Neural - Cerebellum Development: Difference between revisions
No edit summary |
|||
| Line 21: | Line 21: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Preterm delivery disrupts the developmental program of the cerebellum'''< | * '''Preterm delivery disrupts the developmental program of the cerebellum''' | ||
<refname="PMID21858122"><pubmed>21858122</pubmed>| [http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0023449 PLoS One]</ref> "A rapid growth in human cerebellar development occurs in the third trimester, which is impeded by preterm delivery. ...We report that premature birth and development in an ex-utero environment leads to a significant decrease in the thickness and an increase in the packing density of the cells within the external granular layer and the inner granular layer well, as a reduction in the density of bergmann glial fibres. In addition, this also leads to a reduced expression of sonic hedgehog in the purkinje layer. We conclude that the developmental program of the cerebellum is specifically modified by events that follow preterm delivery." | |||
* '''Ontogeny-recapitulating generation and tissue integration of ES cell-derived Purkinje cells''' <ref><pubmed>20835252</pubmed></ref> "Purkinje cells are the sole output neurons of the cerebellar cortex and their dysfunction causes severe ataxia. We found that Purkinje cells could be robustly generated from mouse embryonic stem (ES) cells by recapitulating the self-inductive signaling microenvironments of the isthmic organizer. The cell-surface marker Neph3 enabled us to carry out timed prospective selection of Purkinje cell progenitors, which generated morphologically characteristic neurons with highly arborized dendrites that expressed mature Purkinje cell-specific markers such as the glutamate receptor subunit GluRδ2. Similar to mature Purkinje cells, these neurons also showed characteristic spontaneous and repeated action potentials and their postsynaptic excitatory potentials were generated exclusively through nonNMDA glutamate receptors. Fetal transplantation of precursors isolated by fluorescence-activated cell sorting showed orthotopic integration of the grafted neurons into the Purkinje cell layer with their axons extending to the deep cerebellar nuclei and dendrites receiving climbing and parallel fibers. This selective preparation of bona fide Purkinje cells should aid future investigation of this important neuron." | * '''Ontogeny-recapitulating generation and tissue integration of ES cell-derived Purkinje cells''' <ref><pubmed>20835252</pubmed></ref> "Purkinje cells are the sole output neurons of the cerebellar cortex and their dysfunction causes severe ataxia. We found that Purkinje cells could be robustly generated from mouse embryonic stem (ES) cells by recapitulating the self-inductive signaling microenvironments of the isthmic organizer. The cell-surface marker Neph3 enabled us to carry out timed prospective selection of Purkinje cell progenitors, which generated morphologically characteristic neurons with highly arborized dendrites that expressed mature Purkinje cell-specific markers such as the glutamate receptor subunit GluRδ2. Similar to mature Purkinje cells, these neurons also showed characteristic spontaneous and repeated action potentials and their postsynaptic excitatory potentials were generated exclusively through nonNMDA glutamate receptors. Fetal transplantation of precursors isolated by fluorescence-activated cell sorting showed orthotopic integration of the grafted neurons into the Purkinje cell layer with their axons extending to the deep cerebellar nuclei and dendrites receiving climbing and parallel fibers. This selective preparation of bona fide Purkinje cells should aid future investigation of this important neuron." | ||
|} | |} | ||
Revision as of 22:27, 22 September 2011
Introduction
Neural development is one of the earliest systems to begin and the last to be completed after birth. This development generates the most complex structure within the embryo and the long time period of development means in utero insult during pregnancy may have consequences to development of the nervous system.
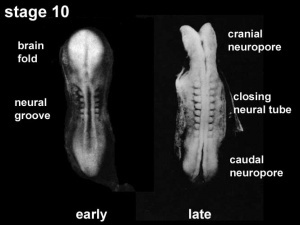
The early central nervous system begins as a simple neural plate that folds to form a groove then tube, open initially at each end. Failure of these opening to close contributes a major class of neural abnormalities (neural tube defects).
Within the neural tube stem cells generate the 2 major classes of cells that make the majority of the nervous system : neurons and glia. Both these classes of cells differentiate into many different types generated with highly specialized functions and shapes. This section covers the establishment of neural populations, the inductive influences of surrounding tissues and the sequential generation of neurons establishing the layered structure seen in the brain and spinal cord.
- Neural development beginnings quite early, therefore also look at notes covering Week 3- neural tube and Week 4-early nervous system.
- Development of the neural crest and sensory systems (hearing/vision/smell) are only introduced in these notes and are covered in other notes sections.
