Neural - Meninges Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (36 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
{{Header}} | |||
==Introduction== | ==Introduction== | ||
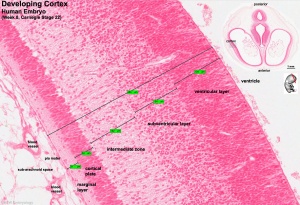
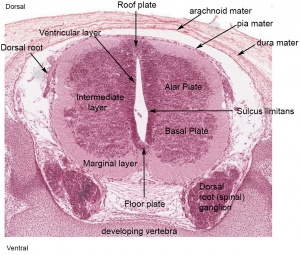
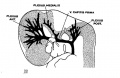
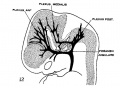
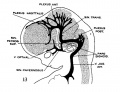
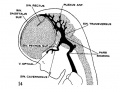
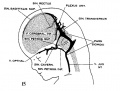
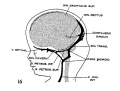
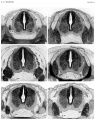
[[File:Stage_22_image_217.jpg|thumb|300px|Human embryo meninges | [[File:Stage_22_image_217.jpg|thumb|300px|Human embryo brain meninges. ([[Week 8]], Carnegie stage {{CS22}})]] | ||
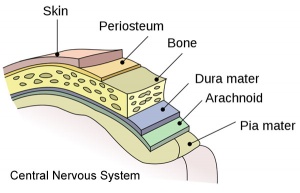
The meninges (singular meninx; Greek, ''meninx'' = membrane) are a complex connective tissue surrounding the central nervous system (brain and spinal cord). The 3 layers from the central nervous outward are: pia mater, arachnoid mater, and the dura mater. All three layers form from the meninx primitiva, a meningeal mesenchyme. | [[File:Human Stage22 spinal cord02.jpg|thumb|300px|Human embryo spinal cord meninges. ([[Week 8]], Carnegie stage {{CS22}})]] | ||
The {{meninges}} (singular meninx; Greek, ''meninx'' = membrane) are a complex connective tissue surrounding the central nervous system (brain and spinal cord). The 3 layers from the central nervous outward are: pia mater, arachnoid mater, and the dura mater. All three layers form initially from the meninx primitiva, a meningeal mesenchyme. The pia mater and the arachnoid can together be called called leptomeninx, and dura mater the pachymeninx. | |||
There have been many theories to the embryonic origins of the three layers that form the meninges, as well as potential differences between species. The safest term would be mesenchymal in origin, but the actual source of this mesenchyme may also differ in the same species at different levels of the central nervous system. The space under the arachnoid layer (subarachnoid space) is filled with cerebrospinal fluid. | |||
: | Recent studies also suggest that rather than acting as a passive connective tissue "neural container" during development, the meninges may also interact and regulate cranial skull and neural development. A second recent change in understanding, is the identification of lymphatic vessels within the {{dura mater}}{{#pmid:29917137|PMID29917137}} that develop in the postnatal period.{{#pmid:31993056|PMID31993056}} | ||
In studying meninges development vascular, lymphatic and cerebrospinal fluid development should also be considered. | |||
Historically, see the 1951 paper describing spinal cord meninges development.<ref name=Sensenig1951>{{Ref-Sensenig1951}}</ref> | |||
:'''Links:''' [[Connective Tissue Development]] | |||
[[ | |||
: | {{Neural Links}} | ||
<br> | |||
{{Neural Links 2}} | |||
<br> | |||
{{Historic Neural}} | |||
== Some Recent Findings == | |||
{| | |||
|-bgcolor="F5FAFF" | |||
| | |||
* '''Review - The Tentorium Cerebelli'''{{#pmid:30305987|PMID30305987}} "The tentorium cerebelli functions as a partition, dispelling the burden of weight from supratentorial structures upon inferior brain matter. Clinicians and neurosurgeons, when assessing pathological findings, should have knowledge regarding the tentorium cerebelli anatomy. This work of literature is a comprehensive review of the tentorium cerebelli, including its anatomy, embryology, and clinical and surgical implications. The evolutionary pattern demonstrates sequential stages to higher mammalian lineage. An understanding of the complexity of the neurovascular structures and the anatomy of the tentorium cerebelli is crucial for surgical procedures by neurosurgeons." | |||
