Paper - Development of the human placenta in the first three months of gestation (1960): Difference between revisions
m (→References) |
m (→References) |
||
| Line 285: | Line 285: | ||
{{Ref-Davies1944b}} | {{Ref-Davies1944b}} | ||
{{Ref-DistzWest1941}} | |||
Distz, J. H. & West, C. M. (1941). A human ovum at the previllous stage. J. Anat., Lond., 75, 269-281. | Distz, J. H. & West, C. M. (1941). A human ovum at the previllous stage. J. Anat., Lond., 75, 269-281. | ||
Revision as of 22:41, 12 August 2020
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Hamilton WJ. and Boyd JD. Development of the human placenta in the first three months of gestation. (1960) J Anat. 94(3): 297-328. PMID14399291 | PDF
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Development of the Human Placenta in the First Three Months of Gestation
By W. J. Hamilton Anatomy Department, Charing Cross Hospital Medical School
and
J.D. Boyd Anatomy Department, University of Cambridge
Introduction
In spite of an extensive literature, going back to the eighteenth century, our knowledge of the details of the structure of the human placenta is still most fragmentary. This is due partly to the fact that much of the work published on its histology has been concerned with late stages of its development and predominantly with placentae separated from the uterus. So far as the earlier stages are concerned, individual investigators have had access only to a very limited number of specimens. Thanks to the invaluable collaboration of a number of obstetricians and pathologists over a number of years, the authors now have in their possession an extensive collection of pregnant uteri at all stages of gestation from about the 11th day after fertilization to full term. Serial sections of a part or the whole of the implantation sites and placental areas of these specimens have been prepared. Consequently the authors have been able to make a full survey of the structure of the human placenta throughout the period of gestation. The present contribution deals with the establishment of the placenta and with its development up to the 60 mm. C.R. length foetal stage. By the end of this period, the general structure of the placenta is essentially that which it will possess throughout its later development. Our aim is to provide a general survey of the early history of the human placenta as a background to more detailed consideration, in later communications, of particular aspects of the relationship between mother and foetus.
In a classical review, published in 1927, Grosser summarized the then existing knowledge of implantation and development of the placenta in Man. The early stages of human development available for study at that time were unsatisfactory . Many of the specimens were incomplete or even fragmentary. For example, only five sections of the Miller ovum (Streeter, 1926) were available for study, and some classical descriptions were undoubtedly based on pathological material (e.g. T. B. 1, Bryce & Teacher, 1908). Since Grosser’s review there have been outstanding contributions on the early stages of human implantation and placental development. Especially noteworthy are the investigations of Hertig and Rock since 1941 (summarized by Hertig, Rock & Adams, 1956). To a lesser extent, a number of other investigators have also added descriptions of specimens at these stages (see Hamilton & Boyd, 1950; Hamilton, Boyd & Mossman, 1952; and Mazanec, 1959, for summaries). Additions in the last thirty years to knowledge of stages of human placental development later than the 18th post-ovulational day and especially of the fully established in situ placenta have been much less extensive. Indeed, as indicated in earlier communications (Boyd & Hamilton, 1950; Hamilton & Boyd, 1951), there is still disagreement and doubt on a number of fundamental points relating to the structure and function of the human placenta at all stages of its development.
The numerous contributions from the Carnegie Laboratory of Embryology on implantation of the blastocyst and on placental development in the macaque monkey (Wislocki & Streeter, 1988; Heuser & Streeter, 1941; Ramsey, 1949, 19546) have provided a remarkably clear picture of foetal-uterine relationships in this primate. In spite of numerous differences, in detail, including a fundamental one in implantation itself, from the conditions obtaining in Man, the data on macaque development constitute an essential comparative basis for any serious consideration of the conditions in the human uterus during gestation. Throughout the study of our material we have found it necessary at every stage to make comparison with the equivalent one in the macaque. It must be stressed, however, that, as Ramsey (1954a) has stated with regard to placental circulation in the two species, the findings in the macaque are not replicas of those found in Man. Conclusions on the morphology of the placenta in the one species cannot, therefore, be transferred automatically to serve as the basis for interpretations of the conditions in the other primate.
A special problem in the description of the development of the foetal components of the placenta is posed by the various appearances presented by the trophoblast at different stages of development and in different regions of the blastocyst wall. Part of the difficulty resides in the variety of terminologies used by investigators in their descriptions. Recently, the authors (Boyd & Hamilton, 1960) have discussed this terminological problem, and it is not considered necessary to review the matter in detail in the present contribution. As the suggestions on terminology put forward by us, however, will be used in our subsequent descriptions, a brief summary of them is required. It must at once be stressed that this terminology is essentially a topographical and descriptive one. Briefly, those cells constituting the wall of the unimplanted blastocyst, and which are individual cellular units, together constitute the blastocystic trophoblast. All of the trophoblast of later stages is derived from it. During the early stages of implantation the blastocystic trophoblast differentiates into primitive syncytiotrophoblast and primitive cytotrophoblast. The former, receiving increments over a considerable period from the latter, is, by the development of the cytotrophoblastic, or trophoblastic, shell, separated into two varieties. First, there is the definitive syncytium which forms a covering for the villi and lines the lacunar and, later, the intervillous spaces. Secondly, there is syncytium on the maternal side of the shell which we call peripheral syncytium. Both varieties of syncytium are augmented until late stages of placental development by further contributions from the related cytotrophoblast. This last named tissue gives origin to: (1) the villous cytotrophoblast (Langhan’s layer); (2) proliferations at the tips of the villi which constitute the cytotrophoblastic columns; and (8) the trophoblastic shell itself. It must be stressed that although there is good evidence that cytotrophoblast from any of the three named subdivisions can give origin to syncytium, reverse differentiation of the latter into the former seems, in normal development, not to occur.
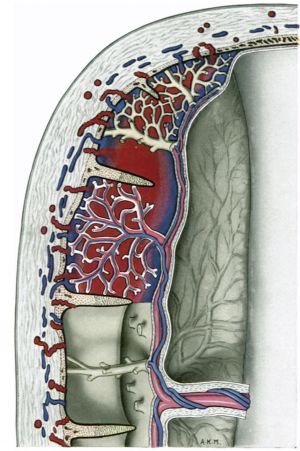
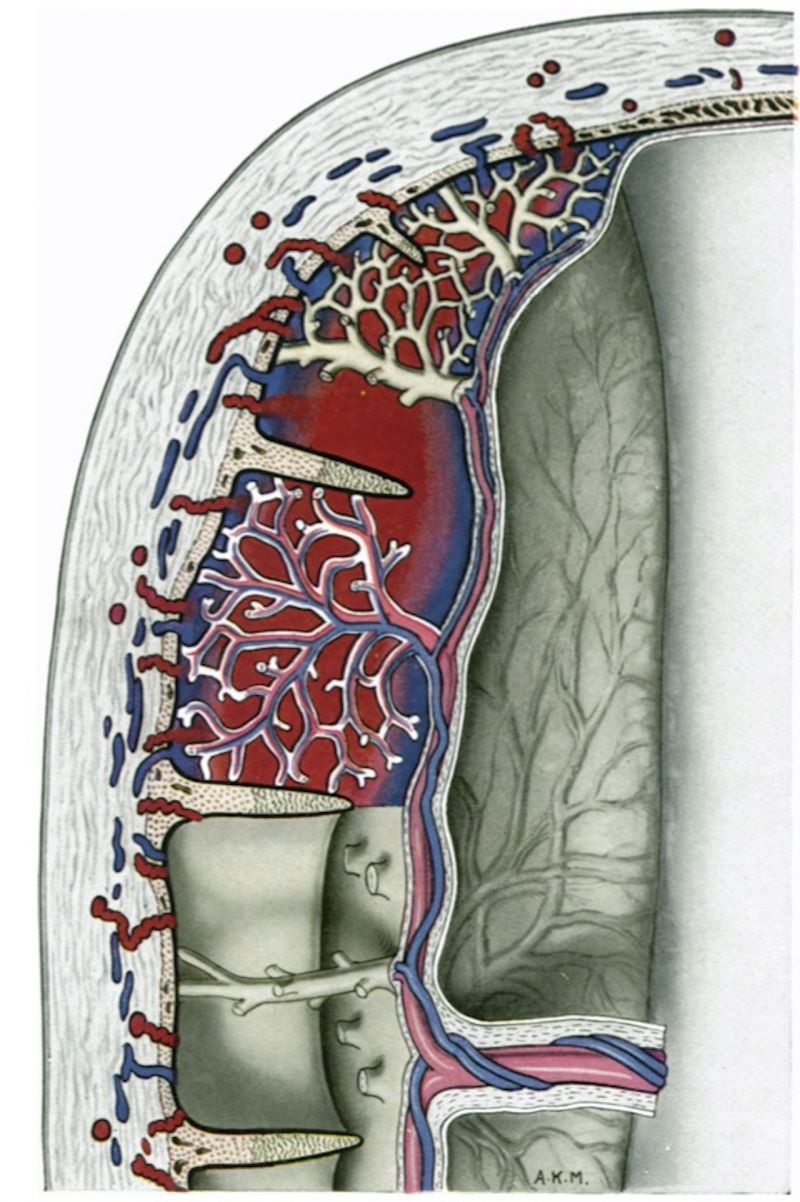
Until the contribution of Spanner (1935) it was generally accepted that, for the most part, the decidual arteries open into the intervillous space, at or near the margins of the septa which separate the cotyledons, and that the uterine veins communicate with the intervillous space through orifices in the interseptal zones of the basal plate. Indeed, Bumm (1893) considered that the openings of the decidual arteries are usually situated quite far down the septa and well away from the basal plate itself. Spanner, however, as a result of observations on placentae injected in situ, concluded that the arteries open into the intervillous space solely through the basal plate and that the blood is only drained away from this space through a specialized peripheral region of it called the marginal sinus. Stieve (1942) and we, ourselves (Hamilton & Boyd, 1950; Boyd, 1956), have criticized Spanner’s concept of the intervillous circulation and have denied the existence of a constant marginal sinus in Spanner’s sense of the term. In particular, it can be shown that there are frequent venous openings through the whole of the basal plate. Ramsey (19540, 1956 a, b) has also shown that uterine venous connexions with the intervillous space can be found in all regions of the uterine surface of the macaque placenta. She points out, indeed, that, in this monkey, such connexions are even less prominent at the margin of the placenta than in Man; she attributes this difference to the superficial implantation in the macaque which ‘ precludes contact between the placental margin and the endometrium when vascular connexions are being established’.
In view of the physiological and clinical importance of the circulation in the intervillous space, and of the conflict of opinion on it, special attention has been given to the morphology of the circulatory arrangements in the placenta at all stages of our study.
Material and Methods
The available in situ specimens in the age group with which we are here concerned have, for the most part, been obtained from hysterectomies (Table 1). In most instances the co-operation of the obstetrician was such that the intact uteri were available within a short time after their removal. Fixation, therefore, has frequently been under optimal conditions. One specimen (42 mm. stage) came from a woman who was accidentally electrocuted. Several others were obtained from material removed during diagnostic curettage. Further details on the history of the younger specimens are given subsequently. In these earlier stages, the embryonic age has been estimated from the coital and menstrual history and from the general state of development of the trophoblast and embryo. In the older specimens, the crownrump measurement has been used in the assessment of the age but, when fully available, the reproductive history has also been taken into account.
In addition to the in situ material we have an extensive collection of sections through early placentae obtained from hysterotomies, curettages and abortions. For certain histological and cytological purposes this disparate material has proved very useful as a supplement to our in situ specimens. We also have available a number of non-pregnant uteri from hysterectomies often at known stages of the uterine cycle.
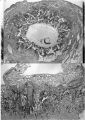
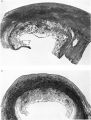
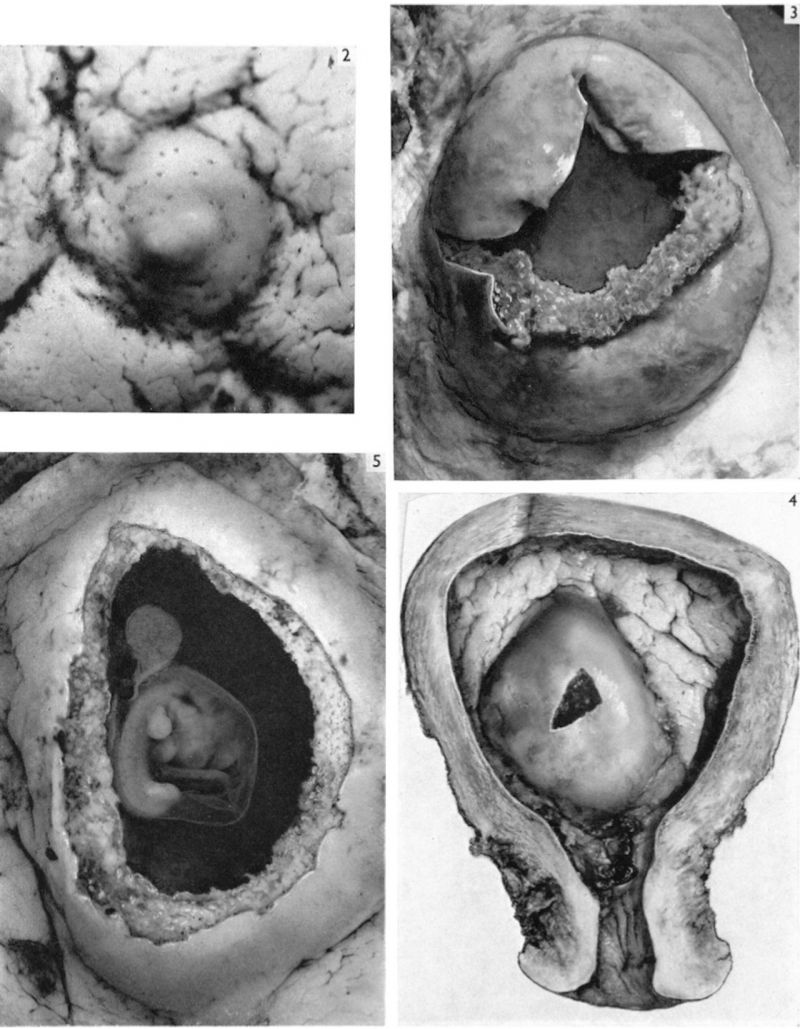
The earliest developmental stage at our disposal is the Barnes embryo, of which a brief description has already been published (Hamilton, Barnes & Dodds, 1948). This specimen, almost completely implanted in the endometrium, was found in a uterus obtained by hysterectomy on the 25th day of a menstrual cycle. Previous cycles were regularly 28 days in duration and the menstrual periods lasted 2-3 days. A history which appears to be reliable establishes that coitus had taken place on the 10th and on the 12th days previous to the operation. Consequently, the maximum conceptional age is not more than 12 days. The uterus was opened at the attachments of the broad ligaments. On examination with a low-power binocular microscope an elevation (Pl. 2, fig. 2) was seen on the posterior uterine wall as a slightly raised translucent area, clearly demarcated from the surrounding endometrium. After fixation, with Bouin—Allen fluid, the area became opaque but was still elevated above the level of the surrounding endometrium. A block of uterine tissue containing the swelling was removed and, after double embedding, was cut serially at 7u. After sectioning, the dimensions of the chorionic sac are approximately 0-931 x 0-770 x 0-737 mm.
