Paper - Contribution to the study of the morphogenesis of the thyroid gland (1941)
| Embryology - 18 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Sgalitzer KE. Contribution to the study of the morphogenesis of the thyroid gland. (1941) J Anat. 75(4): 389-405. PMID 17104869
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Contribution to the Study of the Morphogenesis of the Thyroid Gland
By K. E. Sgalitzer
Hebrew University, Jerusalem
- This work was completed in July 1938.
Introduction
Many problems concerning the development of the thyroid gland still require study for, in spite of numerous investigators, many of the contributions to its embryology have been based on too small a series of embryos to give significant results. Having at: my disposal, through the kindness of Dr V. Patzelt, the large collection of human embryos in the Institute of Histology and Embryology of the University of Vienna I have resurveyed the problem of thyroid development, paying special attention to a peculiar variation in its early anlage and to the question of the developmental history of the thyroglossal duct and its derivatives.
The collection used for this investigation included serial sections of 130 human embryos from 4 to 65 mm. C.R. length and in a good state of preservation. The sections were stained with one or other of the haematoxylin stains or with Mallory’s T.C.T. stain. Only those embryos were studied whose condition and plane of section made exact statement possible. Both graphic and plastic reconstructions were made of important stages.
A Variation in the Thyroid Anlage
Normally, the thyroid gland appears in embryos of about 17-18 pairs of somites (Davis, 1923; Wen, 1928; Politzer, 1928) as an evagination of a specially differentiated part of the wall of the pharynx into the underlying mesenchyme. It is connected to the pharyngeal cavity by a hollow stalk. In an embryo of 22 pairs of somites, however, Wen described the thyroid primordium as follows: “Just caudal to the first pharyngeal pouch and the bifurcation of the aorta there is a thick walled median inyagination of the pharynx, which histologically resembles the thyroid anlage of embryos at this stage of development. It has a core of mesodermal’ tissue and an endothelial sprout from the ventral aortic trunk extending into it.” Wen illustrated this abnormality with a diagram of one section and a mid-sagittal reconstruction.
Our collection of embryos included two embryos showing this abnormality: embryo Bs with 26-27 pairs of somites and embryo F with 28 pairs of somites. Unlike Wen’s embryo, of which the head had been removed and in which the thyroid abnormality might be regarded as an artefact, both of our embryos are sectioned in a complete series and both are excellently fixed and stained. The condition of the tissues permits a reliable diagnosis of the thyroid primordia.
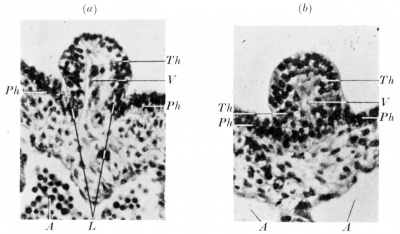
Fig. 1 shows the mid-sagittal reconstruction of a part of the pharynx of embryo F (4 mm., 28 somites). The cavities of the first and second pharyngeal pouches of the right side have been projected on to the midsagittal plane in order to show that the thyroid anlage is situated at its normal level. The latter, however, instead of showing the normal evagination, is represented by a small nodule which projects into the pharyngeal cavity. It is formed by an epithelium which comparison shows to be similar in appearance to a normal thyroid primordium (fig. 2a). There projects into the nodule a mesodermal core containing a small vessel which arises from the aortic sac. The epithelial margins of the nodule are projected like lips towards the underlying connective tissue, and the outer part of these lips merges into the pharyngeal epithelium.
lies partly on the free surface of the pharyngeal epithelium which separates it from the underlying mesenchyme and its conical tip is projected freely into the pharyngeal cavity. The vessel contained in the core of mesenchyme is smaller in this embryo and it could not be followed to its source, it is not represented in the reconstruction. The transition from thyroid to pharyngeal epithelium on the lip-like margins of the nodule is similar to that seen in embryo F.
Although the sagittal reconstructions are different, transverse sections through the thyroid primordia of these two embryos are very similar (fig. 2 a, b). They differ only in the size of the mesodermal core and in the cephalad inclination of the nodule in embryo Bs.
small mesenchymatous core. The nodule
A similar condition is found in the midsagittal reconstruction of the pharynx of embryo Bs (fig. 3). In this embryo the thyroid anlage is smaller than in embryo F and takes the form of a compact nodule with

|
Fig 1 Mid-sagittal graphic reconstruction of the thyroid anlage of embryo F.
The cavities of the first and second pharyngeal pouches of the right side have been projected on to the mid-sagital plane.
A, aortic sac; L, lipped margin of thyroid anlage; M, connective tissue core; Ph, pharyngeal epithelium; Th, thyroid anlage; F, its vessel; I, II, first and second pharyngeal pouches. x250. |
The vessel which supplies the nodule is noteworthy. Normally the developing thyroid gland remains for a long time _without a special vesselland presumably is nourished through the wall of the neighbouring aorta (Hammar & Hellman, 1920). figs. 5 and 6 show the relative -positions of the normal thyroid primordium and the aortic sac. In some sections of normal embryos, indeed, the thyroid primordium is separated from the blood stream by the vascular endothelium alone. Rabl (1923) assumed that there was a chemotropic effect of the thyroid primordium upon the blood vessels, and this assumption may be applicable in an explanation of the origin of the special vessel found in the abnormal primordia.
