Prostate Development
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
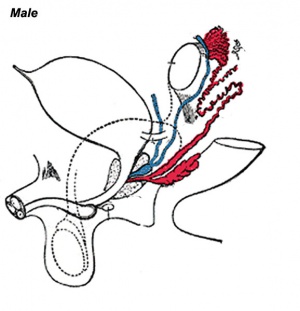
In humans, the male accessory glands are the seminal vesicles, prostate gland, and the bulbourethral glands. The male gonad, the testis, differentiates embryonically initially under the influence of the Y chromosome. Later under the influence the gonad-derived fetal testosterone acting through androgen receptors, a region of the urogenital sinus (UGS) mesenchyme differentiates to form the primordial prostate buds. The buds then signal back to the overlying epithelium, inducing duct formation, this was one of the early studied (1970's) example of an mesenchymal-epithelial interaction in development. Interestingly, the female equivalent gland originally called Skene's gland, then paraurethral gland has now also been renamed the female prostate.
The reproductive function of the prostate becomes active at puberty where prostate secretions contribute the majority by volume of the ejaculate containing spermatozoa.
The prostate gland is generally in the news due to its late postnatal adult growth changes, enlarged due to benign nodular hyperplasia, and the male health effects of prostate cancer. Prostate cancer is the second most common malignant tumor in western males and anatomically involves the prostate peripheral zone.
There are also currently separate pages describing Male | spermatozoa | testis | prostate | Category:Prostate
Historic
Evatt EJ. A contribution to the development of the prostate in man. (1909) Jour. of Anat. and Phys. 43: 314-321.
Lowsley OS. The development of the human prostate gland with reference to the development of other structures at the neck of the urinary bladder. (1912) Amer. J Anat. 13(3): 299-346.
Watson EM. The development of the seminal vesicles in man. (1918) Amer. J Anat. 24(4): 395 - 439.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Prostate Embryology | Prostate Development | Prostatic Urethra Development | Benign Prostatic Hyperplasia | Bulbourethral Embryology | Bulbourethral Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
- Developmental Biology (6th ed.) Gilbert Chapter 14 Intermediate Mesoderm
Prostate Development Overview
- fetal testosterone stimulates urogenital sinus mesenchyme through androgen receptors
- urogenital sinus mesenchyme acts on the overlying epithelium to stimulate cell proliferation
- urogenital sinus epithelium then forms prostate ductal progenitor, the prostatic buds
- prostatic buds then grow into the urogenital sinus mesenchyme
Prostate Bud Growth
- specification phase - instructive developmental cues define where buds will form in the UGS
- initiation phase - prostatic buds begin to form
- elongation phase - proliferation, cell adhesion, and cell migration coordinate outgrowth of prostatic buds into UGM.
Based on the recent review.[6]
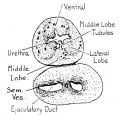
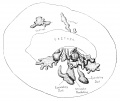
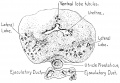
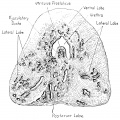
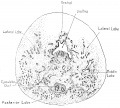
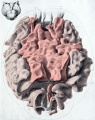
Fetal Prostate
Data and images from a 1912 study by Lowsley.[7] (See all images)
| Table showing number of tubules of each Prostate Lobe opening into prostatic urethra, the number of Albarran' s tubules, and the number of subtrigonal tubules. | |||||||
| Size of Fetus Crown-Rump |
Middle Lobe |
Lateral Lobes |
Posterior Lobe |
Anterior Lobe |
Total No. of Prostatic Tubules |
Subcervical Glands of Albarran |
Subtrigonal Glands |
|---|---|---|---|---|---|---|---|
| 7.5 | 12 | 39 | 11 | 12 | 74 | 0 | 0 |
| 8 | 7 | 27 | 6 | 13 | 53 | 0 | 0 |
| 12.5 | 10 | 46 | 4 | 14 | 74 | 8 | 0 |
| 19 | 0 | 42 | 10 | 7 | 59 | 11 | 5 |
| 27 | 11 | 36 | 9 | 8 | 64 | 9 | 4 |
| 36 | 9 | 34 | 11 | 2 | 56 | 19 | 9 |
| Averages | 10 | 37 | 8 | 9 | 63 | 12 | 6 |
| The averages are taken from the specimens in which the structure is present in case of middle lobe and the groups of Albarran and the subtrigonal group. | |||||||
- Human Fetal Prostate
- Links: Fetal Development
Genital Development Overview
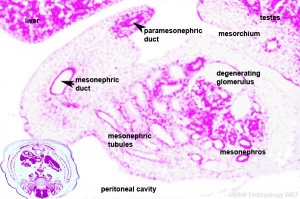
Three main stages during development, mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while.
