Neural - Cerebrum Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 24: | Line 24: | ||
* '''Correlation of diffusion tensor imaging with histology in the developing human frontal cerebrum'''<ref name=PMID19622880><pubmed>19622880</pubmed></ref> "Transient early cerebral laminar organization resulting from normal developmental events has been revealed in human beings through histology and imaging studies. DTI studies have postulated that the fractional anisotropy (FA)-based differentiation of different laminar structures reflects both differing cellular density over the glial fibers and fiber alignment in respective regions. The aim of this study was to correlate FA values in these transient zones with histology. Brain DTI was performed on 50 freshly aborted human fetuses with gestational ages (GA) ranging from 12 to 42 weeks. Regions of interest were placed on the cortical plate, subplate, intermediate and germinal matrix (GMx) zones of the frontal lobe to quantify FA values. Glial fibrillary acidic protein (GFAP), neurofilament (NF) and neuron-specific enolase (NSE) immunohistochemical analyses were performed for the cortical plate, intermediate zone and GMx. In the cortical plate, a significant positive correlation was observed between FA values and percentage area of GFAP expression in fetuses <or=28 weeks of GA (r = 0.56, p = 0.01). FA values showed a significant positive correlation with the percentage area of NF expression in the intermediate zone (r = 0.54, p = 0.05). A significant positive correlation was also observed between FA and the number of NSE-positive cells per mm(2) in the GMx (r = 0.76, p < 0.01) and subplate (r = 0.59, p = 0.03) zones. The results of our study suggest that the FA can be used as noninvasive marker of neurodevelopmental events in the frontal lobe of human fetal brain." | * '''Correlation of diffusion tensor imaging with histology in the developing human frontal cerebrum'''<ref name=PMID19622880><pubmed>19622880</pubmed></ref> "Transient early cerebral laminar organization resulting from normal developmental events has been revealed in human beings through histology and imaging studies. DTI studies have postulated that the fractional anisotropy (FA)-based differentiation of different laminar structures reflects both differing cellular density over the glial fibers and fiber alignment in respective regions. The aim of this study was to correlate FA values in these transient zones with histology. Brain DTI was performed on 50 freshly aborted human fetuses with gestational ages (GA) ranging from 12 to 42 weeks. Regions of interest were placed on the cortical plate, subplate, intermediate and germinal matrix (GMx) zones of the frontal lobe to quantify FA values. Glial fibrillary acidic protein (GFAP), neurofilament (NF) and neuron-specific enolase (NSE) immunohistochemical analyses were performed for the cortical plate, intermediate zone and GMx. In the cortical plate, a significant positive correlation was observed between FA values and percentage area of GFAP expression in fetuses <or=28 weeks of GA (r = 0.56, p = 0.01). FA values showed a significant positive correlation with the percentage area of NF expression in the intermediate zone (r = 0.54, p = 0.05). A significant positive correlation was also observed between FA and the number of NSE-positive cells per mm(2) in the GMx (r = 0.76, p < 0.01) and subplate (r = 0.59, p = 0.03) zones. The results of our study suggest that the FA can be used as noninvasive marker of neurodevelopmental events in the frontal lobe of human fetal brain." | ||