Fig. 2. Transverse section through the thyroid anlage of embryo F (a) and of embryo Bs (b). A, aortic sac; L, lip margin of thyroid anlage; Ph, pharyngeal epithelium; Th, thyroid epithelium; V, vesselntli thyroid. x 290.
Fig. 3. Mid-sagittal reconstruction of the thyroid anlage of embryo Bs. x 285.
The difference between the condition shown by these two embryos and the normal tuberculum thyroideum must be emphasized. Kallius (1903) described the median thyroid anlage-of the rabbit as an epithelial thickening which extends over all of the mesobranchial region. It may become so thick that it forms a hump which projects into the pharyngeal cavity. But where it does so the epithelium forms a cone in the opposite direction reaching the aortic sac. This thickening, called by Kallius the tuberculum thyroideum, is a transient formation which he regarded as serving to “ collect” material for the thyroid anlage. It disappears in subsequent development so completely that a small depression appears in its place. It can be seen from Kallius’_s description and.figures that it is a formation quite different from the above-described abnormal primordia. In Wen’s embryo, and in mine, there is a distinct invagination of the whole thyroid mass, at the normal level of the anlage, into the cavity of the pharynx. But the tuberculum thyroideum of Kallius is an elevation of the pharyngeal epithelium which extends from the first pair of branchial arches to the level of the third pair of pharyngeal pouches. Moreover, in these human embryos, there is no epithelial cone,reaching towards the aortic sac. This cone, which is clearly seen in the rabbit embryo, is found in all mammals at that point where the thyroid gland is formed and represents the chief part of its primordium. finally, there is nothing in the rabbit embryo corresponding to the mesodermal core and the early vascularization by a special vessel. I conclude, therefore, that the tuberculum thyroideum in the rabbit must» not be identified with the rarely occurring invagination of the thyroid primordium into the pharyngeal cavity.
The thyroid primordium described here appears to be a transient formation. It has been found three times among the relatively few described embryos of this stage (22-28 pairs of somites), while no similar formation is known in older embryos. Hence it is concluded that soon after the formation of the nodule_a readjustment of the abnormal development is instituted so that the nodule sinks into the underlying mesenchyme and a normal thyroid gland develops. It might possibly be supposed that the anomaly known as struma interlaryngotrachealis could be explained by assuming its origin from the abnormal thyroid primordium described in this section. But struma interlaryngotrachealis is extremely rare, and its connexion with the main thyroid gland and secondary penetration through the wall of the trachea has been demonstrated by Hiickel (1932), and further, in my embryos the thyroid nodule is found at the level of the normal thyroid formation and well anterior to the respiratory tract.
The Thyroglossal Duct and its Derivatives
In his original description (1885) of the development of the thyroid gland His assumed that the thyroglossal duct passes behind the hyoid bone primordium. He based this opinion on the observation that the pyramidal process of the adult gland is often fixed to the dorsal surface of the hyoid bone. Streckeisen (1886) stated that the thyroglossal duct in development is always ventral to the hyoid, and he considered that the frequent attachment of the pyramidal process to its deep surface is secondary. Further, he showed that in many cases thyroid glandular material could be found on the ventral surface and above the hyoid bone (pre- and suprahyoid glands). These accessory glands were shown to be frequently connected to the upper end of the pyramidal process by vascular connective tissue bands, by cords of glandular tissue, or by cystic formations which Streckeisen showed to be derived from the thyroglossal duct. He also foundin a number of cases a connexion between the suprahyoid gland and the ductus excretorius linguae (Bochdalek, 1866). The suprahyoid gland, or part of it, was found occasionally to be incorporated in the hyoid bone, and transitional forms, from reduplication of the periosteum and inclusion of the gland within its layers to complete incorporation of the glandular material in the bone, were observed. He showed that this incorporation does not occur earlier than the stage of ossification of the body of the hyoid. In 1891 His recanted his former opinion and adopted Streckeisen’s, as he found, in a 16 mm. human embryo, that the situation of the fragments of the duct was such as to make a Ventral course of the thyroglossal duct, relative to the hyoid bone, more probable. He wrote: “One gets the impression that the growing hyoid bone had pushed the series of epithelial corpuscles ventrally. I cannot decide whether this impression corresponds to the real process, but I wish to stress one relation——the straight line from the lower end of the cord attached to the tongue to the highest of the epithelial corpuscles beyond the hyoid bone passes directly through the anlage of the cartilage.” His described the thyroglossal duct in its upper part, neighbouring the foramen caecum, as a thick stump to which a much thinner thread is attached, the latter being enclosed by a sheath of connective tissue cells.
