Trophoblast: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 24: | Line 24: | ||
:'''Links:''' [[Blastocyst]] | [[Implantation]] | [[Week 2]] | [[Week 3]] | [[Placenta Development]] | [[Prenatal Diagnosis]] | [[Trophoblast - Protein Expression]] | [[Ectopic Implantation Research]] | :'''Links:''' [[Blastocyst]] | [[Implantation]] | [[Week 2]] | [[Week 3]] | [[Placenta Development]] | [[Prenatal Diagnosis]] | [[Trophoblast - Protein Expression]] | [[Ectopic Implantation Research]] | ||
<br> | |||
{{Blastocyst Links}} | |||
<br> | |||
{{Placenta Links}} | {{Placenta Links}} | ||
[[File:Extravillous trophoblasts week 5.5.jpg|thumb|alt=Extravillous trophoblasts week 5.5|Extravillous trophoblasts (week 5.5)<ref name=PMID28081266><pubmed>28081266</pubmed></ref>]] | [[File:Extravillous trophoblasts week 5.5.jpg|thumb|alt=Extravillous trophoblasts week 5.5|Extravillous trophoblasts (week 5.5)<ref name=PMID28081266><pubmed>28081266</pubmed></ref>]] | ||
| Line 31: | Line 33: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Gene expression patterns associated with human placental trophoblast differentiation''' | * '''Gene expression patterns associated with human placental trophoblast differentiation'''{{#pmid:29329728|PMID29329728}} "Cell fusion is a hallmark of placental trophoblast cell differentiation and the mature syncytiotrophoblasts play essential roles for fetal-maternal exchange and production of pregnancy-related hormones. Using a well-established in vitro trophoblast differentiation model, we performed a microarray analysis on mRNA expression in trophoblast and syncytiotrophoblast cell cultures. Dramatic changes in gene expression patterns were detected during trophoblast differentiation. Real-time PCR analysis confirmed the reliability of the microarray data. As many as 3524 novel and known genes have been found to be up- or down-regulated for >2-fold. A number of cell cycle regulator including CDC6, CDC20, Cyclins B2, L1 and E2, were down-regulated in the syncytiotrophoblast, providing a mechanism for the loss of mitotic activity during trophoblast differentiation." | ||
* '''A comparative study of five physiological key parameters between four different human trophoblast'''-derived cell lines{{#pmid:28724925|PMID28724925}} "Here, we present a comparative study on the functional aspects of the choriocarcinoma cell lines BeWo, JAR and Jeg-3, as well as the first trimester trophoblast cell line ACH-3P as placental in vitro barrier models for endocrine and transport studies. Functional assays including tight junction immunostaining, sodium fluorescein retardation, trans epithelial resistance, glucose transport, hormone secretion as well as size-dependent polystyrene nanoparticle transport were performed using the four cell types to evaluate key functional parameters of each cell line to act a relevant in vitro placental barrier model." | |||
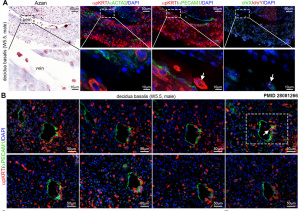
* '''Human extravillous trophoblasts penetrate decidual veins and lymphatics before remodeling spiral arteries during early pregnancy'''{{#pmid:28081266|PMID28081266}} "To study the invasion of maternal endometrium between (W) 5.5 - 12 weeks of gestation by EVTs, we combined fluorescence in situ hybridization, immunofluorescence and immunohistochemistry to determine the presence of (male) EVTs in the vasculature of the maternal decidua. We observed that interstitial mononuclear EVTs directly entered decidual veins and lymphatics from W5.5. This invasion of decidual veins and lymphatics occurred long before endovascular EVTs remodelled decidual spiral arteries." | |||
