Paper - On the development of the amnion and exocoelomic membrane in the previllous human ovum
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hertig AT. On the development of the amnion and exocoelomic membrane in the previllous human ovum. (1945) Yale J Biol Med. 18:107-15. PubMed 21007544
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
On the Development of the Amnion and Exocoelomic Membrane in the Previllous Human Ovum
Eighth Harry Burr Ferris Lecture delivered at Yale University on May 2, 1945. From the Free Hospital for Women, Brookline, Mass., the Departments of Pathology and of Obstetrics, Harvard Medical School, and the Department of Embryology, Carnegie Institution of Washington, Baltimore.
Introduction
An embryologist, as implied by his name, is primarily interested in the development of the embryo as a whole or at least in one or more of its intra-embryonic structures. Being a pathologist, Whose interest in embryology has been stimulated by contact with the pathological problems of The Boston Lying-in Hospital, I have, perforce, become more interested in those extra-embryonic structures, the placenta and its associated membranes. It is the purpose of this lecture to discuss the origin and early development of two such extra-embryonic structures, the amnion and the exocoelomic membrane. Due to the fortunate circumstance of having spent a stimulating year as a National Research Fellow with Dr. George L. Streeter (your Ferris Lecturer in 1936), my embryological status might be described as semi-professional. Certainly -by no stretch of the imagination would my embryological colleagues consider me a full-fledged professional, and I suspect they probably regard me as a rank amateur. Having thus explained why a combination pathologist and obstetrician has ventured into the anatomical specialty of embryology I now turn to the problem of the amnion and the exocoelomic membrane. If one regards the fertilized ovum, as does Dr. Streeter, from its inception to its death at the end of the Biblical three score and ten years, both these extra-embryonic structures are temporary, the exocoelomic membrane being far more evanescent than the amnion. The former is present but 4 to 5 days during the pre-villous stage and is the possible precursor of still another temporary structure, the yolk sac. The amnion, by contrast, appears early and persists throughout gestation — a veritable Methuselah as extra-embryonic structures go. Its life span is rivaled only by the chorion, from whose trophoblast it takes origin.
Studies on the origin of other primate extra-embryonic structures such as the primary mesoderm and the chorionic blood vessels first interested the author[1] in 1935. At that time, the material available for study consisted of five early Macaque ova ranging from 10 to 17 days of age together with 15 human ova (12 pre-somite and 3 somite) in the Carnegie Embryological collection. Morphologic evidence showed a simultaneous origin of angioblasts and primary mesoderm from the chorionic trophoblast of such primate ova. These early connective tissue elements of the chorion, arising by in situ delamination and differentiation, continue to develop as long as primary villi are being formed. The isolated vascular primordia thus formed soon possess power of independent growth and lumen formation, resulting not only in the vascularization of the chorion and primitive villi but the secondary villi as well.
Since these earlier studies it has been possible for the author, in collaboration with Dr. John Rock,[2][3][4][5] to obtain a series of 18 early human ova from uteri removed surgically prior to the first missed menstrual period. Eleven of these specimens are normal while the remainder show varying degrees of abnormality. The general features of many of these specimens have already been reported or descriptions are in press. All of these ova, even the pathological ones (with the exception of a 4-day segmenting morula), throw some light upon the origin of the amnion and exocoelomic membrane. However, the nine normal pre-Villous specimens ranging from 7.5 to 12.5 days of age (8th to 13th days) will be discussed in some detail, because it is in these ‘hitherto unknown stages of human development that the beginning of these extraembryonic structures is first seen.
