2011 Group Project 6
| Note - This page is an undergraduate science embryology student group project 2011. |
Tetralogy of Fallot
Introduction
Tetralogy of Fallot (TOF) is a congenital heart disease affecting approximately 3 in 10000 people. It is the most common form of congenital heart disease, accounting for about 1 in 10 cases. [1] TOF was named after Etienne-Louis Fallot for his description of the anatomy and physiology of the defects associated with the disease[2]. TOF is believed to be caused by one or more genetic mutation on different chromosomes, which includes chromosomes 5,20 and 25, which impact the development of the neonatal heart. These genetic mutations contribute to the four characteristic defects of TOF:
- Pulmonary stenosis: a narrowing of the blood flow path from the right ventricle to the lungs.
- Overiding aorta: an aorta which is continuous with both ventricles instead of just the left one.
- Ventricular septal defect: the septum dividing the left and right ventricles is incomplete
- Right ventricular hypertrophy: enlargement of the cells in the right ventricle. [3]
TOF patients are given the name 'blue babies' because they often have a slightly blue appearance due to the decreased circulating oxygen levels. Some common symptoms in TOF patients include turning blue while crying, collapsing due to tet spells during exercise, dizziness and fatigue.
Etienne Fallot, Helen Taussig and Alfred Blalock have significantly contributed to the understanding of the cardiac anomalies and treatment options associated with TOF. Currently, surgery is the most successful treatment option, however palliative care and medical treatment is also available. Due to technology advancements, doctors are able to detect TOF more accurately and treat patients quicker, allowing an increase in survival rate and a favourable long term prognosis.
Whilst the understanding of this disease has grown greatly, future treatment options are continuously being explored. These include developing durable and efficient surgical procedures, shunts and valves.
History
Early History
In 1671, Niels Stenson was the first to anatomically describe Tetralogy of Fallot. However, the precise descriptions of the anatomy of Tetralogy of Fallot were done in 1784 by William Hunter at St Georges Hospital Medical School in London. His description is as follows[2] :
| “…the passage from the right ventricle into the pulmonary artery, which should have admitted a finger, was not so wide as a goose quill; and there was a hole in the partition of the two ventricles, large enough to pass the thumb from one to the other. The greatest part of the blood in the right ventricle was driven with that of the left ventricle into the aorta, or great artery, and so lost all the advantage which it ought to have had from breathing”[2] |
Hunter’s description of the defected heart’s anatomy along with its resulting physiology was further specified and advanced by Etienne-Louis Fallot. However it was Maude Abbott from Canada who coined the term ‘Tetralogy of Fallot’ in 1924[2].
Contemporary History
In 1938 there was a dawn in surgical therapy for people with heart defects as Gross and Hubbard closed off the patent ductus arteriosus in a girl who was aged 7.[4]
Later, it was the work of Helen Taussig with her using a fluoroscope which allowed her to determine that children suffering from cyanosis, especially those with TOF, had decreased blood flow to the pulmonary circulation and thus more blood was needed to enter into this circulation. She then put forward a concept that shows the benefits of an artificial ductus; as she believed the closing of the patent ductus arteriosis lead to babies having cyanosis and cyanotic spells weeks after the baby was born[4].
It was in the morning of 1943 in which Taussig explained to Blalock and Thomas that she believed it would be possible to direct more blood into the pulmonary circulation by conducting surgical methods. It was only a year later in which this belief came into practice as a cyanotic congenital heart experienced the first successful treatment due to surgical means. This technique was conducted on another two cases with the same defect and thus due to such success from the surgeries, the concept had received worldwide success.[4]
During the 1950s, the rise of open heart surgery began and the era of closed-heart surgery dominance had ceased. It was also during the 1950s to 1970s that surgeons realised the great variance in the anatomy of TOF, thus they had to work out new surgical techniques to accommodate this variation. Additionally in this period, there was an appreciation by surgeons that whilst conducting such operations in the heart, there needs to be precision in how they conduct their work anatomically. Thus in this 20 year period, there was a number of surgeons who had successfully conducted intracardiac surgeries to infants[4].
During her late years of work, Dr Taussig had hypothesised that congenital cardiac defects had arisen from the expression of genetic defects found in the infants. At the time the hypothesis received skepticism, however today studies show us that this idea is quite real [4].
Surgical History
The first procedures for Tetralogy of Fallot were being conducted from around the 1950s. Due the advances in the areas of medicine which support and maintain surgeries, children born with Tetralogy of Fallot now have a great chance of survive to adulthood[2]. In retrospective studies it shows how surgical procedures have improved. From the 1950s till today, the mortality rate from surgery dropped from 50% to less than 2%[5].
It was in 1945 that Blalock and Taussig explained the ‘systemic artery-to-pulmonary artery shunt’ and in 1954 that Varco and Lillehei repaired a TOF heart whilst doing a open heart surgery[5].
During the surgeries in the past the surgeons will place a shunt between the pulmonary artery and a systemic artery of a child with Tetralogy of Fallot as this will allow some improvement in the oxygenation of the infant’s blood. It will be later when the individual grows up that the shunt will be removed then the heart will be repaired. Now surgeons prefer to repair the heart in the initial operation to repair the infants heart[6].