The opinion of both of these workers soon received opposition. Kanthack (1891), basing his studies on adult material, did not recognize any genetic connexion between the foramen caecum and lingual duct on the one hand and the thyroid gland and its pyramidal process on the other. This opinion was criticized by Marshall (1892) who, in a case of median fistula colli, observed a duct originating in the fistula and connected with the apex of the pyramidal process. From the junction a cord of connective tissue, diflicult to distinguish from its surroundings, ran behind the hyoid bone towards the root of the tongue. The upper part of this cord contained a lumen which opened into the oral cavity. On the basis of this finding Marshall repudiated Kanthack’s opinion, and, in opposition to His and Streckeisen, he alleged that the thyroglossal duct was situated behind the hyoid bone. However, the basis for this conclusion was only a cord of connective tissue which the author himself admitted was not easily to be defined from its surroundings. Marshall’s case has been frequently, but incorrectly, quoted as an example of complete retrohyoid thyroglossal duct.
Konig (1896) described some cases of median fistula colli. He showed that the thyroglossal duct may be situated in front of the hyoid bone or within two layers of periosteum, or may even be enclosed in the bone. His description and figures correspond with those of Streckeisen on the incorporation of the prehyoid gland in the hyoid bone, but despite this his cases have been interpreted as showing that the thyroglossal duct was originally within the area where, later, the hyoid bone is formed, that is, that the bone develops around the duct.
In 1896 Schmidt gave a detailed description of the derivatives of the upper part of the thyroglossal duct (the lingual duct or ductus excretorius linguae). In addition he described a thyroid gland with two pyramidal processes, one arising from each lateral lobe. The latter were connected by connective tissue only, a glandular isthmus being absent. The apex of the right pyramidal process terminated abruptly, but that of the left side was continued into a cyst which lay in front of the hyoid bone and reached the lower surface of the genieglossus muscle. The wall of this cyst contained thyroid tissue in its ventral part.
Particular attention attaches to the case published by Matti (1911). Here the thyroglossal duct, which ran behind the thyroid hyoid bone, was patent from the foramen caecum to the pyramidal process and, just above its junction with the latter, opened into the fistula. Matti, referring to His’s work, and concluding from his own case, stated that the position of the thyroglossal duct may vary from individual to individual, being situated either in front of or behind the hyoid bone. He suggested that the former relation was the commoner.
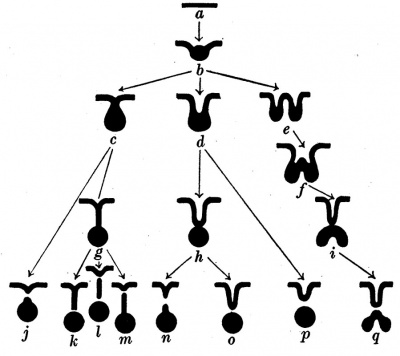
Norris (1918) described the early morphogenesis of the thyroid gland. He found three different forms of the thyroid primordium from which he derived different forms of thyroglossal duct. His important investigations will be discussed more fully later in this paper.
Patzelt (1923) described the remains of the thyroglossal duct of some embryos and of one adult woman. He concluded, from the position of the rest of the duct, that it was originally situated in a prehyoid position.
H. Rabl’s (1923) ideas on the origin and development of the thyroglossal duct were based on his work on the guinea-pig embryos. He stated that the thyroid gland is not the product of the thyroglossal duct as, for example, a salivary gland is the product of its excretory canal. He wrote: “Obviously the thyroid anlage, when growing caudally,. drags behind itself the epithelium of the oral cavity, thus forming the duct. But it is not to be believed that the epithelium of the duct is indifferent, as far as it is adjacent to the specific thyrogenic zone.. . .The tissue which forms the thyroglossal duct is thus a transition between thyrogenic epithelium and the completely indifferent epithelium of the oral cavity.”
Wegelin (1926) acknowledged the point of view of Streckeisen and of His, but as one certain case of retrohyoid thyroglossal duct had been observed by Matti, and as Marshall’s case had been interpreted in the same Way, he concluded: “If the thyroglossal duct fragments the hyoid bone pushes the parts ventrally. But if, when the hyoid bone is formed, the duct is still unbroken, the latter is situated so far dorsally that the bone develops ventral to it.”
Fischel (1929) expressed the prevailing view on the development of the thyroglossal duct. He wrote: “The separation of the thyroid gland from the pharynx does not usually happen in the earliest stages, in embryos of 3-7 mm. length the thyroid anlage is still connected to ‘it.. . .In most cases the thyroglossal duct undergoes involution by losing its lumen in 5 mm. embryos, so that it becomes a solid epithelial cord. In embryos of 7 mm. length this cord is fragmented into several corpuscles of which, in 16 mm. embryos, only rests can be found. These also, for the most part, vanish but it may happen that parts of the duct are preserved.”
Schweitzer (1929) described a complete median fistula colli in which he could determine the position of the duct by means of X-ray pictures. Hiickel (1932) has cited this case as one of a complete thyroglossal duct in a prehyoid position, but Schweitzer’s description, which is not accompanied by any figures, is not clear about the relation between the duct and the hyoid bone. Moreover, the case had been complicated by abscesses and, consequently, cannot be regarded as proof of the original relationship.