Table 1
| Table 1 | ||
|---|---|---|
| Specimen | Age | History |
| Barnes | 11-12 days | Hysterectomy |
| Missen | 14 days | Curettage |
| Gar | 18 days | Hysterectomy |
| Shaw | 18 days | Hysterectomy |
| H. 712 (Camb.) | 23 days | P.M. |
| H. 710 (Camb.) | 26 days | Hysterectomy |
| 100 (CX.) | 10 mm. | Hysterectomy |
| H. 33 (Camb.) | 10 mm. | P.M. |
| H. 757 (Camb.) | 10 mm. | Abortion |
| 101 (CX.) | 10 mm. | Hysterectomy |
| H. 186B (Camb.) | 14 mm. | P.M. |
| 102 (CX.) | 15 mm. | Hysterectomy |
| 103 (CX.) | 22 mm. | Hysterectomy |
| H. 594 (Camb.) | 22 mm. | Hysterectomy |
| H. 767 (Camb.) | 22 mm. | Hysterectomy |
| 104 (CX.) | 25 mm. | Hysterectomy |
| H. 719A (Camb.) | 27-5 mm. | Hysterectomy |
| 105 (CX.) | 29 mm. | P.M. |
| H. 630 (Camb.) | 30 mm. | Hysterectomy |
| 106 (CX.) | 10 weeks | (embryo Hysterectomy not available) |
| 107 (CX.) | 31 mm. | Hysterectomy |
| H. 808 (Camb.) | 33 mm. | Abortion |
| H. 643 (Camb.) | 83-5 mm. | Hysterectomy |
| 108 (CX.) | 34 mm. | Hysterectomy |
| H. 789 (Camb.) | 36 mm. | Hysterectomy |
| 109 (CX.) | 40 mm. | Hysterectomy |
| 110 (CX.) | 42 mm. | P.M. |
| H. 223 (Camb.) | 42 mm. | Hysterectomy |
| H. 802 (Camb.) | 45 mm. | Hysterotomy |
| H. 653 (Camb.) | 46 mm. | Hysterectomy |
| H. 220 (Camb.) | 47 mm. | Hysterectomy |
| H. 721 (Camb.) | 48 mm. | Hysterectomy |
| 111 (CX.) | 50 mm. | Hysterectomy |
| H. 691 (Camb.) | 59 mm. | Hysterectomy |
| 112 (CX.) | 60 mm. | Hysterectomy |
| 113 (CX.) | 60 mm. | Hysterectomy |
| H. 201 (Camb.) | 60 mm. | Hysterectomy |
The next available stage of development is an embryo presented by Dr G. A. K. Missen (late of the Post-Graduate Medical School and now of Guy’s Hospital Medical School, London). The patient’s menstrual history had been irregular for the previous 83 months. The last menstrual period began on 19 July 1953; the curettage was performed on 21 August. The part of the endometrium in which the embryo was subsequently found was fixed immediately in formol-sublimate saline. The block of tissue was cut serially at 54. Owing to the irregularity of the previous menstrual cycles and lack of detail on coital history, it is not possible to determine the precise age of this embryo. The maximum diameters of the chorionic sac (measuring from the edge of the trophoblastic shell) are 1-66 x 1-43 mm. The embryonic disc, which is cut obliquely, measures 0-280 x 0-214 mm. The primitive streak is differentiating and the first indication of Hensen’s node and the primordium of the head process have appeared. The specimen, therefore, corresponds in state of development to such early embryos as Carnegie 1801 (Heuser, Rock & Hertig, 1945) and the EdwardsJones—Brewer (Brewer, 1937, 1938); these embryos fall into the period of 183-15 days of development. Through the kindness of Prof. T. B. Johnston of Guy’s Hospital we had access to the implanted embryo, H. R. 1, described by him (Johnston, 1940, 1941). Prof. R. J. Keller of Edinburgh presented us with a single section through an implanted ovum obtained by curettage. The two specimens probably also fall within the same age-group. The ‘Gar’ embryo, described by the late Prof. C. M. West, has been loaned to us by Prof. J. S. Baxter, of Cardiff. This embryo was obtained as a result of hysterectomy. From the menstrual and coital history, West (1952) concluded that the embryo was 18 days old. The developmental state of this specimen corresponds closely to that of other embryos known to be of this age. The chorionic sac was embedded in the anterior uterine wall close to the ostium of the left uterine tube. It appeared as a hemispherical haemorrhagic swelling 5 mm. in diameter and raised 4 mm. above the surface of the adjacent endometrium. A block of uterine wall including the myometrium and endometrium in which the embryo was embedded, was removed and sectioned serially. In the sections the chorionic sac is somewhat triangular in shape, tapering to a blunt point towards the myometrium. The widest internal measurement of the sac is 2mm. The external measurement at this level is 4-1 mm. The embryonic disc measures 0-56 mm. antero-posteriorly and 0-6 mm. from side to side.
The next older specimen, the Shaw embryo, full details of which have been given in an earlier publication (Hamilton & Gladstone, 1942), was originally estimated from the coital history to be 20-23 days old. From its general development, however, we consider that its age is distinctly younger than this estimation and that it is about 18 days old. The uterus of this specimen was opened immediately after a subtotal hysterectomy. A small elevation about 1 cm. in diameter was observed on the posterior uterine wall. This swelling was excised with a block of surrounding tissue and fixed in Carnoy-sublimate. A complete series of sections, 104 thick, was obtained. The measurements of the chorionic sac are as follows: maximum external diameter, 11 mm.; maximum internal diameter, 8 mm. The embryonic disc measured 1:05 x 1-34 mm.
The next specimen (H. 712) was an implantation site in a uterus removed for multiple fibromyomata. The history is unprecise as there had been menstrual irregularity for some years before the operation. The recorded L.M.P. was 3 months before the operation. The embryo, however, measured 1-8 mm. in total length and possessed seven somites. We place it at about the 28rd day of gestation. Unfortunately, the specimen only became available after the intact uterus, which had been in formalin for 8 days, was opened by a pathologist in the absence of knowledge of a possible pregnancy. Consequently, the implantation site is incomplete though the embryo itself was undamaged.
The last specimen (H. 710) on which specific comment is required is a complete implantation site from a uterus removed for menorrhagia. The menstrual history was irregular and it is not possible to give a precise date for ovulation. The coital history is also uncertain. The L.M.P. commenced on 14 December 1956 and the operation took place on 23 January 1957. Assuming ovulation on the 14th day of the cycle the ovulational age would be 26 days; comparison with the histories of the few other described somite embryos at comparable stages of development suggests that, in fact, H. 710 may be a little older than this. The uterus was opened immediately after operation. A pronounced swelling projected from the posterior wall of the uterus near the fundus. Half-strength Bouin’s fluid was dropped on the swelling and a cruciate incision was made in the decidua capsularis to remove the embryo. A block of tissue containing the collapsed chorionic sac together with the underlying endometrium and uterine musculature was removed, embedded, and cut in serial sections. One half of the block was sectioned at right-angles to the surface of the endometrium and the other parallel to it.
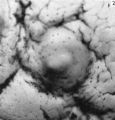
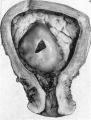
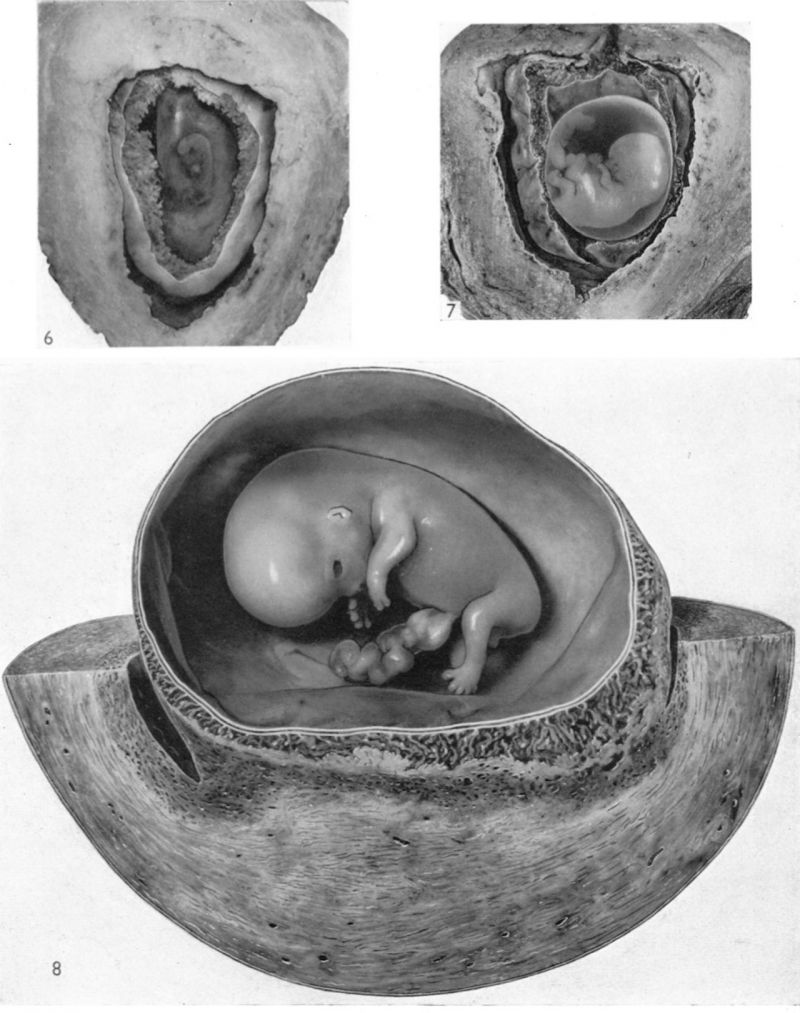
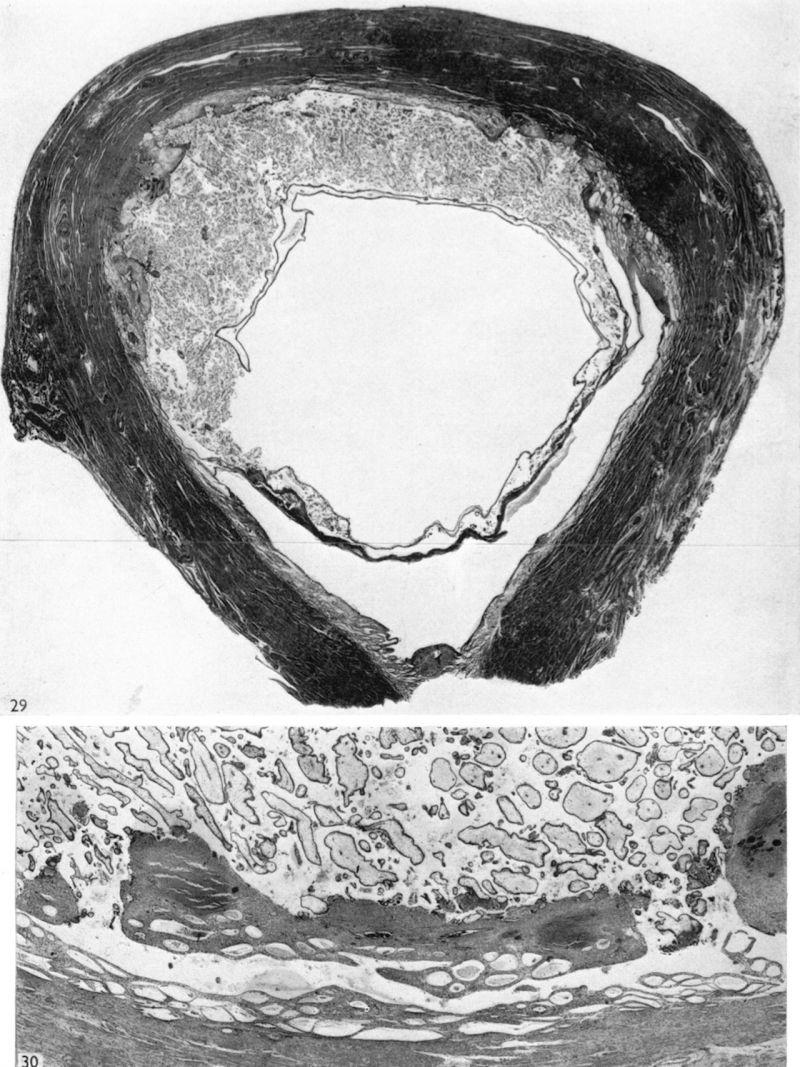
The histories of the patients from whom the placentae associated with embryos of between the 10 and 60 mm. stages were obtained do not require special comment. The salient points of the specimens are given in Table 1. Some of the specimens were fixed by perfusion either from the uterine vessels or by way of the umbilical cord. In several of the specimens indian ink in plasma was injected either by way of the uterine arteries or directly into the intervillous space. Representative specimens of this period are illustrated in Pl. 2, figs. 4 and 5, and in PI. 3, figs. 6-8.
Description Of Specimens
11- to 12-day Stage
The Barnes embryo
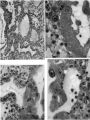
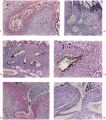
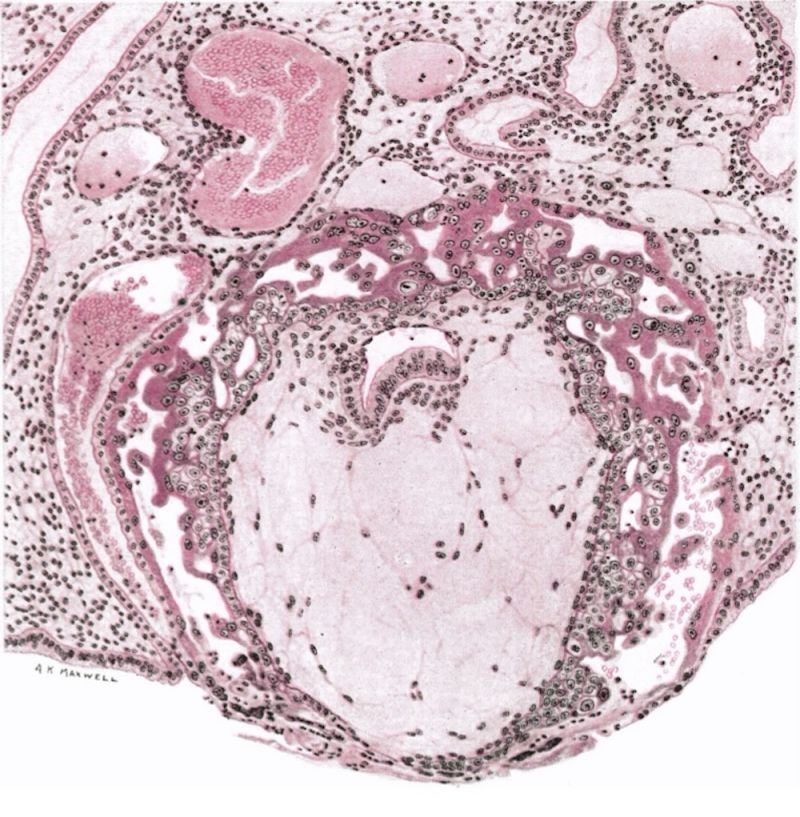
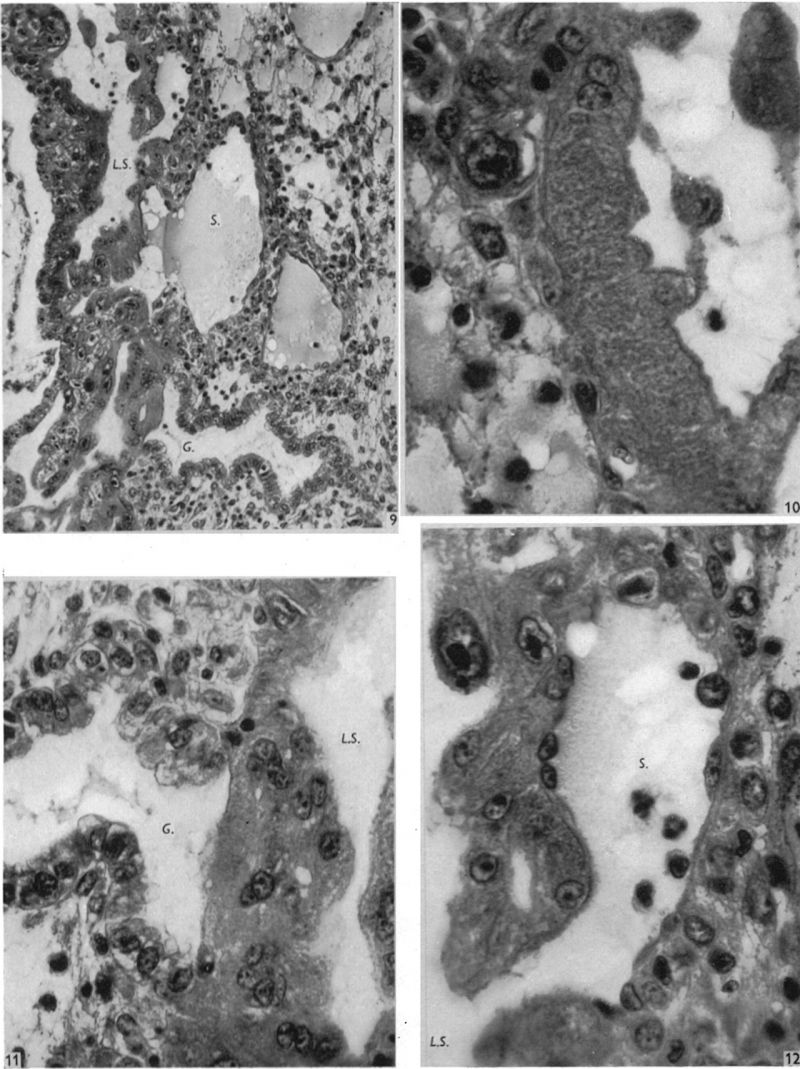
This embryo is almost completely implanted, the site of entry being covered with a thin layer of endometrium though the flattened uterine epithelium has not extended entirely over it (Pl. 1, fig. 1). The regional differentiation of the trophoblast is not uniform. Both primitive syncytium and cytotrophoblast are present but no definite indications of transitional cell types between the two have been seen. In the abembryonic region, towards the uterine cavity, the blastocyst wall is thin and composed almost exclusively of cytotrophoblast with only a small amount of syncytium, and here there is no lacunar formation. Elsewhere, however, the main mass of trophoblast is clearly differentiated into a cytotrophoblastic component and a spun-out primitive syncytium in which the intercommunicating lacunae, containing a small number of maternal blood corpuscles, are well established (Pl. 4, fig. 9). A wax-plate reconstruction of the implantation site indicates that the majority of the lacunae are in free communication with each other. When compared with the maternal blood cells elsewhere in the sections those in the lacunar spaces stain very poorly; presumably some influence operating on them in the spaces has markedly diminished their tinctorial affinities. The primitive syncytium possesses a granular cytoplasm; where it is in contact with the maternal endometrial tissue the latter frequently shows marked oedema and appearances of degeneration (PI. 4, fig. 10). Where the syncytium forms the boundary of the lacunae it is usually irregular with surface projections of vacuolated protoplasmic fronds and streamers. In some regions, however, there is the appearance of a not very well defined brush border. In no part of the primitive syncytium were any mitotic figures observed. No evidence for amitotic division was found but frequently syncytial nuclei are closely apposed or clumped together (PI. 4, fig. 11), although long stretches of protoplasm may be devoid of them (PI. 4, fig. 10). The syncytial nuclei vary considerably in size (Pl. 4, fig. 12). The cytotrophoblast completely covers the embryonic mesoderm, which is, in fact, separated from this covering by a space which is a fixation artefact. There are in addition, however, aggregates of cytotrophoblastic cells, particularly round the equator of the chorion, which project into the syncytium. These aggregates may represent regional proliferative activity of the cytotrophoblast though no sign of special mitotic activity is shown by them. The cytotrophoblastic proliferations are always covered by syncytium and are similar in appearance to those described in other embryos at this stage of development (Stieve, 19386; Dible & West, 1941; Hertig & Rock, 1941, 1944; Wilson, 1954). Such proliferations have often been regarded as forming the early primordia of the chorionic villi and they will be discussed later. It must be stressed that, at this stage, the presence of the lacunae within the syncytium gives the latter a sponge-like structure. Consequently, the system of lacunar spaces is primarily labyrinthine and, as there are no free villi, the term ‘intervillous space’ is not quite appropriate.
