Primordial Germ Cell Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 15: | Line 15: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''MicroRNA dynamics at the onset of primordial germ and somatic cells sex differentiation during mouse embryonic gonad development'''<ref name=PMID29187591><pubmed>29187591</pubmed></ref> "In mammals, commitment and specification of germ cell lines implies involves complex programs that include sex differentiation, control of proliferation and meiotic initiation. Regulation of these processes is genetically controlled by fine-tuned mechanisms of gene regulation in which microRNAs (miRNAs) are involved. We have characterized, by small-RNAseq and bioinformatics analyses, the miRNA expression patterns of male and female mouse Primordial Germ Cells (PGCs) and gonadal somatic cells at embryonic stages: E11.5, E12.5 and E13.5. Differential expression analyses revealed differences in the regulation of key miRNA clusters such as miR-199-214, miR-182-183-96 and miR-34c-5p whose targets have defined roles during gonadal sexual determination in both germ and somatic cells." [[Molecular Development - microRNA]] | |||
* '''Review - Key Signaling Events for Committing Mouse Pluripotent Stem Cells to the Germline Fate'''<ref name=PMID26674564><pubmed>26674564</pubmed></ref> "The process of germline development carries genetic information and preparatory totipotency across generations. The last decade has witnessed remarkable successes in the generation of germline cells from mouse pluripotent stem cells, especially induced germline cells with the capacity for producing viable offspring, suggesting clinical applications of induced germline cells in humans. However, to date, the culture systems for germline induction with accurate sex-specific meiosis and epigenetic reprogramming have not been well-established. In this study, we primarily focus on the mouse model to discuss key signaling events for germline induction. We review mechanisms of competent regulators on primordial germ cell induction and discuss current achievements and difficulties in inducing sex-specific germline development. Furthermore, we review the developmental identities of mouse embryonic stem cells and epiblast stem cells under certain defined culture conditions as it relates to the differentiation process of becoming germline cells." | * '''Review - Key Signaling Events for Committing Mouse Pluripotent Stem Cells to the Germline Fate'''<ref name=PMID26674564><pubmed>26674564</pubmed></ref> "The process of germline development carries genetic information and preparatory totipotency across generations. The last decade has witnessed remarkable successes in the generation of germline cells from mouse pluripotent stem cells, especially induced germline cells with the capacity for producing viable offspring, suggesting clinical applications of induced germline cells in humans. However, to date, the culture systems for germline induction with accurate sex-specific meiosis and epigenetic reprogramming have not been well-established. In this study, we primarily focus on the mouse model to discuss key signaling events for germline induction. We review mechanisms of competent regulators on primordial germ cell induction and discuss current achievements and difficulties in inducing sex-specific germline development. Furthermore, we review the developmental identities of mouse embryonic stem cells and epiblast stem cells under certain defined culture conditions as it relates to the differentiation process of becoming germline cells." | ||
* '''Sall4 is Essential for Mouse Primordial Germ Cell Specification by Suppressing Somatic Cell Program Genes'''<ref name=PMID25263278><pubmed>25263278</pubmed></ref> "The Sall4 zinc finger protein is a critical transcription factor for pluripotency in embryonic stem cells (ESCs). ...Given that Sall4 and Prdm1 are known to associate with the histone deacetylase repressor complex, our findings suggest that Sall4 suppresses the somatic cell program possibly by recruiting the repressor complex in conjunction with Prdm1; therefore, it is essential for PGC specification." | * '''Sall4 is Essential for Mouse Primordial Germ Cell Specification by Suppressing Somatic Cell Program Genes'''<ref name=PMID25263278><pubmed>25263278</pubmed></ref> "The Sall4 zinc finger protein is a critical transcription factor for pluripotency in embryonic stem cells (ESCs). ...Given that Sall4 and Prdm1 are known to associate with the histone deacetylase repressor complex, our findings suggest that Sall4 suppresses the somatic cell program possibly by recruiting the repressor complex in conjunction with Prdm1; therefore, it is essential for PGC specification." | ||
Revision as of 10:15, 18 January 2018
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
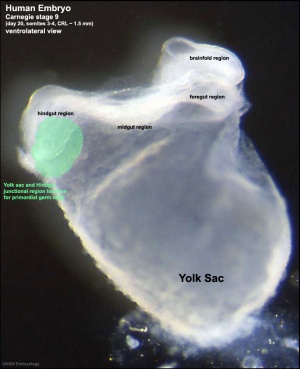
Early in development at the time of gastrulation a small group of cells are "put aside" to later form oocytes and spermatozoa. This population of cells is described as the primordial germ cells (PGCs). These cells also migrate initially into the posterior endoderm that forms the hindgut and from there into the genital ridge that will be the site of the developing gonad. The maintenance of pluripotency within this cell population may arise through epigenetic modifications that suppress somatic differentiation programs.