Fig. 4. Scheme of the development of the thyroid gland and thyroglossal duct (after Norris).
Kampmeier (1937), in a study on the supernumerary thyroid glands in various mammals, stated that thyroid nodes cephalad to the main gland are characteristic of human embryos. He emphasized that all stages of development may be observed from the fragmentation of the thyroglossal duct into nodes to the differentiation of these nodes into typical thyroid tissue.
In his work on the development of the thyroid gland Norris described three forms of thyroid primordium, one of them being solid (fig. 4); My material did not confirm this statement for the primordium was always, at least partly, hollow.
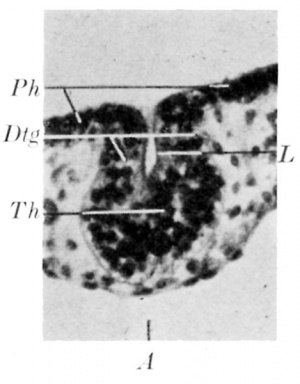
In the embryo B (18 pairs of somites) the thyroid anlage is represented by a small pit with thick walls (fig. 5). All the embryos (eleven in number) in my series between this one and a 6-5 mm. embryo have a hollow-duct. fig. 6 illustrates such an embryo (Fu, 28-29 somites) which shows an extension of the pharyngeal cavity into the thyroid primordium. This stage corresponds to the stage d in Norris’s. scheme (fig. 4). The distal part of the primordium is solid and contains cells more compactly arranged than in the proximal part. The latter can be considered as the anlage of the thyroglossal duct, while the distal part is probably that of the gland itself. The epithelium of both the glandular and duct portions of the primordium is always different from that lining the pharynx. Comparison between figs. 5 and 6 shows that the duct and glandyare formed simultaneouslyas a small pit with a differentiated epithelial lining. They can be distinguished from each other only at that stage when, by deepening and narrowing of the cavity, the pit becomes a narrow passage between the pharynx and the gland primordium. At the same time the histological differentiation commences. Later the thyroglossal duct becomes longer and thinner and its lumen narrower. The lumen is limited to the duct and never enters the gland primordium. At this stage the appearance is similar to that of stage g in Norris’s scheme. The embryo Bb of 6.5 mm. length and all the older embryos in my material have a solid duct.

|
|
| Fig. 5. Section through the thyroid anlage of embryo B. The wall of the pit represents the anlage of both the thyroid gland and the thyroglossal duct. | Fig. 6. Section through the thyroid anlage of embryo Fu with 28 pairs of somites. A, aortic sac; Dtg, thyroglossal duct; L, its lumen; Ph, pharyngeal epithelium; Th. thyroid gland. x 290. |
Before fragmentation the thyroglossal duct consists of a specific epithelium which, as development proceeds, becomes longer and, at the same time, much thinner, especially in its intermediate part, and its lumen is obliterated. finally, in transverse sections, it is represented by a few cells only. These cells are large and vesicular with a palely staining protoplasm and a central nucleus. The duct can be distinguished only with difliculty from the surrounding connective tissue. The glandular tissue, on the contrary, is formed by small cells with darkly staining cytoplasm and can readily be recognized even in small cords. At those points where the thyroglossal duct is about to fragment the basement membrane is not 1 recognizable and the cells show degenerative changes.
Table 1
| Length of embryo in mm | No. of embryos with complete duct | No. of embryos showing forms | No. of embryos with duct absent | ||
|---|---|---|---|---|---|
| l | m | n | |||
| 7 | 3 | / | 1 | 2 | |
| 7.5 | 3 | / | 1 | / | |
| 8 | 2 | / | / | 1 | / |
| 8.5 | 1 | / | 2 | / | / |
| 9 | 3 | / | 2 | 2 | / |
| 9.5 | 1 | / | 2 | / | / |
| 10 | 2 | 1 | 1 | 4 | / |
| 10.5 | / | / | / | 1 | / |
| 11 | / | / | 4 | 4 | 2 |
| 12 | / | 1 | 1 | 9 | 1 |
| 13 | 1 | / | 2 | 4 | 1 |
| 14 | / | / | 1 | 3 | / |
| 15 | / | / | 1 | / | / |
| 16 | l | / | / | 3 | / |
| 17 | / | / | 2 | 2 | / |
| 18 | / | / | 1 | 1 | / |
| 19 | / | / | / | 2 | 1 |
| 20 | / | / | 1 | 4 | / |
| 21 | / | / | 1 | 1 | / |
| 22 | / | / | 2 | 1 | / |
| Total | 17 | 2 | 25 | 44 | 5 |
If the length of an embryo was between two successive numbers it was ascribed to the smaller length.
Norris distinguished eight forms in which the thyroid gland may be separated from the pharynx (fig. 4 j-q). Only three of these are shown in my material, l, m and n. Table 1 shows the relative frequencies of each of these: l is more frequent than m, while the other forms are very rare. The table also shows that only two unfragmented thyroglossal ducts were found in embryos of more than 10 mm. length.
