Zebrafish Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 13: | Line 13: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
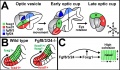
* '''Construction of a vertebrate embryo from two opposing morphogen gradients'''<ref name="PMID24700857"><pubmed>24700857</pubmed></ref> "Here, we show that opposing gradients of bone morphogenetic protein (BMP) and Nodal, two transforming growth factor family members that act as morphogens, are sufficient to induce molecular and cellular mechanisms required to organize, in vivo or in vitro, uncommitted cells of the zebrafish blastula animal pole into a well-developed embryo." | * '''Construction of a vertebrate embryo from two opposing morphogen gradients'''<ref name="PMID24700857"><pubmed>24700857</pubmed></ref> "Here, we show that opposing gradients of bone morphogenetic protein (BMP) and Nodal, two transforming growth factor family members that act as morphogens, are sufficient to induce molecular and cellular mechanisms required to organize, in vivo or in vitro, uncommitted cells of the zebrafish blastula animal pole into a well-developed embryo." [[Developmental_Signals_-_Bone_Morphogenetic_Protein|BMP]] | ||
* '''FishFace: interactive atlas of zebrafish craniofacial development at cellular resolution'''<ref name="PMID23714426"><pubmed>23714426</pubmed></ref> "We present the [https://www.facebase.org/fishface/home Fish Face Atlas], an online, 3D-interactive atlas of craniofacial development in the zebrafish Danio rerio. Alizarin red-stained skulls scanned by fluorescent optical projection tomography and segmented into individual elements provide a resource for understanding the 3D structure of the zebrafish craniofacial skeleton." See also [[#Online Atlases|Online Zebrafish Atlases]] | * '''FishFace: interactive atlas of zebrafish craniofacial development at cellular resolution'''<ref name="PMID23714426"><pubmed>23714426</pubmed></ref> "We present the [https://www.facebase.org/fishface/home Fish Face Atlas], an online, 3D-interactive atlas of craniofacial development in the zebrafish Danio rerio. Alizarin red-stained skulls scanned by fluorescent optical projection tomography and segmented into individual elements provide a resource for understanding the 3D structure of the zebrafish craniofacial skeleton." See also [[#Online Atlases|Online Zebrafish Atlases]] | ||
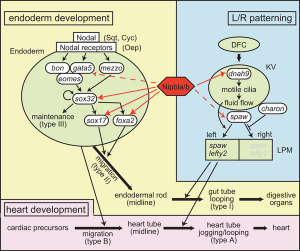
* '''Multifactorial Origins of Heart and Gut Defects in nipbl-Deficient Zebrafish, a Model of Cornelia de Lange Syndrome'''<ref name="PMID22039349"><pubmed>22039349</pubmed></ref> "Cornelia de Lange Syndrome (CdLS) is the founding member of a class of multi-organ system birth defect syndromes termed cohesinopathies, named for the chromatin-associated protein complex cohesin, which mediates sister chromatid cohesion. Most cases of CdLS are caused by haploinsufficiency for Nipped-B-like (Nipbl), a highly conserved protein that facilitates cohesin loading. ... These findings support the view that birth