Talk:SH Lecture - Respiratory System Development
Virtual Slides
UNSW Virtual Slides
http://vslides.unsw.edu.au/VirtualSlideV2.nsf/id/F51ED7
Virtual Slidebox
http://www.path.uiowa.edu/virtualslidebox/nlm_histology/content_index_db.html
http://www.path.uiowa.edu/cgi-bin-pub/vs/fpx_browse.cgi?cat=o_lung&div=nlm
Blue Histology
http://www.lab.anhb.uwa.edu.au/mb140/CorePages/Respiratory/respir.htm
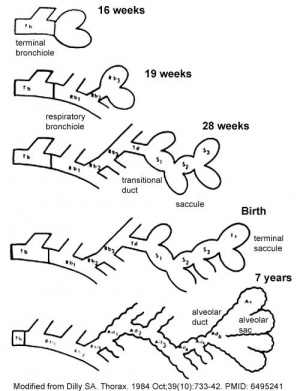
Lung Development Stages
Text from: <pubmed>10852845</pubmed>| PMC1637815 | Environ Health Perspect.
Embryogenesis
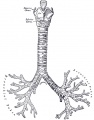
The lungs in humans first appear at the end of the first month of gestation as an evagination of epithelium from the foregut. The bud rapidly divides as a series of branching tubes in a dichotomous pattern. These tubular branches invade and interdigitate with mesenchymal tissues. Branching morphogenesis during this period forms the most proximal portions of the future tracheobronchial tree. As these tissues grow, they push into the future pleuroperitoneal cavity of the embryo. During embryogenesis, transcription factors play an important role in gene expression and regulation. Transcription factors are essential in both the stimulation and inhibition of gene expression to regulate the proper temporal and spatial patterning of lung development. Hepatocyte nuclear factor- 3 (12) and the homeobox gene TTF-1 (13) are examples of transcription factors serving as important regulators of early differentiation of the pulmonary epithelium during this period. Lung development is also highly dependent on interactions between the epithelium and mesenchyme. This dual origin of lung tissues is critical in development. Removal of mesenchyme from the tip of a lung bud during early phases of development with transplantation to the side of a higher ordered segment abolishes further branching at the site of removal while stimulating growth of a
Pseudoglandular stage.
Tubular branching of the human lung airways continues from the fifth to the seventeenth week of gestation. As early as 2 months of gestational age, all segmental bronchi are present. During this period, the lungs take on the appearance of a glandlike structure. This stage is the most critical for the formation of all conducting airways. During this period, the airway tubular structures are lined with tall columnar epithelium, whereas the more distal structures are lined with cuboidal epithelium. A number of signals arising from epithelial mesenchymal interactions during this time continue to modulate cellular proliferation temporally as well as spatially (4). These regulatory signals lead to further branching morphogenesis by affecting the rate of cellular proliferation (15). The presence of extracellular matrix molecules, including collagen, fibronectin, laminin, glycosaminoglycans, and proteoglycans, as well as cell membrane-bound integrins, also plays an important role in directing lung development by influencing the rates of cellular proliferation and differentiation (3,16,1/). Mechanical distention exerted on the lung as well as on specific cell types can also significantly affect gene expression and, ultimately, lung growth and development (4). A variety of growth factors and growth factor receptors are also important in controlling cellular functions (3). Epidermal growth factor, transforming growth factor-a, and retinoic acid all act to affect branching morphogenesis and cellular differentiation (18,19). Epithelial differentiation of ciliated, goblet, and basal cells first appears in the most central airways during this stage of development. Cartilage and smooth muscle cells are also first noted in the trachea and extend more peripherally with progressive growth of the lungs. During this stage of
Canalicular stage.
This stage lasts from week 16 to week 24 in the human fetus. Lung morphology changes dramatically during this time because of differentiation of the pulmonary epithelium, resulting in the formation of the future air-blood tissue barrier. Surfactant synthesis and the canalization of the lung parenchyma by capillaries begin. During this stage, the future gas exchange regions can be easily distinguished from the future conducting airways of the lungs.
Saccular stage.
The saccular stage of lung development in humans lasts from week 24 to near term. The most peripheral airways form widened airspaces, termed saccules. These saccules widen and lengthen the airspace, in large measure by the addition of new generations. During this stage, the future gas exchange region expands significantly. Populations of fibroblastic cells also undergo differentiation during this stage. These fibroblast-like cells are responsible for the production of the extracellular matrix, collagen, and elastin. It is also presumed that they play an important role in epithelial differentiation and control of surfactant secretion in connection with the growth of the gas exchange region during this stage. The vascular tree also grows in length and diameter during this time.