Timeline
| Year | Milestones |
| 1628 | William Harvey published his work De Motu Cordis which portrayed a groundbreaking understanding that the two criculatory systems, pulmonary and systemic, and independant of each other.[4] |
| 1671 | Stenson anatomically described Tetralogy of Fallot[2] |
| 1784 | William Hunter gave a precise description of the anatomy of Tetralogy of Fallot[2]. |
| 1847 | Chevers acknowledged the absence of pulmonary valve in Tetralogy of Fallot hearts.[4] |
| 1850s | Peacock was a pioneer in using a stethoscope to discover a cardiac murmur with a heart with pulmonary stenosis.[4] |
| 1888 | Fallot destroyed the idea that cyanosis had always occured due to inability of the foramen ovale to close. Also, he was the one to use the term tetralogie, and furthermore acknowledged that there was no therapeutic treatments for patients of TOF during his time[4]. |
| 1888 | Etienne-Louis Fallot specified and advanced the description of Tetralogy of Fallot’s heart anatomy and its resulting physiology[4] |
| 1924 | Maude Abbott coined the term “Tetralogy of Fallot”[2] |
| 1930s | Helen Taussig using fluoroscope determined that TOF patients suffer cyanosis because of decreased blood flow to the pulmonary circulation. She then proposed the benefits of an “artificial ductus” in TOF neonates, as she believed the closing of the patent ductus arteriosis lead to babies having cyanosis and cyanotic spells[4]. |
| 1936 | Abbot demonstrated the Chest X-ray, 3-lead electrocardiogram and circulatory and auscilatory diagrams that were produced from a Tetralogy of Fallot heart[4]. |
| 1938 | Gross and Hubbard closed off the patent dusctus arteriosus in a girl who was aged 7[4] |
| 1943 | Taussig explained to Blalock and Thomas that she believed it would be possible to direct more blood into the pulmonary circulation by conducting surgical methods[4]. |
| 1945 | Blalock and Taussig explained the ‘systemic artery-to-pulmonary artery shunt’[5] |
| 1950 | Beginning of the rise of open heart surgery[4] |
| 1954 | Varco and Lilehei repaired a TOF heart whilst doing an open heart surgery[5] |
| 1950-70 | Realisation of variance in the anatomy of TOF. This became the period of success for intracardiac surgeries to infant[4]. |
| mid 1970s | Advancements in Infant surgery, introduction of prostaglandin therapy and rise in electrocardiography signficantly affected the patients with Tetralogy of Fallot[4] |
| 1990s | Dr Taussig hypothesised that congenital cardiac defects had arisen from the expression of genetic defects found in infants[4]. |
Epidemiology
Tetralogy of Fallot is considered to be a rare genetic disorder. It makes up around 7%-10% of cardiac congenital defects. Moreover, epidemiological studies show that it occurs in 3 out of 10000 live births that are delivered. After the neonatal age, Tetralogy of Fallot is the most frequent cause of cyanotic heart disease. Additionally, Tetralogy of Fallot slightly affects more males than females.[7]
The incidence of TOF seems to occur sporadically, even though there is a 3% risk of reoccurrence in a sibling (without any other first degree relatives affected by TOF) [2]. However, mothers who are 35 years and older have an increased risk that their offspring will have TOF, whilst on the other hand mothers who have 2 or more children have a decreased risk[8].
Below is a table which shows the incidence rates of TOF in different races[9]:
| Race | Incidence of TOF malformation (per 10,000) |
| Whites | 2.85 |
| Hispanics | 2.31 |
| Asians | 2.83 |
| Blacks | 3.81 |
| Other | 3.80 |
In regards to genetics, one study showed that in 25% of the cases of TOF there is a microdeletion on Chromosome 22 which has the q11 region within it. Moreover, TOF is associated and frequently diagnosed along side Velocardiofacial Syndrome and DiGeorge Syndrome. Interestingly, another study showed that there is a rate of 25% in which patients with this 22q11 mutation eventually develop schizophrenia [2].
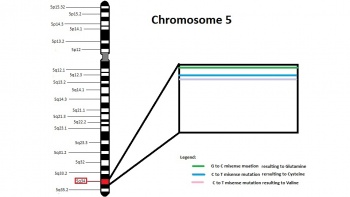
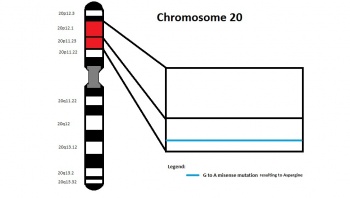
Tetralogy of Fallot is also associated with the chromosomal anomalies Trisomy 21, 18 and 13, and also associated with Alagille Syndrome which has a JAG1 mutation[7]. The most frequently associated genetic anomalies with Tetralogy of Fallot is Trisomy 21 and 22q11.2 deletion. Furthermore, mutations in the 5q35 section (which codes for NKX2.5) were found in 4% of TOF patients which are classified as non-syndromic TOF[10].
In respect to clinical features, 35% of patients with TOF have ventricular and atrial arrhythmias. A 30 year follow up period of patients with TOF have shown that 6% of them die due to sudden cardiac death [6].
TOF is treated with surgery (see Treatment/Management section below). When this is conducted before the age of six months, there is a low mortality rate associated with it (i.e. 2%). After surgical intervention is conducted, there is a survival rate of 85-90%[7]. It is clearly possible that improvements in the surgical field has allowed the reduction in surgical mortality which was 50% in the late 1950’s to 2% with contemporary surgical methods[5]. On the other hand, if a TOF patient decides not to correct their condition, there is a 10% chance they will survive till their age is in the 30s and a 3% chance they will survive till their forties or older. Before surgical interventions were possible, TOF patients usually died within the first few years after birth, and it was rather unusual to see a patient who had survived beyond 30 years of age[2].
In regards to survival statistics, after surgery is conducted to correct TOF there is a [11]:
- 97% chance that the patient will live one year after the surgery
- 98% chance that the patient will survive 20 years after the surgery if they were alive 30 days post surgery
- 90% chance that the patient will survive 30 years post surgery if they were operated on whilst they were children
From epidemiological studies conducted in regards to mortality rates in respect to congenital heart disease in the US between 1979-2005, there was a 40% reduction in mortality that was linked to Tetralogy of Fallot. It is thought that this reduction in mortality was associated to the ‘earlier recognition and treatment of heart failure and arrythmia’ which allowed this change in mortality rates to occur.[12]
Signs and Symptoms
Cyanosis
Normally, red blood cells carry oxygen around the body that is nearly completely saturated with oxygen held by the haemoglobin. When blood loses its oxygen, the colour changes from bright red colour to a colour that is a mix of dark blue and red. This event is known as cyanosis. Cyanosis could occur in situations in which the lungs are compromised due to underlying reasons such as the lungs being infected with chronic obstructive pulmonary disease, pneumonia and exposure to air at high altitudes. In the case of TOF patients, one of the heart defects (i.e. ventricular septal defect, explained further below) allows cyanosis to occur as the blood oxygen concentration is decreased due to the mix of oxygenated and deoxygenated blood in the TOF heart’s ventricles[13].
The most important sign of TOF patients is cyanosis. This is when the lips, fingernails and skin of the patient turns a bluish colour[14]. Additionally during cyanosis, the mucous membranes of the TOF patient may turn blue. Therefore, the cases where dark-skinned individuals may experience cyanosis, the cyanotic event could be observed by change in colour in the nails and mucous membranes, which include lips, around the eyes and gums [13]. Cyanosis is considered to occur due to decreased amounts of oxygenated haemoglobin found within the blood when cyanosis occurs [15].