Ova of the eighth day
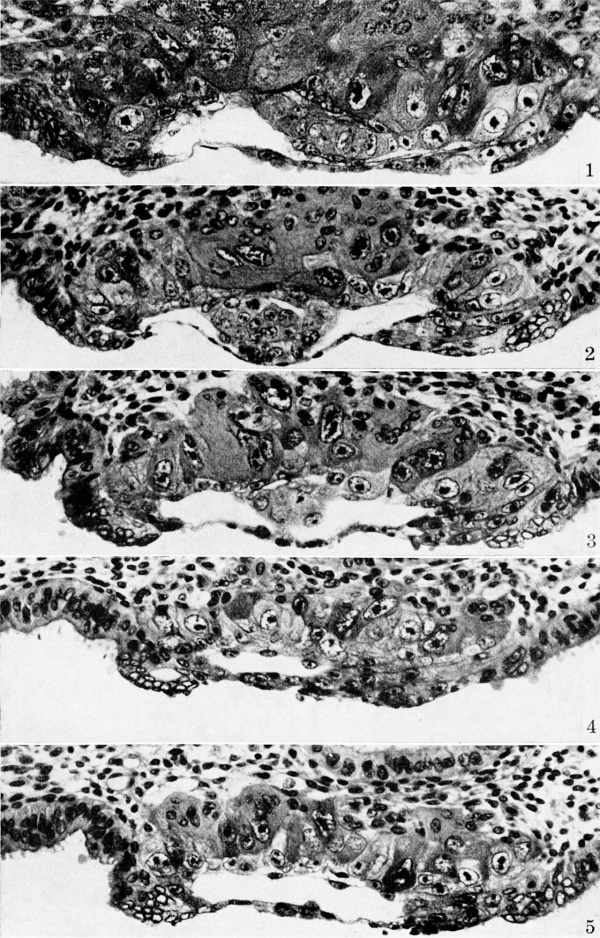
The material in this stage consists of two specimens, both in approximately the same stage of development, Whose essential form is that of a superficially but Well-attached blastocyst containing a bilaminar germ disk. The solid trophoblast, composed of primitive syncytiotrophoblast and cytotrophoblast, is invading the physiologically edematous endometrium characteristic of the 21st to 22nd day of the menstrual cycle. Plate 1 shows the essential features displayed by these ova with respect to the formation of the amnion and extraembryonic mesoblast.
In both specimens, the amniotic cavity is a tiny irregular or multilocular slit lying between the ectoderm and the adjacent trophoblast. The cells of the latter immediately dorsal to the amniotic cavity differ in appearance from those of the remaining trophoblast, they are smaller, flatter, and darker. This is best seen in the specimen (Carnegie 8225) shown on plate 1, fig. 1. These amniogenic cells, however, are still attached to their parent trophoblast and appear to be delarninating in situ therefrom.
Both specimens show isolated instances of in situ delamination of primitive mesoblasts. This process is best seen in the specimen (Carnegie 8020) shown on plate 1, figs. 4 and 5. These spindle-shaped mesoblasts are in one instance (fig. 4) still obviously a part of their parent trophoblast, whereas in another section (fig. 5) these maturing cells have almost separated from their tissue of origin. Such difierentiating mesoblasts tend to occur principally on the inner surface of the trophoblast at the embryonic pole although they are present on the abembryonic pole but in lesser numbers. This may possibly be so because the abembryonic portion of the blastocyst wall is less mature at this stage in the development of the ovum. This primitive chorionic connective tissue is apparently the precursor of the exocoelomic membrane which will ultimately, but only temporarily, line the chorionic cavity of the older pre-villous ova. Its origin in the macaque is shown by Heuser[6][7] to be associated with both the chorionic trophoblast and the primitive endoderm of the embryo. This apparent double origin may -be due merely to continuity of mesoblast with primitive endoderm, since Heuser[7] states that two types of cells are apparently involved. While the membrane does ultimately become attached to the margins of the germdisk, both in the monkey and in man, it appears to the author that in man the exocoelomic (Heuser’s) membrane arises entirely from primitive mesoblast which in turn arises by delamination from the trophoblast. Morphologic evidence for this will be shown in older pre-villous specimens.