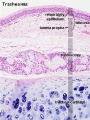
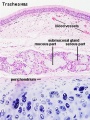
Columnar cells that are undifferentiated characterize the first epithelial cells lining fetal lung tubules. The first epithelial cells to differentiate in the trachea are neuroendocrine cells, followed closely by ciliated cells, and finally basal and secretory cells in rapid sequence. This process of differentiation covers a developmental period ranging from days to months. In rodents including the mouse, rat, and hamster, complete epithelial differentiation of the trachea occurs in as little as 2 days. In primate trachea, cellular differentiation takes up to 6 months to be complete. In most species, epithelial cell differentiation of the trachea usually is not complete until just before birth. For more peripheral airway generations, cellular differentiation is likely to continue into the early postnatal period. Fetal epithelial cells are typically filled with glycogen that is gradually replaced with a granular cytoplasm filled with numerous organelles during cellular differentiation. These glycogen-filled cells are found throughout the tracheobronchial tree as well as into the most peripheral saccules. Differentiation of the epithelium is highly site specific, giving rise to more than 10 different cell types. For example, within the saccules of the lungs, cells lining these surfaces differentiate to form both squamous type 1 cells as well as cuboidal type 2 cells. The presence of glycogen within these cells may persist into early postnatal life.
Original Lecture Page
Developmental Overview
Week 4 - laryngotracheal groove forms on floor foregut.
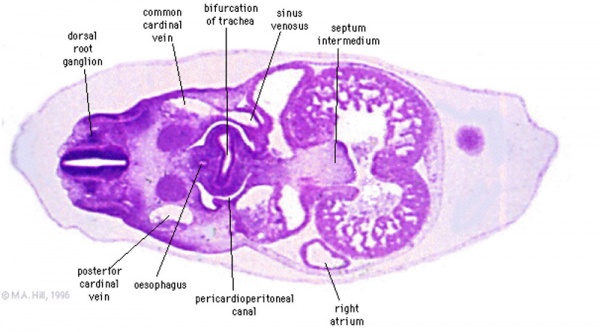
Week 5 - left and right lung buds push into the pericardioperitoneal canals (primordia of pleural cavity)
Week 6 - descent of heart and lungs into thorax. Pleuroperitoneal foramen closes.
Week 7 - enlargement of liver stops descent of heart and lungs.
Month 3-6 - lungs appear glandular, end month 6 alveolar cells type 2 appear and begin to secrete surfactant.
Month 7 - respiratory bronchioles proliferate and end in alveolar ducts and sacs.
Lung Development
- week 4 - 5 embryonic
- week 5 - 17 pseudoglandular
- week 16 - 25 canalicular
- week 24 - 40 terminal sac
- late fetal - 8 years alveolar
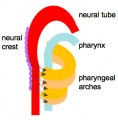
Germ Layers
- Endoderm form epithelium of conducting and alveoli.
- Ectoderm will contribute the neural innervation.
- Mesoderm contributes connective tissues, blood vessels, smooth muscle, also contributes the supporting musculoskeletal components.
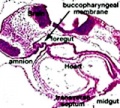
Foregut development
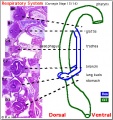
From the oral cavity the next portion of the foregut is initially a single gastrointestinal (oesophagus) and respiratory (trachea) common tube, the pharynx which lies behind the heart. Note that the respiratory tract will form from a ventral bud arising at this level.
