Neural Crest Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 11: | Line 11: | ||
Historically identified as "neural crest" by Arthur Marshall in 1879.<ref name=Marshall1879> | Historically identified as "neural crest" by Arthur Marshall in 1879.<ref name=Marshall1879>{{Ref-Marshall1879}}</ref> (see [[#Historic|history below]] and [[Paper - 1879 The Morphology of the Vertebrate Olfactory Organ|original article]]). | ||
| Line 18: | Line 18: | ||
== Some Recent Findings == | == Some Recent Findings == | ||
[[File:Zebrafish_neural_crest_model.jpg|thumb|alt=Zebrafish neura crest model|Zebrafish neura crest model | [[File:Zebrafish_neural_crest_model.jpg|thumb|alt=Zebrafish neura crest model|Zebrafish neura crest model{{#pmid:23028350|PMID23028350}} | ||
{| | {| | ||
| Line 39: | Line 39: | ||
|- | |- | ||
| | | | ||
* '''Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells''' | * '''Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells'''{{#pmid:26934293|PMID26934293}} "Neural crest (NC) cells are a group of cells located in the neural folds at the boundary between the neural and epidermal ectoderm. Cranial NC cells migrate to the branchial arches and give rise to the majority of the craniofacial region, whereas trunk and tail NC cells contribute to the heart, enteric ganglia of the gut, melanocytes, sympathetic ganglia, and adrenal chromaffin cells. ...These BMP4-treated NC cells were capable of differentiation into osteocytes and chondrocytes. The results of the present study indicate that BMP4 regulates cranial positioning during NC development." [[Developmental Signals - Bone Morphogenetic Protein|Bone Morphogenetic Protein]] | ||
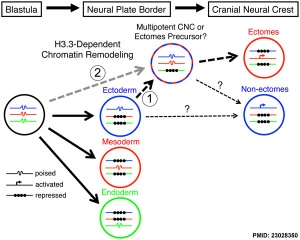
* '''An essential role of variant histone h3.3 for ectomesenchyme potential of the cranial neural crest''' | |||
* '''Dbx1-expressing cells are necessary for the survival of the mammalian anterior neural and craniofacial structures''' | * '''An essential role of variant histone h3.3 for ectomesenchyme potential of the cranial neural crest'''{{#pmid:23028350|PMID23028350}} "The neural crest (NC) is a vertebrate-specific cell population that exhibits remarkable multipotency. Although derived from the neural plate border (NPB) ectoderm, cranial NC (CNC) cells contribute not only to the peripheral nervous system but also to the ectomesenchymal precursors of the head skeleton. ...Surprisingly, embryo-wide expression of dominant mutant H3.3 had little effect on embryonic development outside CNC, indicating an unexpectedly specific sensitivity of CNC to defects in H3.3 incorporation. Whereas previous studies had implicated H3.3 in large-scale histone replacement events that generate totipotency during germ line development, our work has revealed an additional role of H3.3 in the broad potential of the ectoderm-derived CNC, including the ability to make the mesoderm-like ectomesenchymal precursors of the head skeleton." | ||
* '''Analysis of early human neural crest development''' | |||
* '''Cranial neural crest migration: new rules for an old road.''' | * '''Dbx1-expressing cells are necessary for the survival of the mammalian anterior neural and craniofacial structures'''{{#pmid:21552538|PMID21552538}} "Development of the vertebrate forebrain and craniofacial structures are intimately linked processes, the coordinated growth of these tissues being required to ensure normal head formation. In this study, we identify five small subsets of progenitors expressing the transcription factor dbx1 in the cephalic region of developing mouse embryos at E8.5. ... Our results demonstrate that dbx1-expressing cells have a unique function during head development, notably by controlling cell survival in a non cell-autonomous manner." | ||
* '''Derivation of neural crest cells from human pluripotent stem cells.''' | |||