* '''The trophoblast plug during early pregnancy: a deeper insight'''{{#pmid:27510415|PMID27510415}} "During the first trimester of pregnancy, foetal endovascular trophoblasts invade into maternal spiral arteries, accumulate and form plugs in the lumen of the vessels. These plugs only allow blood plasma to seep through. Hence, during the first trimester of pregnancy, a first flow of fluids through the placental intervillous space is established, resulting in a physiological oxygen gradient between mother and foetus. The trophoblast plugs block spiral arteries until the beginning of the second trimester (11-14 weeks)." | |||
* ''' | * '''MSX2 Induces Trophoblast Invasion in Human Placenta'''{{#pmid:27088357|PMID27088357}} "Normal implantation depends on appropriate trophoblast growth and invasion. Meanwhile, overexpression of MSX2 and treatment with the MSX2-specific siRNA resulted in decreased and increased E-cadherin expression, respectively, in JEG-3 cells. Lastly, the protein expression levels of MSX2 were significantly lower in human pre-eclamptic placental villi than in the matched control placentas. Collectively, our results suggest that MSX2 may induce human trophoblast cell invasion, and dysregulation of MSX2 expression may be associated with pre-eclampsia." | ||
* '''OVO-like 1 regulates progenitor cell fate in human trophoblast development'''<ref>Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development [http://www.pnas.org/content/112/45/E6175.abstract.html?etoc PNAS]</ref> "Epithelial barrier integrity is dependent on progenitor cells that either divide to replenish themselves or differentiate into a specialized epithelium. This paradigm exists in human placenta, where cytotrophoblast cells either propagate or undergo a unique differentiation program: fusion into an overlying syncytiotrophoblast. Syncytiotrophoblast is the primary barrier regulating the exchange of nutrients and gases between maternal and fetal blood and is the principal site for synthesizing hormones vital for human pregnancy. How trophoblast cells regulate their differentiation into a syncytium is not well understood. In this study, we show that the transcription factor OVO-like 1 (OVOL1), a homolog of Drosophila ovo, regulates the transition from progenitor to differentiated trophoblast cells. OVOL1 is expressed in human placenta and was robustly induced following stimulation of trophoblast differentiation. Disruption of OVOL1 abrogated cytotrophoblast fusion and inhibited the expression of a broad set of genes required for trophoblast cell fusion and hormonogenesis. OVOL1 was required to suppress genes that maintain cytotrophoblast cells in a progenitor state, including MYC, ID1, TP63, and ASCL2, and bound specifically to regions upstream of each of these genes." [http://www.omim.org/entry/602313 OMIM - OVOL1] | * '''OVO-like 1 regulates progenitor cell fate in human trophoblast development'''<ref>Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development [http://www.pnas.org/content/112/45/E6175.abstract.html?etoc PNAS]</ref> "Epithelial barrier integrity is dependent on progenitor cells that either divide to replenish themselves or differentiate into a specialized epithelium. This paradigm exists in human placenta, where cytotrophoblast cells either propagate or undergo a unique differentiation program: fusion into an overlying syncytiotrophoblast. Syncytiotrophoblast is the primary barrier regulating the exchange of nutrients and gases between maternal and fetal blood and is the principal site for synthesizing hormones vital for human pregnancy. How trophoblast cells regulate their differentiation into a syncytium is not well understood. In this study, we show that the transcription factor OVO-like 1 (OVOL1), a homolog of Drosophila ovo, regulates the transition from progenitor to differentiated trophoblast cells. OVOL1 is expressed in human placenta and was robustly induced following stimulation of trophoblast differentiation. Disruption of OVOL1 abrogated cytotrophoblast fusion and inhibited the expression of a broad set of genes required for trophoblast cell fusion and hormonogenesis. OVOL1 was required to suppress genes that maintain cytotrophoblast cells in a progenitor state, including MYC, ID1, TP63, and ASCL2, and bound specifically to regions upstream of each of these genes." [http://www.omim.org/entry/602313 OMIM - OVOL1] | ||