- Differentiation of gonad (Sex determination)
- Differentiation of internal genital organs
- Differentiation of external genital organs
The 2nd and 3rd stages dependent on endocrine gonad. Reproductive development has a long maturation timecourse, begining in the embryo and finishing in puberty. (More? Puberty Development)
Mouse Prostate Development
The mouse has been used extensively as a model of prostate embryonic development. A similar androgenic regulation occurs of in ventral epithelial bud affecting number and pattern forming in the mouse urogenital sinus.[8]
- begins in fetal mice ductal progenitors (or buds) emerge from urogenital sinus epithelium
- prostatic buds develop in response to androgens, which activate androgen receptors in UGS mesenchyme
- two rows of 3-4 prostatic buds at birth
- Links: Mouse Development
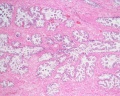
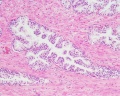
Prostate Histology

|
|
|
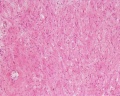
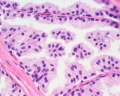
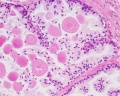
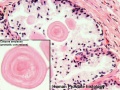
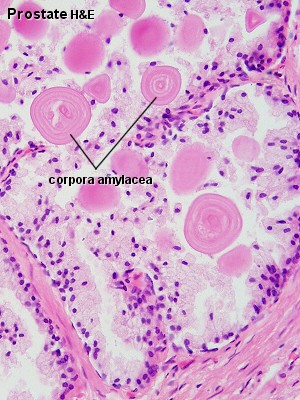
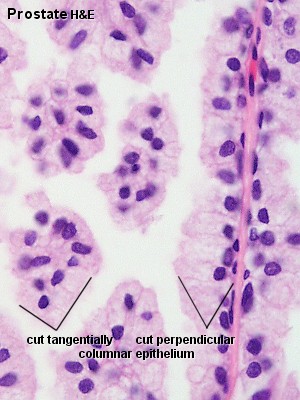
| Human prostate histology | Corpora Amylacea | Submucosal gland |
| (adult, (Stain - Haematoxylin Eosin) low power overview) | (adult, (Stain - Haematoxylin Eosin) detail) | (adult, (Stain - Haematoxylin Eosin) high power detail) |
Anatomy
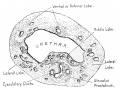
The prostate is the largest accessory sex gland in men (about 2 × 3 × 4 cm). It contains 30 - 50 tubuloalveolar glands, which empty into 15 - 25 independent excretory ducts. These ducts open into the urethra. The glands are embedded into a fibromuscular stroma, which mainly consists of smooth muscle separated by strands of connective tissue rich in collagenous and elastic fibres. The muscle forms a dense mass around the urethra and beneath the fairly thin capsule of the prostrate.
Macroscopically the prostrate can be divided into lobes, but they are inconspicuous in histological sections. In good histological sections it is possible to distinguish three concentric zones, which surround the prostatic part of the urethra.
- peripheral zone contains large (main glands) whose ducts run posteriorly to open into the urethra
- internal zone consists of the so-called submucosal glands
- innermost zone contains mucosal glands
Secretory Glands
The secretory alveoli of the prostate are very irregularly shaped because of papillary projections of the mucosa into the lumen of the gland. The epithelium is cuboidal or columnar. Basal cells are again present, and the epithelium may look pseudostratified where they are found. The secretory cells are slightly acidophilic and secretory granules may be visible in the cytoplasm. Small extensions of the apical cytoplasm into the lumen of the alveoli may represent cells which release their secretory products (secretion is apocrine/merocine). The secretion of the prostate contains citric acid, the enzyme fibrinolysin (liquefies the semen), acid phosphatase, a number of other enzymes and lipids. The secretion of the prostate is the first fraction of the ejaculate.
