Cardiovascular System - Blood Vessel Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 277: | Line 277: | ||
{{Footer}} | {{Footer}} | ||
[[Category:Cardiovascular]] [[Category:Blood]] [[Category:Mesoderm]] | [[Category:Blood Vessel]][[Category:Cardiovascular]] [[Category:Blood]] [[Category:Mesoderm]][[Category:Smooth Muscle]] | ||
Revision as of 12:41, 24 August 2015
| Embryology - 16 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Blood develops initially within the core of "blood islands" in the mesoderm. During development, there follows a series of "relocations" of the stem cells to different organs within the embryo. In the adult, these stem cells are located in the bone marrow. At the time when blood first forms, there are no bones!
Angioblasts initially form small cell clusters (blood islands) within the embryonic and extraembryonic mesoderm. These blood islands extend and fuse together making a primordial vascular network. Within these islands the peripheral cells form endothelial cells while the core cells form blood cells (haemocytoblasts).
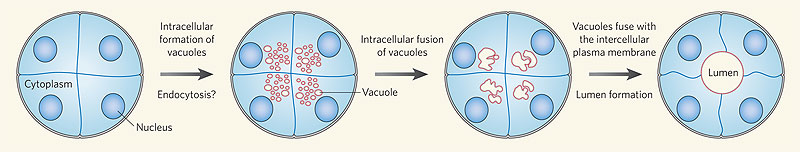
Recent work has shown that the formation of the initial endothelial tube is by a process of coalescence of cellular vacuoles within the developing endothelial cells, which fuse together without cytoplasmic mixing to form the blood vessel lumen.
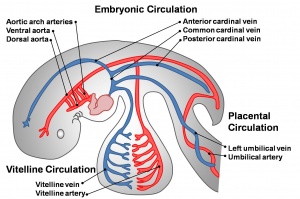
See also the related pages Placental Villi Blood Vessels and Coronary Circulation Development.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Blood Vessel Embryology <pubmed limit=5>Blood Vessel Embryology</pubmed> |
Endothelial Progenitors
Recent work has shown that the formation of the initial endothelial tube is by a process of coalescence of cellular vacuoles within the developing endothelial cells, which fuse together without cytoplasmic mixing to form the blood vessel lumen. [5]
Endothelial Tube Formation
Vessel Specification
The following data is from a recent review.[2]
Arterial Specification
| Factor | Function |
| Shh | Loss of Shh results in lack of arterial identity in zebrafish. Shh acts upstream of VEGF. |
| VEGF | VEGF acts downstream of Shh signaling to activate Notch via the PLCγ/ERK pathway in zebrafish. Mutant mice expressing only VEGF188 lack arterial differentiation. |
| Nrp1 | Null mice display impaired arterial differentiation. Nrp1 is involved in a positive feedback loop of VEGF signaling. |
| Notch | Notch acts downstream of Shh and VEGF signaling in zebrafish. Notch1; Notch4 mutant mice have abnormal vascular development. |
| Dll4 | Null mice lack arterial specification. |
| Dll1 | Null mice fail to maintain arterial identity. |
| Hey1/2 (Grl) | Null mice lack arterial specification. Lack of grl in zebrafish results in loss of arterial specification. |
| Foxc1/c2 | Foxc1; Foxc2 mutant mice lack arterial specification. Foxc1 and Foxc2 directly regulate Dll4 and Hey2 expression. Foxc1 and Foxc2 are also involved in lymphatic vessel development. |
| Sox7/18 | Lack of Sox7/18 results in loss of arterial identity in zebrafish. |
| Snrk-1 | Snrk-1 acts downstream or parallel to Notch signaling in zebrafish. |
| Dep1 | Dep1 acts upstream of PI3K in arterial specification in zebrafish. |
| Crlr | Shh regulates VEGF activity by controlling crlr expression in zebrafish. |
| EphrinB2 | Null mice lack boundaries between arteries and veins. EphrinB2 is involved in lymphatic vascular remodeling and maturation. |
Venous Specification
| Factor | Function |
| COUP-TFII | COUP-TFII suppresses arterial cell fate by inhibiting Nrp1 and Notch. COUP-TFII also interacts with Prox1 to regulate lymphatic gene expression. |
| EphB4 | Null mice lack boundaries between arteries and veins. |
Lymphatic Specification
| Factor | Function |
| Sox18 | Null mice fail to specify lymphatic endothelial cells. Sox18 induces Prox1 expression. |
| Prox1 | Prox1 induces lymphatic markers and maintains lymphatic cell identity. |
Vascular Endothelial Growth Factor
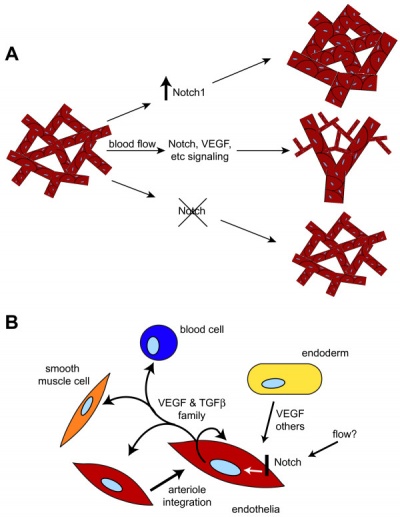
Growing blood vessels follow a gradient generated by tagret tissues/regions of Vascular Endothelial Growth Factor (VEGF) to establish a vascular bed. Recent findings suggest that Notch signaling acts as an inhibitor for this system, preventing sprouting of blood vessels.