* '''Establishment of Trophoblast Stem Cells under Defined Culture Conditions in Mice''' | |||
* '''Establishment of Trophoblast Stem Cells under Defined Culture Conditions in Mice'''{{#pmid:25203285|PMID25203285}} "Here, we report that addition of FGF2, activin A, XAV939, and Y27632 are necessary and sufficient for derivation of TS cells from both of E3.5 blastocysts and E6.5 early postimplantation extraembryonic ectoderm. Moreover, the undifferentiated TS cell state can be stably maintained in chemically defined culture conditions. Cells derived in this manner expressed TS cell marker genes, including Eomes, Elf5, Cdx2, Klf5, Cdh1, Esrrb, Sox2, and Tcfap2c; differentiated into all trophoblast subtypes (trophoblast giant cells, spongiotrophoblast, and labyrinthine trophoblast) in vitro; and exclusively contributed to trophoblast lineages in chimeric animals. This delineation of minimal requirements for derivation and self-renewal provides a defined platform for precise description and dissection of the molecular state of TS cells." | |||
|} | |} | ||
| Line 55: | Line 61: | ||
|- | |- | ||
| | | | ||
* '''Cell fusion induced by ERVWE1 or measles virus causes cellular senescence.''' | * '''Cell fusion induced by ERVWE1 or measles virus causes cellular senescence.'''{{#pmid:24186980|PMID24186980}} "Cellular senescence limits proliferation of potentially detrimental cells, preventing tumorigenesis and restricting tissue damage. However, the function of senescence in nonpathological conditions is unknown. We found that the human placental syncytiotrophoblast exhibited the phenotype and expressed molecular markers of cellular senescence. During embryonic development, ERVWE1-mediated cell fusion results in formation of the syncytiotrophoblast, which serves as the maternal/fetal interface at the placenta." | ||
* '''Early Expression of Pregnancy-Specific Glycoprotein 22 (PSG22) by Trophoblast Cells Modulates Angiogenesis in Mice''' | |||
* '''Involvement of transcription factor NR2F2 in human trophoblast differentiation''' | * '''Early Expression of Pregnancy-Specific Glycoprotein 22 (PSG22) by Trophoblast Cells Modulates Angiogenesis in Mice'''{{#pmid:22423048|PMID22423048}} "Mouse and human pregnancy-specific glycoproteins (PSG) are known to exert immunomodulatory functions during pregnancy by inducing maternal leukocytes to secrete anti-inflammatory cytokines that promote a tolerogenic decidual microenvironment. Many such anti-inflammatory mediators also function as pro-angiogenic factors, which, along with the reported association of -murine PSG - with the uterine vasculature, suggest that PSG may contribute to the vascular adaptations necessary for successful implantation and placental development. ...Therefore, our findings indicate that despite the independent evolution and expansion of human and rodent PSG, members in both families have conserved functions, which include their ability to induce anti-inflammatory cytokines and pro-angiogenic factors as well as to induce the formation of capillary structures by endothelial cells. In summary, our results indicate that PSG22, the most abundant PSG expressed during mouse early pregnancy, is likely a major contributor to the establishment of a successful pregnancy." | ||
* '''Angiogenic growth factors''' | |||
* '''Involvement of transcription factor NR2F2 in human trophoblast differentiation'''{{#pmid:20195529|PMID20195529}} "During the in vitro differentiation of human villous cytotrophoblast (CTB) cells to a syncytiotrophoblast (STB) phenotype, mRNA levels for the nuclear hormone receptor NR2F2 (ARP-1, COUP-TFII) increase rapidly, reaching a peak at day 1 of differentiation that is 8.8-fold greater than that in undifferentiated CTB cells." | |||