The secretory ducts of the prostate are lined by a simple columnar epithelium, which changes to a transitional epithelium near the openings of the ducts into the urethra.
Corpora Amylacea
A characteristic feature of the prostate is the appearance of corpora amylacea in the secretory alveoli. They are rounded eosinophilic bodies. Their average diameter is about 0.25 mm (up to 2 mm). They appear already in the seventh month of foetal development. Their number increases with age - in particular past 50. They may undergo calcification. Corpora amylacea may appear in semen.
Additional Histology Images
- Above text and images modified from: Blue Histology - Prostate
Movies
Genital development animations
| Urogenital Sinus | Urogenital Septum |
| Male External | Testis Descent |
Historic Images of Genital Changes
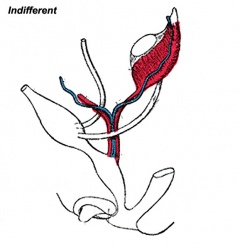
|
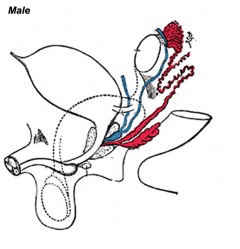
|
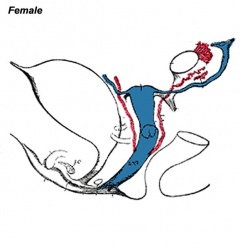
|
| Urogenital indifferent | Urogenital male | Urogenital female |
Historic Gray's Anatomy - The Prostate
(Prostata; Prostate Gland)
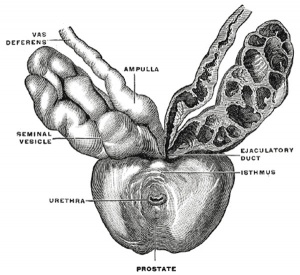
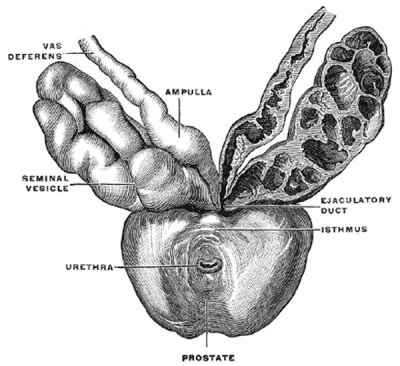
The prostate (Fig. 1160) is a firm, partly glandular and partly muscular body, which is placed immediately below the internal urethral orifice and around the commencement of the urethra. It is situated in the pelvic cavity, below the lower part of the symphysis pubis, above the superior fascia of the urogenital diaphragm, and in front of the rectum, through which it may be distinctly felt, especially when enlarged. It is about the size of a chestnut and somewhat conical in shape, and presents for examination a base, an apex, an anterior, a posterior and two lateral surfaces.
The base (basis prostatæ) is directed upward, and is applied to the inferior surface of the bladder, The greater part of this surface is directly continuous with the bladder wall; the urethra penetrates it nearer its anterior than its posterior border.
The apex (apex prostatæ) is directed downward, and is in contact with the superior fascia of the urogenital diaphragm.
Surface
The posterior surface (facies posterior) is flattened from side to side and slightly convex from above downward; it is separated from the rectum by its sheath and some loose connective tissue, and is distant about 4 cm. from the anus. Near its upper border there is a depression through which the two ejaculatory ducts enter the prostate. This depression serves to divide the posterior surface into a lower larger and an upper smaller part. The upper smaller part constitutes the middle lobe of the prostate and intervenes between the ejaculatory ducts and the urethra; it varies greatly in size, and in some cases is destitute of glandular tissue. The lower larger portion sometimes presents a shallow median furrow, which imperfectly separates it into a right and a left lateral lobe: these form the main mass of the gland and are directly continuous with each other behind the urethra. In front of the urethra they are connected by a band which is named the isthmus: this consists of the same tissues as the capsule and is devoid of glandular substance.
The anterior surface (facies anterior) measures about 2.5 cm. from above downward but is narrow and convex from side to side. It is placed about 2 cm. behind the pubic symphysis, from which it is separated by a plexus of veins and a quantity of loose fat. It is connected to the pubic bone on either side by the puboprostatic ligaments. The urethra emerges from this surface a little above and in front of the apex of the gland.