* '''Physiology and molecular biology of barrier mechanisms in the fetal and neonatal''' brain{{#pmid:29774535|PMID29774535}} "Properties of the local internal environment of the adult brain are tightly controlled providing a stable milieu essential for its normal function. The mechanisms involved in this complex control are structural, molecular and physiological (influx and efflux transporters) frequently referred to as the "blood-brain barrier". These mechanisms include regulation of ion levels in brain interstitial fluid essential for normal neuronal function, supply of nutrients, removal of metabolic products and prevention of entry or elimination of toxic agents. A key feature is cerebrospinal fluid secretion and turnover. This is much less during development, allowing greater accumulation of permeating molecules." | |||
= | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! More recent papers | |||
|- | |||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | |||
: | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Meninges+Development ''Meninges Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Pia+Development ''Pia Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Arachnoid+Development ''Arachnoid Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Dura+Development ''Dura Development''] | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''Postnatal development of lymphatic vasculature in the brain meninges'''{{#pmid:29493038|PMID29493038}} "Traditionally, the central nervous system (CNS) has been viewed as an immune-privileged environment with no lymphatic vessels. This view was partially overturned by the discovery of lymphatic vessels in the dural membrane that surrounds the brain, in contact with the interior surface of the skull. We here examine the distribution and developmental timing of these lymphatic vessels. ...We have found that between birth and postnatal day (P) 13, lymphatic vessels extend alongside dural blood vessels from the side of the skull toward the midline. Between P13 and P20, lymphatic vessels along the transverse sinuses reach the superior sagittal sinus (SSS) and extend along the SSS toward the olfactory bulb." [[Mouse Development]] | |||
== | * '''Intermediate filament protein nestin is expressed in developing meninges'''{{#pmid:25428542|PMID25428542}} " Nestin is a type VI intermediate filament protein known as a marker for progenitor cells that can be mostly found in tissues during the embryonic and fetal periods. ...In this study, in the human meninges intense nestin expression was detected as early as in the 9th week of development. Intensity of this expression gradually decreased in later stages of development and nestin expression still persisted in a small population of newborn meningeal cells." | ||
|} | |||
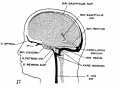
[[File:Meninges_cartoon.jpg|thumb|Meninges simplified cartoon]] | |||
==Pia Mater== | |||
The {{pia mater}} forms innermost part of the meningial layers and is a fine connective tissue covering of the central nervous system. Lies beneath the {{arachnoid mater}}and then tough outer dura mater layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space overlying the pia mater (subarachnoid space) is filled with [[C#cerebrospinal fluid|cerebrospinal fluid]]. The pia mater has close contact with the spinal cord and brain, in the brain it follows down into the sulci and fissures of the cortex. This layer also fuses with the membranous lining of the ventricles ({{ependyma}}) forming the {{choroid plexus}}. | |||
==Arachnoid Mater== | |||
The arachnoid mater (Greek, ''arachne'' = spider + ''-oeides'' = form) forms the central part of the {{meninges}} layers as a meshwork (spider web-like) connective tissue covering of the central nervous system. Lying between the tough outer {{dura mater}} layer and the inner fine {{pia mater}} layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space underlying the arachnoid mater (subarachnoid space) is filled with cerebrospinal fluid. | |||
==Dura Mater== | |||
= | The {{dura mater}} (Latin, ''dura mater'' = hard mother) forms the outer tough connective tissue layer of the {{meninges}} 3 layers that cover the central nervous system. of 3 layers (overlays the {{arachnoid mater}} middle layer and {{pia mater}} inner layer). All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. | ||