The endometrial stroma, immediately round the implantation site, shows the predecidual reaction, and is markedly oedematous. The endometrial cells themselves do not yet show the histological changes distinctive of later stages. The intercellular spaces are distended with a slightly eosinophilic exudate containing occasional blood cells. The uterine glands are tortuous and dilated with large amounts of secretion. Those adjacent to the implantation site are deflected aside by the proliferating trophoblast. Glands near and in contact with the trophoblast show degenerative changes. In some instances the glandular epithelium has disappeared at the point of contact with the trophoblast which thus comes to plug the glandular lumen (PI. 4, figs. 9, 11). Maternal blood corpuscles are present in the lumina of many of the glands but the precise source of such blood cells is not apparent.
Round the implantation site, and especially on its deep surface, a number of thinwalled, intercommunicating sinusoids, lined with endothelium, are in free communication with the lacunar spaces. At the points of communication the primitive syncytium is directly continuous with the endothelium lining the sinusoids (PI. 4, fig. 12). There is no evidence at these points of junction of endothelial reaction to the syncytium. The sinusoids can be traced to veins in the deeper part of the endometrium. Typical coiled arteries are found in the endometrium; they are surrounded by regions of specialized, non-oedematous, decidua. Several of the arteries approach and become intimately related to the implantation site. We have, however, been unable to satisfy ourselves that there is any open communication with the lacunar spaces. Many of the arteries appear to end blindly in the endometrium. No evidence of alterations in the walls of the coiled arteries was observed in this specimen.
14- to 18-day Stages
The Missen embryo
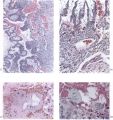
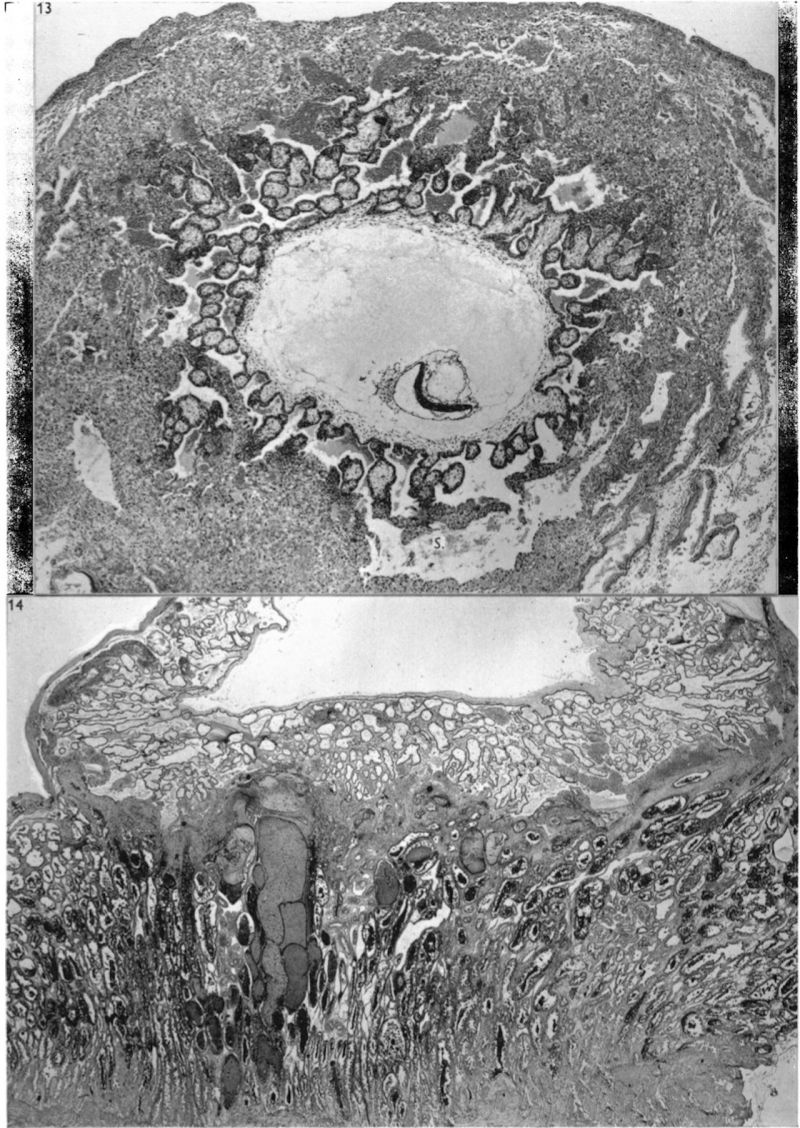
The general arrangement of the trophoblast and related endometrium in this embryo is shown in Pl. 5, fig. 13. It will be seen that the cytotrophoblast has increased very considerably in amount when compared with the 12-day specimen. The trabeculae of peripheral syncytial trophoblast of the previous stage have been invaded by the cytotrophoblast or have been transformed partially into it; certainly the latter tissue now forms a more or less continuous trophoblastic shell round the conceptus. In this peripheral shell there are to be found many spaces of varying size and shape. Some of these spaces contain maternal blood, others possess variously shaped masses of syncytium which is often vacuolated and shows phagocytosis of maternal blood cells. The nuclei of the syncytial masses frequently possess extremely large eosinophilic nucleoli. The spaces in the trophoblastic shell communicate only infrequently with the main portion of the lacunar system. On the maternal side of the trophoblastic shell many strands of peripheral syncytium are present; they extend as streamers into the surrounding endometrium making contact with the sinusoids, the coiled arteries and the glands. The presence of these syncytial streamers makes the external aspect of the trophoblast irregular. On the embryonic side of the trophoblastic shell, many of the trabeculae (‘villi’), separated by the dilated lacunae, can be traced to the chorionic plate. These trabeculae are mainly arranged radially and are covered by a thin layer ofsyncytium. They now possess a mesodermal core. At the periphery of those of the radial strands which reach the trophoblastic shell there is a concentration of small, darkly staining, cytotrophoblastic cells which may well be the original source of the existing shell and which are doubtless the source of subsequent additions to it. The appearance of cytotrophoblast and of mesoderm within the trabecular syncytium does not alter the fundamentally labyrinthine nature of the lacunar spaces of earlier stages. Not all of these trabeculae, however, reach the trophoblastic shell for a number of them arise from the chorionic plate, or even from the main trabeculae, to terminate as free villi. It is such finger-like proliferations of trophoblast, with the contained mesoderm, that lay the basis for the subsequent villous structure of the placenta. At this stage, however, the human placenta clearly presents a combination of labyrinthine and villous characters. It must be stressed that the main trabeculae, which we will call primary villous stems, were never free villi. From this stage forwards, however, it would be pedantic to avoid calling the lacunar system the intervillous space.
The endometrium adjacent to the trophoblast in this embryo shows a well marked decidual reaction. Portions of the uterine glands in the immediate vicinity of the trophoblast are eroded or destroyed. Other glands, situated more remotely, show degenerative changes and maternal blood in their distended cavities. The endothelially lined maternal sinusoids in the implantation region are dilated and filled with plasma and some maternal blood cells. At a number of points, especially in the basal region, the intervillous space communicates with the sinusoids by way of channels through the trophoblastic shell (Pl. 6, fig. 15). Some of these channels are in part lined by syncytium but many of them possess walls formed by cytotrophoblast alone. The sinusoids can be traced away from the implantation site to become continuous directly with endometrial veins. As it is only the superficial part of the endometrium that is available in this specimen few details can be provided for the coiled arteries. Several of them, however, could be traced through the endometrium adjacent to the trophoblastic shell and into the latter where they terminate blindly. None was found to enter directly into the intervillous space; indeed, none could be traced into the spaces in the cytotrophoblast. In the walls of several arteries in the decidua compacta, which were separated by several hundreds of microns from the trophoblastic shell, there were signs of degeneration. A number of cells of syncytial origin were found in close relation to these endometrial arteries.
Embryo H.R. 1
In embryo H.R. 1 (Johnston, 1940, 1941) and in Prof. Keller’s curettage specimen conditions very similar to those described above for the Missen implantation site were found. In both of these specimens, however, the mesodermal cores of the villi are less well developed and the syncytium has perhaps undergone less retrogression (PI. 6, fig. 16). In neither of them could coiled arteries be traced into the intervillous space.
The Gar embryo
This embryo with its implantation site has been described in considerable detail by West (1952). Here we restrict observations on it to points directly relevant to placental development.
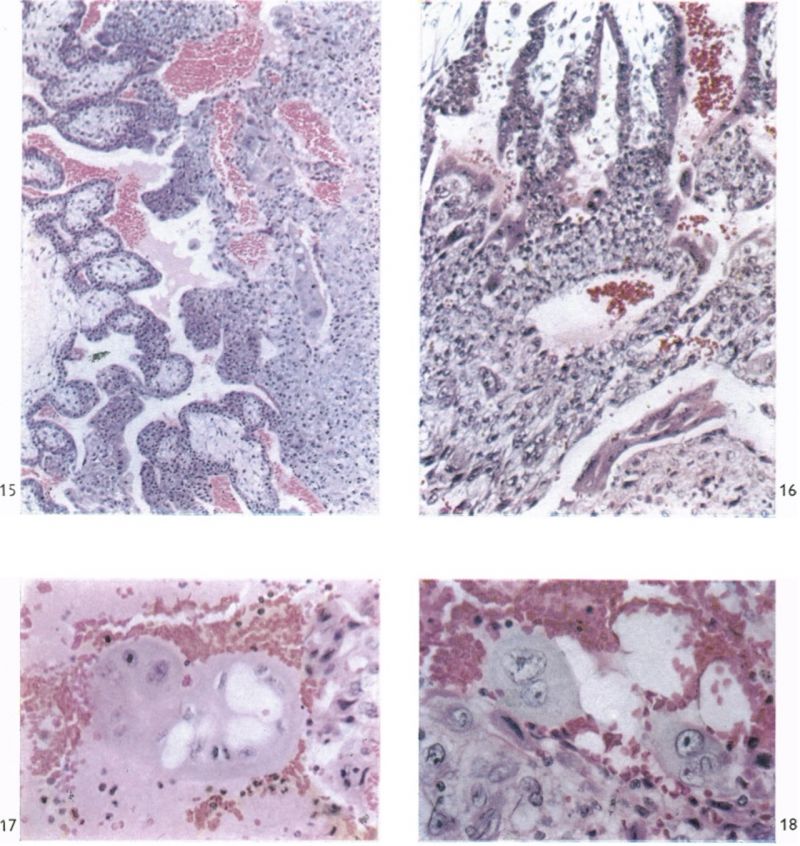
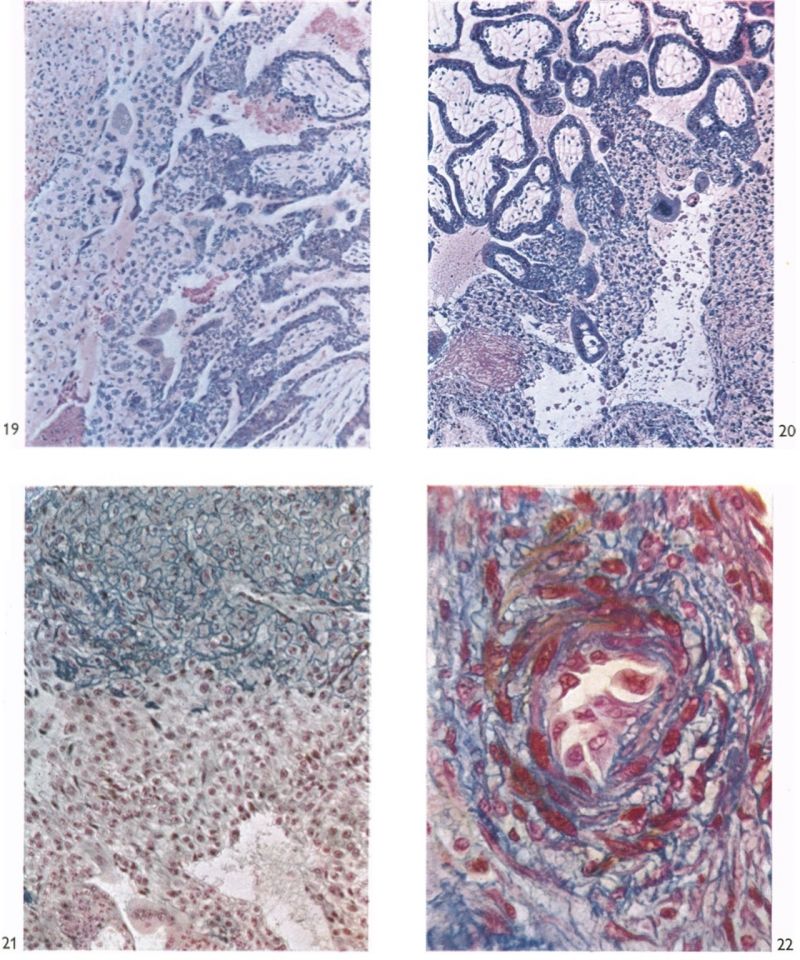
The chorionic villi in this specimen are of two types. There are, first, those which have free endings in the intervillous space and which take their origin from the chorionic plate or from other villi near to their chorionic attachment. Many of these villi are well developed and branch as often as three or four times. The second villous type includes those which extend from the chorionic plate to the trophoblastic shell with which they are continuous. Such primary villous stems are, at least in part, persistent portions of the original trabeculae of the earlier stages. Apart from the small syncytial sprouts, which are attached to each type, the villi possess a welldeveloped core of mesoderm. They are covered by syncytial trophoblast deep to which a layer of cytotrophoblastic cells (Langhan’s cells) can be identified. Early embryonic blood vessels are now present in the mesenchymal cores. At the periphery of the primary villous stems, the cytotrophoblast has broken through the syncytial layer and, by lateral expansion and fusion, constitutes a thick trophoblastic shell (Pl. 7, fig. 19). In the shell there are many labyrinthine irregular spaces, the walls of which are formed for the most part of cytotrophoblast alone. In these spaces there are many maternal blood corpuscles and such elements can also be found lying between the loosely arranged cytotrophoblastic cells themselves. The peripheral cells of the trophoblastic shell are intimately intermingled with the decidua and it is impossible to define a distinct foetal-maternal junction. Syncytial streamers from the trophoblast are occasionally found amongst the decidual cells. The syncytium frequently shows active phagocytosis (Pl. 6, fig. 17). The decidua basalis in this specimen is oedematous and, adjacent to the implantation site, blood corpuscles in large numbers are found amongst the endometrial cells. Coiled arteries can be seen in the deeper parts of the endometrium. As they approach the implantation site the walls of these arteries show necrotic changes; none was, in fact, found to open directly into the cytotrophoblastic lacunar spaces, nor into the intervillous space. The arteries adjacent to the implantation site have apparently been pushed laterally by the expanding chorion; some of them can, however, be traced to the decidua capsularis.