The lateral surfaces are prominent, and are covered by the anterior portions of the Levatores ani, which are, however, separated from the gland by a plexus of veins.
The prostate measures about 4 cm. transversely at the base, 2 cm. in its antero-posterior diameter, and 3 cm. in its vertical diameter. Its weight is about 8 gm. It is held in its position by the puboprostatic ligaments; by the superior fascia of the urogenital diaphragm, which invests the prostate and the commencement of the membranous portion of the urethra; and by the anterior portions of the Levatores ani, which pass backward from the pubis and embrace the sides of the prostate. These portions of the Levatores ani, from the support they afford to the prostate, are named the Levatores prostatæ.
The prostate is perforated by the urethra and the ejaculatory ducts. The urethra usually lies along the junction of its anterior with its middle third. The ejaculatory ducts pass obliquely downward and forward through the posterior part of the prostate, and open into the prostatic portion of the urethra.
Structure
The prostate is immediately enveloped by a thin but firm fibrous capsule, distinct from that derived from the fascia endopelvina, and separated from it by a plexus of veins. This capsule is firmly adherent to the prostate and is structurally continuous with the stroma of the gland, being composed of the same tissues, viz.: non-striped muscle and fibrous tissue. The substance of the prostate is of a pale reddish-gray color, of great density, and not easily torn. It consists of glandular substance and muscular tissue.
The muscular tissue according to Kölliker, constitutes the proper stroma of the prostate; the connective tissue being very scanty, and simply forming between the muscular fibers, thin trabeculæ, in which the vessels and nerves of the gland ramify. The muscular tissue is arranged as follows: immediately beneath the fibrous capsule is a dense layer, which forms an investing sheath for the gland; secondly, around the urethra, as it lies in the prostate, is another dense layer of circular fibers, continuous above with the internal layer of the muscular coat of the bladder, and blending below with the fibers surrounding the membranous portion of the urethra. Between these two layers strong bands of muscular tissue, which decussate freely, form meshes in which the glandular structure of the organ is imbedded. In that part of the gland which is situated in front of the urethra the muscular tissue is especially dense, and there is here little or no gland tissue; while in that part which is behind the urethra the muscular tissue presents a wide-meshed structure, which is densest at the base of the gland—that is, near the bladder—becoming looser and more sponge-like toward the apex of the organ.
The glandular substance is composed of numerous follicular pouches the lining of which frequently shows papillary elevations. The follicles open into elongated canals, which join to form from twelve to twenty small excretory ducts. They are connected together by areolar tissue, supported by prolongations from the fibrous capsule and muscular stroma, and enclosed in a delicate capillary plexus. The epithelium which lines the canals and the terminal vesicles is of the columnar variety. The prostatic ducts open into the floor of the prostatic portion of the urethra, and are lined by two layers of epithelium, the inner layer consisting of columnar and the outer of small cubical cells. Small colloid masses, known as amyloid bodies are often found in the gland tubes.
Vessels and Nerves
The arteries supplying the prostate are derived from the internal pudendal, inferior vesical, and middle hemorrhoidal. Its veins form a plexus around the sides and base of the gland; they receive in front the dorsal vein of the penis, and end in the hypogastric veins. The nerves are derived from the pelvic plexus.
Gray H. Anatomy of the human body. (1918) Philadelphia: Lea & Febiger.
Bulbourethral Gland
The bulbourethral gland (Cowper's gland) in the male is located beneath the prostate gland at the beginning of the internal portion of the penis. Prior to ejaculation these small paired pea-shaped glands produce a thick clear mucus, that drains into the spongy urethra, and functions to neutralize any traces of acidic urine in the urethra.[9] The prostate and bulbourethral glands develop from the urethra in response to DHT. In the mouse model, BMP signaling during bulbourethral gland development.[10]
Abnormalities
Benign Nodular Hyperplasia
A postnatal adult ageing effect with an onset about 45 years of age, the prostate becomes enlarged due to benign nodular hyperplasia. By 60 years of age and older about 3/4 of the males are affected of which half will be symptomatic. This condition affects the mucosal glands.
