User:Z3332337: Difference between revisions
| Line 14: | Line 14: | ||
Lab 5 | Lab 5 | ||
--[[User:Z3332337|Z3332337]] 10:19, 22 August 2012 (EST) | --[[User:Z3332337|Z3332337]] 10:19, 22 August 2012 (EST) | ||
Lab 6 | |||
--[[User:Z3332337|Z3332337]] 10:15, 29 August 2012 (EST) | |||
---- | ---- | ||
Revision as of 10:15, 29 August 2012
Lab Attendance
Lab 1 --Z3332337 11:49, 25 July 2012 (EST)
Lab 2 --Z3332337 10:15, 1 August 2012 (EST)
Lab 3 --Z3332337 10:17, 8 August 2012 (EST)
Lab 4 --Z3332337 10:24, 15 August 2012 (EST)
Lab 5 --Z3332337 10:19, 22 August 2012 (EST)
Lab 6 --Z3332337 10:15, 29 August 2012 (EST)
Lab 1 Assessment
Article Summary
Effect of the method of conception and embryo transfer procedure on mid-gestation placenta and fetal development in an IVF mouse model.
This article discusses the effects of using In Vitro Fertilisation (IVF) against "In Vivo" mouse models on the fetal development, using birthweight as the a quantifiable variable.
This article showed that IVF fetuses had a lower birthweight than fetuses from In vivo pregnancies.
They implanted sites for all IVF mice had similar implantion sites, however this study shows that IVF mice have higher abortion rates along with a lower birthweight when compared with in vivo embryos. It shows here that IVF embryos are more likely to be delayed in reaching the blastocyst stage when compared to "in vivo". They also show delayed development after the blastocyst stage which may be related to the low birthweight of IVF embryos.
Reference: Delle Piane, L., Lin, W., Liu, X., Donjacour, A., Minasi, P., Revelli, A., Maltepe, E. and Rinaudo, P.F. (2010). Effect of the method of conception and embryo transfer procedure on mid-gestation placenta and fetal development in an IVF mouse model. European Society of Human Reproduction and Embryology , 25 (8), 2039-2046.
IVF Nobel Prize
Professor Robert Edwards won the Nobel Prize in Medicine in 2010.
http://www.abc.net.au/news/2010-10-04/father-of-ivf-wins-nobel-prize/2285078
Lab 2 Assessment
Detection of methylation by zygote staining
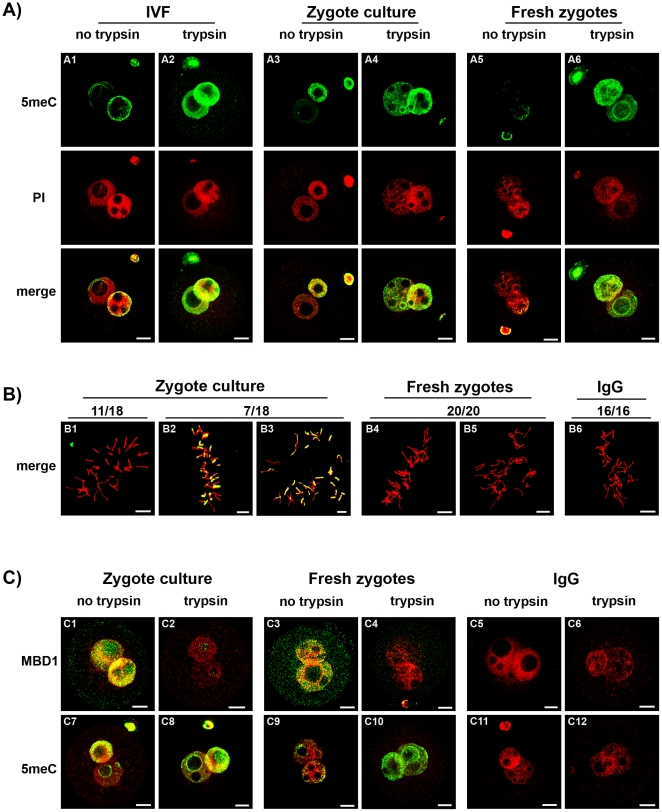
Asymmetric anti-5meC staining of the male and female pronucleus after acid-pretreatment has been reported [5], [6], [20], [21] yet was not confirmed by this study. The zygotes used in past studies were commonly generated by in vitro fertilization or subjected to culture in vitro (which provides logistic advantages for the feasibility of such studies). After antigenic unmasking with acid, the smaller (female) pronucleus in zygotes produced by in vitro fertilization (Fig 7A1) and or cultured in vitro (Fig 7A3) showed more anti-5meC staining compared to those collected directly from the oviduct (Fig 7A5). After antigenic unmasking by acid and trypsin, however, high levels of anti-5meC staining were consistently observed in both pronuclei of IVF (Fig 7A2), cultured (Fig 7A4) and fresh PN5 zygotes (Fig 7A6). Analysis of metaphase zygotes showed that culture from the early zygote stage caused variable levels of anti-5meC staining to persist in acid-only treated zygotes (Fig 7B). The level of methylation was assessed further by comparing staining with anti-MBD1 and anti-5meC in fresh and cultured zygotes (Fig 7C). This analysis showed that a similarly high level of MBD1 staining was observed in PN5 cultured (Fig 7C1) and fresh (Fig 7C3) zygotes, yet 5meC staining persisted in an asymmetrical fashion in cultured (Fig 7C7) but not fresh (Fig 7C9) zygotes. After acid and trypsin unmasking the MBD1 staining was lost from both treatments (Fig 7C2,4) and resulted in a similarly high level of staining with anti-5meC in both cultured and fresh zygotes (Fig 7C8,10). No staining was detected with non-immune control antisera for either antibody (Fig 7C5,6 and C11,12). The current results show that manipulation of the early embryo interferes with the maturational changes in zygotic chromatin that results in acid-resistant antigenic masking of 5meC, and this reduced level of masking was greatest in the female pronucleus giving an artifactual appearance of asymmetric demethylation.