Large venous sinusoids lie adjacent to the implantation site; they anastomose freely with each other and receive a number of communications from the lacunar labyrinth by way of clefts in the trophoblastic shell. The most conspicuous communications between the intervillous space and the maternal veins in the Gar specimen are situated round the equatorial region of the trophoblastic shell. The uterine glands in the region of the implantation site are frequently invaded by the trophoblast with resultant destruction of theglandular epithelium. Indeed, in some regions syncytium is found within the lumina of the glands (PI. 6, fig. 18). In many places the glands are filled with maternal blood.
The Shaw embryo
This specimen has already been described in some detail (Hamilton & Gladstone, 1942). Part of the decidua capsularis had been damaged previous to fixation and there is some distortion in the region of the decidua basalis. Notwithstanding these deficiencies, the specimen shows certain features relevant to understanding the development of the villi and the trophoblast. On the deep aspect of the chorionic sac, the wall of the implantation cavity is formed by an incomplete trophoblastic shell in which spaces containing small masses of syncytium are present (see pl. 3, fig. 6, in Hamilton & Gladstone, 1942). On its embryonic side, the shell is continuous with the cytotrophoblastic cell columns proliferated from the tips of anchoring villi, some of which are presumably villous stems. Freely ending villi, similar to those found in the Gar embryo, are also present. Syncytiotrophoblast of varying thickness covers the cell columns and the intervillous aspect of the trophoblastic shell. The mesenchymal cores of the villi are in the form of a loose reticulum in which there are some, for the most part empty, endothelially lined vascular spaces. These spaces, which are the precursors of the foetal villous vessels, are best developed in the region of attachment of the connecting stalk to the chorionic plate.
A wide space, the basal sinus (which may, in this specimen, be partly artefact), separates the decidua basalis from the trophoblastic shell and communicates very freely with the intervillous space. Owing to the small amount of maternal tissue removed with the conceptus, it has not been possible to trace the endometrial arteries or to determine with accuracy their mode of termination.
Somite Stages (20-30 days)
Embryo H. 712
(7 somites, c. 23 days). Only a portion of the implantation site of this embryo is available. The trophoblastic shell is thinner than in the earlier specimens but in parts it can be seen to possess spaces. Villi are well developed over the whole of the chorionic sac. In those villi which reach and fuse with the cy totrophoblast the average length is about 1-5 mm. The villi are more branched than heretofore and consequently the intervillous space possesses a more complicated structure. The glands in the decidua show signs of secretory activity. The available portions of the specimen, unfortunately, do not permit of comment on either the spiral arteries or the uterine veins.
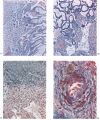
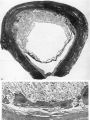
Embryo H. 710
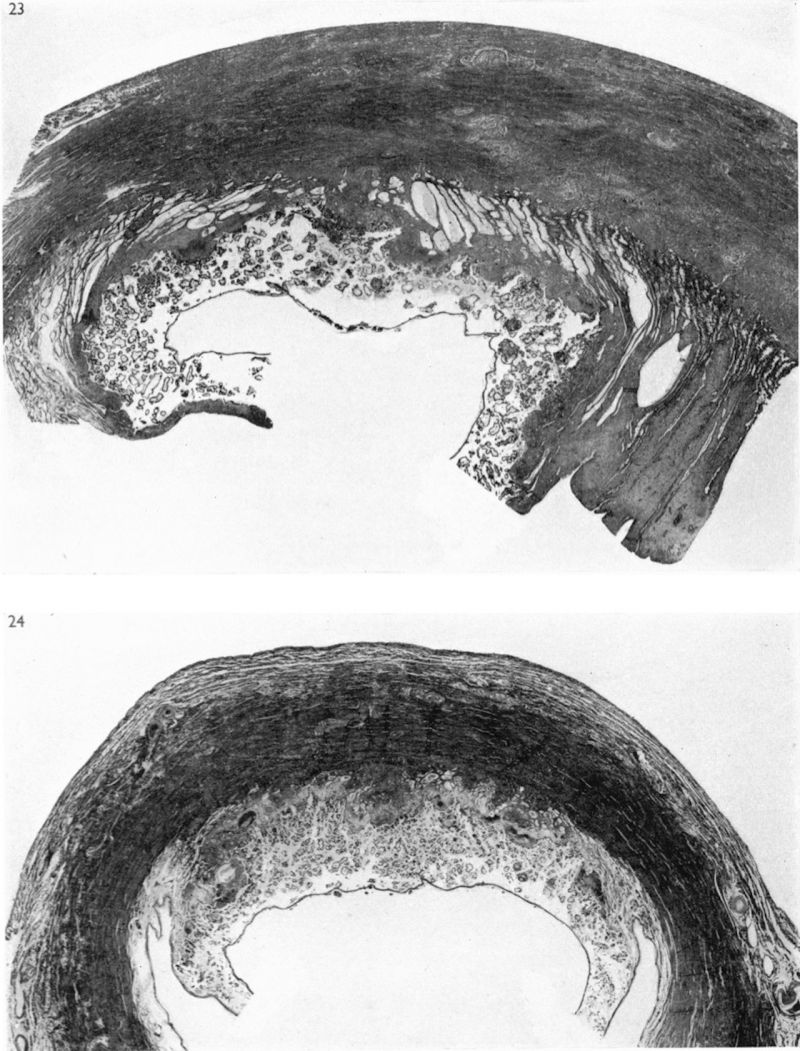
(27 somites, c. 26 days). The implantation site in this specimen is shown in Pl. 2, fig. 8, and in Pl. 5, fig. 14. The embryo was removed by way of a cruciate incision through the decidua capsularis. On the deep aspect of the chorion the main villi have an average length of about 1:75 mm. They are also present, although less well developed, in the region of the decidua capsularis. All of the villi are covered by a continuous band of syncytium deep to which a fully established Langhan’s layer of cytotrophoblast is present (PI. 7, fig. 20). Well-established blood vessels are found in many of the villous mesodermal cores. The intervillous space contains some maternal blood. Peripherally many of the villi are attached to the attenuated trophoblastic shell by columns of cytotrophoblast. There are also islands of cytotrophoblast scattered, in an apparently random fashion, throughout the villous tree. The trophoblastic shell shows many spaces (Pl. 7, fig. 20). Some of these communicate with each other and others can be seen to open into the adjacent intervillous space. The spaces often contain cellular debris, glandular secretion and, occasionally, maternal blood cells. Many of these spaces also contain masses of syncytium which are often markedly vacuolated. The cytotrophoblastic cells of the shell are loosely arranged; debris and maternal corpuscles can frequently be seen between these cells.
The peripheral margin of the trophoblastic shell is intimately related to the decidua compacta and it is difficult to establish the precise line of junction between the two tissues. With suitable staining (e.g. Masson’s trichrome technique or reticulin impregnation), however, it is always possible to identify the junction between maternal and foetal cells. The decidual cells are surrounded by collagenous and reticular fibrils; the cytotrophoblastic cells, however, have no fibrillar material between them (PI. 7, fig. 21). Apart from some stray strands of decidual collagen which are apparently being digested by the cytotrophoblastic cells there are no intercellular fibrils in the trophoblastic shell. In the decidua basalis there are many | dilated glands, often with large amounts of maternal blood in their lumina. Those in the marginal zone of the implantation site are distorted as they pass to their openings situated on the decidua capsularis (PI. 5, fig. 14). The glands situated directly below the implantation, in the decidua basalis, have had their connexions with the uterine lumen interrupted. They terminate blindly in the junctional zone and many of them have their cavities invaded by cytotrophoblast. The secretion of such glands can in some instances be traced into the interstices between the cytotrophoblastic cells. Frequently, however, the appearance is presented of the damming back of secretion owing to the pressure exerted by the expanding conceptus. In those glands which flank the lateral margins of the implantation site there are frequently marked signs of degeneration. That part of a gland wall closer to the trophoblast shows cellular destruction while the opposite wall may possess a lining of intact glandular epithelial cells. (Illustrations of the relationship of the glands to the trophoblast can be found in Boyd, 1959.)
Many coiled arteries are found in the decidua adjacent to the implantation site. These vessels can be traced through the junctional region to the peripheral cytotrophoblast where most of them terminate, apparently blindly. Some of the arteries, however, have open endings which give the appearance of having permitted the escape of maternal blood into the interstices between the cells of the trophoblastic shell. None of the arteries, in spite of most careful search, could be found to discharge directly into the intervillous space itself. The walls of the terminal arteries show signs of degeneration with disappearance of their muscle cells and marked swelling of their endothelium (PI. 7, fig. 22). Such striking changes in the endothelium can be found in loops of the coiled arteries quite remote from the junctional zone.
Openings from the intervillous space to endometrial veins are present over the basal surface of the implantation site; several large communications drain its equatorial region.
Specimens of in situ placentae with embryos of the 10 mm Stage (87-38 days)
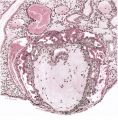
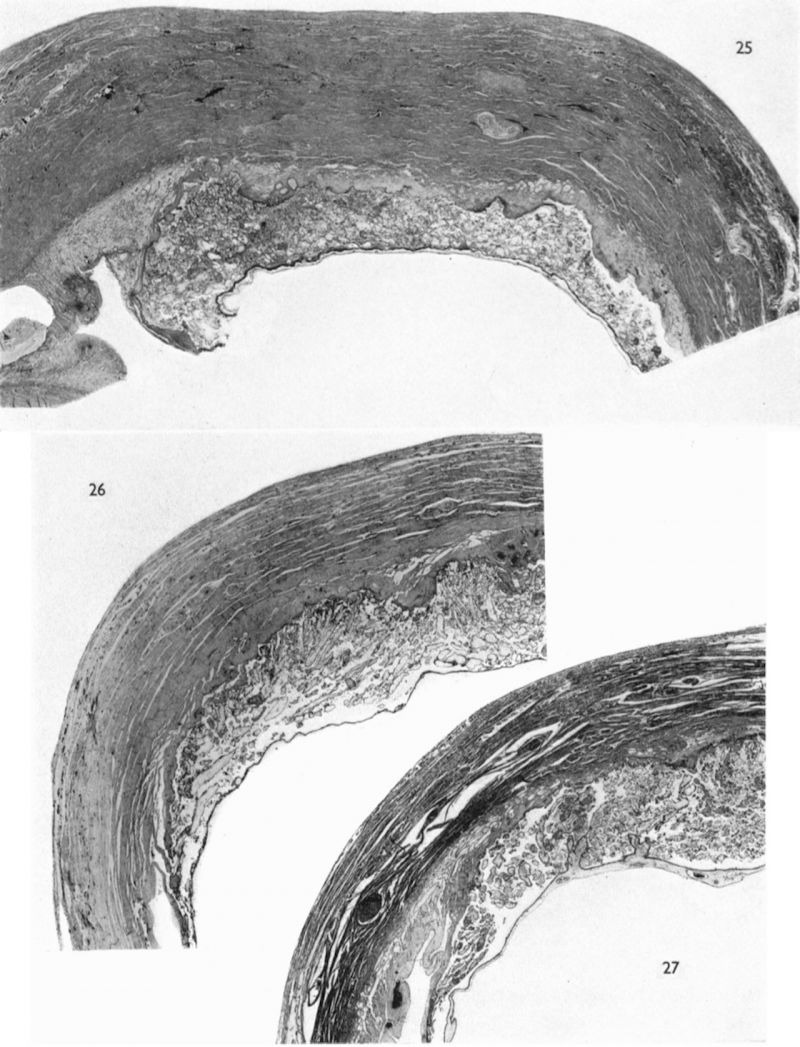
Implantation sites of four embryos at this stage of development are available (Table 1). The general features of the utero-placental junction and of the chorionic villi are very similar in all four specimens. A photomicrograph (Pl. 8, fig. 23) of a section through one of them shows that, in comparison with earlier stages, the decidua basalis is now distinctly reduced. The decidua parietalis, however, is if anything even thicker than in younger specimens. The trophoblastic shell varies in thickness. Over extensive areas of the basal plate it has become attenuated; deep to such areas there is a fibrinoid deposit in the most superficial part of the decidua. In some regions this fibrinoid material shows a lymphocytic invasion. The shell still remains thick, however, where the well-developed cellular columns at the tips of the main villous stems are attached. No clear distinction can, in fact, be made, in such regions, between the cytotrophoblastic cells of the shell and those of the villous. extremities. Between such cytotrophoblastic cells there are many of the spaces to which attention has already been drawn. Some of these spaces are in communication with the intervillous space, but most of them are isolated small cavities. Villi, both attaching and free, are distributed over the entire surface of the chorion. Opposite to the decidua basalis they have a slightly greater average length and are more luxuriant and better vascularized than those in relation to the capsularis. The cytoplasm of the trophoblastic components of the basal villi is generally more basophilic than that of the equivalent cells in the capsular villi.
Aggregations of cytotrophoblastic cells, in the form of the so-called cytotrophoblastic islands, are present in relation to the villi. Such islands are not restricted to, but are much larger on, the basal side. These islands will become increasingly apparent in later stages. Their superficial cells are in direct contact with the contents of the intervillous space.
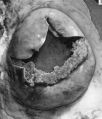
The glands in the decidua basalis are very much distorted and the long axes of most of them are now orientated at an angle to the myometrium. They still show signs of secretory activity, however, and their lumina are frequently distended. Evidence of glandular breakdown in the neighbourhood of the implantation site is still apparent. There are many coiled arteries in the decidua and those in the basal part can be traced into the junctional zone. The walls of the dilated terminal parts. of these vessels show a very marked fibrinoid degeneration with complete disappearance of the histological features of an artery. Some of these terminal arterial segments extend up to the trophoblastic shell and here the continuity of the vessel wall is often interrupted. Blood from these arteries and secretion and debris from the glands can be traced through gaps in the peripheral part of the shell to the cytotrophoblastic lacunae and, thence, to the intervillous space. Communications from the intervillous space to the endometrial veins are evident over the whole of the basal plate but, in one of the specimens, there is a preferential drainage from the zone of transition between the decidua basalis and decidua capsularis (PI. 8, fig. 23). When the uterus of this 10 mm. specimen was opened, it was seen that the decidua capsularis projected across the uterine lumen and made contact with the decidua parietalis of the opposing wall. Here there was no fusion of the two deciduae but, as subsequent section showed, the epithelium of the decidua parietalis in the area of contact had become markedly thinned and the underlying endometrial capillaries and veins were markedly dilated.
Specimen of an in situ placenta with an embryo at the 15 mm Stage (41-43 days)
A section through the central part of the implantation site of this specimen is shown in PI. 8, fig. 24. The diameter of the actual attachment area measures 26 mm. The thickness of the placenta proper varies between 3 and 6 mm. The combined thickness of the chorion and decidua capsularis is about 2 mm. and there are welldeveloped capsular villi. By this stage there is a striking reduction in the thickness of the decidua basalis but its glands continue to show secretory activity. Many of them open directly into the intervillous space and it is noteworthy that these are, for the most part, empty. Other glands, however, which do not communicate with the space, are distended with secretion; their cells are flattened and a number of them contain maternal blood. Except at the site of attachment of the cytotrophoblastic columns at the villous tips the thickness of the trophoblastic shell has become much reduced, often to a single layer of cells. On the decidual side of the shell there is frequently a fibrinoid deposit which is scattered in an irregular fashion over the surface of the basal plate.
Numerous arteries ensheathed by decidua can be traced from the myometrium towards the intervillous space. The terminal portions of these vessels show the hyaline degenerative changes described for earlier specimens and all of them are being invaded by cytotrophoblast. There is also a marked thickening of their endothelial linings in the deeper part of the decidua. The actual terminations of the arteries are in communication with spaces in the thickened parts of the trophoblastic shell. No direct arterial communications with the intervillous space itself have been observed. ‘It would appear, therefore, that, at this stage, blood from these arteries could only have reached the intervillous space by percolation through the interstices situated between the cytotrophoblastic cells (Pl. 12, fig. 31). A striking feature of this specimen is the paucity of maternal veins underlying the central area of the implantation site. Consequently the main drainage of the intervillous space is through communications which open into veins in the region of junction of the decidua capsularis with the decidua parietalis. The villi themselves are clothed by both syncytium and cytotrophoblast; their mesodermal cores contain well differentiated. foetal blood vessels.