- Oral cavity
- Pharynx (esophagus, trachea)
- Respiratory tract
- Stomach
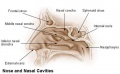
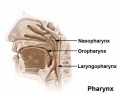
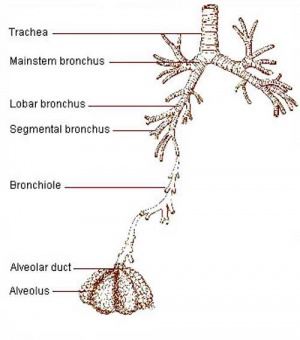
Upper respiratory tract
- part of foregut development
- anatomically the nose, nasal cavity and the pharynx
- the pharynx forms a major arched cavity within the pharyngeal arches (pharyngeal arches will be described in head development lecture)
| File:Endoderm 002 icon.jpg</wikiflv> | 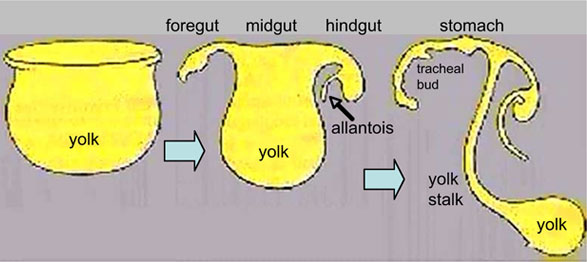
|
Lower respiratory tract
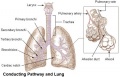
- The lungs go through an embryonic and 4 distinct histological phases of development
Growth initially of branched "conducting" system of bronchial tree, followed by later development of the "functional units" of the alveoli.
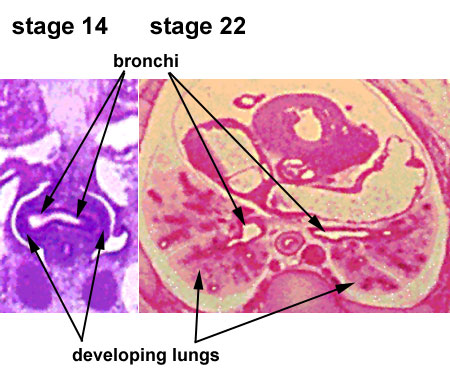
- embryonic - week 4 - 5 (stage 14 above)
- pseudoglandular - week 5 - 17 (stage 22 above)
- canalicular - week 16 - 25
- terminal sac - week 24 - 40
- alveolar - late fetal - 8 years (Latin, alveus = cavity or hollow)
Lung morphogenesis
Embryonic stage
- Lung buds ( endoderm epithelial tubes) grow/push into splanchnic mesenchyme covered with pleural cells (lung border)
- embryonic tissue (adult tissue) organised inside to out
- endoderm (future respiratory epithelia) - splanchnic mesoderm (connective tissue, blood vessels, smooth muscle) - splanchnic mesothelium (visceral pleura) - pericardioperiotoneal canals (pleural cavity) - somatic mesothelium (parietal pleura) - somatic mesoderm (body wall CT, skeleton)
- generates a tree-like network by repeated:
- elongation
- terminal bifurcation
- lateral budding
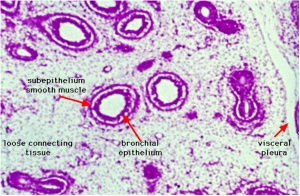
Pseudoglandular stage
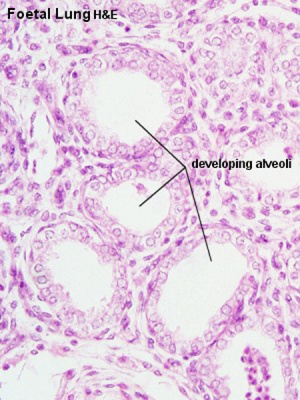
- week 5 - 17 (late embryonic, fetal)
- tubular branching of the human lung airways continues (16-25 generations of branching)
- by 2 months all segmental bronchi are present.
- lungs have appearance of a glandlike structure.
- stage is critical for the formation of all conducting airways.
- lined with tall columnar epithelium, the more distal structures are lined with cuboidal epithelium.
Canalicular stage
- week 16 - 24 (fetal to end of second trimester)
- Lung morphology changes dramatically
- differentiation of the pulmonary epithelium results in the formation of the future air-blood tissue barrier.
- Surfactant synthesis and the canalization of the lung parenchyma by capillaries begin (week 17).
- future gas exchange regions can be distinguished from the future conducting airways of the lungs.
- 1 respiratory bronchiole gives rise to 3-6 alveolar ducts.
Saccular stage
- week 24 to near term (fetal third trimester)
- most peripheral airways form widened airspaces, termed saccules.
- saccules widen and lengthen the airspace (by the addition of new generations).
- future gas exchange region expands significantly.
- Fibroblastic cells also undergo differentiation, they produce extracellular matrix, collagen, and elastin.
- May have a role in epithelial differentiation and control of surfactant secretion
- The vascular tree also grows in length and diameter during this time.
Alveolar stage
- Thyroid hormone required for differentiation and stimulate surfactant production.
