in the mid abdomen [47] [46].
There are typically two main theories outlining the cause of this anomaliy. The first theory suggests that while the kidneys have not yet begun their decent during week 4 of gestation (the metanephric stage), and are still located in the pelvis, the two inferior poles come into contact and thus fusion occurs in the midline, resulting in the formation of a horseshoe kidney, with a fibrous isthmus. As the kidneys are still yet to form a renal capsule, the fusion of the kidneys is also accompanying with the fusion of the nephrogenic blastemas, which is caused by abnormal growth of the spine and organs in the pelvis[46].
The fusion is also suggested to be associated with malrotation. Kidney rotation normally occurs during week 7 and 8 of gestation, as the kidneys migrate from the pelvis and rotate medially. However, in horseshoe kidneys, the inferior mesenteric artery blocks the isthmus and the kidneys fail to ascend and remain at a lower positioning [42][46].
The second theory proposes that the abnormality is caused by a teratogenic event, and is caused by an abnormal migration of posterior nephrogenic cells that later form the parenchymal isthmus.
Horseshoe kidney is typically associated with other congenital defects, including:
Duplicated Ureter
Duplicated ureters are the most common renal abnormality, estimated to occur in 1% of the total population, and is found to be more common in females and is a condition in which more than one ureter drains one kidney[49]. Ureteral duplications can be complete - in which the individual has two ureters from one kidney entering the bladder, or be incomplete, where there are two separate ureters leaving the one kidney that fuse into one tube before entering the bladder [42]. Incomplete duplicated ureters often have no clinical significance other than a higher risk for urinary tract infections (UTI), however, completely duplicated ureters can present with some complications such as one of the ureters joining to the vagina or urethra instead of the bladder or to its complimentary duplicated ureter[50].
The development of the ureter begins at around 4 weeks into gestation when the ureteric bud branches from the Wolffian duct to extend into the nephrogenic blastema[49]. A study carried out on 30 children with duplicated ureters by Atwell et al. (1976) showed that 66% of children with an ureteral duplication had first degree relatives with either a complete or incompletely duplicated ureter, leading to the conclusion the defect is inherited in an autosomal dominant manner[51].
References
- ↑ Kim Ann Zimmermann (2013). “Urinary System: Facts, Functions & Diseases” Feb 11 2013
- ↑ Stanford (2014). “Anatomy of the urinary system”
- ↑ <pubmed>20388228</pubmed>
- ↑ 4.0 4.1 4.2 4.3 <pubmed>21079243</pubmed>
- ↑ Foster, M., Balfour, F. M., Sedgwick, A., & Heape, W. (1883). The Elements of Embryology. (2nd ed.). Organs from Mesoblast. London: Macmillan and Co.
- ↑ Keith, A. (1902). Human Embryology and Morphology. Uro-genital System. London: Edward Arnold.
- ↑ Heisler, J.C. (1907). A Text-book of Embryology. (3rd ed.). Genito-urinary System. Philidelphia and London: W.B. Saunders Company
- ↑ Keibel, F. and Mall, F.P. (1912). Manual of Human Embryology II. The Development of the Urinogenital Organs. J. B. Lippincott Company, Philadelphia
- ↑ Bailey, F.R. and Miller, A.M. (1921). Text-Book of Embryology. Urogenital. New York: William Wood and Co.
- ↑ Potter, E.L., Thierstein, S.T. (1943). Glomerular Development in the Kidney as an Index of Fetal Maturity. The Journal of Pediatrics, 22(6), 695-706
- ↑ Robert L. Vernier, Aksel Birch-Andersen. (1962). Studies of the human fetal kidney: I. Development of the glomerulus. The Journal of Pediatrics, 60(5), 754-768
- ↑ 12.0 12.1 <pubmed>12623969</pubmed>
- ↑ <pubmed>25058584</pubmed>
- ↑ <pubmed>25279991</pubmed>
- ↑ <pubmed>24013366</pubmed>
- ↑ <pubmed>18061157</pubmed>
- ↑ <pubmed>24855634</pubmed>
- ↑ 18.0 18.1 18.2 18.3 18.4 <pubmed>24656820</pubmed>
- ↑ <pubmed>25088264</pubmed>
- ↑ 20.0 20.1 <pubmed>24488483</pubmed>
- ↑ <pubmed>24011574</pubmed>
- ↑ <pubmed>24781774</pubmed>
- ↑ <pubmed>24623338</pubmed>
- ↑ <pubmed>24022365</pubmed>
- ↑ <pubmed>25036148</pubmed>
- ↑ 26.0 26.1 Keeling, J.W. & Khong, T.Y. (2007). The Urinary System, Fetal and Neonatal Pathology. London: Springer. 4th Ed, pp. 623
- ↑ Moore: the developing human 9th edition. Saunders 2011. An imprint of Elsevier
- ↑ <pubmed>19871238</pubmed>
- ↑ Schoenwolf: Larsen's human embryology, 4th ed. chapter 15
- ↑ 30.0 30.1 30.2 30.3 30.4 <pubmed>23408557</pubmed>
- ↑ Ludwikowski B, Oesch Hayward I, Brenner E, Fritsch H. The development of the external urethral sphincter in humans. BJU Int. 2001 Apr;87(6):565-8.
- ↑ 32.0 32.1 <pubmed>17928823</pubmed>
- ↑ <pubmed>23123402</pubmed>
- ↑ 34.0 34.1 <pubmed>3627353</pubmed>
- ↑ <pubmed>23371862</pubmed>
- ↑ 36.0 36.1 36.2 36.3 <pubmed>2621133</pubmed>
- ↑ <pubmed>22535797</pubmed>
- ↑ 38.0 38.1 38.2 38.3 <pubmed>23169372</pubmed>
- ↑ 39.0 39.1 39.2 <pubmed>17878895</pubmed>
- ↑ <pubmed>18252215</pubmed>
- ↑ 41.0 41.1 <pubmed>11746154</pubmed>
- ↑ 42.0 42.1 42.2 42.3 <pubmed>25313840</pubmed>
- ↑ <pubmed>16767405</pubmed>
- ↑ <pubmed>18059107</pubmed>
- ↑ 45.0 45.1 <pubmed>24469670</pubmed>
- ↑ 46.0 46.1 46.2 46.3 <pubmed>24178305</pubmed>
- ↑ <pubmed>22970063</pubmed>
- ↑ <pubmed>16407023</pubmed>
- ↑ 49.0 49.1 J. Gatti, J. Murphy, J. Williams, H. Koo (2013) emedicine overview, Ureteral Duplication, Ureteral Ectopia, and Ureterocele
- ↑ <pubmed>9017803</pubmed>
- ↑ <pubmed>1013379</pubmed>