defects in CdLS arise from collective effects of quantitative changes in gene expression. Interestingly, both the phenotypes and gene expression changes in nipbl morphants differed from those in mutants or morphants for genes encoding cohesin subunits, suggesting that the transcriptional functions of Nipbl cannot be ascribed simply to its role in cohesin loading. (OMIM - [http://omim.org/entry/122470 CDLS1] | [http://omim.org/entry/300590 CDLS2] | [http://omim.org/entry/610759 CDLS3]) | * '''Multifactorial Origins of Heart and Gut Defects in nipbl-Deficient Zebrafish, a Model of Cornelia de Lange Syndrome'''<ref name="PMID22039349"><pubmed>22039349</pubmed></ref> "Cornelia de Lange Syndrome (CdLS) is the founding member of a class of multi-organ system birth defect syndromes termed cohesinopathies, named for the chromatin-associated protein complex cohesin, which mediates sister chromatid cohesion. Most cases of CdLS are caused by haploinsufficiency for Nipped-B-like (Nipbl), a highly conserved protein that facilitates cohesin loading. ... These findings support the view that birth defects in CdLS arise from collective effects of quantitative changes in gene expression. Interestingly, both the phenotypes and gene expression changes in nipbl morphants differed from those in mutants or morphants for genes encoding cohesin subunits, suggesting that the transcriptional functions of Nipbl cannot be ascribed simply to its role in cohesin loading. (OMIM - [http://omim.org/entry/122470 CDLS1] | [http://omim.org/entry/300590 CDLS2] | [http://omim.org/entry/610759 CDLS3]) | ||
Revision as of 17:25, 22 April 2014
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Zebrafish or zebra danio (danio rerio) are seen as the latest "model' for embryological development studies. These embryos have the great advantage that they develop as "see through" embryos, that is, all internal development can be clearly observed from the outside in the living embryo. Much of the early modern work using this embryo model began with the papers of Kimmel.[1][2]
Several large laboratories in the US are now developing large breeding programs to carry out "knockouts" and to find spontaneous mutants of interest.
| Fish Links: Zebrafish Development | Medaka Development | Salmon Development | Movie - Zebrafish Heart | Student Group Project - Zebrafish | Recent References | Category:Zebrafish | Category:Medaka |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Zebrafish Embryology <pubmed limit=5>Zebrafish Embryology</pubmed> |
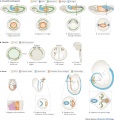
Timeline and Stages of Embryonic Development
| Duration | Period Name | Image |
| 0 - 0.75 hrs | Zygote Period | 
|
| 0.75 - 2.25 hrs | Cleavage Period | 
|
| 2.25 - 5.25 hrs | Blastula Period | 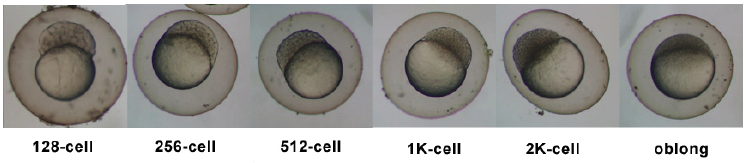
|
| 5.25 - 10.33 hrs | Gastrula Period | 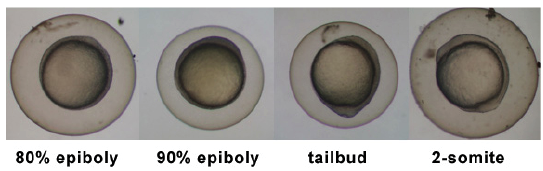
|
| 10.33 - 24 hrs | Segmentation Period | 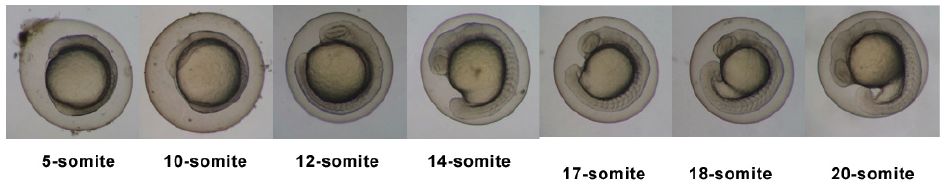
|
| 24 - 48 hrs | Pharyngula Period | 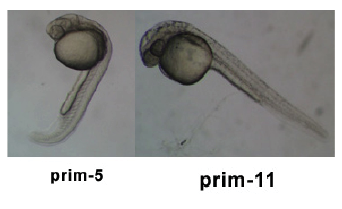