* '''Angiogenic growth factors'''{{#pmid:20338637|PMID20338637}} "Fourteen angiogenic growth factors were measured by multiplex growth factor analysis or ELISA in tissue culture supernatants from EVT and CTB from pregnancies at 8-10 and 12-14 weeks' gestation. Gestational age and cell type differences were observed. Extravillous trophoblast (EVT) and villous cytotrophoblast (CTB) are major producers of angiogenic growth factors that likely contribute to placental vascular development and spiral artery remodelling." | |||
|} | |} | ||
==Trophoblast and Placental Villi== | ==Trophoblast and Placental Villi== | ||
[[File:Early placental structure.jpg|Early placental structure.jpg]] | [[File:Early placental structure.jpg|Early placental structure.jpg]] | ||
Early placental development cartoon showing trophoblast contribution to placental villi. | Early placental development cartoon showing trophoblast contribution to placental villi.{{#pmid:20107601|PMID20107601}} | ||
'''Legend''' | '''Legend''' | ||
| Line 73: | Line 82: | ||
==Cytotrophoblasts== | ==Cytotrophoblasts== | ||
Hyperglycosylated human Chorionic Gonadotropin (hCG) promotes the growth of cytotrophoblast cells and the endometrial invasion by these cells during implantation. | Hyperglycosylated human Chorionic Gonadotropin (hCG) promotes the growth of cytotrophoblast cells and the endometrial invasion by these cells during implantation.{{#pmid:20735820|PMID20735820}} | ||
==Extravillous Cytotrophoblasts== | ==Extravillous Cytotrophoblasts== | ||
Maintain open maternal spiral arteries emptying into the maternal lacunae | Maintain open maternal spiral arteries emptying into the maternal lacunae{{#pmid:20359743|PMID20359743}} | ||
At term, depending on maternal decidual localization, there are several identifiable subtypes: | At term, depending on maternal decidual localization, there are several identifiable subtypes:{{#pmid:28081266|PMID28081266}} | ||
* interstitial mononuclear (and multinuclear) - dispersed in decidual mesenchyme | * interstitial mononuclear (and multinuclear) - dispersed in decidual mesenchyme | ||
* endovascular - in spiral arteries lumen (or replacing endothelial cells) | * endovascular - in spiral arteries lumen (or replacing endothelial cells) | ||
Revision as of 14:00, 21 March 2018
| Embryology - 19 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
(Greek, trophe = "nutrition" and -blast, a primordial cell) During early development the trophoblast cells have an important contribution to extraembryonic tissues (fetal placenta and membranes) and processes of early development (adplantation, implantation and endocrine support of pregnancy).
|
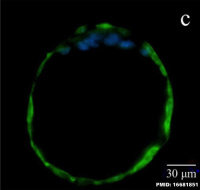
|
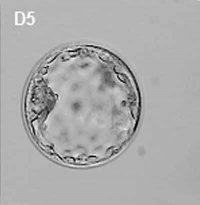
| Human Blastocyst (day 5), trophoblast cells form the peripheral flattened epithelial layer of cells directly under the zona pellucida.[1] | Mouse blastocyst labelled with trophoblast marker.[2] |
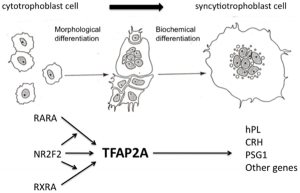
In humans, week 1 blastocyst formation the outer layer of cells (adjacent to the zona pellucida) form a flat squamous epithelial layer of cells, the trophoblast layer. Week 2 following blastocyst hatching the trophoblast layer is involved with initial adhesion to the uterine wall and subsequent implantation within the wall. During this period the trophoblast layer proliferates and differentiates into two distinct layers (syncitiotrophoblast and cytotrophoblast).
Following implantation, trophoblast cells continue to contribute to the placenta. Prenatal diagnosis by invasive chorionic villus sampling and [[ non-invasive cervical cell sampling[3] uses mainly DNA from these cells.
