Placenta - Maternal Decidua: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 4: | Line 4: | ||
This page gives an overview of aspects of maternal component of placental development, formed initially by the decidualization of the endometrium. | This page gives an overview of aspects of maternal component of placental development, formed initially by the decidualization of the endometrium. | ||
In week 2, the trophoblast shell cells proliferate and form a syncitiotrophoblast and cytotrophoblast layer around the conceptus. Syncitiotrophoblast cells migrate into the uterine wall, forming maternal blood-filled spaces (lacunae). | In week 2, the trophoblast shell cells proliferate and form a syncitiotrophoblast and cytotrophoblast layer around the conceptus. Syncitiotrophoblast cells migrate into the uterine wall, forming maternal blood-filled spaces (lacunae). | ||
Decidualization is the process of converting endometrial stromal cells into decidual cells and requires at least 8–10 days of hormone stimulation. A similar "decidual" cellular change, but less significant, also occurs in the uterine lining after ovulation during the secretory phase of the non-pregnant uterus. | Decidualization is the process of converting endometrial stromal cells into decidual cells and requires at least 8–10 days of hormone stimulation. A similar "decidual" cellular change, but less significant, also occurs in the uterine lining after ovulation during the secretory phase of the non-pregnant uterus. | ||
| Line 12: | Line 14: | ||
* in response to elevated progesterone levels | * in response to elevated progesterone levels | ||
* acts mainly through progesterone receptor (PR) PR-A (other isoform is PR-B) | * acts mainly through progesterone receptor (PR) PR-A (other isoform is PR-B) | ||
Placentation begins once the conceptus begins to implant in the uterine wall and the placenta will have both a fetal and a maternal component. | Placentation begins once the conceptus begins to implant in the uterine wall and the placenta will have both a fetal and a maternal component. | ||
| Line 30: | Line 33: | ||
* '''Disordered IL-33/ST2 activation in decidualizing stromal cells prolongs uterine receptivity in women with recurrent pregnancy loss'''<ref name=PMID23300625><pubmed>23300625</pubmed>\ [http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0052252 PLoS One.]</ref> "Decidualization renders the endometrium transiently receptive to an implanting blastocyst although the underlying mechanisms remain incompletely understood. Here we show that human endometrial stromal cells (HESCs) rapidly release IL-33, a key regulator of innate immune responses, upon decidualization. In parallel, differentiating HESCs upregulate the IL-33 transmembrane receptor ST2L and other pro-inflammatory mediators before mounting a profound anti-inflammatory response that includes downregulation of ST2L and increased expression of the soluble decoy receptor sST2. We demonstrate that HESCs secrete factors permissive of embryo implantation in mice only during the pro-inflammatory phase of the decidual process. IL-33 knockdown in undifferentiated HESCs was sufficient to abrogate this pro-inflammatory decidual response. Further, sequential activation of the IL-33/ST2L/sST2 axis was disordered in decidualizing HESCs from women with recurrent pregnancy loss. Signals from these cultures prolonged the implantation window but also caused subsequent pregnancy failure in mice. Thus, Il-33/ST2 activation in HESCS drives an autoinflammatory response that controls the temporal expression of receptivity genes. Failure to constrain this response predisposes to miscarriage by allowing out-of-phase implantation in an unsupportive uterine environment." | * '''Disordered IL-33/ST2 activation in decidualizing stromal cells prolongs uterine receptivity in women with recurrent pregnancy loss'''<ref name=PMID23300625><pubmed>23300625</pubmed>\ [http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0052252 PLoS One.]</ref> "Decidualization renders the endometrium transiently receptive to an implanting blastocyst although the underlying mechanisms remain incompletely understood. Here we show that human endometrial stromal cells (HESCs) rapidly release IL-33, a key regulator of innate immune responses, upon decidualization. In parallel, differentiating HESCs upregulate the IL-33 transmembrane receptor ST2L and other pro-inflammatory mediators before mounting a profound anti-inflammatory response that includes downregulation of ST2L and increased expression of the soluble decoy receptor sST2. We demonstrate that HESCs secrete factors permissive of embryo implantation in mice only during the pro-inflammatory phase of the decidual process. IL-33 knockdown in undifferentiated HESCs was sufficient to abrogate this pro-inflammatory decidual response. Further, sequential activation of the IL-33/ST2L/sST2 axis was disordered in decidualizing HESCs from women with recurrent pregnancy loss. Signals from these cultures prolonged the implantation window but also caused subsequent pregnancy failure in mice. Thus, Il-33/ST2 activation in HESCS drives an autoinflammatory response that controls the temporal expression of receptivity genes. Failure to constrain this response predisposes to miscarriage by allowing out-of-phase implantation in an unsupportive uterine environment." | ||
|} | |} | ||
{| class="wikitable collapsible collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! More recent papers | ! More recent papers | ||
|- | |- | ||
Revision as of 16:25, 15 July 2014
| Embryology - 18 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This page gives an overview of aspects of maternal component of placental development, formed initially by the decidualization of the endometrium.