* '''Analysis of early human neural crest development'''{{#pmid:20478300|PMID20478300}} "The outstanding migration and differentiation capacities of neural crest cells (NCCs) have fascinated scientists since Wilhelm His described this cell population in 1868. Today, after intense research using vertebrate model organisms, we have gained considerable knowledge regarding the origin, migration and differentiation of NCCs. However, our understanding of NCC development in human embryos remains largely uncharacterized, despite the role the neural crest plays in several human pathologies. Here, we report for the first time the expression of a battery of molecular markers before, during, or following NCC migration in human embryos from Carnegie Stages (CS) 12 to 18. Our work demonstrates the expression of Sox9, Sox10 and Pax3 transcription factors in premigratory NCCs, while actively migrating NCCs display the additional transcription factors Pax7 and AP-2alpha. Importantly, while HNK-1 labels few migrating NCCs, p75(NTR) labels a large proportion of this population. However, the broad expression of p75(NTR) - and other markers - beyond the neural crest stresses the need for the identification of additional markers to improve our capacity to investigate human NCC development, and to enable the generation of better diagnostic and therapeutic tools." | |||
* '''Cranial neural crest migration: new rules for an old road.'''{{#pmid:20399765|PMID20399765}} "In this review, we discuss recent cellular and molecular discoveries of the CNCC migratory pattern. We focus on events from the time when CNCCs encounter the tissue adjacent to the neural tube and their travel through different microenvironments and into the branchial arches. We describe the patterning of discrete cell migratory streams that emerge from the hindbrain, rhombomere (r) segments r1-r7, and the signals that coordinate directed migration." | |||
* '''Derivation of neural crest cells from human pluripotent stem cells.'''{{#pmid:20360764|PMID20360764}} "Here we provide protocols for the step-wise differentiation of human embryonic stem cells (hESCs) or human induced pluripotent stem cells (hiPSCs) into neuroectodermal and NC cells using either the MS5 coculture system or a novel defined culture method based on pharmacological inhibition of bone morphogenetic protein and transforming growth factor-beta signaling pathways." (More? [[Stem Cells]]) | |||
|} | |} | ||
[[File:Neural crest formation stages 01.jpg|800px]] | [[File:Neural crest formation stages 01.jpg|800px]] | ||
Neural crest formation stages and gene regulatory networks | Neural crest formation stages and gene regulatory networks.{{#pmid:25903629|PMID25903629}} | ||
{{Neural Crest table}} | {{Neural Crest table}} | ||
| Line 56: | Line 61: | ||
{| | {| | ||
| width=505px|[[File:Human neural crest cell migration-in vitro.jpg|500px|alt=Human neural crest cell migration-in vitro]] | | width=505px|[[File:Human neural crest cell migration-in vitro.jpg|500px|alt=Human neural crest cell migration-in vitro]] | ||
| valign=top|Human neural crest cell migration (in vitro) | | valign=top|Human neural crest cell migration (in vitro){{#pmid:18689800|PMID18689800}} | ||
* '''A''' Human neural tube (NT) from an embryo at [[Carnegie stage 13]]. | * '''A''' Human neural tube (NT) from an embryo at [[Carnegie stage 13]]. | ||
* '''B''' After 16 h, most hNCC have migrated away from the dorsal NT. | * '''B''' After 16 h, most hNCC have migrated away from the dorsal NT. | ||
| Line 147: | Line 152: | ||
|} | |} | ||
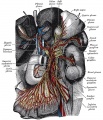
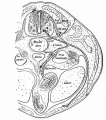
Carotid body are chemoreceptors in the wall of the common carotid (3rd pharyngeal arch) | Carotid body are chemoreceptors in the wall of the common carotid (3rd pharyngeal arch) {{#pmid:8400021|PMID8400021}}{{#pmid:22902512|PMID22902512}} | ||
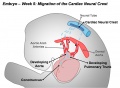
===Cardiac neural crest=== | ===Cardiac neural crest=== | ||
| Line 169: | Line 174: | ||
====para-aortic body==== | ====para-aortic body==== | ||
(organ of Zuckerkandl, OZ) A neural crest derived chromaffin body, anatomically located at the bifurcation of the aorta or at the origin of the inferior mesenteric artery. Thought to act as a fetal regulator of blood pressure, secreting catecholamines into the fetal circulation. | (organ of Zuckerkandl, OZ) A neural crest derived chromaffin body, anatomically located at the bifurcation of the aorta or at the origin of the inferior mesenteric artery. Thought to act as a fetal regulator of blood pressure, secreting catecholamines into the fetal circulation.{{#pmid:13107111|PMID13107111}} In human, reaches its maximal size at 3 years of age and then regresses either by death, dispersion or differentiation.<ref name="PMID23078542"><pubmed>23078542</pubmed></ref> | ||