'Tet Spells' and Fatigue
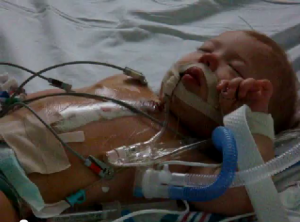
The word ‘tet spells’ is derived from Tetralogy of Fallot, as it is an event experieced by individuals who suffer from TOF. Babies who have TOF can at times enter into these ‘tet spells’, in which there is a sudden drop in oxygen saturation of the blood, causing the baby to turn blue. These ‘tet spells’ become apparent when the baby does certain activities such as crying[14]. An image portraying a 'tet spell' in an infant can be found here: Cyanotic 'Tet spell'
In addition to experiencing ‘tet spells’, the baby could pass out, become unresponsive to their parents calling or touch, develop fatigue and finally have dyspnoea. Furthermore, children with TOF would develop fatigue quickly and possibly pass out, thus surgeons now repair TOF hearts during infancy and not at adult to prevent such events.[14]
Heart Murmur
Heart murmurs are sound waves which are clearly audible that come from the vascular system or from the heart which have a round range between 20-2000Hz. It is considered to be produced as a consequence of blood flow within the cardiovascular system which is that is turbulent[16]. In the case of Tetralogy of Fallot, a heart murmur is a common sign which occurs due to the defected heart’s abnormal blood flow through it. However it should be mentioned that the heart murmur is not a hallmark for congenital heart defects as many hearts of healthy children also have murmurs[14]. An audio sample of a heart murmur that would be heard in a TOF patient is found in the link below. Also below is a link to the sound of a normal heart beat without murmur and without TOF.
Normal Heart: First and Second Heart Sounds - Normal & Unsplit - Waveform and Audio -- TOF Heart:Tetralogy of Fallot - Waveform and Audio
The types of murmurs that can be present in the heart include [6]:
- 1. Pulmonary Insufficiency Murmur - which occurs briefly and low pitched in the diastolic phase of the heart beat. This murmur is often missed during a physical examination of the heart because it is heard to hear it and its duration is short
- 2. Aortic Insufficency Murmur
- 3. Right Ventricular Outflow Murmur – can also have a pansytolic (throughout the systole of the heart beat) murmur if there is a presence of ventricular septal defect found in the heart [6].
Abnormal Growth and Clubbing
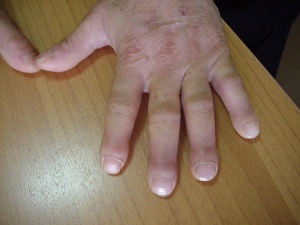
Children with TOF do not grow at the rate of normal children as whilst breastfeeding in infancy, the babies would get tired quicker and during the growth of the infant, normal functionability of the heart and oxygen saturated blood is needed for proper growth [14]. TOF children may also have clubbing. [14] This clubbing would be evident as there would be enlargenemt of the bone or the skin around the fingernails of the patient. [17]
Cardiac and Visceral Problems
It should also be mentioned that pulmonary insufficiency symptoms developed from a TOF heart varies in the degree of pulmonary insufficiency found in that individual. Thus the symptoms that could be presented are of a wide range which could from decline in function to palpitations. Moreover, late symptoms could also develop from the insufficiency which includes right-sided heart failure, exertional dyspnoea, syncope and palpitations. In an event of which right ventricular failure occurs, signs to indicate it include elevated jugular venous pressure, ascites, hepatomegaly, jugular venous distension and peripheral edema.[6]
Genetics/Aetiology
The genetic etiology of Tetralogy of Fallot (TOF) is still currently unknown and being researched. Even though this is the case, most of the studies done on the disorder have agreed that TOF normally occurs with other developmental disorders, like DiGeorge syndrome, and that the disorder may be caused by multiple mutations in a person’s genome. Here are some of the current suspected genetic mutations that leads to the congenital disease Tetralogy of Fallot.
| Features | 22q11.21 | 5q34 | 20p12.1 - p11.23 |
| Chromosome # | 22 [18] | 5 [19] | 20 [20] |
| Chromosome arm | q (long arm) | q (long arm) | p (short arm) |
| Position | 11.21 | 34 | 12.1 to 11.23 |
| # of base pair | 26,890 | 3,208 | 36,362 |
| Gene Affected | TBX1 | NKX2-5 | JAG1 |
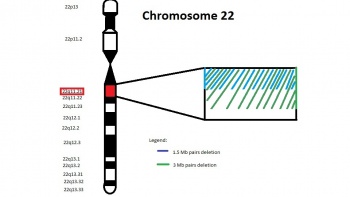
22q11.21
Studies have shown 74% of patients with 22q11.2 microdeletions have Congenital Heart Defect, and out of these 22% have Tetralogy of Fallot (TOF).[21]. This is why mutation in this region of the chromosome is an important consideration for the genetic aetiology of TOF.
This region of chromosome 22 contains the gene for T-box 1 transcription factor (TBX1). As a transcription factor, it regulates the expression of genes by binding to the regulatory region of the DNA and promote/inhibit the transcription of particular genes [22]. Although the genes regulated by this particular transcription factor is still being researched, it has been discovered that mutation in the gene can lead to the development of TOF [23] [10]
The most common genetic mutation of this gene that results in TOF is a microdeletion of 3 or 1.5 Mb of 22q11.2 region on chromosome 22 [10]. The result of this microdeletion is a haploinsufficient gene. This is where there is only a single copy of the gene in one allele, which clinically leads to an abnormal functioning of the protein because a single copy of the gene is incapable of producing enough protein that will allow normal function. [24]
Although microdeletion is the most common mutation in the TBX1 gene that results to TOF, there are other mutational variants that have been observed that resulted in TOF. This variant involve a 30-bp duplication in exon 9c, a region in the TBX1 gene. This leads to the production of non-functioning TBX1 protein because the insertion of the duplicate leads to the expansion of the polyalanine tract of the gene, resulting to a non-functioning TBX1 protein. The proteins produced aggregates within the cytoplasm, leading to decrease in transcription of TBX1 gene, since this gene is dose-dependent, which means that transcription cease as soon as there is enough TBX1 protein. Since the TBX1 proteins are non-functioning transcription of genes that the TBX1 protein is responsible for is not controlled.[10]
Mutation of the TBX1 gene may also lead to other phenotype, which includes:
- DiGeorge Syndrome
- Velocardiofacial Syndrome
- Conotruncal anomaly face syndrome
OMIM number = 602054
5q34
Studies have shown that at least 4% of TOF patients have NKX2-5 mutation. This is why the mutation in this gene is considered in the development of TOF. [25]
This gene encodes the NK2 homeobox 5 transcription factor (NKX2-5 protein). NKX2-5 protein is essential in tissue differentiation and temporal and spatial patterns of development in cardiac tissue. [26]. This means the protein regulates the genes responsible for the formation of the chambers of the heart. This is because studies have shown that the NKX2-5 protein is involved in the development of atrial, ventricular and conotruncal septation, AV conduction and AV valve formation. [27] Atrial and ventricular septation is the formation of the walls that separate the heart into four chambers, while conotruncal septation is the formation of the two great vessels (Aorta and Pulmonary artery) that forms the outflow tracts of the heart.