Prostate Cancer
Prostate cancer is the second most common malignant tumor in western males and anatomically involves the prostate peripheral zone.
The dog has been used as a model of this condition as this species also spontaneously develop prostatic neoplasia. The cell line CT1258 has been derived from a spontaneous canine prostate carcinoma and can induce tumour formation in mice.
(More? "Movember")
References
- ↑ Cunha GR, Vezina CM, Isaacson D, Ricke WA, Timms BG, Cao M, Franco O & Baskin LS. (2018). Development of the human prostate. Differentiation , 103, 24-45. PMID: 30224091 DOI.
- ↑ Rinaldi JC, Santos SAA, Colombelli KT, Birch L, Prins GS, Justulin LA & Felisbino SL. (2018). Maternal protein malnutrition: effects on prostate development and adult disease. J Dev Orig Health Dis , , 1-12. PMID: 29582717 DOI.
- ↑ Brechka H, McAuley EM, Lamperis SM, Paner GP & Vander Griend DJ. (2016). Contribution of Caudal Müllerian Duct Mesenchyme to Prostate Development. Stem Cells Dev. , 25, 1733-1741. PMID: 27595922 DOI.
- ↑ Liu H, Cadaneanu RM, Lai K, Zhang B, Huo L, An DS, Li X, Lewis MS & Garraway IP. (2015). Differential gene expression profiling of functionally and developmentally distinct human prostate epithelial populations. Prostate , 75, 764-76. PMID: 25663004 DOI.
- ↑ Huang L, Pu Y, Hu WY, Birch L, Luccio-Camelo D, Yamaguchi T & Prins GS. (2009). The role of Wnt5a in prostate gland development. Dev. Biol. , 328, 188-99. PMID: 19389372 DOI.
- ↑ Vezina CM, Lin TM & Peterson RE. (2009). AHR signaling in prostate growth, morphogenesis, and disease. Biochem. Pharmacol. , 77, 566-76. PMID: 18977204 DOI.
- ↑ Lowsley OS. The development of the human prostate gland with reference to the development of other structures at the neck of the urinary bladder. (1912) Amer. J Anat. 13(3): 299-346.
- ↑ Allgeier SH, Lin TM, Moore RW, Vezina CM, Abler LL & Peterson RE. (2010). Androgenic regulation of ventral epithelial bud number and pattern in mouse urogenital sinus. Dev. Dyn. , 239, 373-85. PMID: 19941349 DOI.
- ↑ Chughtai B, Sawas A, O'Malley RL, Naik RR, Ali Khan S & Pentyala S. (2005). A neglected gland: a review of Cowper's gland. Int. J. Androl. , 28, 74-7. PMID: 15811067 DOI.
- ↑ Omori A, Harada M, Ohta S, Villacorte M, Sugimura Y, Shiraishi T, Suzuki K, Nakagata N, Ito T & Yamada G. (2011). Epithelial Bmp (Bone morphogenetic protein) signaling for bulbourethral gland development: a mouse model for congenital cystic dilation. Congenit Anom (Kyoto) , 51, 102-9. PMID: 21848994 DOI.
Pubmed Bookshelf
- Madame Curie Bioscience Database Branching Morphogenesis of the Prostate
Reviews
Aaron L, Franco OE & Hayward SW. (2016). Review of Prostate Anatomy and Embryology and the Etiology of Benign Prostatic Hyperplasia. Urol. Clin. North Am. , 43, 279-88. PMID: 27476121 DOI.
Meeks JJ & Schaeffer EM. (2011). Genetic regulation of prostate development. J. Androl. , 32, 210-7. PMID: 20930191 DOI.
Cai Y. (2008). Participation of caudal müllerian mesenchyma in prostate development. J. Urol. , 180, 1898-903. PMID: 18801537 DOI.
Thomson AA. (2008). Mesenchymal mechanisms in prostate organogenesis. Differentiation , 76, 587-98. PMID: 18752494 DOI.
Cunha GR. (2008). Mesenchymal-epithelial interactions: past, present, and future. Differentiation , 76, 578-86. PMID: 18557761 DOI.
Timms BG. (2008). Prostate development: a historical perspective. Differentiation , 76, 565-77. PMID: 18462432 DOI.