* '''Development of laminar organization of the fetal cerebrum'''<ref><pubmed>20981415</pubmed></ref> "Heads of 131 fetal specimens of 14-40 weeks gestational age (GA) were scanned by 3.0T MRI. Eleven fetal specimens of 14-27 weeks GA were scanned by 7.0T MRI. On T(1)-weighted 3.0T MRI, layers could be visualized at 14 weeks GA and appeared clearer after 18 weeks GA. On 7.0T MRI, four zones could be recognized at 14 weeks GA. During 15-22 weeks GA, when laminar organization appeared typical, seven layers including the periventricular zone and external capsule fibers could be differentiated, which corresponded to seven zones in histological stained sections. At 23-28 weeks GA, laminar organization appeared less typical, and borderlines among them appeared obscured. After 30 weeks GA, it disappeared and turned into mature-like structures. The developing lamination appeared the most distinguishable at the parieto-occipital part of brain and peripheral regions of the hippocampus. The migrating thalamocortical afferents were probably delineated as a high signal layer located at the lower, middle, and upper part of the subplate zone at 16-28 weeks GA on T(1)-weighted 3.0T MRI." | * '''Development of laminar organization of the fetal cerebrum'''<ref><pubmed>20981415</pubmed></ref> "Heads of 131 fetal specimens of 14-40 weeks gestational age (GA) were scanned by 3.0T MRI. Eleven fetal specimens of 14-27 weeks GA were scanned by 7.0T MRI. On T(1)-weighted 3.0T MRI, layers could be visualized at 14 weeks GA and appeared clearer after 18 weeks GA. On 7.0T MRI, four zones could be recognized at 14 weeks GA. During 15-22 weeks GA, when laminar organization appeared typical, seven layers including the periventricular zone and external capsule fibers could be differentiated, which corresponded to seven zones in histological stained sections. At 23-28 weeks GA, laminar organization appeared less typical, and borderlines among them appeared obscured. After 30 weeks GA, it disappeared and turned into mature-like structures. The developing lamination appeared the most distinguishable at the parieto-occipital part of brain and peripheral regions of the hippocampus. The migrating thalamocortical afferents were probably delineated as a high signal layer located at the lower, middle, and upper part of the subplate zone at 16-28 weeks GA on T(1)-weighted 3.0T MRI." | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! More recent papers | |||
|- | |||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | |||
Search term: ''Cerebral Cortex Embryology'' | |||
<pubmed limit=5>Cerebral+Cortex+Development</pubmed> | |||
|} | |} | ||
Revision as of 15:39, 17 August 2017
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
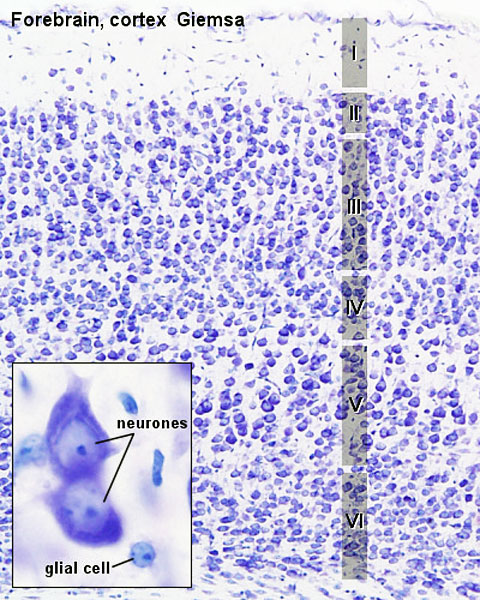
The brain as it is generally recognised. The adult cerebral cortex like other neural structures has a laminar organisation, the mammalian neocortex consists of six layers, while the reptilian and avian cortices have only three layers (equivalent to mammalian layers I, V and VI).
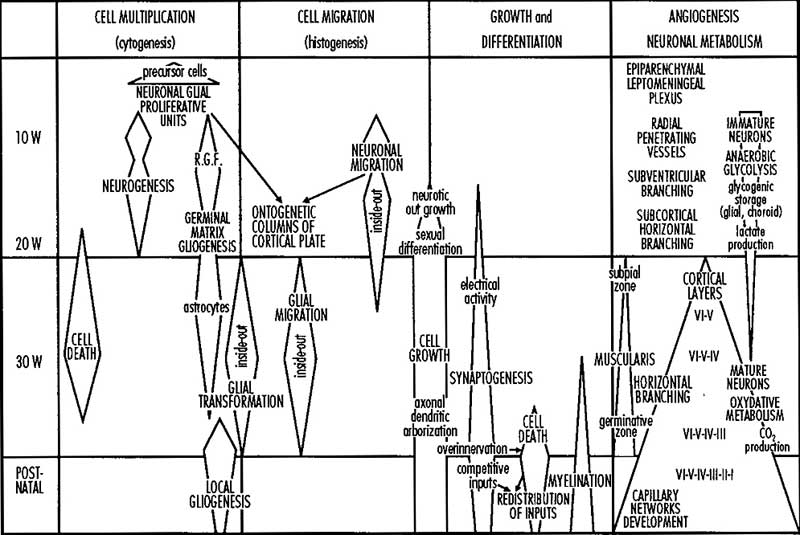
A simplified developmental sequence can be described as cell proliferation, cell migration, and finally cortical organization. In development, lamination occurs in an "inside-out" sequence earlier inside and later born neurons outside. The cortex is divided into areas which serve distinct functions including motor, sensory and cognitive processing. The lamination process requires a range of different signals including; Reelin (Reln, an extracellular protein), Disabled-1 (Dab1, an intracellular signaling molecule), and Cullin-5 (Cul5, an E3 ubiquitin ligase).