In conclusion it can be stated that the thyroglossal duct is formed simultaneously with the gland. During the migration of the gland it becomes longer and thinner and the lumen disappears. finally the duct fragments, in most cases at about its mid-point. This process is usually completed by the 10 mm. stage.
Development of Pyramidal Process
Embryos of 7 mm. length do not yet possess a glandular pyramidal process, the thyroglossal duct passing directly into the thyroid gland. It is only in embryos of 7-10 mm. that the lower part of the duct differentiates into glandular tissue. The pyramidal process thus produced is at first short with its cephalic end continuous with the cord of duct tissue. The latter becomes progressively shorter as the glandular part grows. Apparently the duct tissue differentiates into glandular tissue but the delimitation of the two portions from each other is more or less clear. The total length of pyramidal process and cord of duct tissue fluctuates according to the position at which fragmentation of the thyroglossal duct first occurs. The pyramidal process usually has a broad connexion with the thyroid gland, but the connexion may be represented by a few cords of cells only. Occasionally it may be entirely separated from the gland.
There is considerable variation in the cephalic end of the pyramidal process in embryos of more than 10 mm. length. It may pass into a cord of duct tissue, the upper portion of which may be fragmented, or it may end in a swelling. In older embryos this swelling may no longer be in connexion with the pyramidal process, in which case it is represented by an accessory glandular node in front of the hyoid bone (prehyoid gland).
The pyramidal process is at first median but after the 10 mm stage it comes to lie laterally. The thyroid gland itself is uninfluenced by this displacement of its pyramidal process, the isthmus remaining in the median plane. There are large individual variations, but in older embryos the pyramidal process usually lies far laterally. There is about an equal probability of the displacement being to the right or to the left. In only one older embryo (Ck, 21 mm. long) was the pyramidal process found to lie exactly in the median plane.
The direction of the displaced pyramidal process varies; it may be directed cephalad, parallel to the median plane, or it may pass towards the median plane which it may reach. In the latter case it may continue in a cephalad direction after having reached the median plane. It has never been seen to pass to theother side.
The separated pieces of the thyroglossal duct cephalad to- the pyramidal process may show a lateral displacement to the same side as the pyramidal process or they may lie in the median plane.
One embryo in our collection showed a doubling of the pyramidal process (Bw, 11-5 mm. long). In this embryo both the foramen caecum and the lingual duct are absent. The lobes of the thyroid gland are in a normal position but are not connected by an isthmus. A pyramidal process arises from the medial surface of each lobe and these ‘processes are directed towards the middle line where they meet. Together, therefore, the pyramidal processes form an-arch, the upper part of which is thyroglossal duct tissue While the parts adjoining the thyroid lobes are glandular. *
In conclusion we may say that the pyramidal process develops out of the a lower part of the thyroglossal duct by differentiation of the duct tissue into gland tissue. It is only after this differentiationihas commenced that the lateral displacement of the pyramidal process begins. The accessory thyroid glands, as far as they have taken origin from the pyramidal process, may show an equivalent displacement.
Development of the Lingual Duct
The term lingual duct can only be used after the thyroglossal duct has divided and only if the fragmentation has occurred beneath the surface of the tongue. Table 1, column 11. shows that the lingual duct is found, after division of the thyroglossal duct, in more than half of the specimens.
At first the lingual duct has exactly the same appearance as the thyroglossal duct, being formed by the specific epithelium described above and containing no lumen. It is situated in the median plane, has a variable length but a fairly uniform width. Fragments of tissue may be separated from it, as from the pyramidal process, and these may remain as cords of duct tissue, as accessory thyroid glands or as cysts (see later).
The uniform width of the lingual duct does not persist. His (1891) described the proximal part of the lingual duct of an embryo of 16 mm. length as a thick stump to which a much thinner thread is attached. To this observation of His I should like to add that this broadening of the proximal part of the lingual duct begins in embryos of 10-11 mm. length. Moreover, while the lingual duct is at first formed by the specific epithelium of the duct, in embryos of approximately 13 mm. length the epithelium of the thick proximal part begins to resemble more and more the neighbouring epithelium of the tongue. In embryos of about 20 mm. length a cavity is formed in this thick stump. This cavity represents the definitive foramen caecum and is to be distinguished from the foramen caecum of young embryos which is a transient formation only.
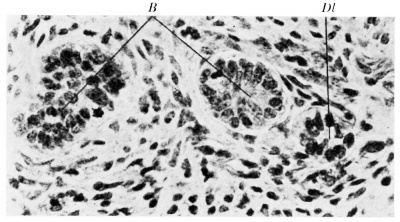
The distal part of the duct does not undergo corresponding changes. It does show some thickening but the thickening is not accompanied by a change in the character of the epithelium. At about the 30 mm. stage, however, small branches of the lingual duct arise from this non-stratified portion of its course. These are the so-called tubes of Bochdalek. They have been found in all of my. embryos more than 30 mm. in length which possess a lingual duct. While the lingual duct itself is situated in the middle line, the branches, which grow ventrally, may curve to the right or to the left. The number of these ducts varies and their length increases with age. Histologically they resemble the A sprouts of salivary glands. The difference between them and the lingual duct is shown in fig. 7. Similar branches may arise from the fragments of the thyroglossal duct which have separated from the caudal end of the lingual duct.