This population of cells when transformed is also thought to give rise to testicular germ cell tumours.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Primordial Germ Cell <pubmed limit=5>Primordial Germ Cell</pubmed> |
| Older papers |
|---|
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
- Developmental Biology (6th ed.) Gilbert Chapter 14 Intermediate Mesoderm
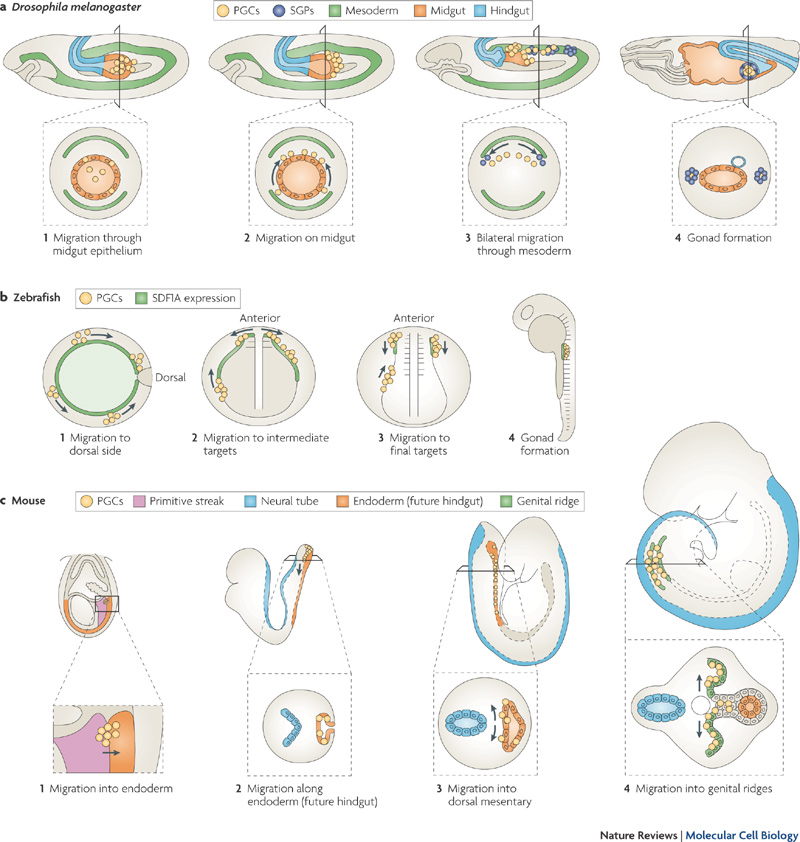
Primordial Germ Cell Migration
Species Comparison of Migration
Stages of primordial germ cell migration[8]
Mouse Migration Movies
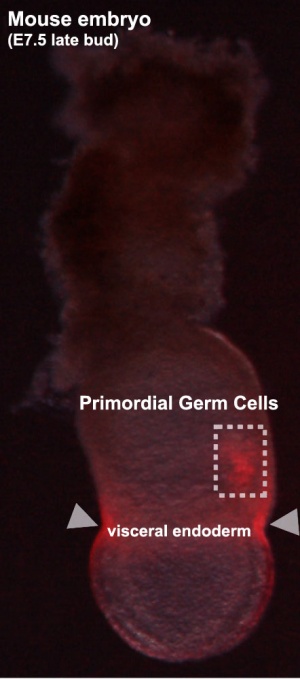
Labeled mouse primordial germ cells (E7.5)[9] |
Labeled mouse primordial germ cells (E10.5) See Mouse Migration Movies]] The movies below show labeled primordial germ cells (green) migrating within the mouse embryo between the periods of E9.0 to E10.5 into the genital ridge region that will later form the gonad. |
| Mouse Primordial Germ Cell Migration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||
- Links: Mouse Development
Fetal Ovary Meiosis
Fetal ovary meiosis[10]
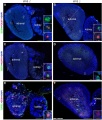