The cortex progenitor cell types are either neuron-restricted or bipotent (neuron-glial) progenitors that generate glial-restricted progenitors at mid- and late neurogenesis.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Cerebral Cortex Embryology <pubmed limit=5>Cerebral+Cortex+Development</pubmed> |
Development Overview
Neuralation begins at the trilaminar embryo with formation of the notochord and somites, both of which underly the ectoderm and do not contribute to the nervous system, but are involved with patterning its initial formation. The central portion of the ectoderm then forms the neural plate that folds to form the neural tube, that will eventually form the entire central nervous system.
- Early developmental sequence: Epiblast - Ectoderm - Neural Plate - Neural groove and Neural Crest - Neural Tube and Neural Crest
| Neural Tube | Primary Vesicles | Secondary Vesicles | Adult Structures |
|---|---|---|---|
| week 3 | week 4 | week 5 | adult |
| prosencephalon (forebrain) | telencephalon | Rhinencephalon, Amygdala, hippocampus, cerebrum (cortex), hypothalamus, pituitary | Basal Ganglia, lateral ventricles | |
| diencephalon | epithalamus, thalamus, Subthalamus, pineal, posterior commissure, pretectum, third ventricle | ||
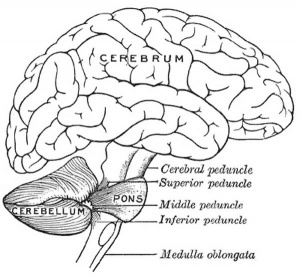
| mesencephalon (midbrain) | mesencephalon | tectum, Cerebral peduncle, cerebral aqueduct, pons | |
| rhombencephalon (hindbrain) | metencephalon | cerebellum | |
| myelencephalon | medulla oblongata, isthmus | ||
| spinal cord, pyramidal decussation, central canal | |||
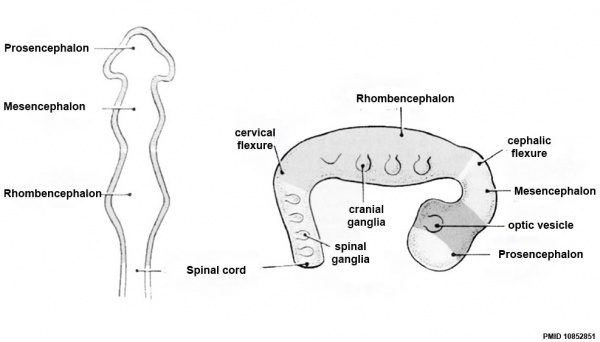
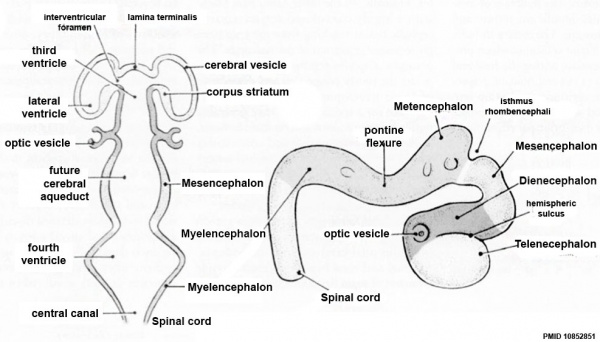
Early Brain Vesicles
Primary Vesicles
Secondary Vesicles
Late Embryonic Brain
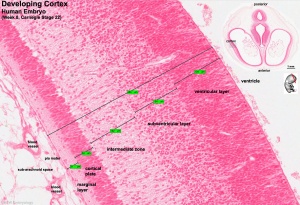
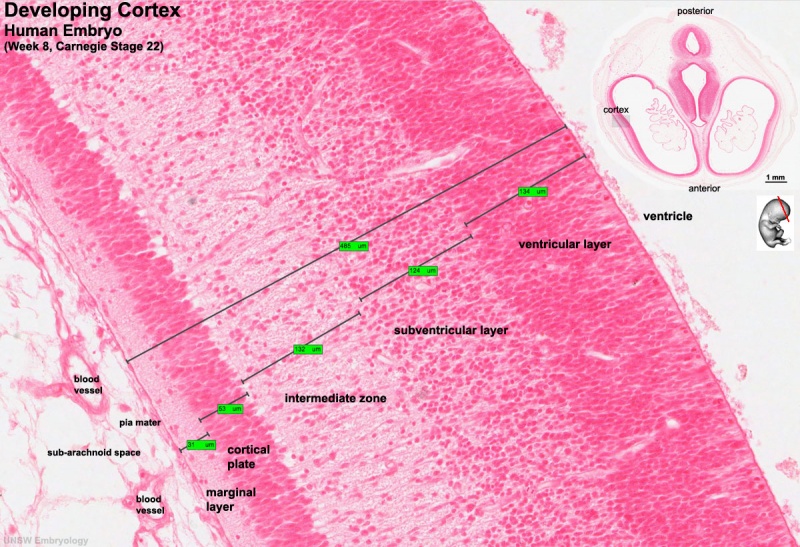
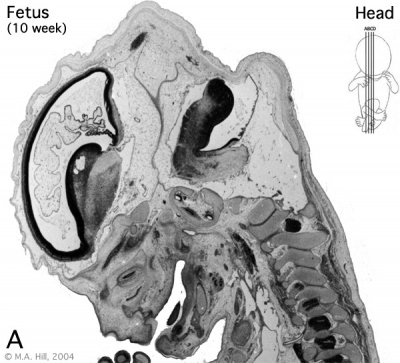
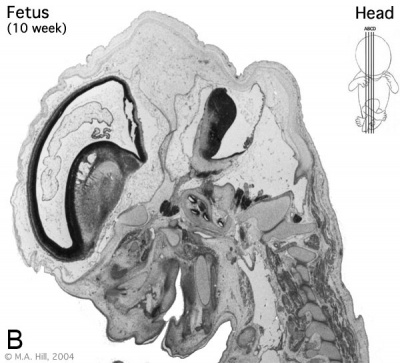
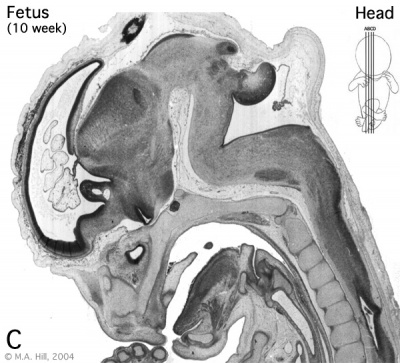
Human embryo developing cortex (Week 8, Carnegie stage 22)
- small embryo shows approximate level of section.
- insert top right shows whole head section.