In conclusion it may be stated that the lingual duct is the upper part of the thyroglossal duct. Its proximal part thickens, changes into an epithelium resembling that of the tongue and develops a cavity. From its distal portion which retains its specific histological character, branches grow out which come to resemble salivary glandular material. Examination of older embryos shows that the transformation of the epithelium gradually proceeds down its distal portion. At the same time the cavity of the foramen caecum lengthens, forming a hollow duct (secondary cavity). Thus the salivary-gland-like ducts come to open into a hollow duct lined by stratified epithelium.
Fig. 7 A Section through the lingual duct of embryo Ez, 31 mm. long. B, branches of the lingual duct; Dl, lingual duct. x 200.
The Position of Thyroglossal Duct Relative to the Hyoid Bone
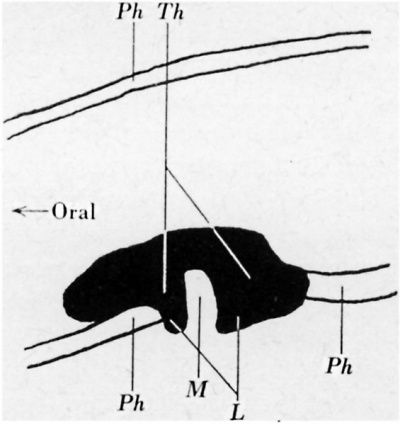
According to my observations the hyoid bone develops dorsal to the thyroglossal duct. Thebone first appears, in embryos of 12-14 mm. length, as a condensation of connective tissue. By this time the thyroglossal duct has already commenced to fragment, but it can be clearly seen that the hyoid bone is limited ventrally by the fragments of the duct. This may be observed in sections in every plane, but especially well in sagittal ones. By the ventral and caudal enlargement of the hyoid bone the cephalic fragments of the duct are displaced ventrally, whilst the caudal ones come to lie behind the hyoid bone. This assumption of His was confirmed by the investigation of a large series of embryos of different stages. Further, the fragments of the thyroglossal duct are often connected with each other ventral to the hyoid bone by cords of connective tissue which represent the sheath formerly enclosing the thyroglossal duct. finally, one of the embryos (Bp, 16 mm. long, fig. 8) retained a complete thyroglossal duct although the hyoid primordium had already appeared. The graphic reconstruction shows clearly that’the duct is displaced ventrally by the hyoid. At the point of greatest displacement the duct is thinnest. In this embryo the hyoid anlage consists of two parts; one is precartilage, the other is still dense connective tissue. Ventrally it can be sharply distinguished from the connective tissue but dorsally it passes gradually into the denser mesenchyme. The examination of the human embryos at my disposal showed that in all of them the position of the fragmented thyroglossal duct corresponds to the relations shown in fig. 8.
Fig. 8. Reconstructed thyroglossal duct of embryo Bp, 16 mm. long; Al, aditus ad laryngem; 0, precartilage part of hyoid anlage; Dtg, thyroglossal duct; E, epidermis; M, connective tissue part of hyoid anlage; Th, thyroid gland.
Discussion
The development of the thyroid gland begins in embryos with 17—18 pairs of somites by the formation of a small pit (figs. 5, 6). in that place where the first pair of pharyngeal pouches meet (Norris represented this stage difi'erently, fig. 4 b, but I think that only cases of double thyroid anlage, in their early stage, correspond to his b, see fig. 9 b2). This pit is lined by a specific epithelium which is the anlage of both thyroid gland and thyroglossal duct. By the lengthening and narrowing of this pit the form represented in fig. 6 (see also fig. 9 d) is produced. Although there is no sharp limit between them the gland anlage and the duct anlage can be easily distinguished from each other.
It appears that the lengthening of the duct is not caused by active growth but by passive extension. This is suggested by the narrowing of the lengthening duct, which loses its lumen in the process, and by the absence of mitoses in its cells at this stage. At some points the cells die and as a result the thyroglossal duct becomes divided into fragments.
Norris stated that the solid thyroglossal duct (fig. 4 g) develops from the solid anlage (c) and that the hollow duct (h) arises from the hollow anlage (d). My findings are contrary to this opinion for all my embryos of less than 6-5 mm. length Had a hollow duct and all the older embryos have solid ducts. Hence I conclude that the solid duct develops from the hollow duct by extension.