| Historical - Who named the trophoblast cell? |
|---|
| From an 1905 paper on tubal implantation - "Hubrecht in 1889 published a monograph on the placentation of the hedgehog, showing that the developing ovum in this animal, after sequestration in a crypt, becomes imbedded in the sub-epithelial portion of the mucoa of the uterus by the action of the non-foetal ectodermal cells of the blastocyst. These cells Hubrecht named trophoblast because of their nutritive function, for by their agency the blastocyst “burrows” into the maternal tissues, both destroying and absorbing them." |
- Links: Blastocyst | Implantation | Week 2 | Week 3 | Placenta Development | Prenatal Diagnosis | Trophoblast - Protein Expression | Ectopic Implantation Research
| Blastocyst Links: blastocyst | morula | fertilization | Week 1 | trophoblast | implantation | Human Day 3-6 Movie | Mouse Model Movie | Category:Blastocyst |
| Molecular: Hippo | Notch |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Trophoblast <pubmed limit=5>Trophoblast</pubmed> |
| Older papers |
|---|
|
Trophoblast and Placental Villi
Early placental development cartoon showing trophoblast contribution to placental villi.[15]
Legend
- SYN - syncytiotrophoblasts
- sCTB - subsyncytial cytotrophoblasts (this layer grows increasingly discontinuous in later trimesters)
- EVT - extravillous cytotrophoblasts (anchor the villous tree in the decidua)
Cytotrophoblasts
Hyperglycosylated human Chorionic Gonadotropin (hCG) promotes the growth of cytotrophoblast cells and the endometrial invasion by these cells during implantation.[16]
Extravillous Cytotrophoblasts
Maintain open maternal spiral arteries emptying into the maternal lacunae[17]
At term, depending on maternal decidual localization, there are several identifiable subtypes:[4]
- interstitial mononuclear (and multinuclear) - dispersed in decidual mesenchyme
- endovascular - in spiral arteries lumen (or replacing endothelial cells)
- intramural - in spiral arteries tunica media
- "epithelial" lines - maternal decidua basalis basal plate with maternal endothelial cells in a mosaic fashion
Syncytiotrophoblasts
- form by fusion of rapidly dividing cytotrophoblast cells
- secrete proteolytic enzymes, enzymes break down extracellular matrix around cells
- Allow passage of blastocyst into endometrial wall, totally surround the blastocyst
- generate spaces, lacunae, that fill with maternal blood
- secrete Human Chorionic Gonadotropin (hCG), hormone, maintains decidua and Corpus Luteum, basis of pregnancy diagnostic test, present in urine is diagnostic of pregnancy
- Later in development placenta will secrete hCG
Cell–cell Fusion Activity
Two pairs of envelope genes of retroviral origin, syncytins, have fusogenic properties. Human endogenous retroviruses (HERVs) make up about 8% of the human genome. Recent study has shown that placentae from intrauterine growth restriction have impaired cell fusion and differentiation that correlates with reduced levels of HERV envelope genes.[18]
Human - syncytin 1 and syncytin 2
- Syncytin 2 is an envelope gene from the human endogenous retrovirus FRD (HERV-FRD)[19]
- Syncytin 2 receptor is Major Facilitator Superfamily Domain Containing 2 (MFSD2) (at chromosomal position 1p34.2)
- belongs to family of presumptive carbohydrate transporters with 10-12 membrane-spanning domains
- Syncytin 1 (-FRD) receptor is ASCT-1/-2
Mouse - syncytin A and syncytin B
- syncytin A is essential for trophoblast cell differentiation and syncytiotrophoblast morphogenesis[20]
- Links: OMIM Syncytin-2
Human Chorionic Gonadotropin
hCG sources - produced by villous syncytiotrophoblast cells, hyperglycosylated hCG produced by cytotrophoblast cells, free beta-subunit made by multiple primary non-trophoblastic malignancies, and pituitary hCG made by the gonadotrope cells of the anterior pituitary.
- levels peak at 8 to 10 weeks of pregnancy, then decline and are lower for rest of pregnancy
- 0-1 week: 0-50 mIU/ml
- 1-2 weeks: 40-300 mIU/ml
- 3-4 weeks: 500-6,000 mIU/ml
- 1-2 months: 5,000-200,000 mIU/ml
- 2-3 months: 10,000-100,000 mIU/ml
- 2nd trimester: 3,000-50,000 mIU/ml
- 3rd trimester: 1,000-50,000 mIU/ml
Non-pregnant females: <5.0 mIU/ml Postmenopausal females: <9.5 mIU/ml
HLA-G
An acronym for histocompatibility antigen, class I, G (also called: Human Leukocyte Antigen G, (HLA-6.0; HLA60, T-CELL A LOCUS, TCA) and is expressed on placental cytotrophoblast cells and other adult tissues. This distinct tissue distribution differs from the other HLA antigens (HLA-A, HLA-B, HLA-C) leading to the description as a non-classical class I antigen. May have a role in protecting the fetus from the maternal immune response. For example, expression of the HLA-G class, instead of HLA-A and HLA-B, may be a mechanism for avoiding clearance by maternal natural killer cells.
