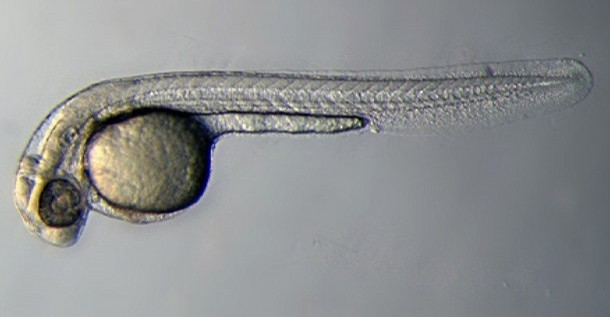
Introduction
Zebrafish or zebra danio (danio rerio) are seen as the latest "model' for embryological development studies. These embryos have the great advantage that they develop as "see through" embryos, that is, all internal development can be clearly observed from the outside in the living embryo. Much of the early modern work using this embryo model began with the papers of Kimmel.[1][2]
Several large laboratories in the US are now developing large breeding programs to carry out "knockouts" and to find spontaneous mutants of interest.
| References
Some Recent Findings
- Multifactorial Origins of Heart and Gut Defects in nipbl-Deficient Zebrafish, a Model of Cornelia de Lange Syndrome[3] "Cornelia de Lange Syndrome (CdLS) is the founding member of a class of multi-organ system birth defect syndromes termed cohesinopathies, named for the chromatin-associated protein complex cohesin, which mediates sister chromatid cohesion. Most cases of CdLS are caused by haploinsufficiency for Nipped-B-like (Nipbl), a highly conserved protein that facilitates cohesin loading. ... These findings support the view that birth defects in CdLS arise from collective effects of quantitative changes in gene expression. Interestingly, both the phenotypes and gene expression changes in nipbl morphants differed from those in mutants or morphants for genes encoding cohesin subunits, suggesting that the transcriptional functions of Nipbl cannot be ascribed simply to its role in cohesin loading.
- The zebrafish transcriptome during early development[4] "The three earliest developmental stages were similar when comparing highly expressed genes, whereas the 50% epiboly stage differed from the other three stages in the identity of highly expressed genes, number of uniquely expressed genes and enrichment of GO molecular functions. Taken together, these observations indicate a major transition in gene regulation and transcriptional activity taking place between the 512-cell and 50% epiboly stages, in accordance with previous studies."
- Genetic analysis of fin development in zebrafish identifies furin and hemicentin1 as potential novel fraser syndrome disease genes[5] " Three of them are due to mutations in zebrafish orthologues of FRAS1, FREM1, or FREM2, large basement membrane protein encoding genes that are mutated in mouse bleb mutants and in human patients suffering from Fraser Syndrome, a rare congenital condition characterized by syndactyly and cryptophthalmos. Fin blistering in a fourth group of zebrafish mutants is caused by mutations in Hemicentin1 (Hmcn1), another large extracellular matrix protein the function of which in vertebrates was hitherto unknown. Our mutant and dose-dependent interaction data suggest a potential involvement of Hmcn1 in Fraser complex-dependent basement membrane anchorage. Furthermore, we present biochemical and genetic data suggesting a role for the proprotein convertase FurinA in zebrafish fin development and cell surface shedding of Fras1 and Frem2, thereby allowing proper localization of the proteins within the basement membrane of forming fins."
- Increased Hox activity mimics the teratogenic effects of excess retinoic acid signaling[6] "Excess retinoic acid (RA) signaling can be teratogenic and result in cardiac birth defects, but the cellular and molecular origins of these defects are not well understood. Excessive RA signaling can completely eliminate heart formation in the zebrafish embryo. However, atrial and ventricular cells are differentially sensitive to more modest increases in RA signaling. Increased Hox activity, downstream of RA signaling, causes phenotypes similar to those resulting from excess RA. These results suggest that Hox activity mediates the differential effects of ectopic RA on atrial and ventricular cardiomyocytes and may underlie the teratogenic effects of RA on the heart."
|
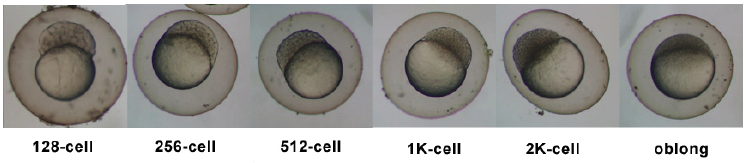
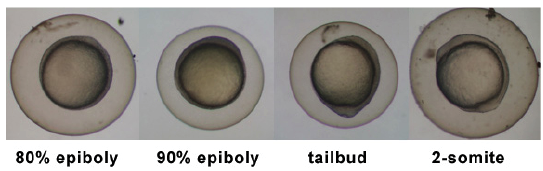
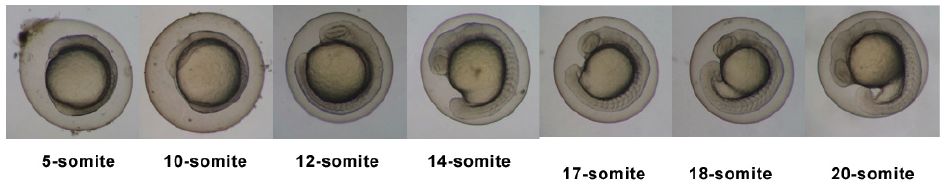
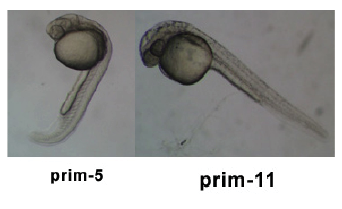
Timeline and Stages of Embryonic Development
Molecular
Fibroblast Growth Factor
- Fgf8 and Fgf3 - regulating the segmentation of the pharyngeal endoderm into pouches. [7]
- Fgf24 and Fgf8 - promotes posterior mesodermal development.[8]
- Sox9 - required for cartilage morphogenesis.[9]
References
- ↑ <pubmed>7229136</pubmed>
- ↑ <pubmed>3077108</pubmed>
- ↑ <pubmed>22039349</pubmed>
- ↑ <pubmed>21609443</pubmed>
- ↑ <pubmed>20419147</pubmed>
- ↑ <pubmed>19384962</pubmed>
- ↑ <pubmed>15509770</pubmed>
- ↑ <pubmed>12925590</pubmed>
- ↑ <pubmed>12397114</pubmed>
Reviews
Articles
Search Pubmed
Search Pubmed: Zebrafish Development
Additional Images
Non-mammalian VEGF Receptors
External Links
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Zebrafish Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Zebrafish_Development
- What Links Here?
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G