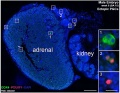
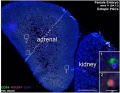
Ectopic PGCs
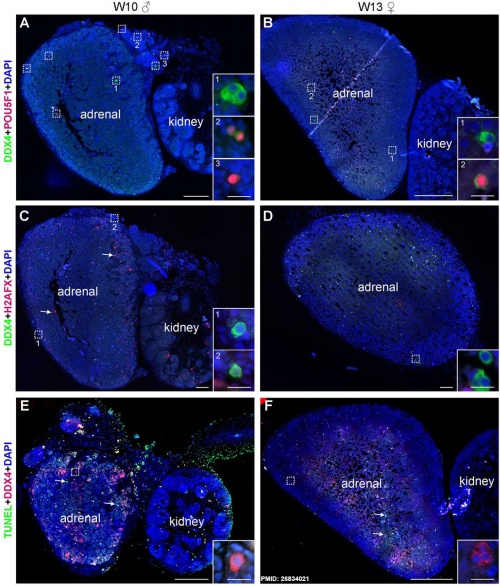
|
Ectopic PGC's in human adrenal gland in first trimester male week 8 (GA week 10) and female week 11 (GA week 13).[10]
|
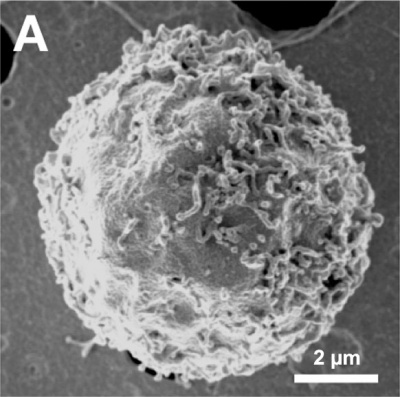
Cell Structure
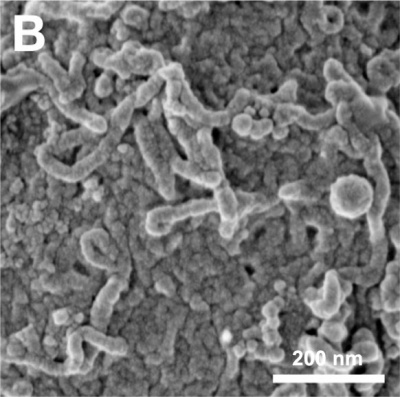
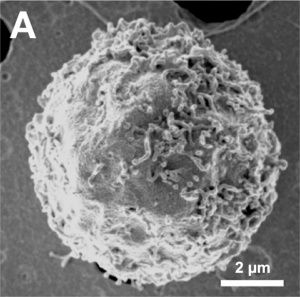
The images below are scanning electron micrographs of the surface of a chicken primordial germ cell that has been grown in culture.[11]
The first image shows the whole cell and the second image shows detail of the cell surface showing extensions.
DNA Methylation
| Mouse primordial germ cell DNA methylation[12]
Demethylation
|
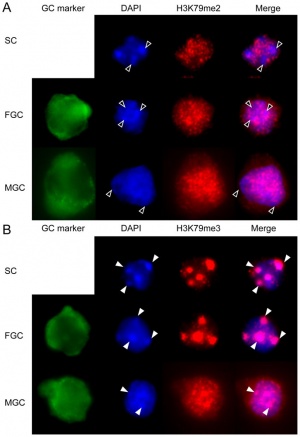
|
X-linked Gene Expression
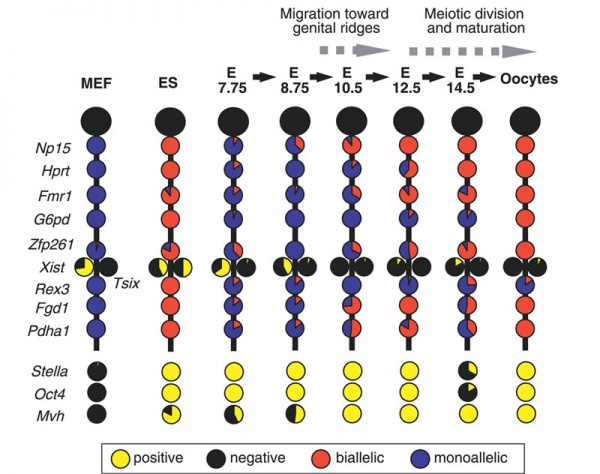
Mouse- X-linked gene expression during primordial germ cell development.[13]
Each circle graph indicates the ratio of cells that are positive (yellow) and negative (black) for each gene, and biallelically (red) and monoallelically (blue) expressed in cells positive for each gene.