Human gene is located at 6p22.1 and there exist several protein isoforms from alternative splicing of messenger RNAs, membrane-bound isoforms (HLA-G 1-4) and soluble soluble (HLA-G 5-6)[21]. The molecule is a heterodimer consisting of both a heavy chain and a light chain (beta-2 microglobulin). The membrane-bound isoform heavy chain is anchored in the membrane and increased expression of the soluble form is related to higher implantation rates. Changes in HLA-G expression have been associated with increased miscarriage rates.[22] Killer cell immunoglobulin-like receptor (KIR) 2DL4 (KIR2DL4) has been shown to act as a receptor for the soluble HLA-G, leading to a stimulation of resting natural killer (NK) cells.[23]
- Links: HLA-G | OMIM142871
Trophoblast Infiltration
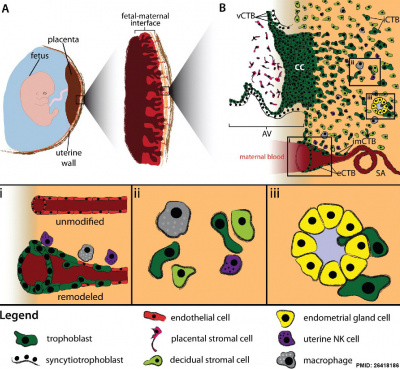
|
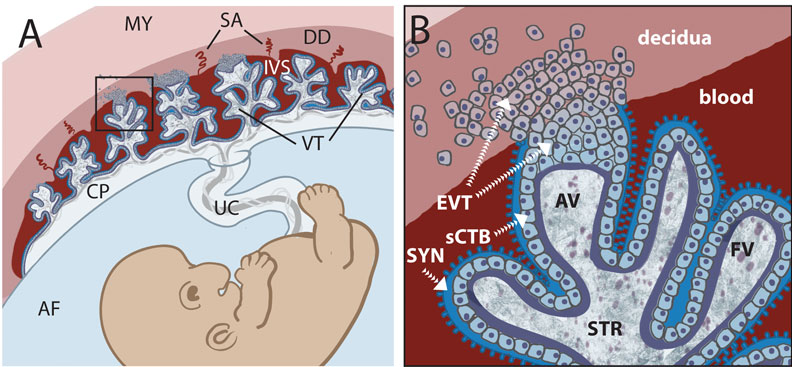
Human trophoblast invasion[24]
(A) The placenta connects the fetus to the uterine wall and establishes a vascular connection between mother and child. The placenta is structured as villous tree and is in direct contact with maternal blood and, thus referred to as hemochorial. The site where the placenta comes in direct contact with the maternal decidua is called the fetal-maternal interface. (B) During early pregnancy, vCTBs fuse to form multinucleated STs, which surround the placental villus. STs transport nutrients and gases from the maternal to the fetal circulation and represent the major endocrine unit of the placenta by secreting hormones such as chorionic gonadotropin, placental growth hormone or placental lactogen. AV form cell columns that attach to the maternal decidua and give rise to the EVT lineage. Invasive EVTs can be divided into iCTBs, which invade the decidual stroma and become terminally differentiated multinucleated GCs, or eCTBs.
|
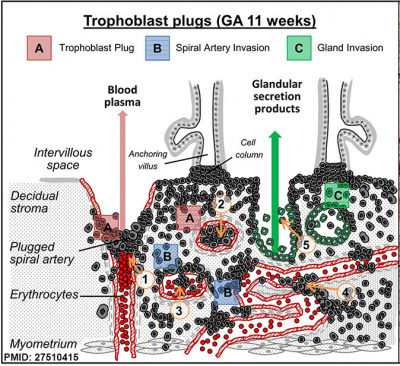
|
Trophoblast plugs within Spiral Arteries Week 9, GA week 11.[7]
|
Trophoblast Cell Lines
A useful component of current placentation and trophoblast research are a number of trophoblast cell lines that have been derived from a number of different sources including term placenta, choriocarcinomas and transformation of primary isolated cells. BeWo cell line is the earliest human choriocarcinoma, established in 1968[25]. Most appear to be examples of extravillous trophoblast cells, some examples of various human "trophoblast" cell lines are listed below.