At the level of the skull, the outer dura layer forms the inner periosteum of the skull and the inner dura layer forms the dural folds (falx and tentorium) that contains the dural sinuses. The dura mater also expresses osteogenic growth factors that may be required for ossification of cranial vault bones. At the level of the spinal cord, the dura is separated from the periosteum of the vertebral canal by an epidural space. | |||
===Dural Lymphatic Vessels=== | |||
Lymphatic vessels are uniquely found within the {{dura mater}}{{#pmid:29917137|PMID29917137}}, and these vessels only develop during the postnatal period.{{#pmid:31993056|PMID31993056}} They are thought to have a role in trafficking {{neural}} macromolecules to the deep cervical lymph nodes. | |||
=== | ===Dural Venous Sinuses=== | ||
The {{dural venous sinuses}} form the major drainage of the brain to the internal jugular veins in the adult. | |||
The dural venous sinuses: | |||
* lie between the dura mater layers (endosteal layer and meningeal) | |||
* run alone not parallel to arteries. | |||
* are valveless allowing for bidirectional blood flow | |||
Unpaired - superior sagittal sinus, inferior sagittal sinus, straight sinus, occipital sinus, intercavernous sinus | |||
Paired - transverse sinus, sigmoid sinus, superior petrosal sinus, inferior petrosal sinus, cavernous sinus, sphenoparietal sinus, basilar venous plexus | |||
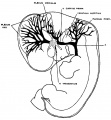
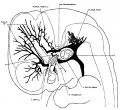
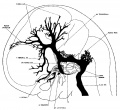
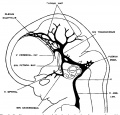
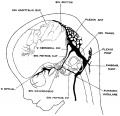
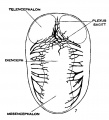
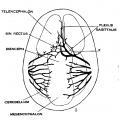
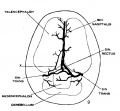
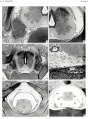
The following figures are from a 1915 study of the venous sinuses of the dura mater in the human embryo.<ref name=Streeter1915>{{Ref-Streeter1915}}</ref> | |||
<gallery> | |||
File:Streeter1915 fig01.jpg|fig 1 embryo 4 mm Embryo No. {{CE588}} | |||
File:Streeter1915 fig02.jpg|fig 2 embryo 13.8 mm Embryo No. {{CE940}} | |||
File:Streeter1915 fig03.jpg|fig 3 embryo 18 mm Embryo No. {{CE144}} | |||
File:Streeter1915 fig04.jpg|fig 4 embryo 21 mm [[:Category:Carnegie Embryo 460|No. 460]] | |||
File:Streeter1915 fig05.jpg|fig 5 embryo 24 mm [[:Category:Carnegie Embryo 632|No. 632]] | |||
File:Streeter1915 fig06.jpg|fig 6 embryo 50 mm [[:Category:Carnegie Embryo 96|No. 96]] | |||
File:Streeter1915 fig07.jpg|fig 7 embryo 13.8 mm [[:Category:Carnegie Embryo 940|No. 940]] | |||
File:Streeter1915 fig08.jpg|fig 8 embryo 20 mm [[:Category:Carnegie Embryo 349|No. 349]] | |||
File:Streeter1915 fig09.jpg|fig 9 fetus 54 mm [[:Category:Carnegie Embryo 458|No. 458]] | |||
File:Streeter1915 fig11.jpg|fig 11 embryo 14 mm [[:Category:Carnegie Embryo 940|No. 940]] | |||
File:Streeter1915 fig12.jpg|fig 12 embryo 18 mm [[:Category:Carnegie Embryo 144|No. 144]] | |||
File:Streeter1915 fig13.jpg|fig 13 embryo 21 mm [[:Category:Carnegie Embryo 460|No. 460]] | |||
File:Streeter1915 fig14.jpg|fig 14 embryo 35 mm [[:Category:Carnegie Embryo 199|No. 100]] | |||
File:Streeter1915 fig15.jpg|fig 15 fetus 50 mm [[:Category:Carnegie Embryo 96|No. 96]] | |||
File:Streeter1915 fig16.jpg|fig 16 fetus 80 mm [[:Category:Carnegie Embryo 234a|No. 234a]] | |||
File:Streeter1915 fig17.jpg|fig 17 adult | |||
</gallery> | |||
===Cerebellar Tentorium=== | |||
The cerebellar tentorium or tentorium cerebelli forms an extension of the {{dura mater}} separating the cerebellum from the inferior portion of the occipital lobes. For a recent review see.{{#pmid:30305987|PMID30305987}} | |||
== References == | |||
<references/> | |||
===Reviews=== | |||
{{#pmid:30801905}} | |||
{{#pmid:29368214}} | |||
{{#pmid:29269062}} | |||
{{#pmid:23381008}} | |||
{{#pmid:22526439}} | |||
{{#pmid:21251809}} | |||
{{#pmid:20099639}} | |||
{{#pmid:19165479}} | |||
{{#pmid:3746345}} | |||