There are 4 known substitution mutation of the NKX2-5 gene in TOF patients, although one of the mutations has been found to be present in non-TOF patients. [25] This includes:
- G to C substitution at base pair 21 = glutamic acid to glutamine amino acid substitution
- C to T substitution at base pair 216 = arginine to cysteine amino acid substitution
- C to T substitution at base pair 219 = alanine to valine amino acid substitution
Mutation of the NKX2-5 gene may also lead to other phenotype, which includes:
- Hypothyroidism
- Congenital non-goitrous
- Atrial septal defect with atrioventricular conduction defects
OMIM number = 600584
20p12.1-p11.23
The typical clinical presentation of mutation in this gene is a disease called Alagille Syndrome (AGS) that have some clinical overlap with that of TOF, which are mainly right heart abnormalities.[28] This makes mutation in this gene of special interest when it comes to TOF
The gene is about 36 kb with 26 exons, which are the coding regions of the DNA,[29] and expresses the gene Jagged-1 protein (JAG1 protein), which is a ligand of the Notch receptor [30]. This means that cells/tissues containing the JAG 1 protein are able to communicate with cells/tissues that contains the Notch receptor on its surface. JAG1 Gene is highly expressed in developing mammalian heart, thus JAG1 proteins are present on cardiac cell membranes [31]. With the Notch receptor present on adjacent cells, JAG1 protein is able to communicate with adjacent cells via intercellular signaling. This is because the bond between the JAG1 protein and the Notch receptor leads to the release of the receptor’s intracellular region from the membrane. This part is transported into the nucleus activating transcription factors, which in turn regulates the transcription of genes that will lead to cellular differentiation and morphogenesis.[30]
There is a missesnse mutation of the JAG1 gene identified at the 821st base pair. This is a G to A base substitution, resulting in glycine to aspartic acid substitution. This may lead to the formation of cysteine residues that results in the formation of abnormal protein because its structure and stability has been compromised.[32] There are 2 types of proteins resulting from the mutation of the allele, a protein that functions abnormally and a protein that functions normally. The abnormal functioning protein is produced at higher temperature[33]:
- Abnormal JAG1 protein = retained intracellularly and not transported to cell surface
- Normal JAG1 protein = retains function as a ligand to Notch receptor
Mutation of the JAG1 gene may also lead to other phenotype, which includes:
- Alagille Syndrome
- Deafness
- Congenital heart defects
- Posterior embryotoxin
OMIM number = 601920
Other Genes
These are the other mutated genes that are found in TOF patients:
| Gene | Location | Protein Product | Gene MIM number |
| ZFPM2 | 8q23.1 | Zinc Finger Protein | 603693 |
| GDF1 | 19p13.11 | Growth/Differentiation Factor 1 | 602880 |
| CITED2 | 6q24.1 | CBP/p300-Interacting Transactivator, with GLU/ASP-rich C-Terminal Domain 2 | 602937 |
| GATA4 | 8p23.1 | GATA-binding protein 4 | 600576 |
| NOTCH2 | 1p12-p11 | NOTCH2 receptor protein | 600275 |
| NOTCH1 | 9q34.3 | NOTCH1 receptor protein | 190198 |
Pathophysiology and Abnormalities
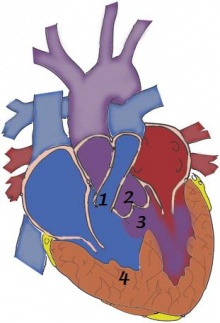
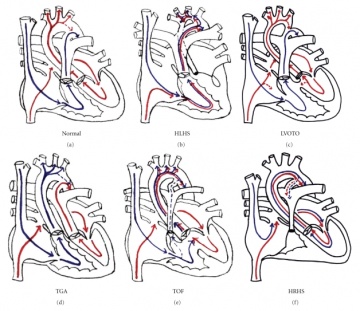
The four pathological features of tetralogy of fallot are:
These features result from the disruption of the aortic and pulmonary outflow tracts. The severity of symptoms is determined by the extent of right ventricular outflow obstruction.[3]
Pulmonary stenosis - Pulmonary stenosis may be caused by a narrowing of the pulmonary valve (valvular stenosis) or the outflow tract of the right ventricle (infundibular stenosis). [34] The narrowing of either the valve or the infundibulum obstructs the flow of blood from the right ventricle into the pulmonary circulation. Consequently, this results in a reduced flow of oxygenated blood in the systemic circulation. If the degree of pulmonary stenosis is mild, a left to right shunt forms. (The higher pressure in the left ventricle causes blood to pass through the septal defect into the right ventricle). However, if the pulmonary stenosis is significant, a right to left shunt will form. [35] This occurs because the stenosis raises the pressure in the right ventricle and forces blood directly into the left ventricle. This is important because deoxygenated blood can now enter the systemic circulation and problems such as cyanosis, dizziness and fainting can occur. The pathophysiology of pulmonary stenosis usually worsens with age because the pulmonary orifice stays the same size despite an increase in the size of the heart. [36]
Overriding aorta - Instead of being positioned directly over the left ventricle, the aorta is displaced anterosuperiorly (in front of and above). The aortic valve is located directly above the interventricular septal defect allowing blood from both the left and right ventricles to pass through the aortic valve. [36] This biventricular connection allows both oxygenated (from left ventricle) and deoxygenated blood (from right ventricle) to enter the systemic circulation. The degree to which the overriding aorta is continuous with the right ventricle determines the severity of symptoms. [37]
Ventricular septal defect - The interventricular septum dividing the left and right ventricles is incomplete at its superior, membranous end. [2] During ventricular contraction, blood from the left ventricle is forced into the right ventricle and then re-enters the pulmonary circulation. This extra volume of blood places pressure of the pulmonary system and compensatory pulmonary hypertension and right ventricular hypertrophy may occur. If the right ventricular pressure exceeds that of the left, the left to right shunt is reversed and the patient will experience cyanosis because deoxygenated blood is bypassing the lungs and entering the systemic circulation. [38]
Right ventricular hypertrophy - Hypertrophy of the right ventricle is a compensatory response to pulmonary stenosis. Because the pulmonary outflow tract is narrowed, the right ventricle must pump harder to meet the oxygen demands of the body. [39]