Marker PC, Donjacour AA, Dahiya R & Cunha GR. (2003). Hormonal, cellular, and molecular control of prostatic development. Dev. Biol. , 253, 165-74. PMID: 12645922
Kim HG, Kassis J, Souto JC, Turner T & Wells A. (1999). EGF receptor signaling in prostate morphogenesis and tumorigenesis. Histol. Histopathol. , 14, 1175-82. PMID: 10506934
Cunha GR & Donjacour AA. (1989). Mesenchymal-epithelial interactions in the growth and development of the prostate. Cancer Treat. Res. , 46, 159-75. PMID: 2577188
Articles
Allgeier SH, Vezina CM, Lin TM, Moore RW, Silverstone AE, Mukai M, Gavalchin J, Cooke PS & Peterson RE. (2009). Estrogen signaling is not required for prostatic bud patterning or for its disruption by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Toxicol. Appl. Pharmacol. , 239, 80-6. PMID: 19523480 DOI.
Allgeier SH, Lin TM, Vezina CM, Moore RW, Fritz WA, Chiu SY, Zhang C & Peterson RE. (2008). WNT5A selectively inhibits mouse ventral prostate development. Dev. Biol. , 324, 10-7. PMID: 18804104 DOI.
Vezina CM, Allgeier SH, Fritz WA, Moore RW, Strerath M, Bushman W & Peterson RE. (2008). Retinoic acid induces prostatic bud formation. Dev. Dyn. , 237, 1321-33. PMID: 18393306 DOI.
Wang H, Leav I, Ibaragi S, Wegner M, Hu GF, Lu ML, Balk SP & Yuan X. (2008). SOX9 is expressed in human fetal prostate epithelium and enhances prostate cancer invasion. Cancer Res. , 68, 1625-30. PMID: 18339840 DOI.
Cook C, Vezina CM, Allgeier SH, Shaw A, Yu M, Peterson RE & Bushman W. (2007). Noggin is required for normal lobe patterning and ductal budding in the mouse prostate. Dev. Biol. , 312, 217-30. PMID: 18028901 DOI.
Letellier G, Perez MJ, Yacoub M, Levillain P, Cussenot O & Fromont G. (2007). Epithelial phenotypes in the developing human prostate. J. Histochem. Cytochem. , 55, 885-90. PMID: 17478449 DOI.
Cunha GR & Lung B. (1978). The possible influence of temporal factors in androgenic responsiveness of urogenital tissue recombinants from wild-type and androgen-insensitive (Tfm) mice. J. Exp. Zool. , 205, 181-93. PMID: 681909 DOI.
Historic
Evatt EJ. A contribution to the development of the prostate in man. (1909) Jour. of Anat. and Phys. 43: 314-321.
Lowsley OS. The development of the human prostate gland with reference to the development of other structures at the neck of the urinary bladder. (1912) Amer. J Anat. 13(3): 299-346.
Watson EM. The development of the seminal vesicles in man. (1918) Amer. J Anat. 24(4): 395 - 439.
Search PubMed
Search Pubmed "Prostate Embryology" Oct 2010 - All (775) Review (113) Free Full Text (171)
Search Pubmed: Prostate Embryology | Prostate Development | Corpora Amylacea |
Additional Images
Historic
Lowsley OS. The development of the human prostate gland with reference to the development of other structures at the neck of the urinary bladder. (1912) Amer. J Anat. 13(3): 299-346.
- Fetal Prostate
Terms
- Albarran glands - (Albarran's glands, glands of Albarran) submucosal glands located in the subcervical region of the prostate gland that empty into the posterior urethra. Named after Joaquin Maria Albarrán y Dominguez (1860 – 1912) a Cuban urologist.
- 5-α-reductase - enzyme that converts testosterone to dihydrotestosterone.
- androgen receptor - (AR)
- benign prostatic hyperplasia
- mesenchyme - embryonic connective tissue
- paraurethral gland - (Skene's gland) - female prostate gland is the correct nomenclature
- prostate gland - Greek, prostates = "one who stands before", "protector", a female prostate gland exists
- prostate cancer
- UGE - urogenital epithelium
- UGS - urogenital sinus
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Medline Plus Prostate Diseases | Prostate Cancer
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 25) Embryology Prostate Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Prostate_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G