===Articles=== | |||
{{#pmid:27250699}} | |||
{{#pmid:26214850}} | |||
{{#pmid:25468011}} | |||
{{#pmid:23437266}} | |||
===Search PubMed=== | |||
'''Search Pubmed:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=Meninges Development Meninges Development] | |||
== | ===Historic=== | ||
{{Ref-Salvi1898}} | |||
{{Ref-Heuser1913}} | |||
{{Ref-HarveyBurr1924}} | |||
{{Ref-HarveyBurr1926}} | |||
{{Ref-O’RahillyMuller1986}} | |||
== External Links == | == External Links == | ||
| Line 144: | Line 161: | ||
<gallery> | <gallery> | ||
</gallery> | |||
===Historic=== | |||
{{Historic Disclaimer}} | |||
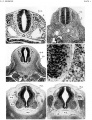
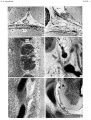
{{Ref-Sensenig1951}} | |||
<gallery> | |||
File:Sensenig1951 plate01.jpg|Plate 1 | |||
File:Sensenig1951 plate02.jpg|Plate 2 | |||
File:Sensenig1951 plate03.jpg|Plate 3 | |||
File:Sensenig1951 plate04.jpg|Plate 4 | |||
</gallery> | </gallery> | ||
Latest revision as of 13:54, 25 July 2020
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The meninges (singular meninx; Greek, meninx = membrane) are a complex connective tissue surrounding the central nervous system (brain and spinal cord). The 3 layers from the central nervous outward are: pia mater, arachnoid mater, and the dura mater. All three layers form initially from the meninx primitiva, a meningeal mesenchyme. The pia mater and the arachnoid can together be called called leptomeninx, and dura mater the pachymeninx.
There have been many theories to the embryonic origins of the three layers that form the meninges, as well as potential differences between species. The safest term would be mesenchymal in origin, but the actual source of this mesenchyme may also differ in the same species at different levels of the central nervous system. The space under the arachnoid layer (subarachnoid space) is filled with cerebrospinal fluid.
Recent studies also suggest that rather than acting as a passive connective tissue "neural container" during development, the meninges may also interact and regulate cranial skull and neural development. A second recent change in understanding, is the identification of lymphatic vessels within the dura mater[1] that develop in the postnatal period.[2]
In studying meninges development vascular, lymphatic and cerebrospinal fluid development should also be considered.
Historically, see the 1951 paper describing spinal cord meninges development.[3]
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Meninges Development | Pia Development | Arachnoid Development | Dura Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Pia Mater
The pia mater forms innermost part of the meningial layers and is a fine connective tissue covering of the central nervous system. Lies beneath the arachnoid materand then tough outer dura mater layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space overlying the pia mater (subarachnoid space) is filled with cerebrospinal fluid. The pia mater has close contact with the spinal cord and brain, in the brain it follows down into the sulci and fissures of the cortex. This layer also fuses with the membranous lining of the ventricles (Template:Ependyma) forming the choroid plexus.
Arachnoid Mater
The arachnoid mater (Greek, arachne = spider + -oeides = form) forms the central part of the meninges layers as a meshwork (spider web-like) connective tissue covering of the central nervous system. Lying between the tough outer dura mater layer and the inner fine pia mater layer. All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin. The space underlying the arachnoid mater (subarachnoid space) is filled with cerebrospinal fluid.
Dura Mater
The dura mater (Latin, dura mater = hard mother) forms the outer tough connective tissue layer of the meninges 3 layers that cover the central nervous system. of 3 layers (overlays the arachnoid mater middle layer and pia mater inner layer). All three layers form from the meninx primitiva, a meningeal mesenchyme that is mesodermal and neural crest in origin.
