Neural - Tectum Development
| Embryology - 19 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Neural development is one of the earliest systems to begin and the last to be completed after birth. This development generates the most complex structure within the embryo and the long time period of development means in utero insult during pregnancy may have consequences to development of the nervous system.
Differences between birds and mammals:
- both - have retinal axons projecting topographically to targets in the brain.
- birds - the visual fibers from the entire retina decussate at the optic chiasm.
- mammals - some axons from the temporal retina diverge at the midline to project ipsilaterally.
Neural development beginnings quite early, therefore also look at notes covering Week 3- neural tube and Week 4-early nervous system. Development of the neural crest and sensory systems (hearing/vision/smell) are only introduced in these notes and are covered in other notes sections.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Tectum Embryology <pubmed limit=5>Tectum Embryology</pubmed> |
| Older papers |
|---|
|
Developmental Signaling Model
Model of interaction of Meis2, Pax3 and Pax7 during dorsal midbrain development. [3]
Development Overview
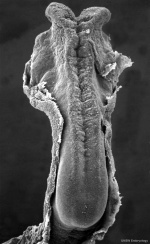
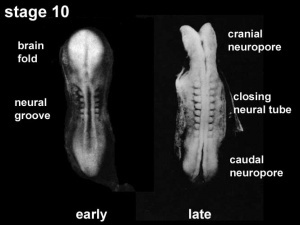
Neuralation begins at the trilaminar embryo with formation of the notochord and somites, both of which underly the ectoderm and do not contribute to the nervous system, but are involved with patterning its initial formation. The central portion of the ectoderm then forms the neural plate that folds to form the neural tube, that will eventually form the entire central nervous system.
- Early developmental sequence: Epiblast - Ectoderm - Neural Plate - Neural groove and Neural Crest - Neural Tube and Neural Crest
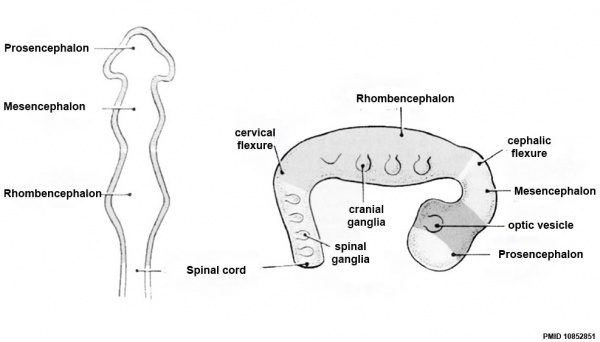
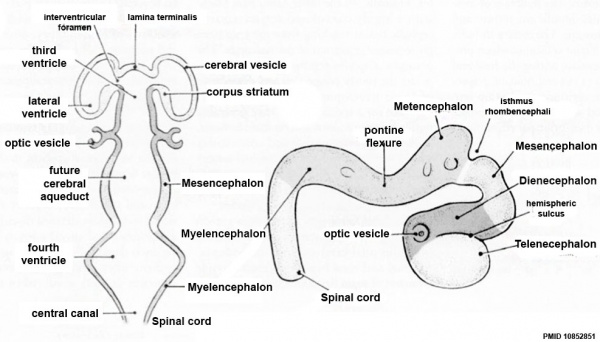
| Neural Tube | Primary Vesicles | Secondary Vesicles | Adult Structures |
|---|---|---|---|
| week 3 | week 4 | week 5 | adult |
| prosencephalon (forebrain) | telencephalon | Rhinencephalon, Amygdala, hippocampus, cerebrum (cortex), hypothalamus, pituitary | Basal Ganglia, lateral ventricles | |
| diencephalon | epithalamus, thalamus, Subthalamus, pineal, posterior commissure, pretectum, third ventricle | ||
| mesencephalon (midbrain) | mesencephalon | tectum, Cerebral peduncle, cerebral aqueduct, pons | |
| rhombencephalon (hindbrain) | metencephalon | cerebellum | |
| myelencephalon | medulla oblongata, isthmus | ||
| spinal cord, pyramidal decussation, central canal | |||
Early Brain Vesicles
Primary Vesicles
Secondary Vesicles
Molecular
Pax
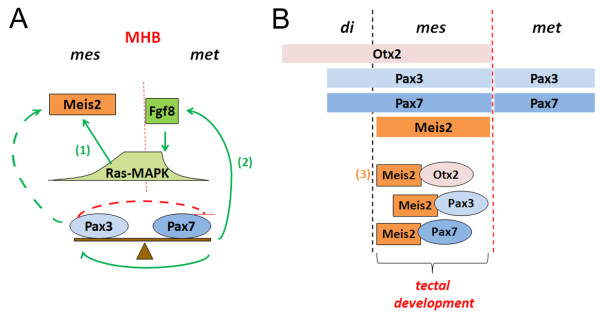
Model of molecular interactions during dorsal midbrain development[3] |
Model for a possible cooperation of Meis2, Pax3, Pax7 and Otx2 during tectal development.