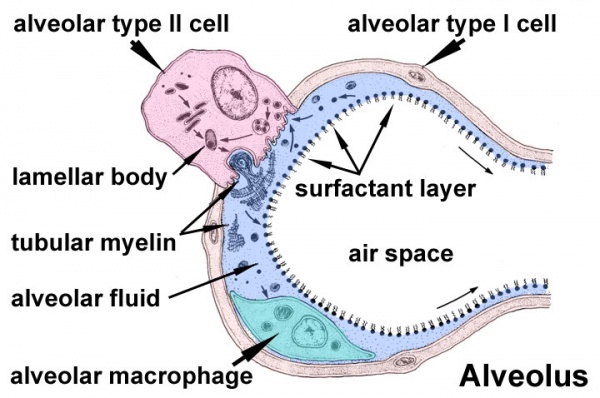
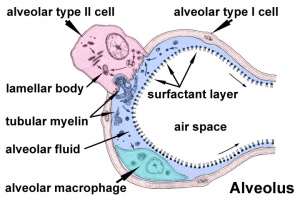
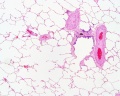
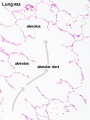
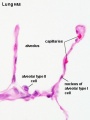
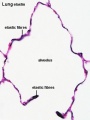
Alveolar type I cells
- small alveolar cells, type I pneumocytes
- very flat cells (thin as 0.05 µm)
- form most of the surface of the alveolar walls
- may contribute epithelium on both faces of the alveolar wall
Alveolar type II cells
- large alveolar cells, type II pneumocytes
- irregular to cuboidal shaped cells
- contain large number of granules called lamellar bodies, these are the precursors to pulmonary surfactant (phospholipid mixture).
- end month 6 alveolar cells type 2 appear and begin to secrete surfactant - premature babies have difficulties associated with insufficient surfactant.
Alveolar macrophages
- remove particulate matter that enters the alveoli with inspired air
- migrate over alveolar epithelium and phagocytose particulate matter
Fetal lung volume
Each lung volume as determined by ultrasound and matched to gestational age (PMID: 16388511)
- 12-13 weeks 0.05 mL
- 19-22 weeks 0.5 mL
- 29-32 weeks 1.9 mL
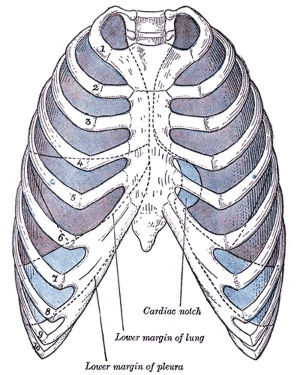
Pleural Cavity
- The anatomical body cavity in which the lungs develop and lie.
- The pleural cavity forms in the lateral plate mesoderm as part of the early single intraembryonic coelom.
- This cavity is initially continuous with pericardial and peritoneal cavities and form initially as two narrow canals
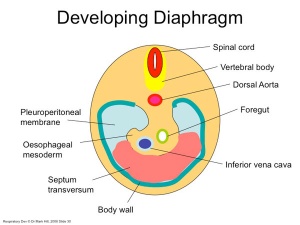
- later becomes separated by folding (pleuropericardial fold, pleuroperitoneal membrane) and the later formation of the diaphragm
pleuropericardial fold - (pleuropericardial membrane) An early embryonic fold which restricts the communication between pleural cavity and pericardiac cavity, contains both the cardinal vein and phrenic nerve.
pleuroperitoneal membrane - An early embryonic membrane that forms inferiorly at the septum transversum to separate peritoneal cavity from pleural cavity.
Pleura
- serous membrane covers the surface of the lung and the spaces between the lobes
- arranged as a closed invaginated sac
- two layers (pulmonary, parietal) continuous with each other, the potential space between them is the pleural cavity
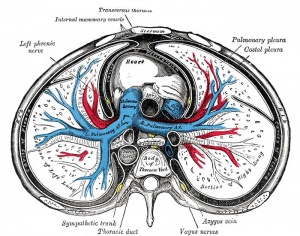
Diaphragm
Not respiratory tract but musculoskeletal development, there are 5 elements that contribute to the diaphragm.
Innervation of the human diaphragm is by the phrenic nerves, arising from the same segmental levels from which the diaphragm skeletal muscles arise, segmental levels C3 to C5.