- BeWo - human choriocarcinoma[25], hypotetraploid (modal number = 86; range = 71 to 178) (ATCC CCL-98)
- JEG-3 - choriocarcinoma HTR8/SVneo a transformed extravillous trophoblast line
- SGHPL-4 - [26][27] SV40 (pSV3neo) transformed primary cell line express cytokeratin-7, HLA class I antigen, HLA-G, BC-1, CD9, human chorionic gonadotrophin, and human placental lactogen.
- TEV-1 - (HPV16) E6/E7 infection first-trimester extravillous trophoblast cell line[28]
- ACH-3P - fusion of primary human first trimester trophoblasts (GA week 12) with a human choriocarcinoma cell line (AC1-1).[29]
- HChEpC1b - retroviral infection by E6/E7/hTERT[30]
History
The name "Trophoblast" was used for the first time by Ambrosius Arnold Willem Hubrecht (1853 – 1915) at the meeting of the Anatomical Congress at Wiirzburg in 1888, and its earliest definition is found in the report of that meeting in Nos. 17 and 18 of the Anatomischer Anzeiger, Bd. III. "We there read, concerning a very early stage of the hedgehog (p. 510) : Die aussere Wand der Keimblase ist verdickt (drei bis vierschichtig) und besitzt wabige Lacunen. Fur diese aussere (epiblastische) Schicht sei der. Name Trophoblast gewahlt."
- Links: The Trophoblast - A Rejoinder (1904) | Ambrosius Arnold Willem Hubrecht | Hubrecht Collection | Embryologists | Embryology History
Abnormalities
Hydatidiform Mole
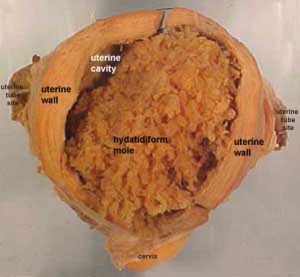
A type of placental abnormality when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Placenta - Abnormalities
Trophoblast Infections
- Malaria (More? Abnormal Development - Malaria)
- Chlamydia trachomatis[31]
- Herpesvirus 8 (HHV-8)[32]
- Group B streptococcus (GBS)[33]
References
- ↑ <pubmed>19924284</pubmed>| PMC2773928 | PLoS One
- ↑ <pubmed>16681851</pubmed>| PMC1479373 | J Transl Med.
- ↑ <pubmed>27807286</pubmed>
- ↑ 4.0 4.1 4.2 <pubmed>28081266</pubmed> Cite error: Invalid
<ref>tag; name 'PMID28081266' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID28081266' defined multiple times with different content - ↑ Jiang SW, Zhou W, Wang J, Little LM, Leaphart L, Jay J, Igbinigie E, Chen H & Li J. (2018). Gene expression patterns associated with human placental trophoblast differentiation. Clin. Chim. Acta , , . PMID: 29329728 DOI.
- ↑ Rothbauer M, Patel N, Gondola H, Siwetz M, Huppertz B & Ertl P. (2017). A comparative study of five physiological key parameters between four different human trophoblast-derived cell lines. Sci Rep , 7, 5892. PMID: 28724925 DOI.