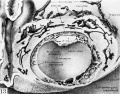
Plate 1. Ova of the eighth day
Fig. 1. The middle cross section of an ovum not over 7.5 days of age. Above the somewhat eccentrically placed germ-disk is a barely discernible cleft—the amniotic cavity. Above the latter are a few dark, flattened amniogenic cells de-lamiznating in situ from the adjacent trophoblast. Toward the left of the chorionic cavity, a few primitive mesoblastic cells are seen which likewise arise from trophoblast by in situ delamination. Carnegie No. 8225, section 20-5-5, X 300 (minus).
Fig. 2. The middle cross section of a 7.5-day ovum. The cleft-like amniotic cavity lies above the germ-disk and beneath amniogenic cells arising from the adjacent trophoblast. A single, flattened meso-blastic cell is seen to the right of the germ-disk and lying upon the inner aspect of the tropho-blast. Carnegie No. 8020, section 6-5-9, X 300 (minus).
Fig. 3. The same ovum as fig. 2 but 7 sections removed. The amniotic cavity is less evident because of its smaller size although the amniogenic cells overlying it are more prominent. Carnegie No. 8020, section 6-5-2, X 300.
Fig. 4. The same ovum as shown in figs. 2 and 3 but the section passes to one side of the germ-disk. Note at the embryonic pole the three prominent mesoblastic cells which are delaminating in situ from the adjacent trophoblast. A somewhat more advanced stage of this process is seen in the two mesoblastic cells shown in fig. 5. Carnegie 8020, section 6-3-6, X 300 (plus).
Fig. 5. A section of a. 7.5-day ovum (same specimen as figs. 2-4) showing a more advanced stage of mesoblastic formation. Note the two, nearly detached mesoblastic cells delaminating from, but still lightly attached to the trophoblast at the embryonic pole of the ovum. Carnegie 8020, section 6-4-3, X 300 (plus).
Fig 1 Carnegie No. 8225
Fig 2 Carnegie No. 8020
Fig 3 Carnegie No. 8020
Fig 4 Carnegie No. 8020
Fig 5 Carnegie No. 8020
Ova of the ninth and tenth days
There are four specimens in this group, representative sections of which are shown on plate 2, figs. 6 to 9. These ova are characterized by progressive embedding of the blastocyst in end-ometrium of the 22nd to 26th day of the cycle with development of a series of intercommunicating lacunae within the trophoblast. Such lacunae, the intervillous space of the future placenta, will later on in the previllous stage be flooded by maternal blood. It is only during the latter part of the present stage that such lacunae connect with endometrial sinusoids, thereby allowing small amounts of maternal blood to flow into the ovum.
The development of the amnion progresses during this stage, the cavity becomes larger and the amniogenic membrane becomes more differentiated. However, not all specimens fit into this orderly pattern. Thus, the youngest specimen (Carnegie 8155, pl. 2, fig. 6) shows a beautifully developed amniotic cavity but relatively little amniogenesis even though there are a few trophoblastic cells of the future amnion lying dorsal to the cavity. The other three specimens, consistent with their more mature development, all show a definite amniotic cavity which is more or less enclosed by a primitive amnion, the latter still attached to its parent trophoblast. With the gradual development of the amnion the cells composing it metamorphose from their cytotrophoblastic prototype to one of fibroblastic or epithelioid type. Coincidentally, they become partially but incompletely detached from the adjacent trophoblast and assume a more or less perfect membranous form. This diaphanous structure, attached to the edge of the primitive embryonic ectoderm, begins to seal off the amniotic from the chorionic cavity.