In week 2, the trophoblast shell cells proliferate and form a syncitiotrophoblast and cytotrophoblast layer around the conceptus. Syncitiotrophoblast cells migrate into the uterine wall, forming maternal blood-filled spaces (lacunae).
Decidualization is the process of converting endometrial stromal cells into decidual cells and requires at least 8–10 days of hormone stimulation. A similar "decidual" cellular change, but less significant, also occurs in the uterine lining after ovulation during the secretory phase of the non-pregnant uterus.
- initiated during the mid-secretory phase of the menstrual cycle
- in response to elevated progesterone levels
- acts mainly through progesterone receptor (PR) PR-A (other isoform is PR-B)
Placentation begins once the conceptus begins to implant in the uterine wall and the placenta will have both a fetal and a maternal component.
During pregnancy, both the maternal blood volume increases by about 50% and the uterine blood flow increases 10 to 12 fold. Flow increase is due to the trophoblast cell invasion of the spiral arteries opening them into blood-filled spaces of the placenta.
For the non-pregnant uterus background see Menstrual Cycle and Uterus Development.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Maternal Decidua <pubmed limit=5>Maternal Decidua</pubmed> |
Maternal Decidua
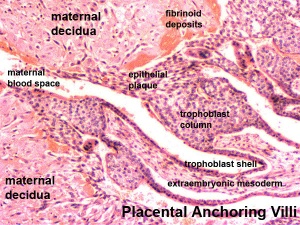
The maternal uterine endometrium stromal cells (fibroblast-like) are transformed by steroid hormones (progesterone) and embryonic signals into the decidua.
The entire maternal decidua is divided into three regions: decidua basalis, decidua capsularis and decidua parietals (decidua vera).
These 3 regions are named by their positional relationship to the conceptus.
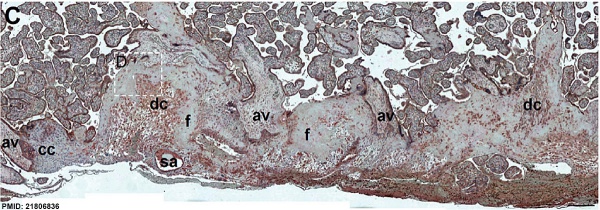
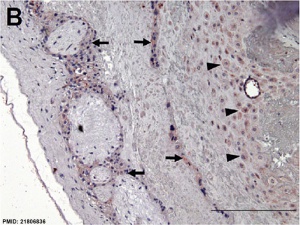
Immunostained placenta and decidua. [2]
SERPINE2 was extensively detected in decidual cells (dc), cytotrophoblasts, extravillous trophoblasts at the junction zone of the cell column (cc) and anchoring villi (av), and the endothelia of the spiral artery (sa); and weak staining was found in fibrinoids (f) and the villous mesenchyme.
Maternal Immune
How does the implanting conceptus avoid immune rejection by the maternal immune system? There are a number of maternal and embryonic mechanisms that are thought to act to prevent immune rejection of the implanting conceptus, though the complete mechanism(s) are unknown. This is particularly relevant to Assisted Reproductive Technologies involving donor eggs.
Below are some examples of research on this topic.
Decidual Immune Cells
- Specialised immune cells.
| Decidual Macrophages (Mϕ) | Decidual T cells | Uterine Natural Killer cells |
|---|---|---|
|
|
|
Chemokine Gene Silencing
- Remove the attraction of maternal immune cells.
A mouse study[3] has shown that the normal immune response to inflammation, accumulation of effector T cells in response to chemokine secretion does not occur during implantation. This is prevented locally by epigenetic silencing of chemokine expression in the decidual stromal cells.
Corticotropin-Releasing Hormone
- Kill the maternal immune cells.
Both maternal and implanting conceptus release CRH at the embryo implantation site. This hormone then binds to receptors on the surface of trophoblast (extravillous trophoblast) cells leading to expression of a protein (Fas ligand, FasL) that activates the extrinsic cell death pathway on any local maternal immune cells ( T and B lymphocytes, natural killer cells, monocytes and macrophages).[4] (Note - This cannot be the only mechanism, as mice with dysfunctional FasL proteins are still fertile).
Decidualization Factors
Preimplantation factor
- Preimplantation factor (PIF) secreted only by viable embryos.
- a 15 amino acid peptide MVRIKPGSANKPSDD
- regulates immunity, promoting embryo-decidual adhesion, and regulating adaptive apoptotic processes.[5]
Activin A
Member of the a transforming growth factor beta (TGFbeta) superfamily, contributes to human endometrial stromal cells (HESC) decidualization and has been localized to decidual cells in the human endometrium. (possibly also BMP2 and TGFbeta1)[6]
Prokineticin 1
Prokineticin 1 (PROK1) signalling via prokineticin receptor 1 (PROKR1) regulates Dickkopf 1 (DKK1) expression, a negative regulator of canonical Wnt signaling.[7]
References
Reviews
Articles
<pubmed>21144801</pubmed>
Search PubMed
Search Pubmed: Maternal Decidua | Decidualization
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 18) Embryology Placenta - Maternal Decidua. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Placenta_-_Maternal_Decidua
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G