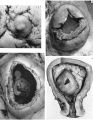
Specimens of in situ placentae with embryos of between 20 and 30 mm Stages (48-60 days)
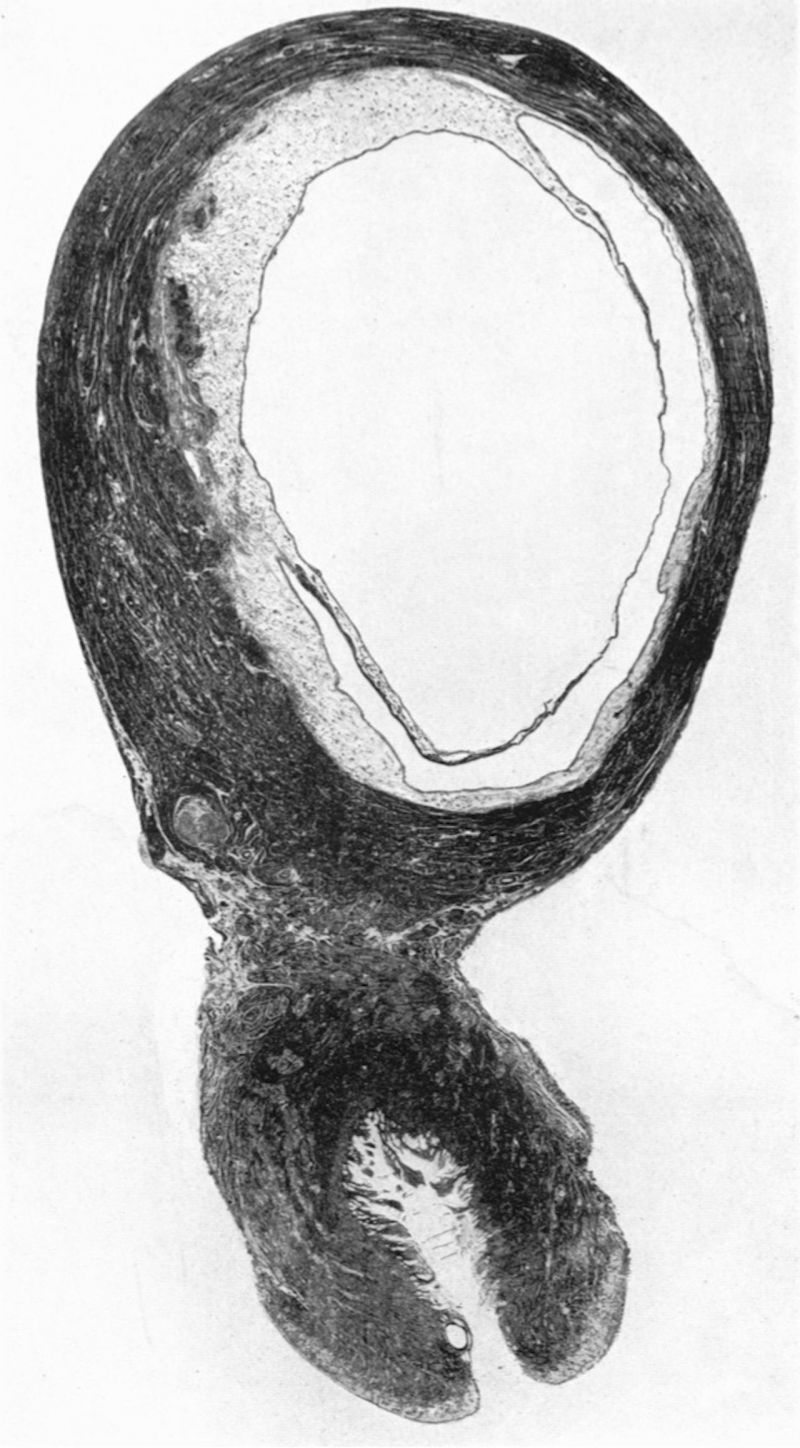
Sections through the whole uterus showing the-developing placenta of the 29 mm. stage and of the 80 mm. stage are shown in Pl. 9, fig. 25, and Pl. 10, fig. 28, respectively. The placentae of these two specimens have diameters of approximately 40 and 47 mm. respectively. It will be seen from these illustrations that there are villi on the part of the chorion related to the decidua capsularis. The latter is now projecting deeply into the uterine lumen but has not yet fused with the decidua parietalis. Indeed, as will be seen from the account of later stages, such fusion does not occur until after the period of development with which we are concerned in this contribution. The trophoblastic shell is now very thin except in the regions of attachment of the terminal cytotrophoblastic cell columns of the anchoring villi. Extensions of syncytium into the decidua basalis are a striking feature of these stages. Such extensions frequently reach the myometrium and isolated syncytial masses can be found in its inner part. Other irregularly shaped plates of syncytium, often highly vacuolated, are present along the basal plate, particularly in the regions near the attachment of the villi.
The villi possess a structure similar to that of earlier stages. They are, however, more branched with resulting increase in the complexity of the intervillous space. The connective tissue in the villi is further differentiated and numerous Hofbauer cells are present.
As in previous stages, the walls of the terminal parts of the spiral arteries show fibrinoid and hyaline degenerative changes. Thickening of the endothelium is still present and in the portions of the vessels nearest to the intervillous space plugs of cytotrophoblast can be identified. Small irregular clefts can be followed from the arterial lumina through the interstices situated between the cytotrophoblastic cells. In some instances, indeed, as, for example, in a 22 mm. specimen, such clefts are represented by narrow, but straight, channels (Pl. 12, fig. 82) passing directly from the vessels to the intervillous space. Venous drainage from the intervillous space now occurs from point to point over the whole of the basal plate. By the end of this period of development centrally placed communications between the intervillous space and the decidual veins are numerous and often very large (PI. 11, fig. 30).
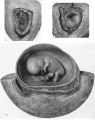
Specimens of in situ placentae with embryos of between the 31 and 60 mm Stages (60-90 days)
We have sixteen specimens of in situ placentae which, on maternal history and foetal size, can be considered to belong to the third month of gestation (PI. 9, figs, 26, 27; Pl. 11, fig. 29). The shape of these placentae is variable but the average transverse diameters range from 50 to 70 mm. There is also considerable variation in placental thickness from specimen to specimen and even in different regions in agiven placenta. Although the chorionic sac becomes much larger during this period the increasingly thinned and stretched decidua capsularis has not, in any of the specimens, yet fused with the decidua parietalis. The villi related to the decidua capsularis, however, show marked signs of retrogression. This retrogression, in the earlier specimens, is most apparent at the pole opposite to the placenta. As older specimens are examined the disappearance of villi is seen gradually to spread from this pole in an equatorial direction so that by the end of the third month nearly the whole of the decidua capsularis is related to a part of the chorion in which the villi are represented only by little stumps. Accompanying the villous retrogression there is a disappearance of the associated intervillous space. The villi in the placenta itself become much more numerous in this period. The main villous stems become thicker and there are more numerous side branches. In the initial part of the third month the cytotrophoblast on most of the villous surfaces constitutes a continuous Langhan’s layer. By the middle of the period, however, areas in which this layer cannot be identified by light microscopy have become apparent and in the oldest specimens it has disappeared in most of the villi.
The trophoblastic shell is very attenuated in all the specimens except in the area of villous attachments. In certain regions the shell may be reduced to a single layer of cells. Throughout the whole period fibrinoid material is distributed irregularly over the whole of the basal plate. Attached to the apical portions of the villi there are many syncytial sprouts. Some of these sprouts are also distributed from point to point throughout the villous tree. In some of the older specimens in this period there are projections from the basal plate into the intervillous space. These projections are the forerunners of the cotyledonary septa which become such important features in the placenta of the second half of pregnancy.
The decidua basalis is slowly but continuously reduced in thickness throughout the third month of gestation. Nevertheless, glandular activity can be detected in all of the specimens. A striking feature is marked development of the uterine glands in the decidua parietalis at the angle of junction of the decidua basalis with the decidua capsularis. Occasional haemorrhage into the basal glands can be detected in all of the specimens. Erosion of some of these glands by cytotrophoblast is also shown in most of them. The decidua basalis in every specimen contains many masses of multinucleated cytoplasm which, on the basis of orientation, distribution and cytology, we (1960, in press) consider to be of trophoblastic origin. Such syncytial masses are often arranged radially as they penetrate through the decidua to reach the inner part of the myometrium where they come to lie between the muscle cells.
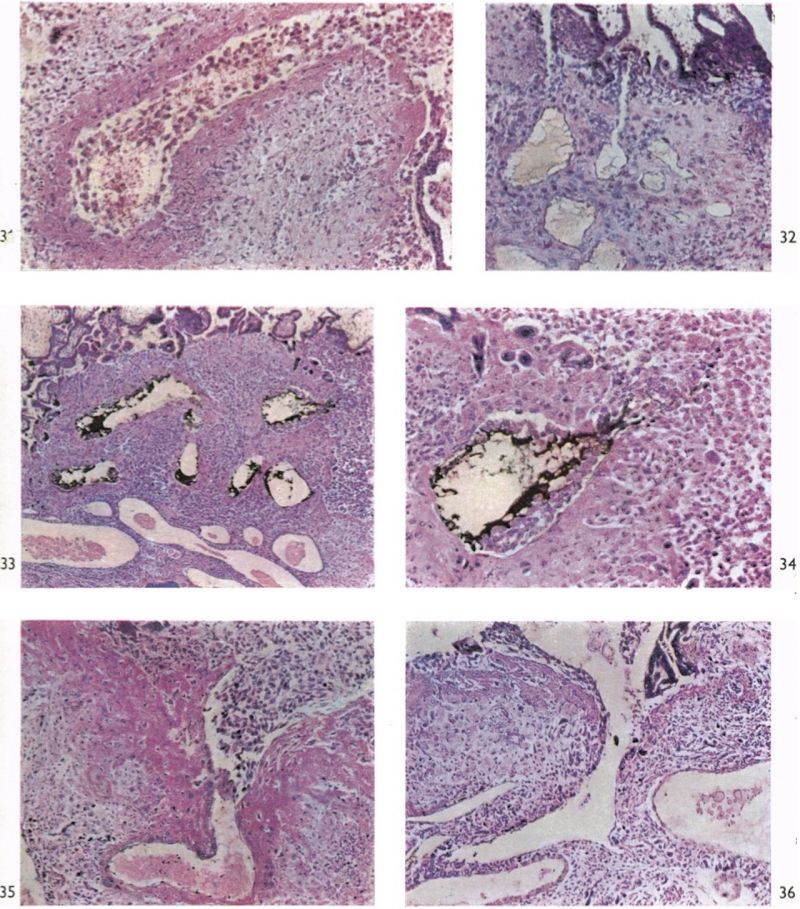
Spiral arteries can be traced through the decidua to the cytotrophoblast. These arteries show the degenerative changes in their walls to which attention has been directed in the descriptions of the earlier stages. In all the specimens these arteries terminate in a mass of cytotrophoblast and cells of foetal origin can be traced up most of the vessels. In no specimen have we found spiral arteries to open freely into the intervillous space. There are always interposed plugs of cytotrophoblast. Such plugs become more loosely arranged as development proceeds and in the oldest specimens in this period (Pl. 12, fig. 35) they give the appearance of being much less capable of acting as an impediment to blood entering the intervillous space. Sections were available of a specimen into which indian ink had been injected into a uterine artery in the broad ligament before the uterus was opened. Such sections (Pl. 12, figs. 88, 84) show ink in the lumina of the spiral arteries from whence it can be traced through intercellular cytotrophoblastic spaces into the intervillous space. In all of the specimens of the third month uterine veins are found to communicate freely with the intervillous space over the whole of the basal plate (Pl. 12, fig. 36).
Discussion
Endometrium and Decidua
At the time of implantation of the blastocyst the human endometrium shows a distinct division into a superficial stratum compactum and a deep, oedematous, stratum spongiosum. It measures about 4-5 mm. (see Krafka, 1941, for details) in thickness and is, of course, under the influence of the luteal hormone secreted by the ovary. After implantation, the endometrial oedema becomes even more marked, particularly in the region underlying, and round, the blastocyst. The oedema, which is presumably to be attributed to alterations in the walls of the blood vessels or in the flow through them, results in an eosinophilic exudate which separates the endometrial cells from each other. The blood vessels near the implantation site are hyperaemic and sinusoidal vascular spaces become apparent; such spaces may be dilated venules or capillaries. Initially, there seems to be little effect on the spiral (coiling) arteries themselves, but the pre-decidual reaction is more intense round them. Wislocki & Streeter (1938), in their studies on implantation in the macaque, have described endometrial changes similar to those just indicated for the human endometrium; in the monkey, however, in the region of attachment of the blastocyst, there is a marked proliferation of the epithelium lining the uterine lumen and in the necks of the glands. This epithelial proliferation is presumably to be associated with the central method of implantation in the macaque.
In our 12-day human specimen the endometrial oedema is well marked. In view of the findings by Hertig e¢ al. (1956) on a number of early specimens, this oedema must be regarded as a normal feature of the endometrium, though Davies (1944) mistakenly considered it to be pathological. Further, in well-fixed specimens such as the Barnes ovum, there is no infiltration of maternal small round cells into the endometrium. Such infiltration is marked in a number of specimens (e.g. Bryce & Teacher, 1908; Davies, 1944) which can, in our opinion, be definitely considered abnormal. Indeed, the absence of lymphocytes from the region surrounding the implantation sites is a striking feature of normal early specimens for it is quite unlike what would be expected if the trophoblast behaved immunogenically as a homograft (Boyd, 1959).
The stromal endometrial cells in the 12-day specimen cannot be regarded as more than pre-decidual in their degree of differentiation. By the 14th day, however, the characteristic and definitive decidual cells have become apparent; these cells persist throughout the stages of development dealt with in this contribution. The decidual cells can always be readily distinguished (PI. 7, fig. 21) from the cytotrophoblastic cells by the fact that the former are surrounded by collagen and reticulin fibrils.
Uterine Glands
In the luteal phase of the uterine cycle the endometrial glands become enormously enlarged. Attachment of an implanting conceptus normally occurs between openings of such glands on the uterine surface (Pl. 2, fig. 2). As the blastocyst becomes interstitially implanted the glands adjacent to it are compressed by the pressure of its circumferential growth. Consequently there is a damming back of contained secretions in the glands concerned and they become much dilated while their necks become stretched, compressed and, finally, occluded. Meanwhile, the actively proliferating trophoblast erodes the endometrial stroma and glandular epithelium, eventually breaking through into the glandular lumen (PI. 6, fig. 18; and see Boyd, 1959, pls. 1, 2). In this manner the contents of the glands become directly available to the trophoblast (PI. 4, figs. 9,11). It is the glands in that part of the endometrium which will become the decidua basalis which are involved in the implantation process and which become dilated. The necks of some of them are also drawn into the lower (i.e. attached) part of the decidua capsularis but the glands in the decidua parietalis persist until a remarkably late stage (PI. 8, fig. 23) inarelatively undistorted condition. As development proceeds, however, glands initially remote from the implantation site are gradually encroached on by the trophoblast. Thus, in the 12-day specimen only a few glands are affected, there are considerably more involved in the 17- to 18-day specimens, and by the 28th day (PI. 5, fig. 14) a very great many, indeed, are included in the process. Gradually the glands in the basal decidua are, for the most part, destroyed. Owing to the nature of placental expansion, however, the glands at the margin of the placenta are much less affected than those in the central region; consequently, well-preserved glands can be found in the marginal zone of the decidua basalis throughout the period with which we are at present concerned. Glandular secretion is apparent in many of the persisting basal glands until comparatively late stages (Hamilton & Boyd, 1951).
Possibly as a result of the glandular penetration by the destructive action of the trophoblast, maternal blood cells are early apparent in many of the glands. Such extravasated blood is already present in several glands in the 12-day specimen (Pl. 1, fig. 1) and is a striking feature in the central part of the decidua basalis of the 28-day specimen (Pl. 5, fig. 14). From the accounts in the literature on early implantations it would seem that such haemorrhage into the glands does not occur earlier than the 12th day. Even in later stages it may be absent (for discussion, see Krafka, 1941). Blood in the glands, however, has been described in so many older specimens that its presence must be considered a normal feature of human implantation from 12 days onwards. Haemorrhage into the uterine glands has also been described in the neighbourhood of the implantation site in the macaque by Wislocki and Streeter (1938); they found that the blood was not present in the glands until the 17th post-ovulational day and that it persisted until the end of the first month of conception. Haemorrhage into glands which are not yet occluded by the pressure of the expanding trophoblast is probably the explanation for the slight external haemorrhage, in both women and the macaque monkey, now well known to occur at, or about, the time of the first ‘missed’ menstruation. This blood loss constitutes the so-called ‘placental sign’ to which attention was first directed by Hartman (1928, 1932).
Trophoblastic Differentiation
Reference has been made in the introduction to certain difficulties in the terminology used to describe the different varieties of trophoblast. In particular, we have avoided terms such as ‘implantation trophoblast’ and ‘resorptive syncytium’ that attribute specific functions to parts of the trophoblast at successive periods of development. There can, of course, be no doubt that there is a functional specialization on the part of the different regions of the trophoblast; until more is known about the details of trophoblastic function, however, terms implying a particular activity by a given region should be avoided. Moreover, as Wislocki & Streeter (1938) have stated, such an adjective as ‘resorptive’ can apply ‘equally well to the entire development of the trophoblast and should not be restricted to any particular stage’.