Formation of primitive mesoblast continues through this stage of ovular development. The ovum is now sufliciently embedded so that most of its wall has begun to form primitive trophoblast. Hence, all specimens of this group show a fairly uniform degree of mesoblast formation at the abembryonic as Well as at the embryonic pole. In two of the specimens (Carnegie 8171 and 8215) shown on plate 2, figs. 7 and 8, there is already an early exocoelomic membrane beginning to form. Perhaps this is associated with the relatively deeper implantation of these two specimens. It is curious that the oldest ovum in this group (Carnegie 8004) shown on plate 2, fig. 9, shows relatively little mesoblast formation even though its amniogenesis is well advanced. This apparent discrepancy may be due to the recent hemorrhage in the chorionic cavity. This maternal blood, undoubtedly an abnormal feature in this otherwise normal ovum, gained entrance through a break in the chorionic membrane. That the hemorrhage had stopped is shown by the clotting of the blood within the lacuna from which the hemorrhage arose. Whether the hemorrhage had a temporarily deleterious efiect on mesoblast formation is impossible to say. In any event, the formation of primitive mesoblast has been slowed up for some reason, although it is not entirely absent. Thus, in plate 2, fig. 9 there are a pair of these primitive connective tissue cells delaminating in situ from the inner surface of the trophoblast lining a the chorionic cavity. Elsewhere in the ovum, n-ot illustrated, there is an imperfect segment of the exocoelomic membrane, already attached to the endoderm of the germ disk.
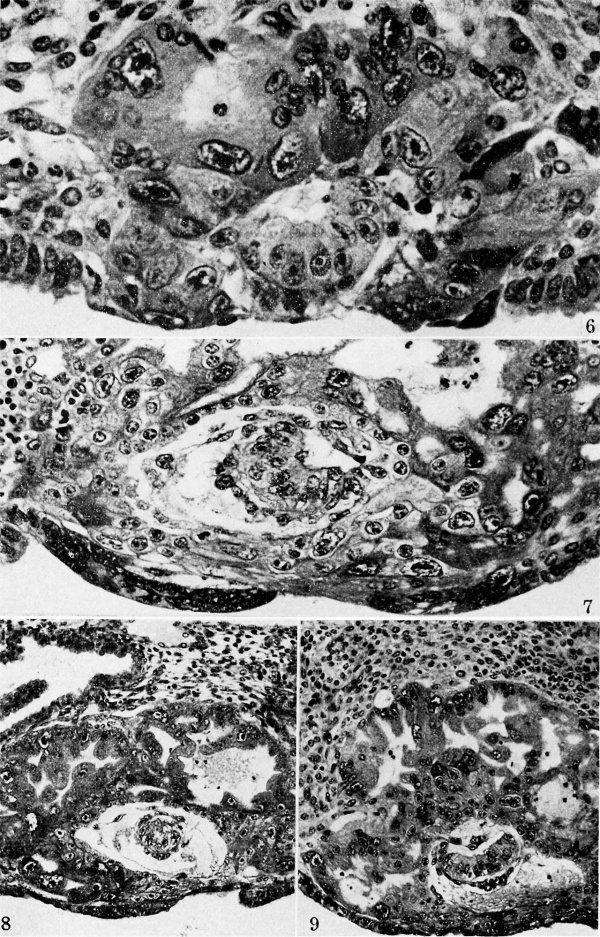
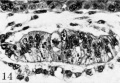
Plate 2. Ova of the ninth and tenth days
Fig. 6. A mid-cross section from an ovum of approximately 8 days of age. Note the prominent amniotic cavity above the primitive ectoderm of the germ-disk but the amniogenic cells have not yet begun to form. There are, however, a few mesoblastic cells to the right of the germ-disk, several of which are still attached to their parent trophoblast. Carnegie 8155, section 4-4-8, X 500.
Fig. 7. A mid-cross section of an ovum 8 to 9 days of age. Note that the amniotic cavity is no more advanced than is that of the younger specimen seen in ii . 6, although there are several amniogenic cells arising from the adjacent trophoblast in the ol er specimen. Likewise, mesoblastic formation is more advanced, as shown by the presence of an early exocoelomic membrane. In places the mesoblastic cells of the latter are still attached to the trophoblast from which they appear to be originating. Carnegie 8171, section 3-2-12, X 300.
Fig. 8. A mid-cross section of an ovum of 8 to 9 days of age. The amniotic cavity is a mere slit although the amniogenic cells are well formed and have become detached from the adjacent trophoblast. The exocoelomic membrane is similar in appearance and stage of development to that of specimen shown in fig. 7. Carnegie 8215, section 12-4-5, X 140.