- Links: X Inactivation | Mouse Development
Molecular
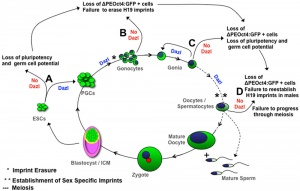
- Prdm1 and Prdm14 - PR domain proteins expressed in mouse (E6.25), suppresses somatic differentiation.
- Sall4 - zinc finger protein, inactivation of this transcription factor in mouse can reduce PGC number.[4]
A study has recently identified 11 genes that are specifically expressed in male and female fetal germ cells, both in vivo and in vitro, but are not expressed in embryonic stem cells.[15]
PGC Markers: alkaline phosphatase-positive, Oct4 (POU5F1), Fragilis (IFITM1)[16], Stella (DPPA3), Dazl, and Vasa (DDX4).
- Steel factor - (KITLG) a ligand for the KIT tyrosine kinase receptor.
- DAZL
- dead end - coding an RNA binding protein mainly expressed in the germ cells of vertebrates.
- Blimp1 - B-Lymphocyte induced maturation protein-1 (PRDM1)
- Prmt5 - protein arginine methyltransferase-5
- Nanog - knockdown induces apoptotic cell death in mouse migrating primordial germ cells.[17]
- AID - Activation-Induced cytidine Deaminase enzyme required for demethylation (removal of CpG methylation). Within the genome, DNA methylation is associated with epigenetic mechanisms and occurs at cytosine residues that are followed by guanines.[18]
Abnormalities
Teratomas
Common group of fetal tumors occuring along the body midline, anywhere from the coccyx to the pineal gland, reflecting the developmental PGC migration pathway (for review see [19]).
- Histologically classified as either mature or immature.
- Immature elements consisting principally of primitive neuroglial tissue and neuroepithelial rosettes and have have a generally favorable prognosis.
- Sacrococcygeal teratomas - most common site (70%–80% of all teratomas).
- classified into four types based on the amount of mass present externally versus internally.
Testicular germ cell tumours (seminoma)
References
- ↑ <pubmed>20886037</pubmed>| PLoS One.
- ↑ <pubmed>29187591</pubmed>
- ↑ <pubmed>26674564</pubmed>
- ↑ 4.0 4.1 <pubmed>25263278</pubmed>
- ↑ <pubmed>20845430</pubmed>
- ↑ <pubmed>19468308 </pubmed>
- ↑ <pubmed>19279135</pubmed>
- ↑ <pubmed> 20027186</pubmed>| Nature Reviews Molecular Cell Biology
- ↑ <pubmed>19997484</pubmed>| PMC2777314 | PLoS Genet.
- ↑ 10.0 10.1 <pubmed>26834021</pubmed>| Biol Open.
- ↑ <pubmed>20886037</pubmed>| PLoS One.
- ↑ <pubmed>21886830</pubmed>| PLoS One.
- ↑ <pubmed>17676999</pubmed>| PMC1950944 | PLoS Genet.
- ↑ <pubmed>19468308</pubmed>| PLoS One.
- ↑ <pubmed>20940145</pubmed>
- ↑ <pubmed>12659663</pubmed>
- ↑ <pubmed>19906868</pubmed>
- ↑ <pubmed>20236475</pubmed>
- ↑ <pubmed>15653597</pubmed>
Reviews
<pubmed>20371640</pubmed>| Reproduction <pubmed>20027186</pubmed> <pubmed>19875497</pubmed> <pubmed>19442193</pubmed> <pubmed>17446386</pubmed> <pubmed>15666347</pubmed> <pubmed>11565804</pubmed> <pubmed>11061420</pubmed>
Articles
<pubmed>19578360</pubmed> <pubmed>18953407</pubmed>
Search PubMed
Search Pubmed: Primordial Germ Cell Migration | Primordial Germ Cell | Testicular germ cell tumours
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Primordial Germ Cell Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Primordial_Germ_Cell_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G