Named in 1901 by Emil Zuckerkandl (1849-1910) a Hungarian-Austrian anatomist at the University of Vienna. | Named in 1901 by Emil Zuckerkandl (1849-1910) a Hungarian-Austrian anatomist at the University of Vienna. | ||
| Line 179: | Line 184: | ||
== Development Overview == | == Development Overview == | ||
The following cranial and trunk data is based upon 185 serially sectioned staged (Carnegie) human embryos. | The following cranial and trunk data is based upon 185 serially sectioned staged (Carnegie) human embryos.{{#pmid:17848161|PMID17848161}} | ||
===Cranial Neural Crest=== | ===Cranial Neural Crest=== | ||
| Line 202: | Line 207: | ||
===Neck and Shoulder=== | ===Neck and Shoulder=== | ||
A mouse study using individually labelled cells of postotic neural crest followed the development of the shoulder girdle (clavicle and scapula) that connects the upper limb to the axial skeleton. | A mouse study using individually labelled cells of postotic neural crest followed the development of the shoulder girdle (clavicle and scapula) that connects the upper limb to the axial skeleton.{{#pmid:16034409|PMID16034409}} | ||
* Clavicle is a neural crest-mesodermal structure, posterior dermal clavicle mesoderm. | * Clavicle is a neural crest-mesodermal structure, posterior dermal clavicle mesoderm. | ||
| Line 216: | Line 221: | ||
| [[File:Mouse-melanoblast migration icon.jpg|200px|link=Quicktime Movie_-_Mouse_Melanoblast_Migration]] | | [[File:Mouse-melanoblast migration icon.jpg|200px|link=Quicktime Movie_-_Mouse_Melanoblast_Migration]] | ||
|- | |- | ||
| Mouse melanocyte migration | | Mouse melanocyte migration{{#pmid:16034409|PMID16277556}} | ||
| Movie Mouse Skin - Melanoblast Migration E14.5 | | Movie Mouse Skin - Melanoblast Migration E14.5{{#pmid:20067551|PMID20067551}} | ||
|} | |} | ||
| Line 227: | Line 232: | ||
===Inhibitors=== | ===Inhibitors=== | ||
* '''versican''' - (VCAN, Chondroitin Sulfate Proteoglycan 2; Cspg2) an extracellular matrix proteoglycan that acts as both an inhibitor of NCC migration and as a guiding cue by forming exclusionary boundaries. | * '''versican''' - (VCAN, Chondroitin Sulfate Proteoglycan 2; Cspg2) an extracellular matrix proteoglycan that acts as both an inhibitor of NCC migration and as a guiding cue by forming exclusionary boundaries.{{#pmid:27241911|PMID27241911}} | ||
| Line 233: | Line 238: | ||
==Historic== | ==Historic== | ||
The paper by Marshall, Morphology of the Vertebrate Olfactory Organ (1879)<ref name=Marshall1879 | The paper by Marshall, Morphology of the Vertebrate Olfactory Organ (1879)<ref name=Marshall1879 />, was historically the first time the term "''neural crest''" was used. In his own earlier papers he had referred to this as a "neural ridge" in describing development of the chicken embryo neural tube. | ||
See [[Paper - 1879 The Morphology of the Vertebrate Olfactory Organ#NeuralCrest|paper text]] and his referenced comment: | See [[Paper - 1879 The Morphology of the Vertebrate Olfactory Organ#NeuralCrest|paper text]] and his referenced comment: | ||
Revision as of 10:40, 15 February 2018
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
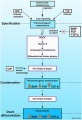
Introduction
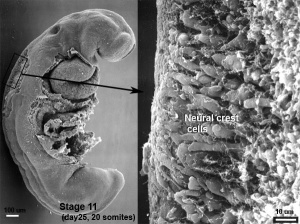
The neural crest are bilaterally paired strips of cells arising in the ectoderm at the margins of the neural tube. These cells migrate to many different locations and differentiate into many cell types within the embryo. This means that many different systems (neural, skin, teeth, head, face, heart, adrenal glands, gastrointestinal tract) will also have a contribution fron the neural crest cells. An in vitro study[1] has shown neural crest cell migration occurs at different rates along the embryo axis between Carnegie stage 11 to 13 in week 4.
In the body region, neural crest cells also contribute the peripheral nervous system (both neurons and glia) consisting of sensory ganglia (dorsal root ganglia), sympathetic and parasympathetic ganglia and neural plexuses within specific tissues/organs.
In the head region, neural crest cells migrate into the pharyngeal arches (as shown in movie below) forming ectomesenchyme contributing tissues which in the body region are typically derived from mesoderm (cartilage, bone, and connective tissue). General neural development is also covered in Neural Notes.