|
Diagnostic Tests
| Diagnostic technique | How the technique works | Presentation in a TOF patient | Image |
| Physical examination | TOF is often diagnosed during fetal life by echocardiography.[2]
If TOF is not detected during fetal life, certain signs and symptoms at birth may alert the need for further investigation. |
Signs and symptoms include mild to moderate cyanosis, which worsens when the baby cries, difficulty feeding and difficulty gaining weight. However, TOF often goes undiagnosed until adult life. Most adult patients will appear normal, however, some may present with cyanosis and clubbing of the fingers. The jugular venous pressure is usually normally (raised jugular venous pressure often indicates right ventricular failure). If the aorta is pushed to the right (so it is continuous with both the left and right ventricle), a lift below the right sternoclavicular joint may be noted. [40] | 
|
| Heart murmurs | Heart murmurs are sounds caused by the turbulent flow of blood. They are heard using a stethoscope. They are often the result of problems including valvular stenosis (narrowing of valves), valvular regurgitation (leakage of valves due to incomplete closure) or defects in the heart wall allowing blood to flow in unusual directions. [41] | Examination of the heart of a patient with TOF may reveal a loud second heart sound (produced by the closure of the pulmonary valve). A harsh systolic ejection murmur may be heard and a palpable thrill may be felt along the left sternal border. [42] These sounds occur because the pulmonary outflow tract is obstructed. Although this murmur is often present, sometimes it may be short or difficult to hear and is often missed on physical examination. A pansystolic (occurring throughout the whole of systole) murmur may also be heard. This type of murmur occurs due to the ventricular septal defect between the left and right ventricles. The increased pressure in the left ventricle forces blood back into the right ventricle, causing a murmur, which lasts the whole of systole (contraction phase). [6] | 
|
| Electrocardiogram | Once TOF is suspected, electrocardiogram and chest radiographs are performed. Electrocardiography is used to assess the electrical activity of the heart. The electrical activity is detected by electrodes, which are placed on the skin of the patient and recorded by an external device. [43] | The electrocardiogram is extremely important in detecting a right bundle branch block (a block in the electrical conducting system of the heart), which is common in patients with TOF. An electrocardiogram will also reveal a heart that is deviated slightly to the right and an enlarged right ventricle due to the ventricular hypertrophy.[40] | 
|
| Chest radiograph | A chest radiograph (or chest x-ray) uses ionising radiation to develop an image of the patient’s chest. It is used to diagnose many conditions including conditions of the thorax and structures within the thoracic cavity including the heart, lungs and major blood vessels entering and leaving the heart. Chest radiographs are often used to screen for certain diseases but further tests are required for a definitive diagnosis.[44] | In a patient with TOF, a chest radiograph will demonstrate a prominent right ventricular shadow, giving the heart a boot-like appearance, which is typical of patients with TOF. The right ventricular hypertrophy causes the apex of the right ventricle to rise on top of the relatively unfilled left ventricle, giving the heart its boot-shaped appearance on examination. [3] The radiograph will also show a right-sided aorta in approximately one-quarter of patients.[40] | 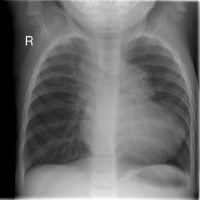
|
| Echocardiogram | An echocardiogram (also called a cardiac ultrasound) is performed to confirm the above findings. Echocardiography uses ultrasound to produce two-dimensional (and now also three-dimensional) images of the heart. It assesses cardiac tissue, valve function, the velocity of blood flow and any abnormal communications within the heart.[45] | The echocardiogram identifies important abnormalities of the heart including obstruction of the pulmonary outflow tract, the size of the pulmonary arteries, the degree of aortic override and the size of the defect in the interventricular septum. [3] | 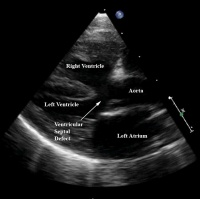
|
| Magnetic resonance imaging | Magnetic resonance imaging (MRI) is evolving as the most important technique for evaluating the size and functioning of the right ventricle. An MRI machine uses a magnetic field to produce a detailed image of the scanned area of the body. It is especially useful in viewing soft tissues as it provides greater contrast than techniques such as x-ray. [46] | MRI’s are important in assessing pulmonary valve competence and the severity of regurgitation (the amount of blood that flows back into the right ventricle due to the incomplete closure of the pulmonary valves) in patients with TOF. It can measure the volume and mass of the right and left ventricles and can assess the degree of pulmonary outflow tract obstruction. Finally, MRIs are important in measuring the degree of ventricular septal defect.[40] | 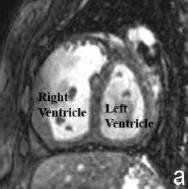
|
Treatment/Management
The treatment for TOF consists of medical therapy, palliative procedures & surgery.
Medical therapy:
Medical therapy is usually used to prepare the infant/adult for surgery. The degree of therapy and what medications are used for treatment depends upon the degree of cyanosis in each patient.
- Patients with acute cyanosis usually carry out knee to chest exercise positions[3] in order to decrease the amount of deoxygenated blood entering circulation. This along with oxygen & intravenous morphine provides more blood flow to the lungs and also decreases ventilator drive. [47]
- Patients with severe cyanosis are usually administered intravenous propranolol, which causes a decrease in muscle spasms that occur in the infundibulum (which causes RVOTO). [2]
- Asymptomatic patients do not require any particular medical treatment.