Notch is a transmembrane receptor protein involved in regulating cell differentiation in many developing systems.
Notch and yolk sac blood vessels model[6]
Links: OMIM - VEGFA | OMIM - Notch
Regulators of Growth
The following data is from a review article on ovary vascular development.[7]
Stimulators of Angiogenisis
|
Inhibitors of Angiogenisis
|
Histology
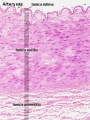
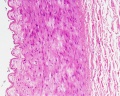
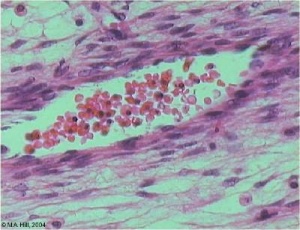
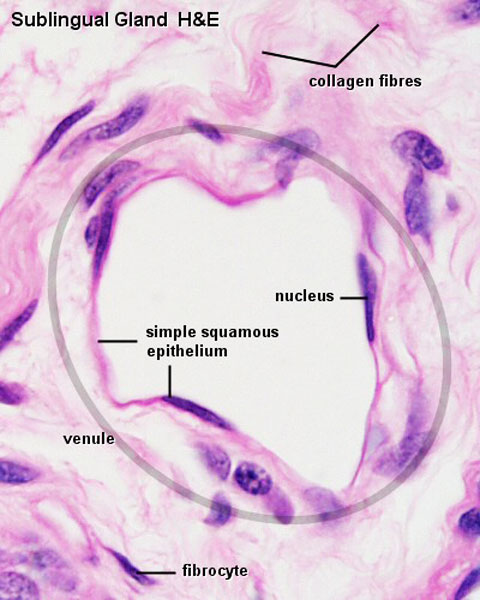
Vein Light Microscopy
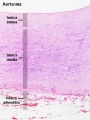
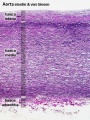
The entire developing and adult cardiovascular system (blood vessels and heart) is lined by a simple squamous epithelium. (Stain - Haematoxylin Eosin)
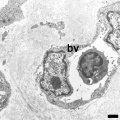
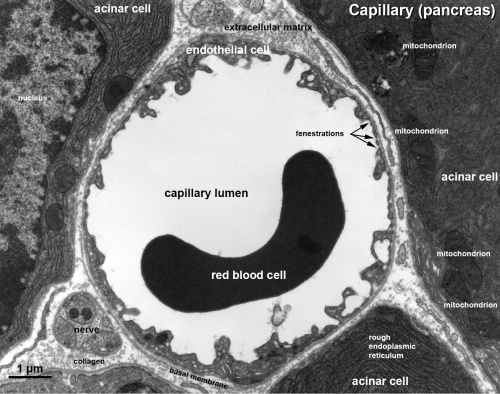
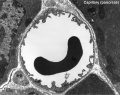
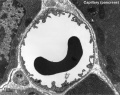
Capillaries
Electron Micrographs
endothelium detail[8]
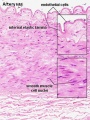
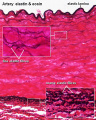
Arteries
Cardiac Blood Vessels
Earliest vessels in the heart wall develop subepicardially (beneath the outside surface of the heart) near the apex at Carnegie stage 15, which then extends centripetally and at stage 17 coronary arterial stems communicate with the aortic lumen.[9]
References
- ↑ <pubmed>25641373</pubmed>
- ↑ 2.0 2.1 <pubmed>20238301</pubmed>| PMC2899674
- ↑ <pubmed>18417617</pubmed>
- ↑ <pubmed>18245384</pubmed>
- ↑ <pubmed>11827993</pubmed>
- ↑ <pubmed>21352545</pubmed>| BMC Dev Biol.
- ↑ <pubmed>11141338</pubmed>
- ↑ <pubmed>21702933</pubmed>| PMC3141733 | BMC Cell Biol.
- ↑ <pubmed>8915616</pubmed>
Reviews
<pubmed></pubmed> <pubmed></pubmed> <pubmed>25641373</pubmed>
Articles
<pubmed></pubmed> <pubmed></pubmed> <pubmed>10948449</pubmed> <pubmed>12406884</pubmed>
Search Pubmed
Click on the listed keywords below (used to search the external database) the most current references on Medline will be displayed.
Search Pubmed: Blood Vessel Development | Blood Vessel embryology | Blood Vessel smooth muscle Development | Blood Vessel smooth muscle Development
Terms
- angioblasts stem cells in blood islands generating endothelial cells
- angiogenesis the formation of blood vessels also called vasculogenesis in the embryo
- blood islands earliest sites of blood vessel and blood cell formation, seen mainly on yolk sac chorion
- vascular endothelial growth factor (VEGF) protein growth factor family that stimulates blood vessel growth, a similar factor can be found in the placenta (PIGF).
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 16) Embryology Cardiovascular System - Blood Vessel Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Blood_Vessel_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G



![endothelium detail[8]](/embryology/images/thumb/6/6a/Blood_capillary_EM_01.jpg/120px-Blood_capillary_EM_01.jpg)