Historically identified as "neural crest" by Arthur Marshall in 1879.[2] (see history below and original article).
| Neural Crest Links: neural crest | Lecture - Early Neural | Lecture - Neural Crest Development | Lecture Movie | Schwann cell | adrenal | melanocyte | peripheral nervous system | enteric nervous system | cornea | cranial nerve neural crest | head | skull | cardiac neural crest | Nicole Le Douarin | Neural Crest Movies | neural crest abnormalities | Category:Neural Crest | |||
|
Some Recent Findings
 Moore, K.L. & Persuad, T.V.N. (2008). The Developing Human: clinically oriented embryology (8th ed.). Philadelphia: Saunders. (chapter links only work with a UNSW connection). * Nervous System * Pharyngeal Apparatus, Face, and Neck
Moore, K.L. & Persuad, T.V.N. (2008). The Developing Human: clinically oriented embryology (8th ed.). Philadelphia: Saunders. (chapter links only work with a UNSW connection). * Nervous System * Pharyngeal Apparatus, Face, and Neck  Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R. and Francis-West, P.H. (2009). Larsen’s Human Embryology (4th ed.). New York; Edinburgh: Churchill Livingstone. The following chapter links only work with a UNSW Library subscription * Chapter 10 - Development of the Peripheral Nervous System * Chapter 16 - Development of the Pharyngeal Apparatus and Face Additional Resources * Developmental Biology. 6th edition. The Neural Crest * Nelms BL, Labosky PA. Transcriptional Control of Neural Crest Development. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Available from: http://www.ncbi.nlm.nih.gov/books/NBK53145/ * Dupin E, Creuzet S, Le Douarin NM. The Contribution of the Neural Crest to the Vertebrate Body. In: Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000-. Available from: http://www.ncbi.nlm.nih.gov/books/NBK6098/
Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R. and Francis-West, P.H. (2009). Larsen’s Human Embryology (4th ed.). New York; Edinburgh: Churchill Livingstone. The following chapter links only work with a UNSW Library subscription * Chapter 10 - Development of the Peripheral Nervous System * Chapter 16 - Development of the Pharyngeal Apparatus and Face Additional Resources * Developmental Biology. 6th edition. The Neural Crest * Nelms BL, Labosky PA. Transcriptional Control of Neural Crest Development. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Available from: http://www.ncbi.nlm.nih.gov/books/NBK53145/ * Dupin E, Creuzet S, Le Douarin NM. The Contribution of the Neural Crest to the Vertebrate Body. In: Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000-. Available from: http://www.ncbi.nlm.nih.gov/books/NBK6098/ Objectives
* Understand the structures derived from ectoderm. * Understand the formation of neural folds. * Identify the initial location of neural crest cells in the trilaminar embryo. * Identify pathways of neural crest migration throughout the embryo. * To know the major tissues to which neural crest cells contribute. * To know how abnormalities in development that result from abnormal neural crest cell migration. * Understand how neural crest cells contribute to the pharyngeal arches and the head structures they form.Neural Crest Derivatives
A key feature of neural crest is the migration into other embryonic tissues to form specific neural and non-neural populations and structures.Cranial neural crest
* migration - dorsolaterally and into pharyngeal arches * craniofacial mesenchyme - cartilage, bone, cranial neurons, glia, and connective tissues of the face * pharyngeal arches and pouches - thymic cells, tooth odontoblasts, middle ear bones (ossicles), stria vascularis cells, and jaw (mandible)| Cochlea - Stria Vascularis | |
|---|---|
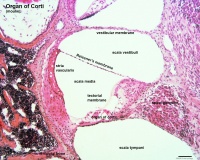 | 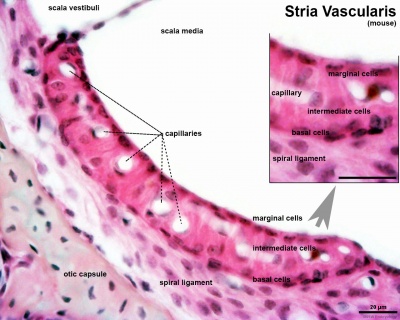 |