I agree with Norris in deriving forms l and m from g, but I do not believe that form n develops directly from form h. I never found a trace of the division of a hollow duct. Further, I observed in many embryos with a solid duct that it was thinnest in its intermediate portion and that there its cells appeared degenerated. It seems, therefore, certain that form 11., as well as forms l and m, develop from g. None of the other forms described by Norris (j, k, 0, p, q) of separation of the thyroid gland from the pharynx were observed in my material. By disregarding them, the scheme of the development of the thyroid gland becomes much simpler according to my findings (fig. 9) than is the one given by Norris (fig. 4).
Fig. 9. Scheme of the development of the thyroid gland and of the various forms of thyroglossal duct rests.
Tourneux & Verdun (1897), having only a few specimens at their disposal, supposed the pyramidal process to appear first in embryos of 26 mm. length. According to them it arises from one of the lateral lobes. This opinion has not been accepted and I could find no evidence for it. Nevertheless, their opinion is understandable for the thyroglossal duct originally joins the gland in the middle line, whilst later the pyramidal process is attached to one of the lateral lobes. But the examination of a large number of embryos of different stages shows clearly the transformation of the duct epithelium into glandular tissue and all stages in the transition from a median to a lateral pyramidal process.
Finally, the embryo Bm, described above, may be considered as a reliable proof against Tourneux & Verdun’s interpretation. In this embryo a thyroglossal duct is preserved throughout its length and shows in its distal portion a glandular transformation and a lateral displacement similar to that of all pyramidal processes at this stage of development.
The rare anomaly of a double pyramidal process is ‘generally explained by assuming a high bifurcation of the thyroid anlage, that is, it is supposed that the growing thyroglossal duct splits at its apex and that each branch gives origin to a lobe of the gland. This view is not supported by the facts that the gland and the duct originate simultaneously, that the gland is not the product of the duct, and that _the duct becomes longer by passive extension only and not by active growth. fig. 25 of Weller (1933) shows a-doubling of the thyroid anlage and Norris presented a similar, and an older, stage in his scheme (fig. 4 e, f). In my opinion such a doubling of the anlage of the thyroid means also a doubling of the thyroglossal duct, for each of the evaginations is gland anlage in its distalportion only, being duct primordium in its proximal part. I consider that from Norris’s stage f a thyroid gland with a double pyramidal process may be easily derived whereas Norris and Weller derive only an early bilobed thyroid gland from it. On my interpretation, the common proximal part of the duct as well as its double distal portion is passively extended and the appearance of bifurcation of the pyramidal process, when it differentiates, results (fig.9; see also Weller, fig. 5). Later the two “branches” of such a bifurcate thyroglossal duct may (ease of Schmidt) or may not (embryo Bw, fig. 9 v) be separated from one another. The examination of the embryo Bw shows further that cords of thyroid cells have grown medially and laterally from each gland primordium. I think it is probable that the medial lobes of each of these glands would later have united to form a secondary isthmus. Thus an isthmus may be found in some cases of double pyramidal process, whilst in others it can be missing (Hilckel’s case).
The fact that the hyoid gland of the guinea-pig, originating from the thyroglossal ‘duct, is slower in its development than the main thyroid gland, and differs from it in structure, led H. Rabl to conclude that the thyroglossal duct develops from a tissue not equivalent to thyroid tissue. He supposed the thyroglossal duct to be oral cavity epithelium subjected to traction by the migrating thyroid gland, and he considered-this epithelium to represent a transition between the thyrogenic and the “indifferent” oral epithelia. On the basis of this observation Rabl supposed that in man the “indifferent” epithelium of the oral cavity also participates in the formation of the upper part of the thyroglossal duct. He considered that the stratified epithelium and the mucous glands derived from the lingual duct supported this view. My observations, however, show that, in the human embryo, the epithelium of the thyroglossal duct in its earlier stages resembles thyrogenic epithelium more than the oral epithelium. The undivided thyroglossal duct is lined by this specific epithelium up to its junction with the tongue. Only after fragmentation of the duct does its proximal portion show a transformation of its epithelium to one similar to the tongue epithelium. It cannot be concluded that the latter is dragged or grows actively into the connective tissue of the tongue, for, after separation of the thyroid gland, there is no fixed point to enable the lingual duct to drag the epithelium and there is no sign of active growth. I conclude, therefore, that the epithelium of the thyroglossal duct is a specific one, but from the point of view of its potentialities, it is intermediate between the thyrogenic and the oral cavity epithelia. Its lower end changes into thyroid tissue and its upper end differentiates to resemble the neighbouring epithelium of the tongue.
The mucous glands of the lingual duct constitute no_proof that the epithelium of the duct is common oral cavity epithelium, for the glandular sprouts arise directly from the specific duct epithelium (fig. 7) and not from stratified epithelium.
In my material no cysts of the lingual duct were found, but one embryo (Ge, 43 mm. in length) showed a separated fragment distal to the lingual duct itself, both being completely lined by stratified epithelium. Branches have grown out from the fragment which is already hollow. It seems probable that after the beginning of secretion by the differentiated mucous glands such a formation becomes a cyst.