- ↑ 7.0 7.1 Weiss G, Sundl M, Glasner A, Huppertz B & Moser G. (2016). The trophoblast plug during early pregnancy: a deeper insight. Histochem. Cell Biol. , 146, 749-756. PMID: 27510415 DOI. Cite error: Invalid
<ref>tag; name 'PMID27510415' defined multiple times with different content - ↑ Liang H, Zhang Q, Lu J, Yang G, Tian N, Wang X, Tan Y & Tan D. (2016). MSX2 Induces Trophoblast Invasion in Human Placenta. PLoS ONE , 11, e0153656. PMID: 27088357 DOI.
- ↑ Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development PNAS
- ↑ Ohinata Y & Tsukiyama T. (2014). Establishment of trophoblast stem cells under defined culture conditions in mice. PLoS ONE , 9, e107308. PMID: 25203285 DOI.
- ↑ Chuprin A, Gal H, Biron-Shental T, Biran A, Amiel A, Rozenblatt S & Krizhanovsky V. (2013). Cell fusion induced by ERVWE1 or measles virus causes cellular senescence. Genes Dev. , 27, 2356-66. PMID: 24186980 DOI.
- ↑ Blois SM, Tirado-González I, Wu J, Barrientos G, Johnson B, Warren J, Freitag N, Klapp BF, Irmak S, Ergun S & Dveskler GS. (2012). Early expression of pregnancy-specific glycoprotein 22 (PSG22) by trophoblast cells modulates angiogenesis in mice. Biol. Reprod. , 86, 191. PMID: 22423048 DOI.
- ↑ Hubert MA, Sherritt SL, Bachurski CJ & Handwerger S. (2010). Involvement of transcription factor NR2F2 in human trophoblast differentiation. PLoS ONE , 5, e9417. PMID: 20195529 DOI.
- ↑ Lash GE, Naruse K, Innes BA, Robson SC, Searle RF & Bulmer JN. (2010). Secretion of angiogenic growth factors by villous cytotrophoblast and extravillous trophoblast in early human pregnancy. Placenta , 31, 545-8. PMID: 20338637 DOI.
- ↑ Robbins JR, Skrzypczynska KM, Zeldovich VB, Kapidzic M & Bakardjiev AI. (2010). Placental syncytiotrophoblast constitutes a major barrier to vertical transmission of Listeria monocytogenes. PLoS Pathog. , 6, e1000732. PMID: 20107601 DOI.
- ↑ Cole LA. (2010). Biological functions of hCG and hCG-related molecules. Reprod. Biol. Endocrinol. , 8, 102. PMID: 20735820 DOI.
- ↑ Whitley GS & Cartwright JE. (2010). Cellular and molecular regulation of spiral artery remodelling: lessons from the cardiovascular field. Placenta , 31, 465-74. PMID: 20359743 DOI.
- ↑ <pubmed>20664994</pubmed>
- ↑ <pubmed>18988732</pubmed>| PNAS
- ↑ <pubmed>19564597</pubmed>
- ↑ <pubmed>16339579</pubmed>
- ↑ <pubmed>24557735</pubmed>
- ↑ <pubmed>16366734</pubmed>
- ↑ <pubmed>26418186</pubmed>
- ↑ 25.0 25.1 <pubmed>5753554</pubmed>
- ↑ <pubmed>9804259</pubmed>
- ↑ <pubmed>16226120</pubmed>
- ↑ <pubmed>15866109</pubmed>
- ↑ <pubmed>18093301</pubmed>
- ↑ <pubmed>19148547</pubmed>
- ↑ <pubmed>19265152</pubmed>
- ↑ <pubmed>19115001</pubmed>
- ↑ <pubmed>18599257</pubmed>
Reviews
<pubmed>19876840</pubmed>
Articles
<pubmed>20081188</pubmed> <pubmed>20033790</pubmed> <pubmed>19617545</pubmed> <pubmed>19564597</pubmed> <pubmed>19582159</pubmed>
Search PubMed
Search October 2010
- Syncytiotrophoblast - All (10230) Review (1104) Free Full Text (2508)
- Cytotrophoblast - All (9828) Review (1062) Free Full Text (2401)
Search Pubmed: Trophoblast | syncytiotrophoblast | cytotrophoblast |
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 19) Embryology Trophoblast. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Trophoblast
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G