Fig. 9. A mid-cross section of a 9.5 -day ovum. The amniotic cavity is well formed, as are the enclosing amniogenic cells although the latter are still attached in places to their parent trophoblast. Mesoblastic formation is scanty and is represented in this section by two delaininating cel s seen toward the lower left, of the chorionic cavity. The granular material in the latter is extravasated blood which is a possible factor in delaying the formation of the mesoblast. Carnegie 8004, section 11-4-4, x 160.
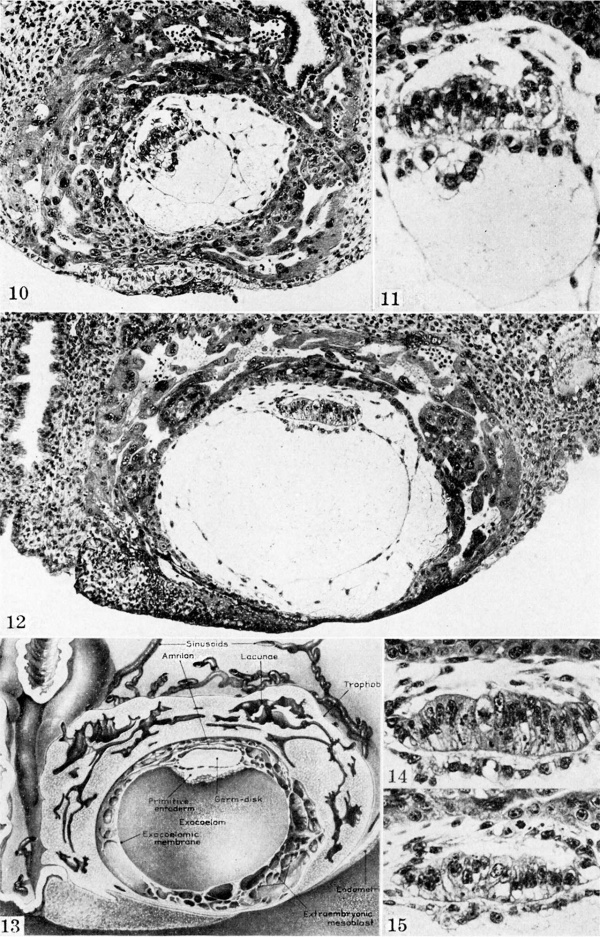
Ova of the twelfth and thirteenth days
Three specimens of this stage are available for study but only two are illustrated. The essential features of amniogenesis and formation of the exocoelomic membrane during this phase of ovular development are shown on plate 3, figs. 10 to 15. These ova are A characterized by the continued differentiation of early primordial villi, most marked in the older of the two specimens. Maternal blood has likewise flooded the lacunae of the latter. The chorionic cavity has become progressively more distended so that embedding of the ovum does not quite keep pace with its general growth. As a result the ovum is elevated above the surrounding endometrium and continues to be imperfectly oovered by endometrium.
Tlhe amniotic cavity is now a distinct dome-shaped or flattened ellipsoidal space. It is as yet imperfectly enclosed by a thin membranous amnion whose cells are still attached to the trophoblast. New amniogenic cells are being added to the definitive amnion as shown by the cell in mitosis (pl. 3, fig. 15 ) which is attached to both the trophoblast and the amnion.
The exocoelomic (Heuser’s) membrane has now reached its peak of development. It is a relatively complete, but by no means an imperforate membrane lining the chorionic cavity. It is attached to the endoderm at the edge of the germ-disk. Its inner surface is relatively smooth but its outer aspect is quite the reverse. Its component cells, of flattened or stellate fibroblastic type, are continuous with a gossamer web of mesoblasts which are even yet delaminating in situ from the inner surface of the surrounding trophoblast. The gross form of this evanescent structure is best seen in the reconstruction of the older specimen (Carnegie 7700) shown on plate 3, fig. 13.