| Inner ear cochlea, showing the stria vascularis intermediate cells that are derived from neural crest. Hearing - Inner Ear Development | |
| Eye - Cornea | |
|---|---|
 | 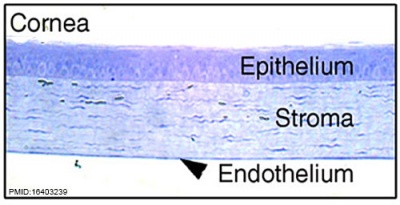 |
| Human embryonic cornea detail (Week 8, Carnegie stage 22) | Mouse cornea layers |
| The adult eye cornea has three layers: an outer epithelium layer (ectoderm), a middle stromal layer of collagen-rich extracellular matrix between stromal keratocytes (neural crest) and an inner layer of endothelial cells (neural crest) | |
Carotid body are chemoreceptors in the wall of the common carotid (3rd pharyngeal arch) [14][15]
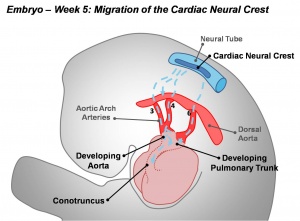
Cardiac neural crest
Trunk neural crest
- migration - two major pathways over somites (dorsolaterally) and between somite and neural tube (ventrolaterally)
- dorsolateral - skin melanocytes
- ventrolaterally - dorsal root ganglia, sympathetic ganglia, adrenal medulla, aortic nerve clusters
para-aortic body
(organ of Zuckerkandl, OZ) A neural crest derived chromaffin body, anatomically located at the bifurcation of the aorta or at the origin of the inferior mesenteric artery. Thought to act as a fetal regulator of blood pressure, secreting catecholamines into the fetal circulation.[16] In human, reaches its maximal size at 3 years of age and then regresses either by death, dispersion or differentiation.[17]
Named in 1901 by Emil Zuckerkandl (1849-1910) a Hungarian-Austrian anatomist at the University of Vienna.
Vagal and sacral neural crest
- migration - ventrally into surrounding splanchnic mesenchyme of gastrointestinal tract
- splanchnic mesenchyme - parasympathetic (enteric) ganglia of the gut
Development Overview
The following cranial and trunk data is based upon 185 serially sectioned staged (Carnegie) human embryos.[18]
Cranial Neural Crest
- stage 9 - an indication of mesencephalic neural crest
- stage 10 - trigeminal, facial, and postotic components
- stage 11 - crest-free zones are soon observable in rhombomere 1, 3, and 5
- stage 12 - rhombomeres 6 and 7 neural crest migrate to pharyngeal arch 3 and then rostrad to the truncus arteriosus
- stage 13 - nasal crest and the terminalis-vomeronasal complex are last of the cranial crest to appear
stages 9-14 - otic vesicle primordium descends
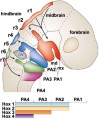
Vagal Neural Crest
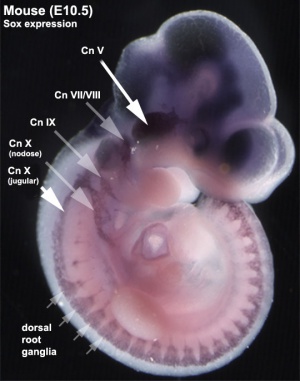
Recent research suggests that the vagal neural crest cells are a transitional population that has evolved between the head and the trunk, taking separate pathways to the both the heart and to the gut.[19][20]
Trunk Neural Crest
Spinal ganglia increase in number over time and are in phase with the somites, though not their centre. There are 3 migratory pathways: ventrolateral between dermatomyotome and sclerotome, ventromedial between neural tube and sclerotomes, and lateral between surface ectoderm and dermatomyotome.
- stage 13 - about 19 present
- stage 14 - about 33 present
- stage 15-23 - 30–35 ganglia
Neck and Shoulder
A mouse study using individually labelled cells of postotic neural crest followed the development of the shoulder girdle (clavicle and scapula) that connects the upper limb to the axial skeleton.[21]
- Clavicle is a neural crest-mesodermal structure, posterior dermal clavicle mesoderm.
- Cryptic cell boundaries traverse apparently homogeneous skeleton of the neck and shoulders.
- Bones and muscles code of connectivity that mesenchymal stem cells of both neural crest and mesodermal origin obey
- Neural crest anchors the head onto the anterior lining of the shoulder girdle
- Hox-gene-controlled mesoderm links trunk muscles to the posterior neck and shoulder skeleton.
- Skeleton identified as neural crest-derived is affected in human Klippel-Feil syndrome, Sprengel's deformity and Arnold-Chiari I/II malformation.