Some Recent Findings
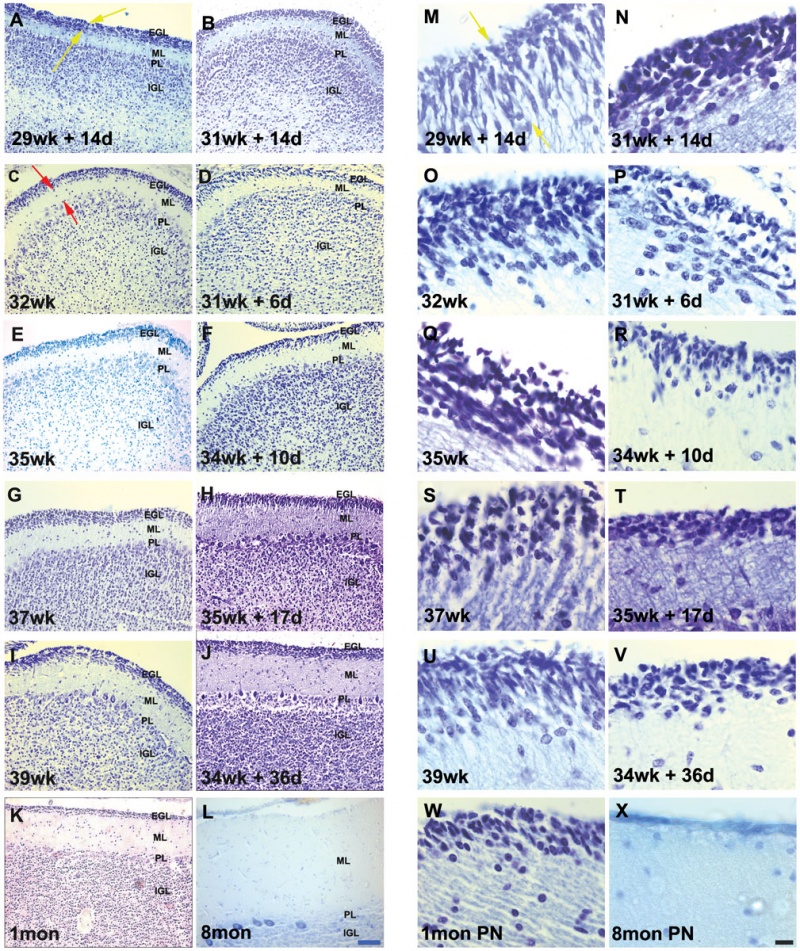
<refname="PMID21858122"><pubmed>21858122</pubmed>| PLoS One</ref> "A rapid growth in human cerebellar development occurs in the third trimester, which is impeded by preterm delivery. ...We report that premature birth and development in an ex-utero environment leads to a significant decrease in the thickness and an increase in the packing density of the cells within the external granular layer and the inner granular layer well, as a reduction in the density of bergmann glial fibres. In addition, this also leads to a reduced expression of sonic hedgehog in the purkinje layer. We conclude that the developmental program of the cerebellum is specifically modified by events that follow preterm delivery."
|
Development Overview
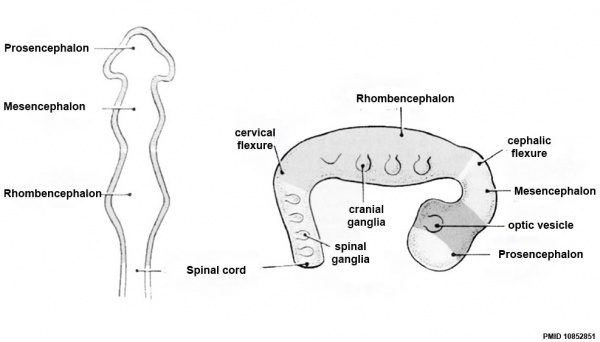
Neuralation begins at the trilaminar embryo with formation of the notochord and somites, both of which underly the ectoderm and do not contribute to the nervous system, but are involved with patterning its initial formation. The central portion of the ectoderm then forms the neural plate that folds to form the neural tube, that will eventually form the entire central nervous system.