- small shaded box on whole section shows region of large image.
- layer thicknesses are shown in microns.
Early Fetal Brain
|
|
|
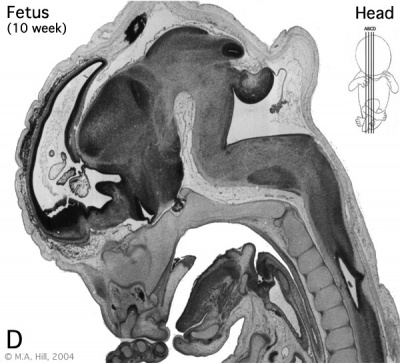
|
The above images are from a week 10 human fetus.
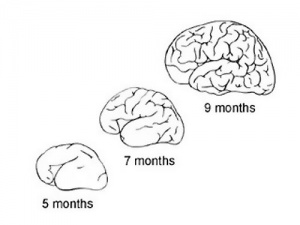
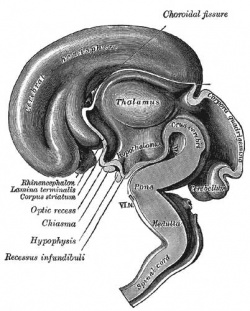
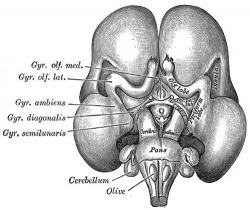
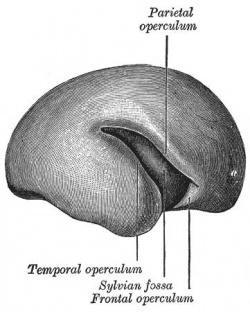
Fetal Brain
|
|
|
| Fetal brain (3 months) | Fetal brain (4 months) | Fetal brain (5 months) |
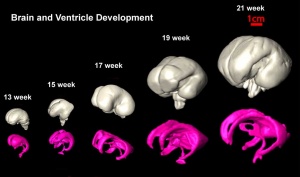
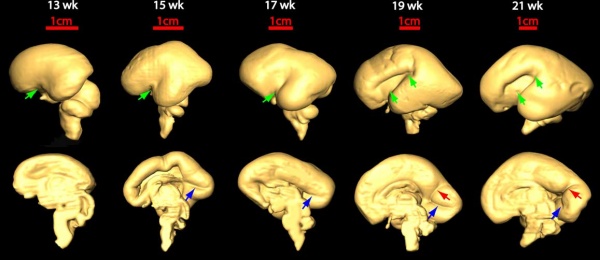
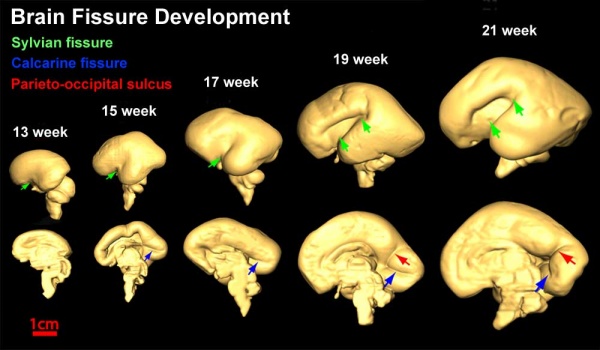
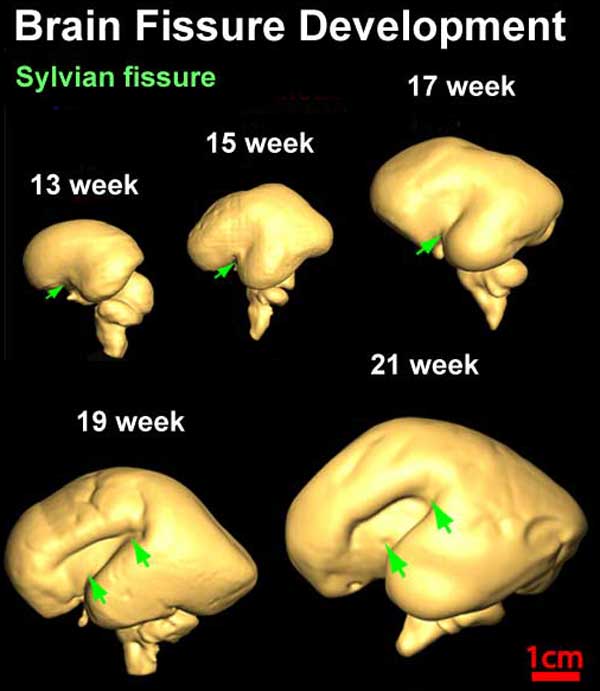
Fissures are the major indentations, sulci (singular sulcus), that divide the brain surface into lobes and appear during fetal development as the brain grows. The images below show MRI analysis of the developing human fetal brain.
- Links: Magnetic Resonance Imaging
Developmental Overview
Cortical Neurons
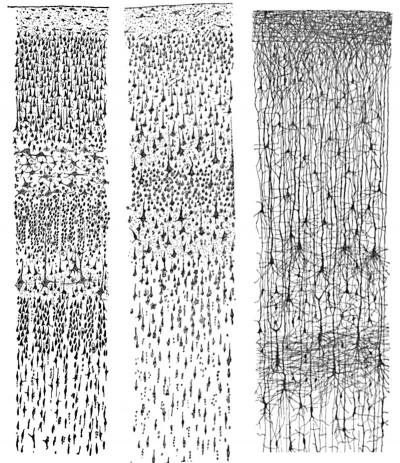
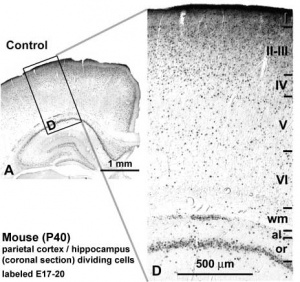
Cortical layers in a historic drawing by Cajal.