|
| 48-72 hrs | Hatching Period | 
|
| 72 hrs - 30 Days | Larval Period | 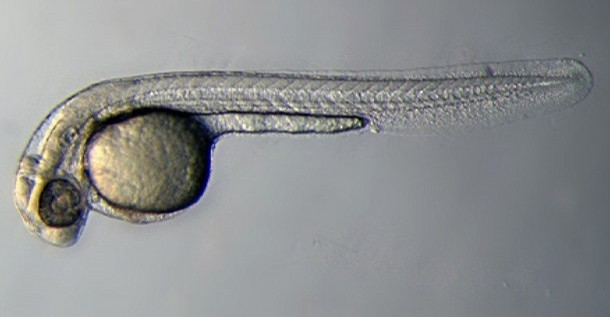
|
Skull

|
Zebrafish Skull Neural Crest Contribution[8]
|
Molecular
Fibroblast Growth Factor
- Fgf8 and Fgf3 - regulating the segmentation of the pharyngeal endoderm into pouches. [9]
- Fgf24 and Fgf8 - promotes posterior mesodermal development.[10]
- Sox9 - required for cartilage morphogenesis.[11]
References
- ↑ <pubmed>7229136</pubmed>
- ↑ <pubmed>3077108</pubmed>
- ↑ 3.0 3.1 <pubmed>22039349</pubmed>
- ↑ <pubmed>24700857</pubmed>
- ↑ <pubmed>23714426</pubmed>
- ↑ <pubmed>21609443</pubmed>
- ↑ <pubmed>20419147</pubmed>
- ↑ Kague E, Gallagher M, Burke S, Parsons M, Franz-Odendaal T, et al. (2012) Skeletogenic Fate of Zebrafish Cranial and Trunk Neural Crest. PLoS ONE 7(11): e47394. doi:10.1371/journal.pone.0047394 PLoS ONE
- ↑ <pubmed>15509770</pubmed>
- ↑ <pubmed>12925590</pubmed>
- ↑ <pubmed>12397114</pubmed>
Journals
Zebrafish "is the only peer-reviewed journal to focus on the zebrafish, which has numerous valuable features as a model organism for the study of vertebrate development. Due to its prolific reproduction and the external development of the transparent embryo, the zebrafish is a prime model for genetic and developmental studies, as well as research in toxicology and genomics. While genetically more distant from humans, the vertebrate zebrafish nevertheless has comparable organs and tissues, such as heart, kidney, pancreas, bones, and cartilage." [jour PubMed listing]
Reviews
<pubmed>21501748</pubmed> <pubmed>20674361</pubmed> <pubmed>19557689</pubmed> <pubmed>19371733</pubmed> <pubmed>18992377</pubmed>
Articles
Search Pubmed
Search Pubmed: Zebrafish Development
Additional Images
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- NIH NIH Zebrafish Initiative
- ZFIN - The Zebrafish Model Organism Database
- Keller at European Molecular Biology Laboratory, Germany Movies - Reconstruction of zebrafish early embryonic development by scanned light sheet microscopy
- YouTube Timelapse recording of about 18 hours of embryonic development of the zebrafish with some annotation
Online Atlases
- Fish Face Atlas 3D-interactive atlas of craniofacial development in the zebrafish Danio rerio.
- Zebrafish Atlas
- 3D Atlas of Zebrafish Vasculature Anatomy
- Zebrafish Brain Atlas
- Atlas of Zebrafish Anatomy
- Atlas of Zebrafish Development
- Zebrafish Anatomy Portal
- FishNet 3D developmental atlas
| Animal Development: axolotl | bat | cat | chicken | cow | dog | dolphin | echidna | fly | frog | goat | grasshopper | guinea pig | hamster | horse | kangaroo | koala | lizard | medaka | mouse | opossum | pig | platypus | rabbit | rat | salamander | sea squirt | sea urchin | sheep | worm | zebrafish | life cycles | development timetable | development models | K12 |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Zebrafish Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Zebrafish_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G