The primordial villi are just beginning to form in the older specimen (pl. 3, fig. 12). Focal accumulations of cytotrophoblast develop in the chorionic membrane and grow peripherally. In so doing, they become invested by syncytiotrophoblast, thereby account ing for the presence of syncytium as the outer element of the mature chorionic villus. Coincident with these growth processes, the core of the future villus differentiates into premature mesoblastic and angioblastic tissue. The details of this phenomenon have been previously reported by the author[1], as mentioned earlier in the lecture. However, the previously studied human material was neither as young nor as perfectly preserved and sectioned as the present specimens so that this confirmation of previous data is worthy of comment.
Plate 3. Ova of the twelfth and thirteenth days
Fig. 10. A mid-cross section of an 11.5-day ovum. The amniotic cavity is more advanced in its development than in the previous specimens although the amniogenic cells now a distinct membrane are still attached to the adjacent trophoblast. The exocoelome is now well contained within its membrane which is continuous above with the primitive endoderm of the germ-disk. Mesoblast formation is still continuing and is laying the foundation for the future connective tissue of the chorion. Carnegie 7699 section 8-5-3, X 100.
Fig. 11. The germ-disk, amnion and exocoeloomic membrane of the 11.5-day ovum at higher magnification to show greater detail. Note the attachment of the amniogenic cells to their parent trophoblast. Carnegie 7699, section 8-S-3, x 250.
Fig. 12. A mid-cross section of a 12.5-day ovum. The amniotic cavity is a cleft-like space above the primitive ectoderm of the germ-disk. It is barely enclosed above by the amniogenic cells which are still arising, in places, from adjacent trophoblast: Details of this process, at higher magnification, are shown in figs. 14 and 15. The exocoelomic (Heuser’s) membrane has reached the high point of its development and is destined to disintegrate within the next few days. Its essential mesoblastic nature is shown by the character of its cells and by their attachment. in places, to the trophoblast from which they arose. Carnegie 7700, section 6-1-S, x 100.
Fig. 13. plastic reconstruction of half the 12.5-day ovum. The cut surface represents the section seen in fig. 12. This picture, by its three dimensional concept serves to emphasize the nature of the amnion, the einocoelomic membrane and the mesoblast lining the chorionic cavity at this stage in their development. Carnegie 7700, reconstruction, x 75.
Fig. 14. A mid-cross section of the amnion and germ-disk of the 12.5-day ovum (same section as fig. 12). The cleft-like amniotic cavity is seen dorsal to the primitive ectoderm and is barely enclosed by the extremely thin amnion above. Carnegie 7700, section 6-1-5, X 250.
Fig. 15. para-median section of the germ-disk and amniotic cavity from the 12.5-day ovum. Note the variable thickness and appearance of the amniogenic cells which are being added to by dividing cells derived from the adjacent trophoblast. Carnegie 7700, section S-7-7, x 250
Fig 10 Carnegie 7699
Fig 11 Carnegie 7699
Fig 12 Carnegie 7700
Fig 13 Carnegie 7700
Fig 14 Carnegie 7700
Fig 15 Carnegie 7700
Villous ova of the fourteenth to seventeenth day
Two specimens from this stage of embryonic development are available (pl. 4, figs. 16 and 17). They have been described in detail by Dr. Heuser in conjunction with Dr. Rock and the author.7 These ova are characterized by the development of chorionic villi which have become simply branched in the older specimen. Mater nal blood is circulating through the intervillous space of the early placenta, but some of it has escaped through the confining membrane of the latter into the uterine cavity. and endometrial glands adjacent to the ovum. The latter is still not completely embedded within the endometrium but is covered by the “Schlusscoagulum” of Peters. Indeed, the younger of the two specimens is essentially similar to the famous embryo described by that author. The exocoelomic membrane as such has almost completely dis appeared although several bubble-like remnants of it are seen throughout the chorionic cavity of the younger specimen. The yolk sac which may be its successor, at least functionally, is now well formed and is larger than the amnion in both specimens. In the younger, it is composed of a single layer of mesoblasts attached to the endoderm, whereas in the older it is formed by a double layer of such cells.