Palliative Procedures:
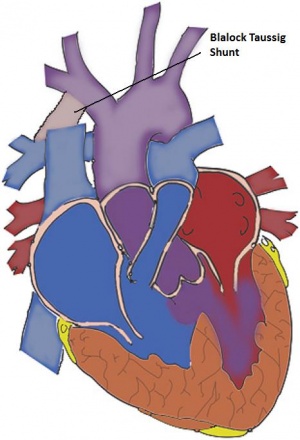
The Blalock-Taussig Shunt:
Total corrective surgery is the most advantageous option for young infants[7]. However, in some instances, infants suffering from pulmonary atresia or other anomalies (in addition to TOF) may require a palliative method instead[48], because the anomaly changes the candidate’s capability to having total corrective surgery. In some instances, infants with very small pulmonary arteries cannot tolerate corrective surgery and require palliation instead. The main aim of palliative surgery is to increase pulmonary blood flow and allow for growth and possibly total correction of the pulmonary artery[49]. By far, the most common procedure is the modified Blalock-Taussig Shut (B-T shunt). This shunt is placed between the pulmonary and subclavian arteries (on the same side)[49]. The original B-T shunt was placed by anastomosing a branch of the transected subclavian artery and the pulmonary artery[49], but the classic B-T shunt had a disadvantage of thrombosis that occurred due to the small size of the vessel.
The major advantage of the modified Blalock-Taussig shunt is that it[48]:
- Directs partially oxygenated blood from the subclavian artery to the pulmonary artery (hence to the lungs for oxygenation), therefore relieving cyanosis
- Allows for preservation of subclavian artery
- The shunt can be used on either side
- Allows for easier control and closure of the shunt at the time of complete repair.
Due to this feature, the shunt has an excellent success rate.
Complications of the B-T shunt are rare, but usually include[50]:
- Blockage of the shunt – causing the blue colour of the patient to return
- Infection – the ‘foreign’ shunt may become a site for bacteria to implant & cause infection.
- Distortion of the pulmonary artery arises as the child grows and the point at which the shunt is attached to the artery may not grow, causing a kink in the artery to develop.
Other shunts such as the Potts, Waterston and Glenn shunts are either not used or infrequently used today (see table below for more details). [51]
| Type of shunt | Descriptions | Reasons for it not being used | Image |
| Pott’s shunt | This shunt is a connection that is established between the descending portion of the aorta (on the left side of chest) to the left branch of the pulmonary artery. [50] | The shunt has a tendency to increase the pulmonary blood flow, pulmonary hypertension, causes blood flow to be preferential to one of the lungs, the shunt causes kinking in the pulmonary artery and it is also very hard to close when complete repair is undertaken. [47] | 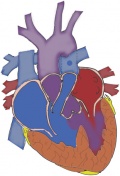
|
| Waterston shunt | This shunt was placed between the back of the aorta, to the right branch of the pulmonary artery. [50] | This shunt’s use is more suited to those with pulmonary artery stenosis and also the procedure is quite difficult to perform. It causes excessive pulmonary blood flow and hypertension and congestive cardiac failure has been found in 20% of patients that have the Waterston or Pott's shunt [52] | 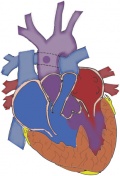
|
| Glenn shunt | The superior vena cava is anastomosed via a shunt to the right pulmonary artery (the classic Glenn shunt). The modified Glenn shunt is where the superior vena cava is attached to the right branch of the pulmonary artery, which is till connected to the main pulmonary artery.[50] | This procedure is very complicated and difficult to perform, also complete repair becomes a laborious task. [47] | 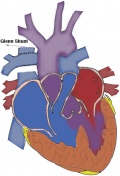
|
Surgery:
Today, Tetralogy of Fallot can only be treated with open-heart surgery, which is usually performed before 6 months of age[3].
The goal of surgery is to be able to treat the four congenital abnormalities associated with TOF. It is also advantageous to perform this surgery at an early age and not wait until the child is older, however the specific timing of the operation is still under some controversy[6].
Surgeries occur under cardiopulmonary bypass (CPB) and are performed either through the atrium (Transatrial) or from the right atrium/from the pulmonary artery (transatrial- transpulmonary) [7]. Surgical approaches are the most common in patients of all ages.
If a patient has had palliative procedures, these shunts must be isolated and taken down [53], before the actual operation can commence. In the surgery itself, the surgeon:
- Widens the narrowed pulmonary blood vessels and the pulmonary valve itself, hence allowing for greater blood flow to the lungs.[2]
- Repairs the ventricular septal defect using a patch to cover the hole in the septum.[2]
Fixing these two defects also solves the other two defects (the hypertrophy of the right ventricle wall and provides oxygenated blood to the Aorta). When the surgery is carried out during infancy, the incision heals in roughly 6 weeks.
More information regarding Treatment and Surgery can be found here: |Tetralogy of Fallot Repair|Treatment Options for Tetralogy of Fallot
Prognosis
If a person has undetected TOF or has not undergone corrective surgery, then the usual outcome depends upon the severity of the Right Ventricular Outflow Tract Obstruction (RVOTO)
A person suffering from severe RVOTO has a 25% chance to die in the first year, 40% chance of dying within 3 years, 70% chance to die by the age of 10 and a 95% chance of death by the age of 40.[7]
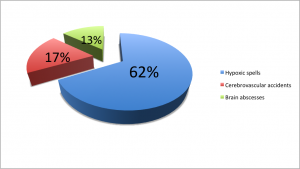
The major causes of death in surgically untreated patients are
- Hypoxia spells – 62%
- Cerebrovascular accidents – 17%
- Brain abscesses – 13% [7]
If a patient undergoes elective surgery then the patient has a low mortality rate of just 2% (in & directly after surgery) and a long-term survival rate of 85-90%[3]. In the absence of other serious complications, patients are able to live normal lives, shown by the possibility to carry out a successful pregnancy.
Whilst this may be the case many patients go on to develop pulmonary valve insufficiency [54] (where upon contraction of the right ventricle, blood flows backwards from the pulmonary arteries in to the right ventricle). This causes the right ventricle to work harder and hence become dilated.
This can lead to hindered function of the right ventricle and cause fatigue.
Another outcome of repair can be a narrowing at the outflow area/branch of Pulmonary arteries[55], which produces high pressure in the right ventricle and can lead to further corrective surgery being undertaken.