Cajal-Retzius Neurons
Cajal-Retzius (CR) cells are some of the earliest generated cortical neurons arising from restricted domains of the pallial ventricular zone, and then migrate from the borders of the developing pallium to cover the cortical primordium. These early forming neurons then control the radial migration of neurons and the formation of cortical layers. In mice, this has been shown by these cells secreting the extracellular glycoprotein Reelin (Reln) and it has been suggested that these cells also fine tune multiple signaling pathways underlying the regulation of cortical regionalization.[6]
Molecular
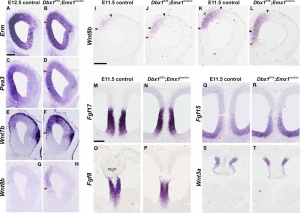
- Fgfr1 and Fgfr2 - control excitatory cortical neuron development within the entire cerebral cortex.[8]
- Fgfr2 - proper formation of the medial prefrontal cortex (mPFC).[8]
- MARCKS - (myristoylated alanine-rich C-kinase substrate protein) a cellular substrate for PKC modulates radial glial placement and expansion.[9]
- MicroRNA - noncoding RNAs that regulate mRNA expression, highly expressed during development.[10]
- Wnt - contribute to the production of basal progenitors (non-surface dividing or intermediate progenitors).[11]
Corpus Callosum
The corpus callosum is the area of the brain which connects the two cerebral hemispheres. Maximum increase in thickness and width of the corpus callosum occurred between 19 and 21 weeks' gestation.[12]
Human Timeline:
- 74 days - callosal axons appear.
- 84 days - subdivisions of the genu and splenium can be identified.
- 115 days - adult morphology is seen.
Agenesis of Corpus Callosum
Agenesis of the corpus callosum (ACC) is a partial or complete absence of the corpus callosum, a rare cerebral malformation.
- Links: NINDS Information
Cerebral Vascular Development
| Overview cartoon | Early vascular changes |
|---|---|
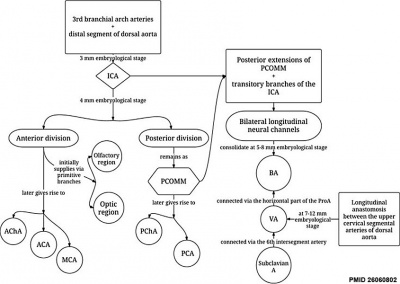
|

|
Chronological development of cerebral brain arteries initially with the rise of the internal carotid artery and subsequently with the development of the posterior circulation.[13]
- Links: Blood Vessel Development | Head Development
Abnormalities
Agenesis of Corpus Callosum
Agenesis of the corpus callosum (ACC) is a partial or complete absence of the corpus callosum, a rare cerebral malformation.
- Links: NINDS Information
Hemimegalencephaly
Increased proliferation
Heterotopia
Ectopic migration
Lissencephaly
A malformations derived from abnormal neuronal migration leading to agyria (convolutions of the cerebral cortex are not fully formed) and pachygyria (convolutions of the cerebral cortex unusually thick ).
Pachygyria (Greek, pachy = "thick")
Microlissencephaly
Decreased proliferation
Polymicrogyria
Abnormal organization, numerous small gyri and a thick disorganized cortical plate lacking normal lamination, disruption of microtubule-based processes underlies a large spectrum of neuronal migration.[14]
Schizencephaly
Abnormal organization
References
- ↑ <pubmed>20108226</pubmed>
- ↑ <pubmed>24972575</pubmed>
- ↑ <pubmed>21414909</pubmed>| J Neurosci.
- ↑ <pubmed>19622880</pubmed>
- ↑ <pubmed>20981415</pubmed>
- ↑ 6.0 6.1 <pubmed>20668538</pubmed>| PLoS Biol.
- ↑ <pubmed>19812240</pubmed>| PMC2871377
- ↑ 8.0 8.1 <pubmed>20410112</pubmed>
- ↑ <pubmed>19666823</pubmed>
- ↑ <pubmed>19914179</pubmed>
- ↑ <pubmed>20215343</pubmed>
- ↑ <pubmed>11778993</pubmed>
- ↑ <pubmed>26060802</pubmed>| J Stroke.
- ↑ <pubmed>19465910</pubmed>
Reviews
<pubmed>19732610</pubmed> <pubmed>19763105</pubmed>
Articles
<pubmed>23727529</pubmed> <pubmed>20161753</pubmed> <pubmed>20040495</pubmed> <pubmed>20410119</pubmed> <pubmed>20410112</pubmed> <pubmed>20215343</pubmed>
Search PubMed
Search Pubmed: Cerebrum Embryology | Cerebrum Development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Zagreb Neuroembryological Collection - contains more than 500 prenatal human brains stained with various classical neurohistological, as well as modern histochemical and immunohistochemical methods. The bank is located at the Croatian Institute for Brain Research. http://www.hiim.hr/nova
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Neural - Cerebrum Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Cerebrum_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G