Neural - Telencephalon Development
Introduction
Neural development is one of the earliest systems to begin and the last to be completed after birth. This development generates the most complex structure within the embryo and the long time period of development means in utero insult during pregnancy may have consequences to development of the nervous system.
The early central nervous system begins as a simple neural plate that folds to form a groove then tube, open initially at each end. Failure of these opening to close contributes a major class of neural abnormalities (neural tube defects).
Within the neural tube stem cells generate the 2 major classes of cells that make the majority of the nervous system : neurons and glia. Both these classes of cells differentiate into many different types generated with highly specialized functions and shapes. This section covers the establishment of neural populations, the inductive influences of surrounding tissues and the sequential generation of neurons establishing the layered structure seen in the brain and spinal cord.
- Neural development beginnings quite early, therefore also look at notes covering Week 3- neural tube and Week 4-early nervous system.
- Development of the neural crest and sensory systems (hearing/vision/smell) are only introduced in these notes and are covered in other notes sections.
Some Recent Findings
|
Development Overview
Neuralation begins at the trilaminar embryo with formation of the notochord and somites, both of which underly the ectoderm and do not contribute to the nervous system, but are involved with patterning its initial formation. The central portion of the ectoderm then forms the neural plate that folds to form the neural tube, that will eventually form the entire central nervous system.
- Early developmental sequence: Epiblast - Ectoderm - Neural Plate - Neural groove and Neural Crest - Neural Tube and Neural Crest
| Neural Tube | Primary Vesicles | Secondary Vesicles | Adult Structures |
|---|---|---|---|
| week 3 | week 4 | week 5 | adult |
| prosencephalon (forebrain) | telencephalon | Rhinencephalon, Amygdala, hippocampus, cerebrum (cortex), hypothalamus, pituitary | Basal Ganglia, lateral ventricles | |
| diencephalon | epithalamus, thalamus, Subthalamus, pineal, posterior commissure, pretectum, third ventricle | ||
| mesencephalon (midbrain) | mesencephalon | tectum, Cerebral peduncle, cerebral aqueduct, pons | |
| rhombencephalon (hindbrain) | metencephalon | cerebellum | |
| myelencephalon | medulla oblongata, isthmus | ||
| spinal cord, pyramidal decussation, central canal | |||
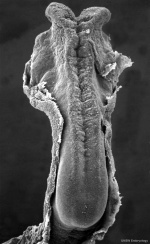
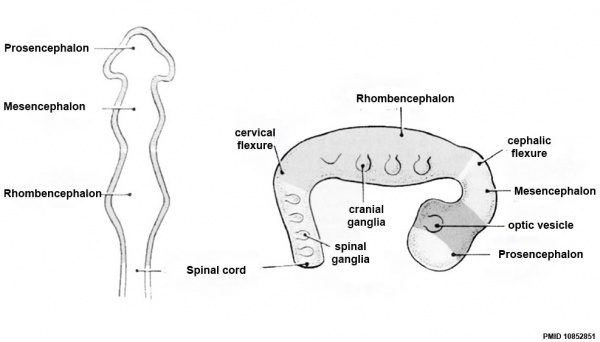
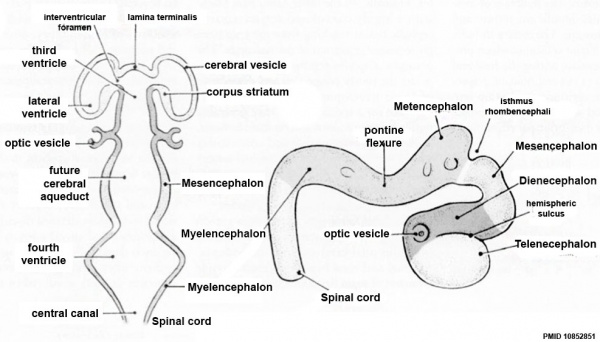
Early Brain Vesicles
Primary Vesicles
Secondary Vesicles
Molecular Development
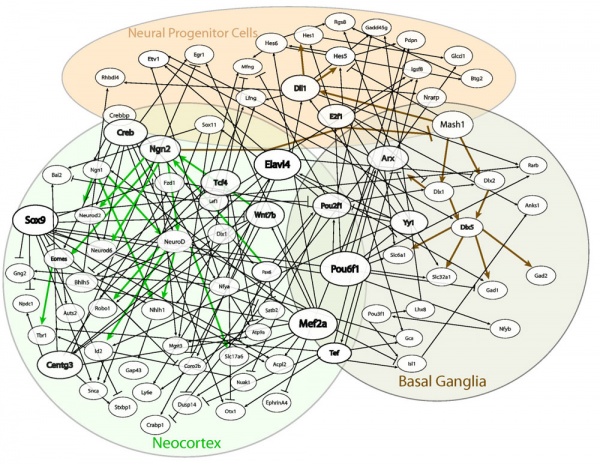
Regulatory Networks
Algorithm-based gene regulatory network structure for dorsal and ventral telencephalon development.[3]
To highlight the key regulators, the nodes representing genes predicted to be the parent of at least nine other genes are largest in size (Sox9, Mef2a, Elavl4 and Pou6f1), whereas those that are predicted to regulate at least five other genes are medium in size (Ngn2, Centg3, Tef, Tcf4, Wnt7b, Pou2f1, Yy1, Dll1, E2f1, Arx, and Creb).
Netrin-1 Signaling
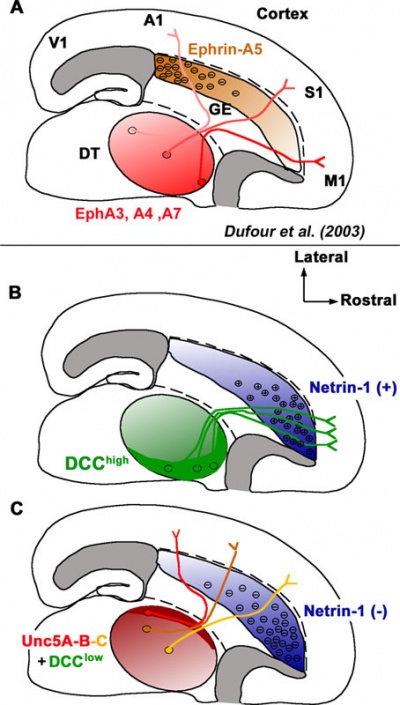
|
Model of the Role of Netrin-1 Signaling in the Topography of Thalamocortical Projections in the Ventral Telencephalon</ref><pubmed>18479186</pubmed>| PMC2584572 |
PLoS Biol.</ref>
ReferencesReviews<pubmed>19210541</pubmed> <pubmed>19143049</pubmed>
Articles<pubmed>19632216</pubmed> <pubmed>18997789</pubmed> <pubmed>18682721</pubmed> <pubmed>17959802</pubmed> Search PubMedSearch Pubmed: Telencephalon Embryology | Telencephalon Development |
Glossary Links
Cite this page: Hill, M.A. (2024, April 25) Embryology Neural - Telencephalon Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Telencephalon_Development
|