Red lines indicate negative regulation, green arrows positive regulation. Dashed lines indicate hypothetical direct regulation of the Meis2 promoter/enhancer by different Pax3 concentrations. Solid lines indicate indirect regulation of Meis2 expression via Pax3/7 mediated induction of Fgf8 as previously reported: |
Drebrin
Optic tectum (chicken) actin-binding protein, with two major isoforms (A, E) produced by alternative splicing from a single DBN1 gene, isoform conversion occurs in parallel with synaptogenesis.[5]
- drebrin E - (5q35.3) widespread but not ubiquitous cell types in various tissues.
- drebrin A - adult brain neuron-specific, concentrated in dendritic spines, and its accumulation level is regulated by synaptic activity.
- OMIM: DBN1 drebrin E
References
- ↑ Watanabe Y, Sakuma C & Yaginuma H. (2018). Dispersing movement of tangential neuronal migration in superficial layers of the developing chick optic tectum. Dev. Biol. , 437, 131-139. PMID: 29548944 DOI.
- ↑ Yang C, Li X, Li Q, Li H, Qiao L, Guo Z & Lin J. (2018). Sonic Hedgehog Regulation of the Neural Precursor Cell Fate During Chicken Optic Tectum Development. J. Mol. Neurosci. , 64, 287-299. PMID: 29285739 DOI.
- ↑ 3.0 3.1 3.2 Agoston Z, Li N, Haslinger A, Wizenmann A & Schulte D. (2012). Genetic and physical interaction of Meis2, Pax3 and Pax7 during dorsal midbrain development. BMC Dev. Biol. , 12, 10. PMID: 22390724 DOI.
- ↑ Pyrgaki C, Trainor P, Hadjantonakis AK & Niswander L. (2010). Dynamic imaging of mammalian neural tube closure. Dev. Biol. , 344, 941-7. PMID: 20558153 DOI.
- ↑ Shirao T & Sekino Y. (2017). General Introduction to Drebrin. Adv. Exp. Med. Biol. , 1006, 3-22. PMID: 28865011 DOI.
Reviews
Takahashi M & Shinoda Y. (2018). Brain Stem Neural Circuits of Horizontal and Vertical Saccade Systems and their Frame of Reference. Neuroscience , , . PMID: 30193861 DOI.
Marachlian E, Avitan L, Goodhill GJ & Sumbre G. (2018). Principles of Functional Circuit Connectivity: Insights From Spontaneous Activity in the Zebrafish Optic Tectum. Front Neural Circuits , 12, 46. PMID: 29977193 DOI.
Avitan L & Goodhill GJ. (2018). Code Under Construction: Neural Coding Over Development. Trends Neurosci. , 41, 599-609. PMID: 29935867 DOI.
Joly JS, Recher G, Brombin A, Ngo K & Hartenstein V. (2016). A Conserved Developmental Mechanism Builds Complex Visual Systems in Insects and Vertebrates. Curr. Biol. , 26, R1001-R1009. PMID: 27780043 DOI.
Zhaoping L. (2016). From the optic tectum to the primary visual cortex: migration through evolution of the saliency map for exogenous attentional guidance. Curr. Opin. Neurobiol. , 40, 94-102. PMID: 27420378 DOI.
Greene ND & Copp AJ. (2009). Development of the vertebrate central nervous system: formation of the neural tube. Prenat. Diagn. , 29, 303-11. PMID: 19206138 DOI.
Articles
Triplett MA, Avitan L & Goodhill GJ. (2018). Emergence of spontaneous assembly activity in developing neural networks without afferent input. PLoS Comput. Biol. , 14, e1006421. PMID: 30265665 DOI.
Lischka K, Yan J, Weigel S & Luksch H. (2018). Effects of early eye removal on the morphology of a multisensory neuron in the chicken optic tectum. Brain Res. , 1691, 9-14. PMID: 29680273 DOI.
Saitsu H & Shiota K. (2008). Involvement of the axially condensed tail bud mesenchyme in normal and abnormal human posterior neural tube development. Congenit Anom (Kyoto) , 48, 1-6. PMID: 18230116 DOI.
Search PubMed
Search Pubmed: Tectum Embryology | Tectum Development
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 19) Embryology Neural - Tectum Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Tectum_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G