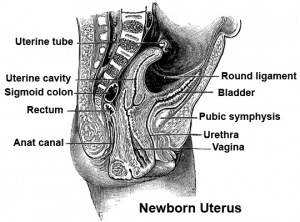
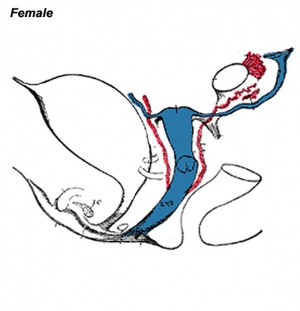
Vagina Development
| Embryology - 10 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The embryonic origin of the vagina has been a historically hotly debated issue with several different contributions and origins described. Current molecular studies show the whole vagina is derived from the Müllerian duct with bone morphogenic protein 4 (BMP4) reshaping the intermediate mesoderm-derived Müllerian duct into the vaginal primordium.[1]
One description shows the vagina arising by downward growth of Wolffian and Mullerian ducts. The sinovaginal bulbs are the caudal ends of the Wolffian ducts. Vaginal development is also under negative control of androgens. An earlier understanding was that the upper part of the vagina derived from Müllerian ducts and the lower part from the sinovaginal bulbs (formed by fusion form the vaginal plate) all derived from the urogenital sinus. The terms sinovaginal bulbs and vaginal plate were first coined by Koff in 1933.[2]
The model rodent vagina has a dual origins, cranial part is from the Müllerian duct and caudal part from the urogenital sinus. Acién's hypothesis, relates to abnormalities and the embryology of the human vagina as deriving from the Wolffian ducts and the Müllerian tubercle.
| Uterus Development Animation
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Vagina Embryology <pubmed limit=5>Vagina Embryology</pubmed> |
Paramesonephic Duct Development
The paired paramesonephic ducts (Müllerian ducts) go through a series of developmental changes recently identified as regulated by a number of molecular factors.
Initiation
Coelomic epithelium Lim1 expressing cells are specified to a duct fate.
- Lim - proteins named for 'LIN11, ISL1, and MEC3,' are defined by the possession of a highly conserved double zinc finger motif called the LIM domain.
- LIM domain-binding factors - interact with the LIM domains of nuclear proteins are capable of binding to a variety of transcription factors.
Invagination
Duct invagination induced by Wnt4 to reach the mesonephric (Wolffian)
Elongation
Cells at the leading tip proliferate and form the duct elongating to reach the cloaca (urogenital sinus). Mesonephric secretes WNT9b to guide duct elongation. Pax2 also acts in elongation and duct maintenance.
- WNT9b - member of the WNT protein family that encode cysteine-rich secreted glycoproteins that act as extracellular signaling factors.
- Pax2 - member of the paired box protein family.
- Links: OMIM - WNT9b | OMIM - Pax2 | OMIM - paired box gene
Postnatal Development
A study in mouse has identified Dicer, a riboendonuclease required for microRNA biosynthesis, to be required for postnatal growth if the female reproductive tract.[5]
Adult Dimensions
A recent study using magnetic resonance imaging (MRI) has accurately measured the dimensions of the adult vagina.[6]
- "Seventy-seven MRI scans were performed on 28 women before gel application to establish baseline vaginal measurements. Average dimensions were calculated for each woman and for the population. The influence of potential covariates (age, height, weight and parity) on these dimensions was assessed. ...Mean vaginal length from cervix to introitus was 62.7 mm. Vaginal width was largest in the proximal vagina (32.5 mm), decreased as it passed through the pelvic diaphragm (27.8 mm) and smallest at the introitus (26.2 mm)."
Abnormalities
Mayer- Rokitansky-Kuster-Hauser syndrome
(MRKH) Abnormality of development of the female genital tract: partial or complete absence (agenesis) of the uterus; absent or hypoplastic vagina; normal fallopian tubes, ovaries, normal external genitalia and normal female chromosome pattern (46, XX). Has an incidence of approximately 1 in 4500 newborn girls and has been associated with a microdeletion at 17q12.[7]
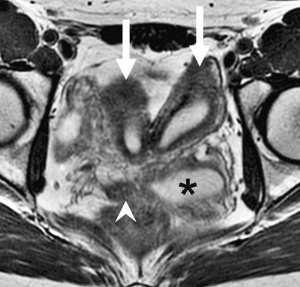
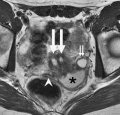
OHVIRA Syndrome
Obstructed HemiVagina and Ipsilateral Renal Anomaly with uterine didelphysis is a syndrome due to lateral non-fusion of the Mullerian ducts with asymmetric obstruction. The presence of vaginal septum also gives rise to other clinical conditions.
OHVIRA Syndrome Magnetic Resonance Images
Endocrine Disruptors
Endocrine disruptors in female reproductive tract development and carcinogenesis.[8]
References
Reviews
<pubmed>19598112</pubmed> <pubmed>16208476</pubmed> <pubmed>15467266</pubmed>
Articles
<pubmed>17532316</pubmed> <pubmed>17070514</pubmed> <pubmed>14695376</pubmed> <pubmed>12740945</pubmed> <pubmed>12449044</pubmed> <pubmed>15086027</pubmed> <pubmed>19598112</pubmed> <pubmed>15821572</pubmed> <pubmed>18391520</pubmed>
Search PubMed
Search Pubmed Oct 2010 "Vagina Embryology" All (3507) Review (251) Free Full Text (228)
Search Pubmed: Vagina Embryology | Vagina Development | vaginal plate development | Mullerian duct
Genital Links: genital | Lecture - Medicine | Lecture - Science | Lecture Movie | Medicine - Practical | primordial germ cell | meiosis | endocrine gonad | Genital Movies | genital abnormalities | Assisted Reproductive Technology | puberty | Category:Genital
| ||||
|
| Uterus Development Animation
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 10) Embryology Vagina Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Vagina_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G