The amnion is now in the stage familiar to all students of embryology — that of a blunted and somewhat flattened cone whose base rests upon the ectoderm of the germ disk. In the younger specimen, its exceedingly thin wall of mesoblasts merges imperceptibly into the primitive connective tissue of the adjacent chorion. In the older specimen, however, it possesses a double-layered wall composed mainly of very thin mesoblastic tissue except at its apex where its less differentiated polyhedral cells are continuous with the adjacent chorionic connective tissue. Here the latter forms a definite adhesion-like strand that is interpreted as being the forerunner of the amniotic duct.
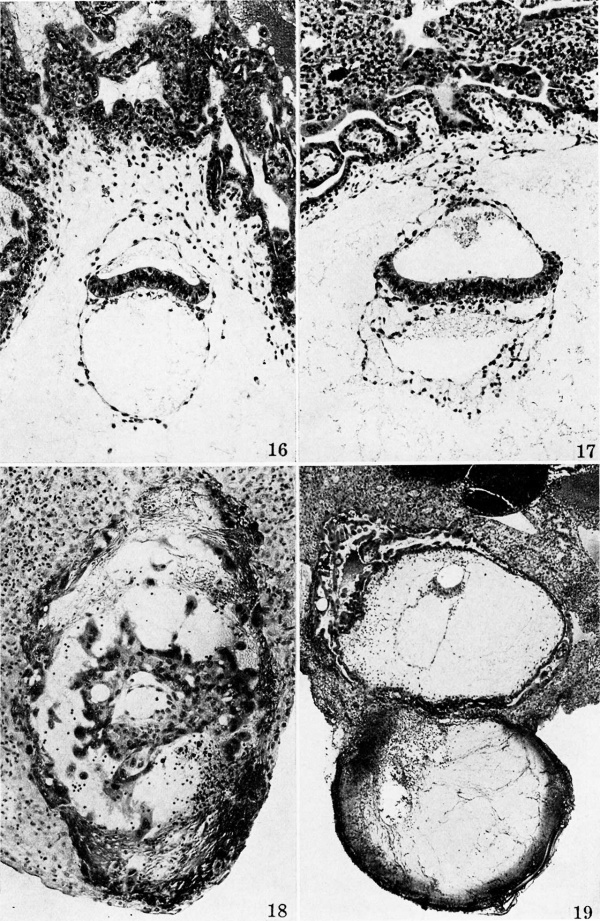
Plate 4 Normal villous ova and pathologic pre-villous ova
Fig. 16. A maid-cross section of the embryo and adjacent chorion of a 14-day ovum. The amniotic cavity is now a flattened cone and is completely enclosed by the thin amniotic membrane above and the primitive ectoderm below. The yolk-sac has now appeared and has already outstripped the amniotic cavity in size although the former has probably not been present for more than 36 ours. Carnegie 7801, section 12-1-3, X 100.
Fig. 17. A mid-cross section of the embryo and adjacent chorion of a 16-day ovum. The doublelayered amnion is attached by mesocblast to the adjacent chorion. The amniotic cavity is approximately equal in size no the yolk-sac cavity. The latter is bounded by the endoderm above and the double-layered yolk-sac elsewhere. Carnegie 7802. section 44-3-5. X 100.
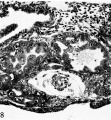
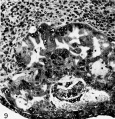
Fig. 18. A mid-cross section of a pathologic, pre-villous ovum of uncertain age but probably 11 to 12 days old Its embryonic mass and hence the amnion are absent although there is an exioocoelomic membrane and some mesoblastic tissue lining the chorionic cavity. Carnegie 7771, sec tion 3-4-I. x 100.
Fig. 19. A mid-cross section of a pathologic ovum of uncertain age. Its relatively normal embryo is equivalent to that of a 14- or possibly 15-day specimen although its trophoblast is defective, especially toward the right side. Note the relatively poor mesoblastic formation associated this region in contrast to the abundant mesoblast associated with the better, but still defective trophoblast on the left side. Carnegie 7800, section 15-1-3, X 35.