Finally, whilst corrective TOF surgery repairs the anomalies, it is crucial that long term follow up with a cardiologist be mandatory to detect any problems, whether they be recurring or new. Hence patients are advised to carry out physical examinations, echocardiography and other exams as the patient matures into a teenager and adult years.[3]
Future Directions
In the last decade, several innovations have been developed for possible treatment methods and also management of TOF, these include:
Percutaneous Pulmonary valve replacement:
This has been the most promising and exciting area for the past decade, with majority of focus going into conduit rehabilitation between the pulmonary artery and right ventricle. The replacement is designed for patients who have already had TOF elective surgery and are in need of pulmonary valve replacement. The percutaneous pulmonary valve is composed of a bovine Internal Jugular Vein, with the native valve (bovine) fixed into a platinum stent it is moved along into the right ventricular outflow via fluoroscopic control. This control allows for the valve to be fixed precisely by inflation, using a balloon in balloon method [2]. If the percutaneous pulmonary valve proves to have great success and long-term durability, it has the potential to allow for earlier intervention in such cases [6] . So far no mortality or late mortality has been registered in the patients tested.
Oral Drug Therapy
Pharmacologic therapy could be used to alter the clinical outcomes that arise with pulmonary insufficiency, which may develop from Tetralogy of Fallot. Scientists and researchers are using MRI imaging technology to see the effect that Nitric Oxide may have upon pulmonary vasodilation and are amidst of trialling similar oral drugs for long-term use. [7].
Genetic Mutation & therapy
Recently, congenital heart patients went under large-scale mutation screenings and several important genes (islet 1, Mef2c etc) [56] involved in cardio genesis, have been identified. Identification of these genes can possibly provide markers for diagnosis of cardiac anomalies such as TOF. Work is also being done for genetic manipulation to be used as a therapeutic tool.
In vitro engineered valves
Finally creation of in vitro tissue engineered valves using human endothelial cells (using allograft techniques) has been shown to have long term durability and also reduce valve degeneration, cusp thickening or pulmonary insufficiency (trivial to mild) [57]. Creation of such valves has great promise for the world of cardiac congenital diseases as it will allow greater options for valve replacement and allow for growth and remodelling as the child continues through somatic growth. [7].
Glossary
Allagile Syndrome - it is a neonatal genetic disorder, which results in liver disease with cholestasis, peripheral pulmonic stenosis and unusual face
Anastomosing - connection made between adjacent blood vessels.
Annular hypoplasia – A ring shaped underdevelopment of tissue/organs
Anomalies - something deviating from normal
Arrythmia - (dysrhythmia) refers to an event where there is abnormal electrical activity of the heart which causes the heart to have either a fast, slow, or irregular heart beat.
Artificial Ductus - it is an instrument inserted into blood vessels to create a channel that would allow blood to flow through.
Ascites – a clinical term which refers to increased amounts of fluid between the tissues that line the abdomen and abdominal organs within the peritoneal cavity.
Cardiopulmonary bypass - deoxygenated blood returning to the heart is diverted to a machine which oxygenated the lung and then pumps it to the arterial system.
Clubbing – event where there is changes in under and around the toenails and fingernails. Also there is changes in the nails is some disorders.
Congenital - a disease/physical abnormality present from birth.
Conotruncal septation – it is the formation of the two great vessels (Aorta and Pulmonary artery) that forms the outflow tracts of the heart.
Cyanosis - Clinical term referring to a situation where there is a blue colouration of the skin and mucous membranes when there is a presence of deoxygenated hemoglobin. Cyanotic heart disease is one of the causes to this situation.
Cyanotic heart disease - Clinical term referring to a congenital heart abnormality (defect) which causes a lack of oxygen which results in cyanosis.
DiGeorge Syndrome - a congenital disorder which results from a microdeletion of the 22q11.2 region of Chromosome 22. It is characterised by hypocalcemia, immunodeficiency, and congenital heart disease.
Dyspnoea – Shortness of breath, difficult or laboured breathing
Edema/Oedema - Swelling caused by the accumulation of abnormally large amounts of body fluid in between the spaces os the body's cells or in the circulatory system.
Electrocardiogram - it is a diagnostic procedure that detects the electrical activity of the heart.
Exon – coding region of DNA.
Flouroscope - an imaging instrument used in fluoroscopy, where an X-ray and a CCD video camera is used to obtain real time moving images.
Foramen Ovale - it is an anatomical channel found on the interatrial septum, which allows the blood to flow between atria in the developing baby.
Haploinsufficient gene – it is where there is only a single copy of the gene in one allele, which clinically leads to an abnormal functioning of the protein because a single copy of the gene is incapable of producing enough protein that will allow normal function.
Heart Failure - condition where the heart is unable to supply blood to the whole body.
Hepatomegaly – Swelling/enlargement of the liver beyond its anatomical normal size.
Hypertension - A medical condition where the pressure within the systemic arteries is raised, causing the heart to work harder than normal.
Hypertrophy - Increase in size of an organ due to an increase in the size of its cells.
Infundibulum - a funnel-shaped cavity.
Intravenous - inside veinous structures.
Microdeletion - it is a mutation whereby a small part of the chromosome/DNA sequence is omitted.
Missense mutation – it is a point mutation, whereby a single nucleotide has been changed resulting to a different codon that will lead to the formation of a different amino acid.
Murmur – sounds which are heard during a heart beat, which includes sounds of blowing, whooshing or rasping. The sound is caused by turbulent blood flow through the heart valves or near the heart.
Palliative care - care that focuses on relieving the suffering of patients rather than treating it.
Palpitation – when the heart beats Unusually, abnormally, rapidly or violently.
Patent Ductus Arteriosus - is a congenital condtion that causes the failure of the ductus arteriosus to close
Prostaglandin - a group of fatty acids which are naturally derived. Some of its roles include regulation of acid secretion from the stomach, induce platelet aggregation, control inflammation, lowering of the blood pressure, and many more.
Pulmonary Atresia - a form of congenital heart disease in which the pulmonary valve does not form properly
Pulmonary valve insufficiency - A condition where the pulmonary valve cannot prevent back flow of blood.
Shunt – A channel created surgically either via surgical reconstruction or synthetic tube. It allows blood or other bodily fluid is diverted from its normal path.
Stenosis - An abnormal narrowing, usually in relation to a tube. Examples of narrowing or a tube include blood vessel, gastrointestinal tract, respiratory tract, etc.