At the level of the skull, the outer dura layer forms the inner periosteum of the skull and the inner dura layer forms the dural folds (falx and tentorium) that contains the dural sinuses. The dura mater also expresses osteogenic growth factors that may be required for ossification of cranial vault bones. At the level of the spinal cord, the dura is separated from the periosteum of the vertebral canal by an epidural space.
Dural Lymphatic Vessels
Lymphatic vessels are uniquely found within the dura mater[1], and these vessels only develop during the postnatal period.[2] They are thought to have a role in trafficking neural macromolecules to the deep cervical lymph nodes.
Dural Venous Sinuses
The dural venous sinuses form the major drainage of the brain to the internal jugular veins in the adult.
The dural venous sinuses:
- lie between the dura mater layers (endosteal layer and meningeal)
- run alone not parallel to arteries.
- are valveless allowing for bidirectional blood flow
Unpaired - superior sagittal sinus, inferior sagittal sinus, straight sinus, occipital sinus, intercavernous sinus
Paired - transverse sinus, sigmoid sinus, superior petrosal sinus, inferior petrosal sinus, cavernous sinus, sphenoparietal sinus, basilar venous plexus
The following figures are from a 1915 study of the venous sinuses of the dura mater in the human embryo.[8]
fig 1 embryo 4 mm Embryo No. 588
fig 2 embryo 13.8 mm Embryo No. 940
fig 3 embryo 18 mm Embryo No. 144
fig 4 embryo 21 mm No. 460
fig 5 embryo 24 mm No. 632
fig 6 embryo 50 mm No. 96
fig 7 embryo 13.8 mm No. 940
fig 8 embryo 20 mm No. 349
fig 9 fetus 54 mm No. 458
fig 11 embryo 14 mm No. 940
fig 12 embryo 18 mm No. 144
fig 13 embryo 21 mm No. 460
fig 14 embryo 35 mm No. 100
fig 15 fetus 50 mm No. 96
fig 16 fetus 80 mm No. 234a
Cerebellar Tentorium
The cerebellar tentorium or tentorium cerebelli forms an extension of the dura mater separating the cerebellum from the inferior portion of the occipital lobes. For a recent review see.[4]
References
- ↑ 1.0 1.1 Jani RH & Sekula RF. (2018). Magnetic Resonance Imaging of Human Dural Meningeal Lymphatics. Neurosurgery , 83, E10-E12. PMID: 29917137 DOI.
- ↑ 2.0 2.1 Bálint L, Ocskay Z, Deák BA, Aradi P & Jakus Z. (2019). Lymph Flow Induces the Postnatal Formation of Mature and Functional Meningeal Lymphatic Vessels. Front Immunol , 10, 3043. PMID: 31993056 DOI.
- ↑ Sensenig EC. The early development of the meninges of the spinal cord in human embryos. (1951) Contrib. Embryol., Carnegie Inst. Wash. Publ. 611.
- ↑ 4.0 4.1 Rai R, Iwanaga J, Shokouhi G, Oskouian RJ & Tubbs RS. (2018). The Tentorium Cerebelli: A Comprehensive Review Including Its Anatomy, Embryology, and Surgical Techniques. Cureus , 10, e3079. PMID: 30305987 DOI.
- ↑ Saunders NR, Dziegielewska KM, Møllgård K & Habgood MD. (2018). Physiology and molecular biology of barrier mechanisms in the fetal and neonatal brain. J. Physiol. (Lond.) , , . PMID: 29774535 DOI.
- ↑ Izen RM, Yamazaki T, Nishinaka-Arai Y, Hong YK & Mukouyama YS. (2018). Postnatal development of lymphatic vasculature in the brain meninges. Dev. Dyn. , , . PMID: 29493038 DOI.
- ↑ Yay A, Ozdamar S, Canoz O, Baran M, Tucer B & Sonmez MF. (2014). Intermediate filament protein nestin is expressed in developing meninges. Bratisl Lek Listy , 115, 718-22. PMID: 25428542
- ↑ Streeter GL. The development of the venous sinuses of the dura mater in the human embryo. (1915) Amer. J Anat.18: 145-178.
Reviews
Dasgupta K & Jeong J. (2019). Developmental biology of the meninges. Genesis , 57, e23288. PMID: 30801905 DOI.