Skin Melanocytes
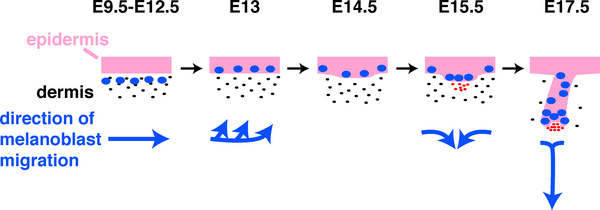
|
|
| Mouse melanocyte migration[22] | Movie Mouse Skin - Melanoblast Migration E14.5[23] |
Neural Crest Migration
A key event in neural crest development is migration from the original site that neural crest cells are generated (edge of the neural plate) to the different anatomical regions within the embryo.
Stimulators
- complement component C3a - (C3a) acts as an autocrine diffusible chemotactic agent attracting NCC toward the self-secreted source.
Inhibitors
- versican - (VCAN, Chondroitin Sulfate Proteoglycan 2; Cspg2) an extracellular matrix proteoglycan that acts as both an inhibitor of NCC migration and as a guiding cue by forming exclusionary boundaries.[24]
- Links: OMIM 118661
Historic
The paper by Marshall, Morphology of the Vertebrate Olfactory Organ (1879)[2], was historically the first time the term "neural crest" was used. In his own earlier papers he had referred to this as a "neural ridge" in describing development of the chicken embryo neural tube.
See paper text and his referenced comment:
- "I take this opportunity to make a slight alteration in the nomenclature adopted in my former paper. I have there suggested the term neural ridge for the longitudinal ridge of cells which grows out from the reentering angle between the external epiblast and the neural canal, and from which the nerves, whether cranial or spinal, arise. Since this ridge appears before closure of the neural canal is effected, there are manifestly two neural ridges, one on either side ; but I have also applied the same term, neural ridge, to the single outgrowth formed by the fusion of the neural ridges of the two sides after complete closure of the neural canal is effected, and after the external epiblast has become completely separated from the neural canal. I propose in future to speak of this single median outgrowth as the neural crest, limiting the term neural ridge to the former acceptation. Thus, while there are two neural ridges, there is only one neural crest, a distinction that will be at once evident on reference to my former figures."
- Links: Embryology History
References
- ↑ 1.0 1.1 <pubmed>18689800</pubmed> Cite error: Invalid
<ref>tag; name 'PMID18689800' defined multiple times with different content - ↑ 2.0 2.1 Marshall AM. The morphology of the vertebrate olfactory organ. (1879) Quarterly Journal of Microscopic Science. 19: 300–340.
- ↑ 3.0 3.1 Cox SG, Kim H, Garnett AT, Medeiros DM, An W & Crump JG. (2012). An essential role of variant histone H3.3 for ectomesenchyme potential of the cranial neural crest. PLoS Genet. , 8, e1002938. PMID: 23028350 DOI.
- ↑ <pubmed>29108781</pubmed>
- ↑ Mimura S, Suga M, Okada K, Kinehara M, Nikawa H & Furue MK. (2016). Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells. Int. J. Dev. Biol. , 60, 21-8. PMID: 26934293 DOI.
- ↑ Causeret F, Ensini M, Teissier A, Kessaris N, Richardson WD, Lucas de Couville T & Pierani A. (2011). Dbx1-expressing cells are necessary for the survival of the mammalian anterior neural and craniofacial structures. PLoS ONE , 6, e19367. PMID: 21552538 DOI.
- ↑ Betters E, Liu Y, Kjaeldgaard A, Sundström E & García-Castro MI. (2010). Analysis of early human neural crest development. Dev. Biol. , 344, 578-92. PMID: 20478300 DOI.
- ↑ Kulesa PM, Bailey CM, Kasemeier-Kulesa JC & McLennan R. (2010). Cranial neural crest migration: new rules for an old road. Dev. Biol. , 344, 543-54. PMID: 20399765 DOI.
- ↑ Lee G, Chambers SM, Tomishima MJ & Studer L. (2010). Derivation of neural crest cells from human pluripotent stem cells. Nat Protoc , 5, 688-701. PMID: 20360764 DOI.
- ↑ Green SA, Simoes-Costa M & Bronner ME. (2015). Evolution of vertebrates as viewed from the crest. Nature , 520, 474-482. PMID: 25903629 DOI.