Pathological ova
Of the seven specimens available for study two illustrated in plate 4 (figs. 18 and 19) are of interest with respect to the formation of .primitive mesoblast and the exocoelomic membrane. Nature has seen fit to perform on each of these ova a teratological experiment which has resulted in the absence of the embryo in one and impaired the development of the trophoblast in the other. The blighted ovum, thus bereft of its embryonic mass, has still been able to form an exocoelomic membrane because its formative trophoblast was relatively unimpaired. It would seem, therefore, that in this instance, at least, the embryo was not a contributory or inductive force in the formation of the exocoelomic membrane. It might be further observed that since there is no embryo there is no amnion because the inductive force of the former with respect to the latter was lacking.
The other specimen, presumably older, shows an absolute hypoplasia of a portion of its trophoblast and only a relative hypoplasia of the remainder. The amount of primitive mesoblast associated with the two portions of trophoblast is strikingly different; the better developed region has given rise to an abundant primitive connective tissue whereas the other has generated practically none. It is apparent from these fortuitous natural experiments that the trophoblast plays the leading role in the formation of extra-embryonic structures of mesoblastic origin; the exocoelomic membrane arising therefrom without induction from the embryo whereas the latter does induce the trophoblast to form its amnion.
Summary and Conclusion
From a series of eleven normal human ova, nine in the pre-villous and two in the villous stage, is demonstrated the progressive in situ delamination of primitive mesoblast, exocoel-omic membrane, and amnion from trophoblast. The primitive mesoblast ultimately becomes the connective tissue of the chorion, Whereas the exocoelomic membrane is an extremely temporary mesoblastic structure lining the chorionic cavity during the ninth to the thirteenth days of ovular development. The amnion begins to delaminate from adjacent trophoblast during the eighth day and gradually encloses the concave space above the germ-disk during the next five days to form the amniotic cavity.
Observations on two pathological pre-villous human ova deficient in embryonic and trophoblastic tissues, respectively, show that the exocoelomic membrane will form in the absence of an embryo, although an amnion will not, and that the degree of mesoblast formation is correlated with the degree of trophoblasftic development.
References
- ↑ 1.0 1.1 Hertig AT. Angiogenesis in the early human chorion and in the primary placenta of the macaque monkey. (1935) Carnegie Instn. Wash. Publ. 459, Contrib. Embryol. 25: 37-81.
- ↑ Hertig AT. and Rock J. Two human ova of the pre-villous stage, having an ovulation age of about eleven and twelve days respectively. (1941) Carnegie Instn. Wash. Publ. 525, Contrib. Embryol., 29: 127-156.
- ↑ Hertig AT. and Rock J. On the development of the early human ovum, with special reference to the trophoblast of the previllous stage: A description of a normal and 5 pathologic human ova. (1944) Amer. J. Obstet Gynecol., 47: 149-184.
- ↑ Hertig AT. and Rock J. Two human ova of the pre-villous stage, having a developmental age of about seven and nine days respectively. (1945) Contrib. Embryol., Carnegie Inst. Wash. Publ. 557, 31: 65-84.
- ↑ Rock J. and Hertig AT. Some aspects of early human development. (1942) Amer. f. Obstet. Gynecol, 44: 973-983.
- ↑ Heuser CH. An intrachorionic mesothelial membrane in young stages of the monkey (Macacus rhesus). (1932) Anat. Rec, 52, Suppl., 15-16.
- ↑ 7.0 7.1 Heuser CH. Early development of the primitive mesoblast in embryos of the rhesus monkey. (1938) Carnegie Inst. (Publ. 501), Cooperation in Research 383-88.
Cite this page: Hill, M.A. (2024, April 24) Embryology Paper - On the development of the amnion and exocoelomic membrane in the previllous human ovum. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_On_the_development_of_the_amnion_and_exocoelomic_membrane_in_the_previllous_human_ovum
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G