Weller RO, Sharp MM, Christodoulides M, Carare RO & Møllgård K. (2018). The meninges as barriers and facilitators for the movement of fluid, cells and pathogens related to the rodent and human CNS. Acta Neuropathol. , 135, 363-385. PMID: 29368214 DOI.
Mortazavi MM, Quadri SA, Khan MA, Gustin A, Suriya SS, Hassanzadeh T, Fahimdanesh KM, Adl FH, Fard SA, Taqi MA, Armstrong I, Martin BA & Tubbs RS. (2018). Subarachnoid Trabeculae: A Comprehensive Review of Their Embryology, Histology, Morphology, and Surgical Significance. World Neurosurg , 111, 279-290. PMID: 29269062 DOI.
Adeeb N, Mortazavi MM, Deep A, Griessenauer CJ, Watanabe K, Shoja MM, Loukas M & Tubbs RS. (2013). The pia mater: a comprehensive review of literature. Childs Nerv Syst , 29, 1803-10. PMID: 23381008 DOI.
Adeeb N, Mortazavi MM, Tubbs RS & Cohen-Gadol AA. (2012). The cranial dura mater: a review of its history, embryology, and anatomy. Childs Nerv Syst , 28, 827-37. PMID: 22526439 DOI.
Siegenthaler JA & Pleasure SJ. (2011). We have got you 'covered': how the meninges control brain development. Curr. Opin. Genet. Dev. , 21, 249-55. PMID: 21251809 DOI.
Patel N & Kirmi O. (2009). Anatomy and imaging of the normal meninges. Semin. Ultrasound CT MR , 30, 559-64. PMID: 20099639
Mack J, Squier W & Eastman JT. (2009). Anatomy and development of the meninges: implications for subdural collections and CSF circulation. Pediatr Radiol , 39, 200-10. PMID: 19165479 DOI.
O'Rahilly R & Müller F. (1986). The meninges in human development. J. Neuropathol. Exp. Neurol. , 45, 588-608. PMID: 3746345
Articles
Tanaka M. (2016). Embryological Consideration of Dural Arteriovenous Fistulas. Neurol. Med. Chir. (Tokyo) , 56, 544-51. PMID: 27250699 DOI.
Yokogawa N, Murakami H, Demura S, Kato S, Yoshioka K, Yamamoto M, Iseki S & Tsuchiya H. (2015). Effects of Radiation on Spinal Dura Mater and Surrounding Tissue in Mice. PLoS ONE , 10, e0133806. PMID: 26214850 DOI.
Baltsavias G, Parthasarathi V, Aydin E, Al Schameri RA, Roth P & Valavanis A. (2015). Cranial dural arteriovenous shunts. Part 1. Anatomy and embryology of the bridging and emissary veins. Neurosurg Rev , 38, 253-63; discussion 263-4. PMID: 25468011 DOI.
Tochitani S & Kondo S. (2013). Immunoreactivity for GABA, GAD65, GAD67 and Bestrophin-1 in the meninges and the choroid plexus: implications for non-neuronal sources for GABA in the developing mouse brain. PLoS ONE , 8, e56901. PMID: 23437266 DOI.
Search PubMed
Search Pubmed: Development Meninges Development
Historic
Salvi G. Meninges histogenesis and structure (L'istogenesi E La Struttura Delle Meningi). (1898) Thèse de Paris.
Heuser CH. The development of the cerebral ventricles in the pig. (1913) Amer. J Anat. 15(2): 215-251.
Harvey SC. and Burr HS. An experimental study of the origin of the meninges. (1924) Proc. Soc. Exper. Biol. and Med. 22: 52-53.
Harvey SC. and Burr HS. The development of the meninges. (1926) Arch Neurol Psychiatry 15:545–567
O'Rahilly R. and Müller F. The meninges in human development. (1986) J Neuropathol Exp Neurol 45: 588–608.
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Additional Images
Historic
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Sensenig EC. The early development of the meninges of the spinal cord in human embryos. (1951) Contrib. Embryol., Carnegie Inst. Wash. Publ. 611.
Terms
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Neural - Meninges Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Meninges_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G