Reference: Li, Y., & O'Neill, C. (2012). Persistence of cytosine methylation of DNA following fertilisation in the mouse. PLoS One., 7(1), e30687. Epub 32012 Jan 30626. Copyright Li, O'Neill. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Identify a Protein Associated with Implantation
A protein associated implantation is Proprotein 6 Convertase (PC6), a serine protease. It is involved in endometrial receptivity and is integral for cellular remodelling by cleavage of scaffolding protein ezrin-radixin-moesin binding phosphoprotein 50 (EBP50). Cleavage event allows for the ezrin protein to integrate the actin cytoskeleton and the plasma membrane, increasing chances of binding and therefore implantation. Knockout mice were used to down-regulate PC6 and results in failure of cleavage of EBP50 and therefore a decrease of healthy embryo implantation when compared to the wild type mice.
Lab 3 Assessment
1. Identify the difference between "gestational age" and "post-fertilisation age" and explain why clinically "gestational age" is used in describing human development.
The "gestational age" is any time between conception and birth, typically measured from the woman's last menstrual period until the current time. Whereas, "post-fertilisational age" is specifically the time after the ovum is fertilised.
"Gestational age" is used to in describing human development because it is measures pregnancy in weeks, as a pregnancy typically lasts 38-42 weeks.
http://www.parentsconnect.com/pregnancy/trying-to-conceive/Gestational_Fetal_Age.html
2. Identify using histological descriptions at least 3 different types of tissues formed from somites.
Somites which develop from the paraxial mesoderm, give rise to 3 different types of tissues: dermatome, myotome and sclerotome tissues.
Dorsolaterally there is the: Dermatome which forms the dermis and and contributing fibroblasts, & Myotome which forms the myoblasts (primordial muscle cells).
Ventromedially there is the: Sclerotome which forms the vertebrae and the ribs.
Lab 4 Assessment
Amniocentesis: is where amniotic fluid is extracted with a needle is inserted into the amniotic cavity through the abdominal wall, guided by an ultrasound. This test detects for levels of metabolic by-products, including alpha-fetoprotein. This test is usually administered between 14-16 weeks gestation.
Elevated levels of the afore-mentioned protein may indicate abnormalities regarding the neural tube, such as spina bifida (unfused vertebrae over spinal cord) and anencephaly (the end of the neural tube fails to fuse).
Chorionic Villus Sampling (CVS): is where tissue is removed from the chorion layer of the uterine wall, by a needle or a catheter through the cervix, also guided by an ultrasound. This tissue is then karyotyped for molecular and genetic testing, for abnormalities such as Trisomy 21 (aka: Down Syndrome). This test is administered usually in weeks 10-12 of gestation.
It should be noted that abnormalities detected in CVS may be confirmed through amniocentesis.
Reference:
Larsen's human embryology 4th ed. Schoenwolf, Gary C; Larsen, William J, (William James). Philadelphia, PA : Elsevier/Churchill Livingstone, c2009.
2) Identify a paper that uses cord stem cells therapeutically and write a brief description of the paper's findings.
Roura, Santiago, Josep-Maria Pujal, and Antoni Bayes-Genis. "Umbilical Cord Blood for Cardiovascular Cell Therapy: From Promise to Fact." Annals of the New York Academy of Sciences 1254, no. 1 (2012): 66-70.
Umbilical Cord Blood (UBC) is a valuable stem cell source and has a great potential for vascular growth and repair. This article indicated that UBC may be used as an alternate source of hematopoietic cytokines (CD133+) which stimulate angiogenesis.
It has been indicated in the therapy for an array of diseases including the reconstitution of a defective immune system, neurological improvement of cerebral palsy, and possibly the treatment of type 1 diabetes. It has also shown improvements in reducing 'graft vs. host' disease in transplant patients due to the presence of Multipotent Stromal Cells (MSCs) found in UCB. MSCs are also found in bone marrow.
Research on animal models through fluorescent angiography has shown that UCB may improve, if not recover, endothelial function from "UCB-MSC differentiation and new microcirculatory vessel formation".