The position of the thyroglossal duct relative to the hyoid bone seems to be quite clearly defined by my material. The duct itself is situated in front of the region where later the hyoid bone is formed. Its fragments lie in front of the_hyoid bone and may be included in the bone, in later stages, either as long cords or accessory glands (Streckeisen, Konig).
Difficulties in the way of accepting this interpretation are provided only by the case described by Matti, and by Wegelin’s supposition, based on this case, that the fragments of the divided thyroglossal duct lie infront of the hyoid bone, but that the duct if unfragmented at the period of formation of the hyoid, lies so far dorsally that the bone develops ventral to it. This statement, like others in Wegelin’s book, shows that he dealt with embryological -problems in a purely theoretical manner. Examination of embryos at different stages shows that the thyroglossal duct before fragmentation lies in the same plane as its fragments afterwards. Only after the formation of the hyoid bone has begun are the fragments pushed ventrally by it. Nevertheless Matti’s case exists and cannotbe denied. But even if, considering this case an anomaly, we set aside the findings of normal embryology and do not take into account the embryo Bp, which has an undivided thyroglossal duct running in front of the hyoid bone, Wegelin’s conclusions must still be opposed on the basis of Matti’s own findings. The duct opened into a fistula. This alone shows that the duct was in an abnormal position, being too ventral in its lower portionland, presumably, too dorsal in its upper. The cause of this is unknown but it must be emphasized that in this case there was an anomaly of position as well as the anomaly of persistence of the duct. And a combined anomaly cannot be in an adequate basis for a general conclusion which would assume the second anomaly to be the rule every time the first anomaly occurs.
Summary
- A variation in the thyroid anIage is described in two human embryos. The variation consisted of an invagination of the thyroid anlage into the pharyngeal cavity and it is suggested that, if development had proceeded, the abnormality would have righted itself and developed into a normal thyroid gland.
- The developmental history of the thyroglossal duct, the lingual duct and the pyramidal process of the thyroid gland, is described for a large series of human embryos from the 4 mm to the 65 mm stage.
References
BOCHDALEK, V. (1866). 0st. z. prakt. Heilk. p. 683.
Davis CL. Description of a human embryo having twenty paired somites. (1923) Carnegie Instn. Wash. Publ. 332, Contrib. Embryol., 15: 1-51.
FISCHEL, A. (1929). Lehrbuch der Entwiclclung-des Menschen. Wien u. Berlin: J. Springer.
HAMMAR, J. A. & HELLMAN, T. J. (1920). Z. ges. Anat. 2. Z. K¢.mstLehre, 5, 218.
His, W. (1885). Anatomic memrchlicher Embryonen, 3. A
- (1891). Arch. Anat. Entw.-Gesch. 26.
HucKEL, R. (1932). Die Entwicklungsstorungen der Schilddriise. In Schwalbe-Gruber, Die
Morphologie der Misebildungen dee Menschen und der Tiere. VIII. Teil, 15. Lieferung, 3. Abteilung, p.- 575. Jena: G. fischer.
KALLIUS, E. (1903). Amt. Am. 23, 35.
KAJDMEIER, O. F. (1937). University of Illinois Bulletin, 35, 12.
KANTHACK, A. (1891). J. Anat. Phyetol. 25, 155.
Konig, F. (1896). Arch. Iclin. Chir. 51, 578.
Marshall AM. Thyro-Glossal duct or “canal of His”. (1881) J Anat. Physiol. 16(3): 305–354. PMID 17231961
MARSHALL, C. F. (1892). J. Amt. Physiol. 28, 94.
MATTI, H. (1911). Arch. lclin. Chir. 95, 112.
NORRIS, E. H. (1918). Amer. J. Anat. 24-, 443.
PATZELT, V. (1923). Anat. Anz. 57, 220. A
PoLITZER, G. (1928). Z. yes. Anat. 1. Z. Amt. EntwGesch. 87, 674.
RABL, H. (1923). Anat. Anz. 57, 157. .
SCHMIDT, M. B. (1896). Feswchrift f. B. Schmidt, p. 91. Jena: G. fischer.
SCHWEITZER, R. (1929). Schweiz. med. Wschr. 10, 250.
SGALITZER, K. (1938). Beitr. pathol. Anal. 100, 285.
STRECKEISEN, A. (1886). Virchows Arch. 108, 131.
TOURNEUX, F. & VERDUN, P. (1897). J. amt. phyaiol. p. 305.
WEGELIN, C. (1926). Schilddriise. In Henke-Lubarsch, Hands. d. spez. pathol. Amt. u. Hist. 8.
Weller GL. Development of the thyroid, parathyroid and thymus glands in man. (1933) Contrib. Embryol., Carnegie Inst. Wash. 24: 93-139.
Wen IC. The anatomy of human embryos with seventeen to twenty-three pairs of somites (1928) J. Comp. Neural., 45: 301-376.
Cite this page: Hill, M.A. (2024, April 18) Embryology Paper - Contribution to the study of the morphogenesis of the thyroid gland (1941). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Contribution_to_the_study_of_the_morphogenesis_of_the_thyroid_gland_(1941)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G