In the 74-day human ovum described by Hertig & Rock (1945) the blastocystic trophoblast has already differentiated at the implantation pole into a thick plaque of proliferating, and intermingled, syncytium and cytotrophoblast. The remaining part of the blastocyst wall is constituted by a thin ‘mesothelial’ membrane which consists of a single layer of flattened blastocystic cells. In the junctional region between the latter and the plaque itself cells intermediate in character between the two are found. In specimens several days older (Hertig & Rock, 1945, 1949), when implantation is nearly achieved, differentiation into primitive cytotrophoblast and primitive syncytiotrophoblast is much clearer. Both of these are alike derived from the blastocystic trophoblast but the syncytium arises indirectly in the sense that it stems from cells which are uninucleate and which, consequently, cannot initially be distinguished from other cytotrophoblastic cells. How the syncytium actually takes its origin is an important question to which, so far, there is no compelling answer. Hither certain cytotrophoblastic cells undergo nuclear division—mitotic or otherwise—without cellular division, or adjacent cytotrophoblastic cells unite and, by their fusion, constitute a multinucleate mass of cytoplasm. We have given much attention, throughout the study of all of our material, to the method of syncytial formation in the trophoblast and have been unable to decide between the two possibilities. The syncytium presents a histological structure quite strikingly different from the cytotrophoblast. Hence, if elements of the latter are included by fusion in the former the cytotrophoblastic cytoplasm must rapidly change in character during the assimilation. On the other hand, if there is nuclear multiplication in the syncytium, the absence of signs of mitosis in the multinucleate cytoplasm suggests the not very satisfactory explanation of amitotic division. Whatever factors do, in fact, cause the appearance of the syncytium, contact with the endometrial tissues does not seem to be an absolutely determining one, for primitive cytotrophoblast is found in direct apposition to the maternal cells. Once it has appeared, however, the primitive syncytium seems to be mainly responsible for the local destruction of the maternal tissues.
Soon after its first appearance the syncytium of the trophoblast shows marked vacuolation. The vacuoles become confluent and by the 12th day a well-established system of lacunar spaces is present throughout the syncytium. Moreover, some of the dilated maternal sinusoids are now in open communication with the lacunae and maternal blood is present in them. The method whereby the sinusoids are tapped is not apparent. It does not appear to be a disordered destruction of maternal tissue that is responsible; the endothelium of the maternal vessels, at the points of communication, is in neat apposition to the peripheral syncytium (PI. 4, figs. 9, 12), and there is no sign of maternal haemorrhage into the decidua surrounding this part of the trophoblast. The maternal blood cells in the lacunar spaces in the 12-day specimen are very palely stained. The difference in the degree of eosinophilia between them and the maternal erythrocytes in blood vessels away from the implantation site is quite striking. It is, therefore, possible that the cells in the lacunar spaces in this stage, due to the stagnant nature of the circulation or to the effect of syncytial secretion on them, are in a special physiological state.
In the 12-day specimen the primitive syncytium covers the cytotrophoblast and the latter is present only in some of the trabecular columns which incompletely separate the lacunae from each other (PI. 1, fig. 1). There is no sign, at this stage, of villus formation by the extension of separate syncytial ‘streamers’ into a maternal blood space. The traditional picture of ‘primary villi’ is one that must be dismissed.
It was based on conclusions arrived at from the study of badly fixed material and on an absence of understanding of the method of formation of the lacunar spaces and of the relationship of the trophoblast to them. Already at the 12th day the human placental arrangement, to use Grosser’s terminology, is haemo-chorial, but at this stage it quite obviously possesses not a villous structure but a labyrinthine one.
In the period from the 14th to the 20th day there is intense proliferative activity in the trophoblast; this activity results in the appearance of the definitive structure of the placenta and in the full establishment of the circulation in the rearranged lacunar spaces which now, collectively, constitute the intervillous space. The first result of the growth processes is that the trophoblastic trabeculae, instead of being arranged quite irregularly, tend, more and more, to become orientated radially round the chorion and, therefore, to take on a villous appearance. Soon these trabeculae, which initially are syncytial in character, come to possess a cellular core by active multiplication of the trophoblastic cells on their deep, or chorionic, surface. It is at this stage that the now highly modified trabeculae have been called ‘primary villi’. It must again be stressed, however, that the primary villous stems, as we have called them earlier, do not arise as individual and separate sprouts from the chorion. With continuing development the primary villous stems increase in length and, concomitantly, their cytotrophoblastic cores extend distally to the region of attachment of the syncytium to the endometrium. Coincidental with the extension of the eytotrophoblast the stems, at their chorionic ends, develop mesodermal cores which follow the cytotrophoblast in its extension towards the decidua. In part, this mesoderm in the villous stems is the result of proliferative activity in the general extraembryonic somatopleuric mesoderm of the chorion. The mesodermal cores, however, seem also to receive contributions of cells derived from the cytotrophoblast in the manner first described by Hertig (1935). Whatever the actual origin of the mesoderm in the villous stems, once it has appeared the stage of the ‘secondary villi’ of earlier authors has been established. The newly formed mesodermal cells of the cores of the villi do not come into direct contact with the syncytium on their surfaces. Proximally (i.e. at their chorionic ends) the villous stems preserve a single layer of cytotrophoblastic cells which, as Langhan’s layer, separates the mesodermal core from the overlying definitive syncytium. Distally the cytotrophoblastic cells, as the cytotrophoblastic columns, form the full thickness of each villous stem. On their free surfaces the columns are covered by definitive syncytium which separates them from the enlarging intervillous space. At their decidual ends, however, active proliferation of the cytotrophoblastic columns results in an expansion of the cytotrophoblast beyond the distal ends of the villous stems where their cells extend into the primitive syncytium. In this position they mushroom outwards from the maternal end of each villous stem with the result that, eventually, they meet equivalent cells from adjacent primary villous stems to form an irregularly complete cytotrophoblastic layer, known as the trophoblastic shell, round the whole conceptus. This shell was first described and named by Siegenbeek van Heukelom (1898) and his description was amplified by Peters (1899). The term is a useful one for it ascribes to the expanded and fused tips of the primary villous stems precisely that feature which is most characteristic of the resulting cytotrophoblastic layer—that it forms a shell for the developing conceptus and placenta.
Any decidual cells included within the trophoblastic shell are soon destroyed, for our own observations confirm those of Jones & Brewer (1935) and Brewer (1937) that no maternal cells persist within it. Immediately on the decidual side of the trophoblastic shell there is the special transformation of the endometrial stroma to which Merrtens (1894) first directed attention, and which has variously been known as the ‘boundary zone’, the Durchdringungszone, Umlagerungszone and the ‘ trophosphere’. It is the compact decidua, immediately surrounding the trophoblastic shell, which presents a different histological picture from the more remotely situated endometrium. In it are the terminations of the maternal blood vessels and through it the blood must pass to reach the intervillous space. In the region of junction between the boundary zone and the shell there appears the fibrinoid deposit which is called Nitabuch’s membrane and which is shown in our 28-somite stage and in all of the older specimens (Pl. 12, figs. 383-36). The boundary zone is traversed by the maternal blood vessels on their way to and from the intervillous space and, in the younger stages, uterine glands pass through it to open into spaces in the trophoblastic shell or even into the intervillous space itself (Frassi, 1908; Boyd, 1959).
By the method of its formation the trophoblastic shell splits, in an irregular fashion, the initial primitive syncytium into a part on the intervillous space aspect of the shell and a part on its decidual aspect. The former of these two portions of the primitive syncytium becomes the lining of that part of the intervillous space in contact with the basal plate of the placenta. The residual syncytium on the decidual side of the shell persists only in part and in an irregularly distributed fashion until late developmental stages; this persisting peripheral syncytium probably gives origin to some of the giant cells of foetal origin which become so widely distributed in the decidua basalis and the adjacent part of the myometrium (Boyd & Hamilton, 1960). The cells of the trophoblastic shell itself, however, also appear to be able to differentiate into syncytial giant cells. Indeed, after it has apparently reached a stage of full differentiation the cytotrophoblast retains the potency to produce syncytium throughout pregnancy.
The trophoblastic shell, though in general consisting of closely packed cells (Pl. 7, fig. 21), possesses many variously shaped, and occasionally quite large, intercellular spaces and gaps to which attention was first drawn by Jung (1908). Many of these spaces come to communicate with the peripheral part of the intervillous space, and some of them do so with the lumina of the uterine glands. In the spaces (Pl. 7, fig. 20) detritus, glandular secretion and, eventually, maternal blood cells are found, often accompanied by portions of syncytium. In our opinion these spaces are of considerable importance in the earlier histiotrophic phase of embryonic nutrition and they are also important in that many of the endometrial spiral arteries will communicate with the intervillous space by way of them. It must be stressed that the cells of the cytotrophoblastic shell never develop mesenchymatous derivatives equivalent to the mesodermal cores of the villous stems and the villi. Hence, when the foetal blood vessels develop in the mesenchymal cores to produce the ‘tertiary villi’, they do not penetrate the shell and do not come into contact with the maternal decidua or blood.
By the establishment of the trophoblastic shell a mechanism is apparently provided, through its interstitial growth, for a rapid circumferential extension of the whole implantation site. The surrounding decidua, including the glands, is encroached upon and, at the same time, the side of the implantation site in contact with the decidua capsularis bulges out into the uterine lumen. Generally the cytotrophoblastic shell becomes markedly thinned from the 10 mm. stage onwards, and, at the same time, the related fibrinoid material increases in amount. In the regions overlying the attachment of the villi, however, its attenuation is much less marked.
As a result of the expansion of the trophoblastic shell the area available for the intervillous space is considerably augmented. Into this enlarged area extend sprouts from the surface of the primary villous stems and also, we think, from the surface of the chorion itself. These sprouts are initially formed of definitive syncytium alone. Soon, however, they increase in size and pass through the stages of villi possessing cytotrophoblastic, mesodermal and, eventually, vascularized mesenchymal cores. Such sprouts are undoubtedly true villi and their presence justifies the ascription of a villous character to the human placenta. Nevertheless, these true villi are a secondary addition to the structure of the placenta. Some of them later become joined together at their free ends by syncytial fusion, but we doubt if this fusion involves more than the syncytium for we have never observed unequivocally the junction, described by Stieve (1941), of separate villi by the fusion of their mesodermal cores or of their constituent blood vessels. We have not, by direct observation, been able to decide if some of the villi arising from the chorionic plate manage to reach the basal plate and to fuse with it. As the placenta grows extremely rapidly in the period between the 5 mm. stage and the 60 mm. stage it seems likely that new major villous stems are continuously added. There may, however, be some longitudinal splitting of the initial primary villous stems. Certainly there is rapid growth in their length and marked branching at their basal extremities.
The earliest specimen available to us, the Barnes ovum, possesses a system of welldeveloped lacunae in the trophoblastic syncytium (PI. 1, fig. 1). Several sinusoids communicate with the lacunae (PI. 4, figs. 9, 12) in which, as has already been stated, maternal blood cells are present. Because of their size we presume that these sinusoids have arisen from endometrial venules; they are too large for a capillary origin to be likely. The sinusoids can be traced away from the implantation site for long distances to become continuous with the basal endometrial venous plexus. Several spiral endometrial arteries are present in the vicinity of the implantation site in the Barnes specimen, but no connexions between them and the lacunar spaces have been found. Hertig & Rock (1941), in their description of a human implantation of the 11th day, have described the related sinusoids as being arterio-venous in nature, considering that there is an arterio-venous anastomotic connexion between an adjacent spiral artery and the sinusoids. We have been unable to identify any such anastomoses, and the work of Bartelmez (1957) makes their presence unlikely. It seems to us, therefore, that the first maternal vessels to be ‘tapped’ by the lacunae are slightly dilated venules or capillaries in the stratum compactum of the decidua. With further extension of the invading syncytium, and more dilatation of the blood vessels, the supply of blood to the lacunae comes from venous sinusoids. That no direct arterial openings into the intervillous space are found in presomite embryos, such as the Edwards-Jones-Brewer (Brewer, 1937), the HR 1 (Johnston, 1940), the Shaw (Hamilton & Gladstone, 1942), the Torpin (Krafka, 1941) and the Gar (West, 1952), would seem to indicate that the flow of blood into the spaces is a ‘controlled seepage’, as Krafka has suggested. It can further be pointed out that, in the macaque monkey (Wislocki & Streeter, 1988), it is not until the 17th day that maternal vessels discharge their contents into the lacunae. A continuous flow of blood through the intervillous space was not considered likely until the 8rd week. These investigators, however, go further for they state: ‘We have no absolute information as to when the maternal blood actually begins to flow through the intervillous space, but it may safely be assumed as occurring gradually and as probably being completed in the main before the time that the fetal circulation is established.’
Whether the blood in the syncytial lacunae is simply ‘pooled’ or is subject to a slow ebb and flow circulation cannot be decided on the evidence. We have already recorded, however, that the maternal blood cells in the lacunae of the 12-day specimen show staining characteristics suggesting that the circulation is, in fact, very sluggish. Mossman (1956) has suggested that, as the perforation into a single sinusoid (he actually instances a capillary) elongates, two openings, an inlet and an outlet, can be achieved. With increase in the trophoblastic invasion, Mossman considers that the maternal vessels would disappear progressively back to the level of true arterioles and venules, thus permitting a ‘through’ circulation.
Whatever the explanation of the precise method whereby the spiral arteries themselves come to open into the intervillous space we have not been able to find communications of the arteries with the space until the 10 mm. stage. Even at this stage the communications are only by way of gaps in the trophoblastic shell. Doubtless capillaries have been tapped long before this stage and as their blood pressure is presumably higher than that of the veins a proper circulation through the intervillous space then becomes possible. In our 27-somite specimen, for example, the probability that such a circulation existed in life seems most likely. Indeed, Ortmann (1938) has described spiral arteries opening directly into the intervillous space in a 9-somite embryo originally described by Veit & Esch (1922). In our wellfixed 28-somite specimen, while we have found appearances very similar to some of those illustrated by Ortmann, we have been unable to convince ourselves that the spiral arteries themselves actually open into the intervillous space. But whether they do or do not so communicate already in the 28-somite specimen, the walls of the terminal portions of the spiral arteries show marked alteration and, in particular, a marked hypertrophy of their endothelial linings (Pl. 7, fig. 22). In subsequent stages the changes in the walls of the arteries become much more marked so that, for long stretches of their length, back from the implantation site, they show a striking degenerative appearance, with disappearance of their muscle cells, hyaline alterations in their walls, and dilatation of the terminal parts of their lumina. Further, from the period when the spiral arteries can actually be found to communicate with the intervillous space, there is the appearance of cells within their lumina (Boyd & Hamilton, 1956). Some of these intrusive cells are certainly of foetal origin for they possess the same histological characteristics as the cytotrophoblast and, indeed, can be traced directly from the shell into the arteries (Pl. 12, figs. 31, 85). It is possible that some of the intravascular cells in the more proximal part of the spiral arteries are of maternal origin, being derived from the hypertrophied endothelium, the muscle cells, or, even, migrating elements from the adjacent decidua. Our strong impression, however, is that most of the intravascular intrusive cells are of trophoblastic origin.
Up to the 43 mm. stage such cells are found in all of the arteries that pass through the transitional zone and they are also present in many of these vessels until much later stages (see illustrations in Boyd, 1956). The presence of these cells within the spiral arteries during human pregnancy has been recorded by a number of investigators (for summary, see Grosser, 1927). Similar intravascular cells have been reported in the pregnant uteri of other mammals (for summary, see Orsini, 1954). The discussion in the literature has chiefly related to the possible origin of these intra-arterial cells; little consideration has been given to their possible functional import. The constancy with which they are present in the human subject seems to us to justify the conclusion that they play some part in the placental mechanism, probably by cutting down very considerably the pressure in the blood which reaches the intervillous space. In several of our specimens in which we injected indian ink into the main uterine arteries, subsequent histological examination of the placentae showed that the ink passed from the spiral arteries through the restricted spaces between the intravascular cells and then by way of very narrow chinks in the cytotrophoblast before reaching the intervillous space itself (Pl. 12, figs. 33, 34). Under such conditions the pressure of the blood reaching the intervillous space must be very much reduced. The extension of the trophoblast into the arteries is obviously not due merely to the growth of this foetal tissue. The invasion of the arteries is selective for it can be affirmed quite dogmatically that, in the human placenta, the veins draining from the intervillous space never show such an invasion.
As has been stated previously, the sinusoids draining the early trophoblastic lacunae can readily be traced to the basal endometrial veins. As development continues the sinusoids increase in number and, indeed, cannot be distinguished from veins. In our earlier specimens the veins are found draining at irregular intervals from the whole of the basal surface of the developing placenta. Ortmann (1938), in his somite specimen, describes and figures the venous drainage as being exclusively from the central part of the basal plate. In our somite specimens, however, the drainage is both central and from the margin. As development proceeds the central veins are retained (Pl. 11, fig. 30) and are progressively added to in number as the placenta increases in size. In none of our placentae up to the 60 mm. stage is there an exclusive venous drainage from the marginal placental zone.