Stethoscope - an instrument used, especially by medical doctors, to hear ausculatory sounds made in the body, generally in the thoracic area.
Syncope – an event where there is a brief loss of consciousness which is associated with transient cerebral anemia, heart block, sudden lowering of the blood pressure, etc.; usually results in fainting.
Systolic ejection murmur - A heart murmur heard during the contraction phase of the heart.
Tet spell - A hypoxic attack due to a lack of circulating oxygen. A tet spell is characterised by shortness of breath and brieff loss of consciousness.
Thrombosis - a Coagulation or clotting in a part of the circulatory system.
Transcription factor - it regulates the expression of genes by binding to the regulatory region of the DNA and promote/inhibit the transcription of particular genes.
Trisomy - it is a type of genetic mutation where there is an extra chromosome, in this case a total of 3.
Velocardiofacial Syndrome - is a syndrome that causes cleft palate, heart defects, abnormal facial structure and learning problems, which is due to a congenital malformation.
Ventricular Septation – it is the formation of the interventricular septum in the adult heart, which separates the left and right ventricle.
Internal links
- Cardiovascular development abnormalities - a page which explains about diferent abnormalities that can arise during Cardiovascular development. Also explains about TOF.
- Advanced cardiac embryology - a page which describes, at an advanced level, the way the Cardiac system develops during embryogenesis.
- Cardiovascular system development - notes which describes the development of the Cardiovascular system.
- Cardiovascular development lecture - a lecture given by Dr Hill in 2009 in regards to cardiovascular development.
- Heart Development - Heart development lecture delivered in 2011 which shows the genetic influences on cardiac anomalies such as TOF.
- Cardiovascular System - Movies - a page which has movies on the development of Cardiovascular system
References
- ↑ <pubmed>1689816</pubmed>
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 <pubmed>19683809</pubmed>
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 <pubmed></pubmed>19144126 Cite error: Invalid
<ref>tag; name 'PMID19144126' defined multiple times with different content - ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 <pubmed>7888802</pubmed>
- ↑ 5.0 5.1 5.2 5.3 5.4 <pubmed>21251297</pubmed>
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 <pubmed>21048055</pubmed>
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 <pubmed>20091166</pubmed>
- ↑ <pubmed>12632214</pubmed>
- ↑ <pubmed>12632215</pubmed>
- ↑ 10.0 10.1 10.2 10.3 <pubmed>19948535</pubmed>
- ↑ <pubmed>16908723</pubmed>
- ↑ <pubmed>19853711</pubmed>
- ↑ 13.0 13.1 http://www.nlm.nih.gov/medlineplus/ency/article/003215.htm
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 <http://www.nhlbi.nih.gov/health/health-topics/topics/tof/signs.html
- ↑ http://www.ncbi.nlm.nih.gov/books/NBK367/
- ↑ <pubmed>14979389</pubmed>
- ↑ http://www.nlm.nih.gov/medlineplus/ency/article/001567.htm
- ↑ http://ghr.nlm.nih.gov/gene/TBX1
- ↑ http://ghr.nlm.nih.gov/gene/NKX2-5
- ↑ http://ghr.nlm.nih.gov/gene/JAG1
- ↑ <pubmed>11339373</pubmed>
- ↑ <pubmed>9570129</pubmed>
- ↑ <pubmed>20937753</pubmed>
- ↑ http://ghr.nlm.nih.gov/glossary=haploinsufficiency
- ↑ 25.0 25.1 <pubmed>1714651</pubmed>
- ↑ <pubmed>7665173</pubmed>
- ↑ <pubmed>10587520</pubmed>
- ↑ <pubmed>12427653</pubmed>
- ↑ <pubmed>9268641</pubmed>
- ↑ 30.0 30.1 <pubmed>11869892</pubmed>
- ↑ <pubmed>10556292</pubmed>
- ↑ <pubmed>11152664</pubmed>
- ↑ <pubmed>12649809</pubmed>
- ↑ <pubmed>14132270</pubmed>
- ↑ <pubmed>15934690 </pubmed>
- ↑ 36.0 36.1 <Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology. 8th edn. Saunders/Elsevier: Philadelphia, PA.>
- ↑ <pubmed>11520451</pubmed>
- ↑ Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology. 8th edn. Saunders/Elsevier: Philadelphia, PA.
- ↑ <pubmed>3068155</pubmed>
- ↑ 40.0 40.1 40.2 40.3 <pubmed>8272784</pubmed>
- ↑ http://www.nhlbi.nih.gov/health/health-topics/topics/heartmurmur/
- ↑ Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology. 8th edn. Saunders/Elsevier: Philadelphia, PA
- ↑ Braunwald E. (Editor), Heart Disease: A Textbook of Cardiovascular Medicine, Fifth Edition, p. 108, Philadelphia, W.B. Saunders Co., 1997
- ↑ http://www.nlm.nih.gov/medlineplus/ency/article/003804.htm
- ↑ http://www.nlm.nih.gov/medlineplus/ency/article/003869.htm
- ↑ Squire LF, Novelline RA (1997). Squire's fundamentals of radiology (5th ed.). Harvard University Press
- ↑ 47.0 47.1 47.2 <http://emedicine.medscape.com/article/2035949-treatment#aw2aab6b6b3>
- ↑ 48.0 48.1 <http://emedicine.medscape.com/article/2035949-treatment#a1156>
- ↑ 49.0 49.1 49.2 <pubmed>19040408</pubmed>
- ↑ 50.0 50.1 50.2 50.3 <http://www.chdinfo.com/aa/aa112397.htm>
- ↑ http://emedicine.medscape.com/article/2035949-treatment#a1156
- ↑ <pubmed>4813185</pubmed>
- ↑ < http://emedicine.medscape.com/article/2035949-treatment#aw2aab6b6b6>
- ↑ <http://emedicine.medscape.com/article/2035949-treatment#aw2aab6b6b9>
- ↑ <http://www.nhlbi.nih.gov/health/health-topics/topics/tof/livingwith.html>
- ↑ <pubmed>17178242</pubmed>
- ↑ <pubmed>16820562</pubmed>
2011 Projects: Turner Syndrome | DiGeorge Syndrome | Klinefelter's Syndrome | Huntington's Disease | Fragile X Syndrome | Tetralogy of Fallot | Angelman Syndrome | Friedreich's Ataxia | Williams-Beuren Syndrome | Duchenne Muscular Dystrolphy | Cleft Palate and Lip