- ↑ Woodhoo A & Sommer L. (2008). Development of the Schwann cell lineage: from the neural crest to the myelinated nerve. Glia , 56, 1481-90. PMID: 18803317 DOI.
- ↑ Barraud P, Seferiadis AA, Tyson LD, Zwart MF, Szabo-Rogers HL, Ruhrberg C, Liu KJ & Baker CV. (2010). Neural crest origin of olfactory ensheathing glia. Proc. Natl. Acad. Sci. U.S.A. , 107, 21040-5. PMID: 21078992 DOI.
- ↑ <pubmed>10683170</pubmed>
- ↑ Smith P, Scraggs M & Heath D. (1993). The development of the nerve network in the fetal human carotid body and its subsequent function in cardiac disease. Cardioscience , 4, 143-9. PMID: 8400021
- ↑ Hempleman SC & Warburton SJ. (2013). Comparative embryology of the carotid body. Respir Physiol Neurobiol , 185, 3-8. PMID: 22902512 DOI.
- ↑ WEST GB, SHEPHERD DM, HUNTER RB & MACGREGOR AR. (1953). The function of the organs of Zuckerkandl. Clin Sci , 12, 317-25. PMID: 13107111
- ↑ <pubmed>23078542</pubmed>
- ↑ O'Rahilly R & Müller F. (2007). The development of the neural crest in the human. J. Anat. , 211, 335-51. PMID: 17848161 DOI.
- ↑ <pubmed>20962585</pubmed>
- ↑ Bryan R. Kuo, Carol A. Erickson Vagal neural crest cell migratory behavior: A transition between the cranial and trunk crest. Volume 240, Issue 9, pages 2084–2100, September 2011 Dev Dynamics
- ↑ Matsuoka T, Ahlberg PE, Kessaris N, Iannarelli P, Dennehy U, Richardson WD, McMahon AP & Koentges G. (2005). Neural crest origins of the neck and shoulder. Nature , 436, 347-55. PMID: 16034409 DOI.
- ↑ Matsuoka T, Ahlberg PE, Kessaris N, Iannarelli P, Dennehy U, Richardson WD, McMahon AP & Koentges G. (2005). Neural crest origins of the neck and shoulder. Nature , 436, 347-55. PMID: 16034409 DOI.
- ↑ Mort RL, Hay L & Jackson IJ. (2010). Ex vivo live imaging of melanoblast migration in embryonic mouse skin. Pigment Cell Melanoma Res , 23, 299-301. PMID: 20067551 DOI.
- ↑ Szabó A, Melchionda M, Nastasi G, Woods ML, Campo S, Perris R & Mayor R. (2016). In vivo confinement promotes collective migration of neural crest cells. J. Cell Biol. , 213, 543-55. PMID: 27241911 DOI.
Books
Trainor, P. (ed) Neural crest cells: evolution, development and disease. ISBN: 978-0-12-401730-6 ScienceDirect Nelms BL, Labosky PA. Transcriptional Control of Neural Crest Development. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. PMID 21452438
Reviews
Bronner ME & Simões-Costa M. (2016). The Neural Crest Migrating into the Twenty-First Century. Curr. Top. Dev. Biol. , 116, 115-34. PMID: 26970616 DOI.
Green SA, Simoes-Costa M & Bronner ME. (2015). Evolution of vertebrates as viewed from the crest. Nature , 520, 474-482. PMID: 25903629 DOI.
Lee YH & Saint-Jeannet JP. (2011). Sox9 function in craniofacial development and disease. Genesis , 49, 200-8. PMID: 21309066 DOI.
Kish PE, Bohnsack BL, Gallina D, Kasprick DS & Kahana A. (2011). The eye as an organizer of craniofacial development. Genesis , 49, 222-30. PMID: 21309065 DOI.
Jiang M, Stanke J & Lahti JM. (2011). The connections between neural crest development and neuroblastoma. Curr. Top. Dev. Biol. , 94, 77-127. PMID: 21295685 DOI.
PubmedParser error: The PubmedParser extension received invalid XML data. ()
Articles
O'Rahilly R & Müller F. (2007). The development of the neural crest in the human. J. Anat. , 211, 335-51. PMID: 17848161 DOI.
Search PubMed
Search Pubmed: Neural Crest Development
NCBI - Policies and Guidelines | PubMed | Help:Reference Tutorial
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Neural Crest Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_Crest_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G