From our observations on the arterial supply to, and the venous drainage from, the intervillous space in the period with which we are at present directly concerned it seems to us justifiable to conclude that the circulation of maternal blood through the early placenta is under conditions which suggest that the pressure of the blood is low and its rate of flow slow. Moreover, as the maternal arteries and veins are connected with the intervillous space at random over the whole of the basal plate, the circulation in the space cannot be one that follows a very fixed path. There may, perhaps, be some preferential channels through the intervillous space but our attempts to establish their presence have been quite unsuccessful. This overall picture of blood entering and leaving the intervillous space at random is not very satisfying, particularly if one is looking for an optimum method for gaseous exchange between the maternal and the foetal organism. Nevertheless, we consider that it corresponds with what is to be found from an unprejudiced survey of the in situ placenta.
In an earlier communication (Hamilton & Boyd, 1950), based on a study of such human placentae, we indicated that a re-assessment of the circulation within the intervillous space seemed to us to be necessary. Until the contribution of Spanner (1935) the generally accepted view on this circulation was the one put forward by Bumm (1898) who considered that, in the mature placenta, the orifices of the endometrial arteries into the intervillous space are situated in the placental septa some distance from the basal plate and that the openings of the uterine veins draining the space are through the basal plate about the middle of each cotyledon. This view was questioned by Kermauner (1912). More recently Ramsey (1956), 1959), in a series of papers on the macaque placenta, using modern techniques and with abundant material has shown that, in fact, the arterial openings are independent of the septa. She considers that what Bumm mistook for arteries in the septa were artefacts, being the remnants of glands, lymphatic channels or localized foci of necrosis. Bumm had postulated that the blood flowed back from the arterial openings in the septa, through the intervillous space, to the venous openings in the basal plate; he considered that, as a result, each cotyledon of the placenta functioned as a separate circulatory unit.
Spanner (19385), from his studies of the placenta after injection of the maternal circulation with celluloid and later digesting away the soft tissues, reached quite a different conclusion on the intervillous space circulation. He considered that the endometrial arteries only opened through the basal plate and that all the blood was drained away from the intervillous space through a constant, dilated, peripheral portion of this space which he designated the marginal sinus. In his view it was only at the placental margin, i.e. at the marginal sinus, that the intervillous space communicated with the uterine veins. In criticizing Spanner’s theory of the circulation Ramsey (1956a, 1959) points out the possibility that, in Spanner’s material, the injection medium occluded the slender, oblique central venous drainage channels while accumulating in certain wider more marginal channels. Whatever the explanation of Spanner’s conclusions we can substantiate our earlier statement (Hamilton & Boyd, 1951) that at all stages from the late somite embryo until stages up to foetuses of 60 mm. (and, for that matter, until full term) there are, in the basal decidua, many centrally placed veins draining the intervillous space. We do not deny that, here and there, a vein may occasionally drain from the peripheral region of the intervillous space; indeed we have not infrequently observed such drainage. Nevertheless, we agree with Ramsey (1956a) that this marginal drainage in the human placenta can be relegated to an unimportant role in the explanation of the circulation in the intervillous space.
In the stages of placental development with which we are at present concerned the septa, which are such striking features of the mature placenta, have not yet become established. It is obvious, however, that the septa are only a secondary modification and that a fully functional circulation can exist in their absence. Their absence in the earlier stages removes, of course, real substance from Bumm’s interpretation which demands the presence of the septa as an effective part of the circulatory mechanism. It also detracts strongly from Spanner’s interpretation of the septa as baffles essential for ensuring that the blood from the openings of the arteries in the basal plate passes down to the chorionic plate, where, as the so-called subchorial lake, it is directed, for drainage, to the marginal sinus. Indeed, we consider that the course of the circulation in the mature intervillous space is probably much as we depict it diagrammatically in Pl. 18. Such a circulation would result when the septa are added to the arrangement we find present up to the stage of placental development in the 60mm. c.R. length foetus. It also corresponds closely to Ramsey’s (1956 a, 1959) interpretation of placental circulation for both monkey and Man. It is supported by the observations on human placentae by Stieve (1941, 1942), Kladetsky-Haubrich (1952), Boe (1958, 1954) and Browne (1954). To the circulatory mechanism existing in the placenta itself must be added, as Hinselmann (1925), Wagner (1929), Kladetsky-Haubrich (1952) and, particularly, Ramsey (1959) have pointed out, myometrial contractions; these may play an important role by causing intermittent elevations in the intervillous space pressure. When such BraxtonHicks’s contractions decrease, or cease, the blood from the space will then drain into the uterine veins. Ramsey has also suggested that the head of pressure in the arterial blood entering the intervillous space is sufficient to ensure that the blood is not short-circuited into the adjacent venous orifices. This mechanism may well operate in the fully established and mature placenta but can hardly do so when the arteries contain the plugs of cytotrophoblast to which we have drawn attention.
Summary
- A description is given of the development and differentiation of the trophoblast in Man from about the 12th day until about the 90th day (i.e. to a stage with an embryo of 60 mm.).
- In the earliest specimen the primitive syncytium forms trabecular columns, which incompletely enclose separate lacunar spaces. At a slightly later stage of development these columns become radially arranged to form the primary villous stems.
- The stems acquire cytotrophoblastic cores as a result of the proliferation of the cytotrophoblast of the chorion; at their chorionic ends, with the development of mesoderm, secondary villi are then produced. There are no ‘free villi’ at this stage and the developing placenta is essentially labyrinthine.
- With continuing development the cytotrophoblastic cells at the peripheral extremities of the secondary villi proliferate to form cell columns; when these reach the primitivesyncytium they mushroom out in it and fuse to form the trophoblastic shell.
- Interstitial growth within the shell results in an increase in size of the implantation site.
- The circulation in the lacunae is at first sluggish. The spiral arteries do not open directly into the intervillous space; the maternal blood percolates through intercellular gaps in the trophoblastic shell and leaves it through randomly distributed venous openings.
- No arterio-venous anastomoses connecting the spiral arteries with the sinusoids have been identified.
- When the maternal arteries come to communicate with the intervillous space they also randomly perforate the basal plate.
- The arterial orifices into the intervillous space are invaded by cytotrophoblastic cells, and such cells can be found in these vessels throughout the remainder of the period of development under consideration.
- The endothelium in the proximal parts of the spiral arteries is often hypertrophied.
- The vascular arrangements found in the period under review do not reveal a marginal sinus in the sense described by Spanner. The probable circulation in the intervillous space is discussed.
We record our indebtedness to the many obstetricians and pathologists who have, often at great personal inconvenience, sent us specimens. We wish to thank Dr G. A. K. Missen who presented us with the 14-day embryo. Prof. J. S. Baxter very generously placed at our disposal the ‘Gar’ embryo, and Prof. T. B. Johnston gave us permission to examine the H.R. 1 embryo; to both we are very grateful. Finally, we wish to record our indebtedness to the Nuffield Foundation, for a grant making possible the preparation of a portion of the material, and to the Central Research Fund of the University of London for a grant to defray the cost of the colour blocks. We have been fortunate in the technical aid rendered to us by Mr E. J. Park, Mr R. H. Watts and Mr J. W. Cash; the photography is the work of Mr J. A. F. Fozzard and Mr K. Iles.
References
Bartelmez GW. The form and the functions of the uterine blood vessels in the rhesus monkey. (1957) Contrib. Embryol., Carnegie Inst. Wash. 36:153—182.
Boe, F. (1953). Studies on the vascularization of the human placenta. Acta obstet. gynec. scand. 32, 1-92.
Bog, F. (1954). Vascular morphology of the human placenta. In The Mammalian Fetus: Physiological Aspects of Development. Cold Spring Harb. Symp. Quant. Biol. 19, 29-35.
Boyd, J. D. (1956). Morphology and physiology of the utero-placental circulation. In Gestation: Trans. 2nd Conf. (Villee, ed.), pp. 182-194. N.Y.: Macy Foundation.
Boyd, J. D. (1959). Some aspects of the relationship between mother and child. Ulster Med. J. 28, 35-46.
Boyd, J. D. & Hamitron, W. J. (1950). Development of the placenta. Int. Anat. Congr., Oxford, Commun., p. 30.
Boyd, J. D. & Hamitton, W. J. (1956). Cells in the spiral arteries of the pregnant uterus. J. Anat., Lond., 90, 595.
Boyd, J. D. & Hamitton, W. J. (1960). The giant cells of the pregnant human uterus. J. Obstet. Gynaec. Brit. Emp. (in the Press).
Brewer JI. A normal human ovum in a stage preceding the primitive streak (The Edwards-Jones-Brewer ovum). (1937) Amer. J Anat., 61: 429-481.
Brewer JI. A human embryo in the bilaminar blastodisc stage (The Edwards-Jones-Brewer ovum). (1938) Contrib. Embryol., Carnegie Inst. Wash. Publ. 496, 27: 85-93.
Browne, J. C. M. (1954). Utero-placental circulation. In The Mammalian Fetus: Physiological Aspects of Development. Cold Spring Harb. Symp. Quant. Biol. 19, 60-71.
Bryce TH. and Teacher JH. Contributions To The Study Of The Early Development And Imbedding Of The Human Ovum 1. An Early Ovum Imbedded In The Decidua. (1908) James Maclehose and Sons. Glasgow.
Bum, E. (1893). Uber die Entwicklung des mutterlichen Blutkreislaufes in der menschlichen Placenta. Arch. Gynak. 43, 181-195.
Davies F. A previllous human ovum, aged nine to ten days (the Davies-Harding ovum). (1944) Trans. Roy. Soc. Edinb. 61: 315-326.
Distz JH. and West CM. A human ovum at the previllous stage. (1941) J Anat. 75: 269-281.
Distz, J. H. & West, C. M. (1941). A human ovum at the previllous stage. J. Anat., Lond., 75, 269-281.
Frassi, L. (1908). Weitere Ergebnisse des Studiums eines jungen menschlichen Eies in situ. Arch. mikr. Anat. Entwick. 71, 667-695.
Grosser, QO. (1927). Friihentwicklung, Eihautbildung und Placentation des Menschen und der Sdugetiere. Minchen: Bergmann.
Hamilton, W. J., BARNES, J. & Dopps, G. H. (1948). Phases of maturation, fertilization and early development in Man. J. Obstet. Gynaec. Brit. Emp. 50, 241-246.
Hamilton, W. J. & Boyd, J. D. (1950). Phases of human development. In Modern Trends in Obstetrics and Gynaecology, pp. 114-137. London: Butterworth.
Hamilton, W. J. & Boyd, J. D. (1951). Observations on the human placenta. Proc. R. Soc. Med. 44, 489-496.
Hamilton WJ. Boyd JD. and Mossman HW. Human Embryology. (1945) Cambridge: Heffers.
Hamilton WJ. and Gladstone RJ. A presomite human embryo (Shaw) - the implantation. (1942) J Anat. 76(2): 187-203 PMID 17104888
Hartman CG. The breeding season of the opossum (didelphis virginiana) and the rate of intrauterine and postnatal development. (1928) J Morphol. 45(1): 143-214.
Hartman, C. G. (1928). The period of gestation in the monkey, Macacus rhesus, first description of parturition in monkeys, size and behaviour of the young. J. Mammal. 9, 181-194.
Hartman CG. Studies in the reproduction of the monkey Macacus (Pithecus) rhesus, with special reference to menstruation and pregnancy. (1932) Contr. Embryol. Carneg. Instn, 23: 1161.
Hartman, C. G. (1932). Studies in the reproduction of the monkey Macacus (Pithecus) rhesus, with special reference to menstruation and pregnancy. Contr. Embryol. Carneg. Instn, 23, 1161.
Hertig AT. Angiogenesis in the early human chorion and in the primary placenta of the macaque monkey. (1935) Carnegie Instn. Wash. Publ. 459, Contrib. Embryol. 25: 37-81.
Hertig, A. T. (1935). Angiogenesis in the early human chorion and in the primary placenta of the macaque monkey. Contr. Embryol. Carneg. Instn, 25, 37-82.
Hertig, A. T. & Rock, J. (1941). Two human ova of the previllous stage, having an ovulation age of about 11 and 12 days respectively. Contr. Embryol. Carneg. Instn, 29, 127-156.
Hertig, A. T. & Rock, J. (1944). On the development of the early human ovum with special reference to the trophoblast of the previllous stage: a description of 7 normal and 5 pathologic human ova. Amer. J. Obstet. Gynec. 47, 149-184. ,
Hertig, A. T. & Rock, J. (1945). Two human ova of the previllous stage, having a developmental age of about seven and nine days, respectively. Contr. Embryol. Carneg. Instn, 31, 67-84.
Hertig, A. T. & Rock, J. (1949). Two human ova of the previllous stage, having a developmental age of about 8 and 9 days respectively. Contr. Embryol. Carneg. Instn, 33, 169-186.
Hertig, A. T., Rock, J. & Adams, E. C. (1956). A description of 34 human ova within the first 17 days of development. Amer. J. Anat. 98, 435-494.
Heuser, C. H., Rock, J. & Hertic, A. T. (1945). Two human embryos showing early stages of the definitive yolk sac. Contr. Embryol. Carneg. Insin, 31, 85-99.
Heuser, C. H. & STREETER, G. L. (1941). Development of the macaque embryo. Contr. Embryol. Carneg. Instn, 29, 15-55.
HINSELMANN, H. (1925). Normales und pathologisches Verhalten der Placenta und des Fruchtwassers. In Biologie und Pathologie des Weibes (Halban & Seitz), 6, 241-574. Berlin: Urban and Schwarzenberg.
Johnston TB. An early human embryo, with 0.55 mm. long embryonic shield. (1940) J. Anat., 75:1-49.
Johnston TB. The chorion and endometrium of the embryo H.R.1. (1941) Amer. J Anat. 75: 153-163.
Jones, H. O. & Brewer, J. I. (1935). Normal human ovum in the primitive streak stage (approximately eighteen and one-half days). Surg. Gynec. Obstet. 60, 657-666.
June, P. (1908). Beitrdge zur friihesten Eieinbettung beim menschlichen Weibe. Berlin: Karger.
KERMAUNER, F. (1912). Uber Plazentarcotyledon und des ‘Blutkreislauf im intervillésen Raum. Arch. Anat. Physiol.; Lpz., pp. 189-192.
KLapetsky-Hausricu, A. L. (1952). Beobachtungen iiber den venésen Abfluss aus der Plazenta an Hand von Befunden an einer in situ fixierten Plazenta aus dem 5 Monat. Acta Anat., Basel, 14, 168-178.
Krarka, J. (1941). The Torpin ovum, a presomite human embryo. Contr. Embryol. Carneg. Instn, 29, 167-193.
Mazan_Ec, K. (1959). Blastogenese des Menschen. Jena: Fischer.
MERRTENS, J. (1894). Beitrige zur normalen und pathologischen Anatomie der menschlichen Placenta. Z. Geburish. Gyndk. 30, 1-22. 324 W. J. Hamilton and J. D. Boyd
Mossman, H. W. (1956). In Discussion on ‘Morphology and physiology of the utero-placental circulation’ (Boyd). In Gestation: Trans. 2nd Conf. (Villee,ed). p.185. N.Y.: Macy Foundation.
Orsini, M. W. (1954). The trophoblastic giant cells and endovascular cells associated with pregnancy in the hamster, Cricetus auratus. Amer. J. Anat. 94, 273-381.
Orrmann, R. (1938). Uber die Placenta einer in situ fixierten menschlichen Keimblase aus der 4 Woche. Z. ges. Anat. 1. Z. Anat. EntwGesch. 108, 427-458.
PETERS, H. (1899). Ueber die Einbettung des menschlichen Eies und das friiheste bisher bekannte menschliche Placentationsstadium. Vienna and Leipzig: Deuticke.
Ramsey EM. The vascular pattern of the endometrium of the pregnant rhesus monkey (Macaca mulatta). (1949) Contr. Embryol. Carneg. Instn, 33, 1138-147.
Ramsey, E. M. (1949). The vascular pattern of the endometrium of the pregnant rhesus monkey (Macaca mulatta). Contr. Embryol. Carneg. Instn, 33, 1138-147.
Ramsey, E. M.(1954a). Circulation in the maternal placenta of primates. Amer. J. Obstet. Gynec. 67, 1-14.
Ramsey, E. M. (19546). Venous drainage of the placenta of the rhesus monkey (Macaca mulatta). Contr. Embryol Carneg. Instn, 35, 151-173.
Ramsey, E. M. (1956a). Distribution of arteries and veins in the mammalian placenta. In Gestation: Trans. 2nd Conf. (Villee, ed.), pp. 229-251. N.Y.: Macy Foundation.
Ramsey, E. M. (19566). Circulation in the maternal placenta of the rhesus monkey and man, with observations on the marginal lakes. Amer. J. Anat. 98, 159-190.