The paired phrenic nerves are mixed containing motor neurons for the diaphragm and sensory nerves for other abdominal structures (mediastinum, pleura, liver, gall bladder).
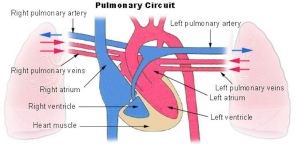
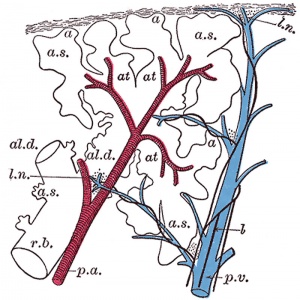
Pulmonary Circulation
- the pulmonary system not "functional" until after birth
- pulmonary arteries - (deoxygenated blood to lung) 6th aortic arch arteries
- pulmonary veins - (oxygenated blood from lung) are incorporated into the left atrium wall
- bronchial arteries - branches from dorsal aorta
Fetal Respiratory Movements
- Fetal respiratory movements (FRM) or Fetal breathing movements (FBM) are regular muscular contrations occurring in the third trimester.
- thought to be preparing the respiratory muscular system for neonatal function (and amniotic fluid are thought to have a role in lung maturation)
- thought to also have a role in late lung development.
The First Breath
- The respiratory system does not carry out its physiological function (gas exchange) prenatally and remain entirely fluid-filled until birth.
- At birth, fluid in the upper respiratory tract is expired and fluid in the lung aveoli is rapidly absorbed this event has also been called "dewatering of the lung".
- The lung epithelia has to now rapidly change from its prenatal secretory function to that of fluid absorbtion.
Exchange of Fluid for Air
- fall in pulmonary vascular resistance
- increase in pulmonary blood flow
- thinning of pulmonary arteries (stretching as lungs increase in size)
- blood fills the alveolar capillaries
In the heart, pressure in the right side of the heart decreases and pressure in the left side of the heart increases (more blood returning from pulmonary).
- Respiratory Rate is higher than adult (30 breaths/minute).
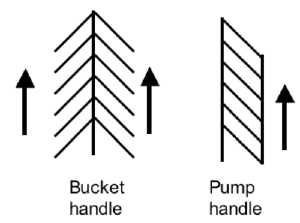
Rib Orientation
- Infant rib is virtually horizontal, allowing only diaphragmatic breathing
- Adult rib orientation is oblique (both anterior and lateral views), allows for pump-handle and bucket handle types of inspiration.
Respiratory Tract Histology
- Histology Links: Blue Histology | UNSW Virtual Slides | UIOWA Virtual Slidebox
Respiratory Tract Abnormalities
Respiratory System - Abnormalities
Tracheoesophageal Fistula
- Tracheo-Oesophageal Fistula, Oesophageal Atresia - Oesophageal Atresia with or without tracheo-oesophageal fistula
- Fistula - an abnormal communication between 2 structures (organs, vessels, cavities) that do not normally connect.
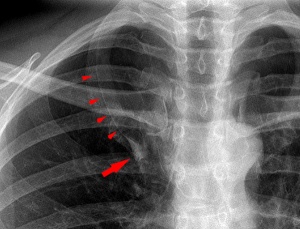
Lobar Emphysema (Overinflated Lung)
- There is an overinflated left upper lobe
- There is a collapsed lower lobe
- The left lung is herniating across the mediastinum
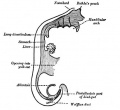
Congenital Diaphragmatic Hernia
- Failure of the pleuroperitoneal foramen (foramen of Bochdalek) to close (left side)
- allows viscera into thorax -iIntestine, stomach or spleen can enter the pleural cavity, compressing the lung.
- rare (Morgagni hernia) -an opening in the front of the diaphragm.
- Links: GeneReviews
Azygos Lobe
- Common condition (0.5% of population).
- The right lung upper lobe expands either side of the posterior cardinal.
- There is also some course variability of the phrenic nerve in the presence of an azygos lobe.
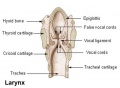
Congenital Laryngeal Webs
- Laryngeal abnormality due to embryonic (week 10) incomplete recanalization of the laryngotracheal tube during the fetal period.
- Rare abnormality occuring mainly at the level of the vocal folds (glottis).