- Early developmental sequence: Epiblast - Ectoderm - Neural Plate - Neural groove and Neural Crest - Neural Tube and Neural Crest
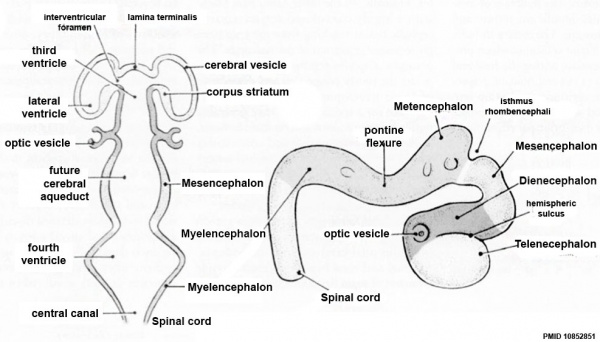
| Neural Tube | Primary Vesicles | Secondary Vesicles | Adult Structures |
|---|---|---|---|
| week 3 | week 4 | week 5 | adult |
| prosencephalon (forebrain) | telencephalon | Rhinencephalon, Amygdala, hippocampus, cerebrum (cortex), hypothalamus, pituitary | Basal Ganglia, lateral ventricles | |
| diencephalon | epithalamus, thalamus, Subthalamus, pineal, posterior commissure, pretectum, third ventricle | ||
| mesencephalon (midbrain) | mesencephalon | tectum, Cerebral peduncle, cerebral aqueduct, pons | |
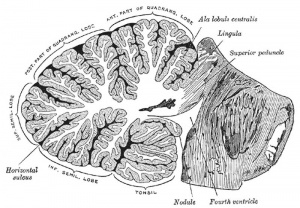
| rhombencephalon (hindbrain) | metencephalon | cerebellum | |
| myelencephalon | medulla oblongata, isthmus | ||
| spinal cord, pyramidal decussation, central canal | |||
Early Brain Vesicles
Primary Vesicles
Secondary Vesicles
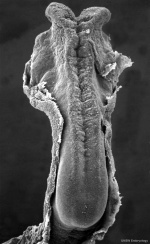
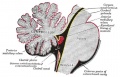
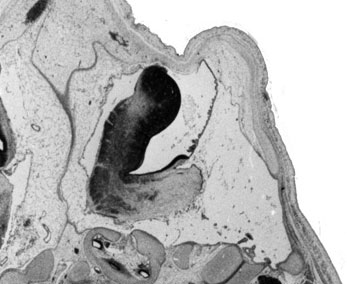
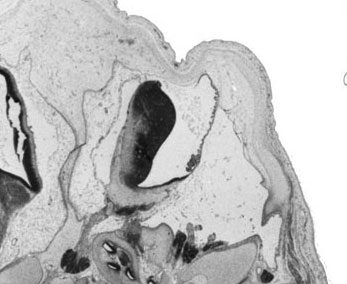
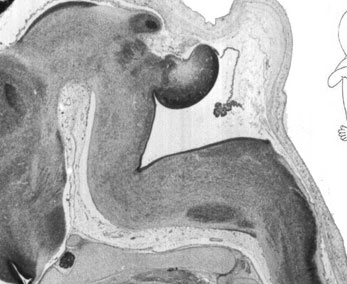
Fetal Cerebellum (Week 10)
|
|
| Plane A (midline) | Plane B (medial) |
|
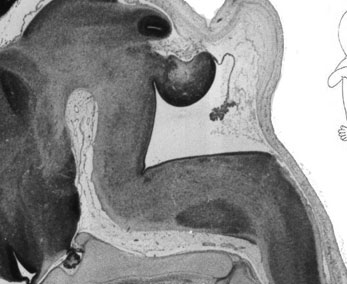
|
| Plane C (lateral) | Plane D (most lateral) |
- Links: 10 week Fetal | Fetal Development
Postnatal
Developing human cerebellum <refname="PMID21858122"><pubmed>21858122</pubmed>| PLoS One</ref>
A greater EGL cell density and reduced EGL thickness were reported in preterms with ex-utero exposure, as compared to their age matched stillborn controls.
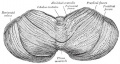
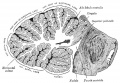
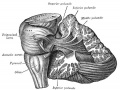
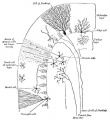
Cerebellum Images
References
- ↑ <pubmed>20835252</pubmed>
Reviews
<pubmed>19732611</pubmed> <pubmed>17408845</pubmed> <pubmed>16243598</pubmed> <pubmed>15610138</pubmed> <pubmed>12843872</pubmed>
Articles
<pubmed>20460306</pubmed> <pubmed>15496441</pubmed>
Search PubMed
Search Pubmed: Cerebellum Embryology | Cerebellum Development
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 25) Embryology Neural - Cerebellum Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Cerebellum_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G