Ramsey, E. M. (1959). Vascular adaptations of the uterus to pregnancy. Ann. N.Y. Acad. Sci. 75, 726-745. :
SIEGENBEEK VAN HEUKELOM, D. E. (1898). Ueber die menschliche Placentation. Arch. Anat. Phys., Lpz., Anat. Abt., pp. 1-36.
SPANNER, R. (1935). Mutterlicher und kindlicher Kreislauf der menschlichen Placenta und seine Strombahnen. Z. ges. Anat. 1. Z. Anat. EntwGesch. 105, 163-242.
StiEvE, H. (1986). Ein ganz junges, in der Gebirmutter erhaltenes menschliches Ei (Keimling Werner). Z. mikr. Anat. Forsch. 40, 281-322.
STIEVE, H. (1941). Die Entwicklung und der Bau der menschlichen Placenta. 2. Zotten, Zottenraumgitter und Gefasse in der zweiten Hilfte der Schwangerschaft. Z. mikr.-anat. Forsch. 50, 1-120.
STIEVE, H. (1942). Anatomie der Placenta und des intervillésen Raumes. In Biologie und Pathologie des Weibes (Seitz & Amreich), 7, 109-146. Berlin: Urban and Schwarzenberg.
Streeter GL. The "Miller" ovum—the youngest normal human embryo thus far known. (1926) Carnegie Instn. Wash. Publ 363, Contrib. Embryol., 18: 31-48.
STREETER, G. L. (1926). The ‘Miller’ ovum—the youngest normal human embryo thus far known. Contr. Embryol. Carneg. Insin, 18, 31-48.
Vert, O. & Escu, P. (1922). Untersuchungen eines in situ fixierten operativ gewonnen menschlichen Eies der vierten Woche. Z. ges. Anat. 1. Z. Anat. EntwGesch. 63, 343-414.
Wacner, G. A. (1929). Der intervillése Raum. Arch. Gynak. 137, 699-708.
West CM. Two presomite human embryos. (1952) J. of Gynaecol. Geat Brit. Emp., 59: 336-351.
West, C. M. (1952). Two presomite human embryos. J. Obstet. Gynaec. Brit. Emp. 59, 386-351.
Wison, K. M. (1954). A previllous ovum of eleven days development. Amer. J. Obstet. Gynec. 68, 63-68.
Wislocki, G. B. & STREETER, G. L. (1938). On the placentation of the macaque (Macaca mulatta), from the time of implantation until the formation of the definitive placenta.. Contr. Embryol. Carneg. Instn, 27, 1-65.
Explanation of Plates
Plate 1
Fig. 1. A drawing ( x 140) of a section through the middle of an implantation site at the 11th-12th day of development (Barnes embryo). The surface view of the implantation site is shown in Pl. 2, fig. 2. The blastocystic trophoblast has differentiated into primitive syncytium and cytotrophoblast ; the latter surrounds the primitive mesoblast which almost completely fills the © original blastocyst cavity. At intervals the cytotrophoblast has proliferated to form projections which are the forerunners of the ‘primary villi’. The primitive syncytium, which has not yet extended completely round the superficial aspect of the implantation site, shows an extensive development of intercommunicating lacunae containing some maternal blood cells. The endometrium surrounding the implanted ovum is oedematous and there is haemorrhage into a uterine gland in contact with the syncytium. Photomicrographs of selected portions of the trophoblast of this specimen are illustrated in Pl. 4, figs. 9-12.
Plate 2
Fig. 2. Photograph ( x 12) of the surface view of the implantation site of the Barnes embryo. The implantation site itself appears as a slightly elevated area of the endometrium on which openings of the uterine glands can be seen. The surface of the endometrium shows characteristic shallow and irregular furrows.
Fig. 3. Photograph ( x 3-5) of the surface view of the partially opened implantation site from which a 28-somite embryo (Camb. H. 710) had been removed. Chorionic villi are seen on the everted cut surface of the decidua capsularis. Photomicrographs of sections through this implantation site are illustrated in Pl. 5, fig. 14 and PI. 7, figs. 20-22.
Fig. 4. Photograph ( x 1-1) of a general view of the interior of the uterus containing the implantation site of a 10 mm. embryo (CX. 100). A small opening has been made through the central part of the decidua capsularis. Note the smoothness of this portion of the decidua in comparison with the furrowed decidua vera (parietalis).
Fig. 5. Photograph ( x 3-5) of the implantation site of a 10 mm. embryo (CX. 100) with the decidua capsularis removed to an extent sufficient to show the embryo, within the collapsed amnion, and the yolk sac. Note the extent of development of the chorionic villi related to the decidua capsularis. This photograph has been rotated in relation to fig. 4 to show the embryo in a suitable orientation. A section through the placenta of this specimen is illustrated in Pl. 8, fig. 23.
Plate 3
Fig. 6. Photograph ( x 1) of a coronal section of a uterus containing a 15 mm. embryo (CX. 102). The decidua capsularis has been opened to show the amnion containing the embryo. A section of the placenta of this specimen is illustrated in Pl. 8, fig. 24.
Fig. 7. Photograph ( x 0-9) of a coronal section of a uterus containing a 29 mm. embryo (CX. 105). Note that the uterine lumen is still widely patent. A section through the uterus and in situ placenta of this specimen is illustrated in Pl. 9, fig. 25.
Fig. 8. Photograph ( x 2) of section through a portion of the uterus to show the placenta and chorionic sac containing a 36 mm. embryo (Camb. H. 789.). The basal placental plate separating the chorionic villi and the intervillous space from the decidua basalis appears as a thin band across the greater part of the attachment area. The decidua basalis, though of variable thickness, is still very distinct. The placental (i.e. lower) half of the decidua capsularis has chorionic villi related to it projecting into an associated extension of the intervillous space. The upper part of the decidua capsularis, together with the abembryonic portion of the chorion, is markedly attenuated. Consequently in this region the interior of the chorionic sac is separated from the uterine lumen by only a very thin double layer of foetal and maternal tissue. The decidua vera can be seen on either side of the placenta extending up to the cut margin of the uterine wall. Note the presence of uterine veins throughout the decidua basalis and the absence of any region of the intervillous space which could be identified as a marginal sinus.
Plate 4
Fig. 9. Photograph ( x 200) of a section through the deep central portion of the implantation site of the Barnes embryo; maternal tissue is on the right of the illustration. The intereommunicating lacunar spaces (L.S.) are shown surrounded by syncytial trophoblast. Uterine venous sinusoids (S.) communicate with these. A plug of syncytium can be seen in direct contact with the lumen of the uterine gland (G.).
Fig. 10. Photomicrograph (x 840) of a section through the primitive syncytium and adjacent oedematous endometrium in the Barnes embryo; maternal tissue is on the left of the illustration. Note the texture of the syncytial cytoplasm, a long stretch of which is devoid of nuclei. In the upper part of the figure there is an irregular projection of syncytium, containing a very large nucleus, into the endometrium; elsewhere the foetal-maternal junction is smooth.
Fig. 11. Photomicrograph ( x 636) of the foetal-maternal junction of the Barnes embryo. A mass of primitive syncytium is in direct contact with the lumen of a uterine gland (G.) in which secretion can be seen. The syncytium separates this lumen from a lacunar space (L.S.).
Fig. 12. Photomicrograph ( x 840) to show an opening of a venous sinusoid (S.) into a lacunar space (L.S.). Note the relations of the endothelium of the sinusoid to the syncytium.
Plate 5
Fig. 13. Photomicrograph ( x 45) of a section of the implantation site of a 14-day embryo (Missen). Chorionic villi with mesodermal cores are now well established round the whole of the chorionic sac. There is an extensive intervillous space which, in this section, communicates at two points with a large venous sinusoid (S.). Details of the differentiation of the trophoblast in this specimen are shown in PI. 6, fig. 15.
Fig. 14. Photomicrograph ( x 10) of a section of the implantation site of a 28-somite human embryo (Camb. H. 710). Villi which cover the whole of the chorionic sac are well established, and possess numerous foetal vessels. There is no indication of a marginal sinus in the intervillous space ; there is, however, venous drainage from the left margin of this space. The decidua basalis is thick and contains many dilated glands; several of the central ones show haemorrhage into their lumina. The effect of expansion of the chorionic sac on the uterine glands is particularly well shown on the right of the illustration where their necks are constricted and stretched, thus damming back the products of secretion. Veins and spiral arteries can be identified at intervals. in the decidua basalis.
Plate 6
Fig. 15. Photomicrograph (x 100) of part of a section (H. & E.) through the foetal-maternal junction of a 14-day embryo (Missen). The chorionic villi (to the left of the illustration) possess mesodermal cores but as yet no definitive blood vessels are present. The villi possess a covering of definitive syncytium overlying Langhan’s layer. At the tips of some of the villi cytotrophoblastic columns, by proliferation, are contributing to the trophoblastic shell. On the maternal side of this shell a number of persisting masses of primitive syncytium can be identified. In the intervillous space there are maternal blood vessels and at about the centre of the figure an isolated syncytial sprout in the intervillous space; maternal blood cells are also present in the intervillous space.
Fig. 16. Photomicrograph ( x 150) of part of a section (H. & E.) through the tips of the chorionic villi (above) and of the adjacent endometrium (below) in a 16-day human embryo. Cytotrophoblastic columns at the tips of the two central villi have proliferated and fused to form part of the trophoblastic shell. Definitive syncytium can be identified on the surface of the villi and primitive syncytium is related to the shell. Maternal blood vessels and a syncytial sprout. can be identified in the intervillous space.
Fig. 17. Photomicrograph ( x 200) of a section (H. & E.) through the peripheral syncytium of an 18-day embryo (Gar). The syncytium shows extensive vacuolation and a maternal red blood corpuscle is present in one of the vacuoles. At another point several such red blood corpuscles are in the process of coming to be included within the syncytium.
Fig. 18. Photomicrograph ( x 270) of a section (H. & E.) through a uterine gland near the implantation site of an 18-day embryo (Gar). The lumen of the gland possesses maternal red blood corpuscles and two masses of multinucleated syncytium.
Plate 7
Fig. 19. Photomicrograph (x 100) of a section (H. & E.) of an 18-day embryo (Gar). Marked proliferation at the tips of the villi (to the right) has resulted in the production of a trophoblastic shell which now extends completely around the foetal tissue. The villi are covered except. at their tips by a Langhan’s layer of cytotrophoblast and a superficial layer of definitive syncytium. At one point a syncytial sprout can be seen arising from the definitive syncytium. Remnants of the primitive syncytium lie within and on the maternal aspect of the trophoblastic shell. Early vasculogenesis is present in the mesodermal cores of the villi.
Fig. 20. Photomicrograph ( x 60) of a section (H. & E.) through the tips of the chorionic villi and trophoblastic shell of a 26-day human implantation site (Camb. H. 710). The portion of the trophoblastic shell illustrated contains a typical irregularly shaped space in which glandular secretion and cellular detritus can be identified. There are several masses of primitive syncytium related to the cytotrophoblast. One of these shows extensive vacuolation and an inclusion within one of the vacuoles.
Fig. 21. Photomicrograph ( x 150) of a section (Masson’s trichrome stain) of the foetal-maternal junction of a 26-day implantation site (Camb. H. 710). The foetal tissue (below) shows the trophoblastic shell, the tip of a villus with a cytotrophoblastic column and some residual syncytium. Interspersed amongst the typical cytotrophoblastic cells of the shell are darkly staining fusiform cells. In the upper portion of the figure the decidua is surrounded by bluestaining collagen in striking contrast to the appearance presented by the cytotrophoblast. 22. Photomicrograph ( x 350) of a section (Azan stain) through an endometrial spiral artery close to the implantation site of a 26-day embryo (Camb. H. 710). The cells of the endothelium of the vessel are markedly hypertrophied.
Plate 8
Fig. 23. Photomicrograph ( x 6) of a section through the uterus and in situ placenta of a 10 mm. embryo (CX. 100). The villi are more or less equivalently developed round the whole of the chorionic sac. The decidua capsularis is still relatively thick. The distortion and dilatation of the glands in the decidua basalis, which has been irregularly penetrated by the trophoblast, are clearly shown. The thick decidua vera is well shown on the right of the illustration.
Fig. 24. Photomicrograph (x 6) of a section through the uterus and in situ placenta of a 15 mm. embryo (CX. 102). The chorionic villi are now distinctly better developed in relation to the decidua basalis and the decidua capsularis is much attenuated. The uterine glands are less apparent than in the specimen illustrated in fig. 28. The very irregular decidua basalis and the decidua vera are both much thinned.
Plate 9
Fig. 25. Photomicrograph ( x 2-5) of a section through the uterus and in situ placenta of a 29 mm. embryo (CX. 105). The foetal-maternal junction is now very distinct and the decidua basalis is thin. The implantation in this specimen was low on the posterior uterine wall and the cervix can be seen to the left of the illustration. On the right there has been some separation of the chorionic villi from the decidua.
Fig. 26. Photomicrograph ( x 3:5) of a section through rather less than half of the in situ placenta of a 42 mm. foetus (CX. 110) and the related uterine wall.
Fig. 27. Photomicrograph ( x 4) of a section through rather less than half of the in situ placenta of a 50 mm. foetus (CX. 111) and the related uterine wall.
Plate 10
Fig. 28. Photograph ( x 1-8) of a uterus which contained a 30 mm. embryo (CX. 106). The decidua capsularis, which is very thin, approaches but does not make contact with the decidua vera. Note the complete absence of any indication of a marginal sinus in the intervillous space.
Plate 11
Fig. 29. Photograph ( x 1-8) of a uterus which contained a 60 mm. foetus (CX. 112). The uterine lumen is still apparent in the lower half of the uterus. The decidua basalis is very thin. Distorted and dilated uterine glands are present in the decidua vera to the right of the area of placental attachment.
Fig. 30. Photomicrograph ( x 18) of a section through the central portion of the in situ placenta of a 30 mm. embryo (CX. 106). Two large openings from the intervillous space into uterine veins are shown. A plug of villi can be seen in the right of these communications.
Plate 12
Fig. 31. Photomicrograph (x 100) of a section (Stain - Haematoxylin Eosin) of the termination of an endometrial spiral artery in the in situ placenta of a 15 mm. embryo (CX. 102). Cytotrophoblast cells are invading and plugging the lumen of the vessel. The wall of the artery shows marked degenerative changes. A portion of syncytium can be seen to the right of the illustration.
Fig. 32. Photomicrograph ( x 100) of a section (Stain - Haematoxylin Eosin) through the foetal-maternal junction in the in situ placenta of a 22 mm. embryo (CX. 103). The terminal coils of an endometrial spiral artery can be seen. A straight channel from the lumen of one of the coils extends almost to the intervillous space. Apices of villi and the cytotrophoblastic shell are seen in the upper portion of the illustration. 328 W. J. Hamilton and J. D. Boyd
Fig. 33. Photomicrograph (x 110) of a section (Stain - Haematoxylin Eosin) through the terminal coils of an endometrial spiral artery and adjacent intervillous space of a 46 mm. embryo (Camb. H. 658). Before fixation a uterine artery in this specimen was injected with indian ink. The ink can be seen in the lumen of the spiral artery and some of it is percolating amongst the cytotrophoblastic cells.
Fig. 34. Photomicrograph ( x 340) of a section through the termination of the spiral artery shown in fig. 33. The percolation of ink between the cytotrophoblastic cells is clearly seen. Note the highly modified arterial wall and the cytotrophoblastic cells within the arterial lumen.
Fig. 35. Photomicrograph ( x 70) of a section through the termination of an endometrial artery in the in situ placenta of a 50 mm. embryo (CX. 111). The arterial wall shows marked degeneration and cytotrophoblastic cells are extending into the vessel. At each side of the arterial opening Nitabuch’s membrane can be seen.
Fig. 36. Photomicrograph ( x 85) of a section of the central region of an in situ placenta of a 50 mm. embryo (CX. 111) showing communication between the intervillous space and a maternal vein. Note the continuity between cytotrophoblastic cells and unaltered venous endothelium. There is no cytotrophoblast within the lumen of the vein but a sectioned syncytial sprout can be seen.
Plate 13
Fig. 37. Scheme to show the essential features in placental structure which are found after the 60 mm. stage. Three cotyledons, including a marginal one, are illustrated; the cotyledons are separated from each other on the maternal side by the septa. They each contain the group of villi which constitute the associated ‘foetal’ cotyledon. The villi branch freely and there are many adhesions between adjacent ones so giving a partially labyrinthine nature to the intervillous space. Such villi are shown in relief in the marginal cotyledon and in section in the adjacent one. The third cotyledon has been dissected to show the arrangement of the septa. The openings of the endometrial arteries and veins into the intervillous space through the basal plate are indicated and an attempt has been made to show the probable degree of oxygenation of the maternal blood in the intervillous space. A marginal sinus in the intervillous space has not been included in the scheme as our material does not show such a feature.
Cite this page: Hill, M.A. (2024, April 25) Embryology Paper - Development of the human placenta in the first three months of gestation (1960). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Development_of_the_human_placenta_in_the_first_three_months_of_gestation_(1960)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G