Meconium Aspiration Syndrome
(MAS) Meconium is the gastrointestinal contents that accumulate in the intestines during the fetal period. Fetal stress in the third trimester, prior to/at/ or during parturition can lead to premature meconium discharge into the amniotic fluid and sunsequent ingestion by the fetus and damage to respiratory function. Damage to placental vessels meconium myonecrosis may also occur.
Newborn Respiratory Distress Syndrome
- Hyaline Membrane Disease (membrane-like substance from damaged pulmonary cells)
- absence of surfactant, if prolonged can be irreversible
- intrauterine asphyxia, prematurity and maternal diabetes
- Links: medline plus | eMedicine
Bronchopulmonary Dysplasia
- A chronic lung disease which can occur following premature birth.
- The definition of bronchopulmonary dysplasia (BPD) has in recent years changed from a severe lung injury and associated repair, to more of a disruption of lung development.
Terms
- antenatal before birth.
- alveoli number at birth - from 20 - 50 million and eventually in the adult 300 million.
- Bronchopulmonary dysplasia - (BPD) the most common serious sequela of premature birth.
- Bronchiolitis - is a viral infection of the lower respiratory tract and most common lower respiratory tract infection in infants. Respiratory syncytial virus (RSV) is responsible for 70 percent of all cases overall and Parainfluenza, adenovirus and influenza account for most of the remaining cases. (HSTAT Management of Bronchiolitis in Infants and Children)
- Chronic obstructive pulmonary disease (COPD) causes include smoking (85–90 percent of all cases), genetic factors (alpha-1 antitrypsin deficiency), passive smoking (children), occupational exposures, air pollution, and hyperresponsive airways. (HSTAT Management of Acute Exacerbations of Chronic Obstructive Pulmonary Disease)
- Clara cells non-ciliated cell found in the small airways (bronchioles) consisting of ciliated simple epithelium, these cells secrete glycosaminoglycans (Clara cell secretory protein, CCSP) to protect the bronchiole lining.
- Congenital Diaphragmatic Hernia (CDH) disorder with an incidence of 1 in 2500 live births.
- fetal breathing-like movements (FBMs) or Fetal respiratory movements are thought to be regular muscular contrations occurring in the third trimester, preparing the respiratory muscular system for neonatal function and to have a role in late lung development.
- glucocorticoid treatment - antenatal therapy to promote the maturation of the human fetal lung. Given as a synthetic glucocorticoid between 24 and 32 weeks of pregnancy to promote lung maturation in fetuses at risk of preterm delivery.
- lamellar bodies the storage form of surfactant in type II alveolar cells, seen as centrically layered "packages" of phospholipid. A count of lamellar bodies can be used as an assay for measuring fetal lung maturity.
- maternal diabetes if not controlled in pregnancy may delay fetal pulmonary maturation.
- Persistent Pulmonary Hypertension of the Newborn (PPHN) serious newborn condition due to due to the failure of closure one of the prenatal circulatory shunts, the ductus arteriosus. Occurs in about 1-2 newborns per 1000 live births and results in hypoxemia. (More? Respiratory Development - Birth)
- Pharyngitis inflammation of the pharynx involving lymphoid tissues of the posterior pharynx and lateral pharyngeal bands.
- pneumocyte or alveolar type I and type II cells.
- pulmonary hypoplasia can be due to anencephaly, renal hypoplasia or abnormalities of the thoracic cage
- pulmonary neuroendocrine cells (PNEC) single or innervated clusters of cells (neuroepithelial bodies) that line the airway epithelium, thought to have a role in regulating fetal lung growth and differentiation. At birth may also act as airway oxygen sensors involved in newborn adaptation. These cells synthesis and release amine (serotonin, 5-HT) and a several neuropeptides (bombesin).
- Respiratory distress syndrome (RDS) due to a surfactant deficiency at birth, particulary in preterm birth.
- secondary alveolar septa formed during the alveolar stage and are formed by projections of connective tissue and a double capillary loop.
- surfactant produced by alveolar type II cells is a mixture of lipids and proteins that both maintains alveolar integrity and plays a role in the control of host defense and inflammation in the lung.
- Surfactant therapy (American Academy of Pediatrics Policy | Canadian Paediatric Society Recommendations)
- thyroid hormone involved in the regulation of fetal lung development.
- vascular endothelial growth factor (VEGF) a secreted growth factor acting through receptors on endothelial cells to regulate vasculogenesis through their development, growth and function.