Lecture - Fertilization
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This lecture will cover male and female gametogenesis and fertilisation.
IN development 1 embryonic cell (zygote) will produce about 1013 (100,000,000,000,000) cells in the adult at any one time (over time with cell death and ongoing replacement this is substantially more).
This is where the first embryonic cell begins! Fertilization is the fusion of haploid gametes, egg (oocyte) and sperm (spermatozoa), to form the diploid zygote. Note though there can be subtle differences in the fertilization process which occurs naturally within the body or through reproductive technologies outside the body, the overall product in both cases is a diplod zygote.
| 2016 Lecture Video Recording |
|---|
| This 2016 lecture video recording is similar in content to the current 2018 lecture and is available to help understand this online content.
<html5media height="600" width="800">File:2016ANAT2341-Lecture-Fertilization.mp4</html5media> Click to play new window - 2016 Lecture Video (54.46 MB) |
| Some Recent Research |
|---|
|
Lecture Archive: 2016 Video | 2016 PDF | 2015 | 2015 PDF | 2014 PDF | 2013 | 2012 | 2011 | 2010 | 2009
Lecture Objectives
- Broad understanding of reproductive cycles.
- Understand the key features of gametogenesis.
- Understand the differences in male and female gametogenesis.
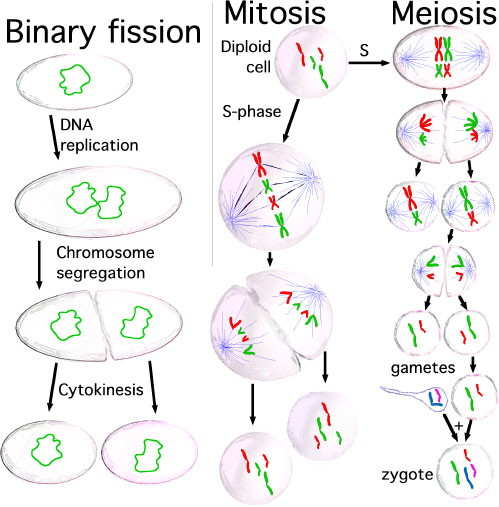
- Brief understanding of the differences between mitosis and meiosis.
- Understanding of the events in fertilization.
Lecture Resources
| Movies | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
|
| |||||||||||||||||||||
|
|
|
|
|
|
| References | ||
|---|---|---|
| Moore, K.L., Persaud, T.V.N. & Torchia, M.G. (2015). The developing human: clinically oriented embryology (10th ed.). Philadelphia: Saunders. | The following chapter links only work with a UNSW connection. | |
| Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R., Francis-West, P.H. & Philippa H. (2015). Larsen's human embryology (5th ed.). New York; Edinburgh: Churchill Livingstone. | The following chapter links only work with a UNSW connection. | |
| Hill, M.A. (2020). UNSW Embryology (20th ed.) Retrieved April 25, 2024, from https://embryology.med.unsw.edu.au |
| |
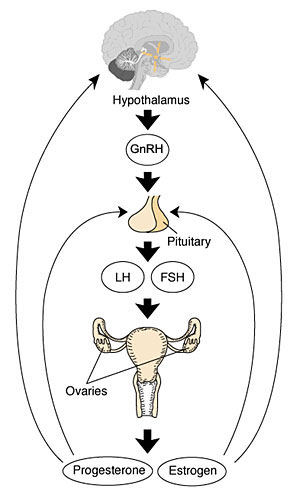
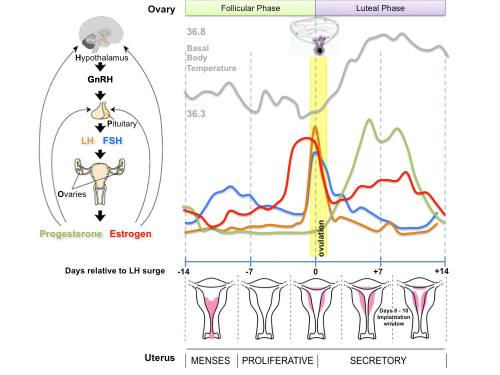
Human Reproductive Cycle
Sexual reproduction in most species is regulated by regular endocrine changes, or cycles, in the female. These cycles begin postnatally, function for variable times and can then decrease or cease entirely.
- Human reproduction is regulated in females by the menstrual cycle, a regular cyclic hormonal change which coordinate changes in the ovary and internal reproductive tract. This cycle commences at puberty and ends at menopause.
- Non-primates (rats, mice, horses, pig) reproduction is regulated in females by the estrous cycle (British spelling, oestrous).
| Female | Male |
|---|---|
|
|
Gametogenesis
The term "gametogenesis" describes the process of development of the haploid germ cells.
meiosis in the gonad (ovary or testis) produces the female and male haploid gametes, oocyte (egg) and spermatozoa (sperm).
Meiosis time course and final gamete number differs between female and male.
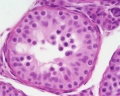
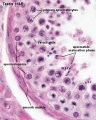
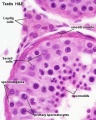
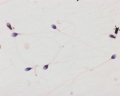
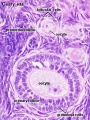
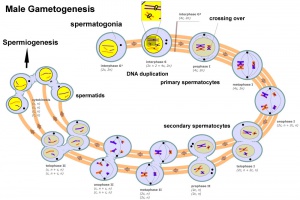
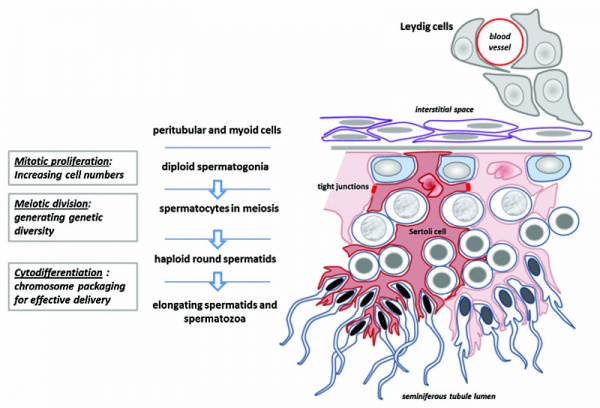
Male - Spermatogenesis
Human spermatozoa (electron microscope)
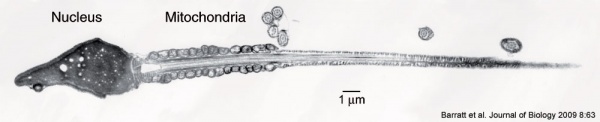
Mouse spermatozoa (electron microscope)
The testes have two functions.
- produce the male gametes or spermatozoa
- produce male sexual hormone, testosterone (internal and external genitalia, sex characteristics)
Human spermatozoa take about 48 days from entering meiosis until morphologically mature spermatozoa.
Sertoli cells (support cells) Interstitial cells or Leydig cells (produce hormone) |
|
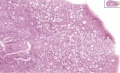
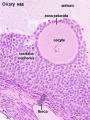
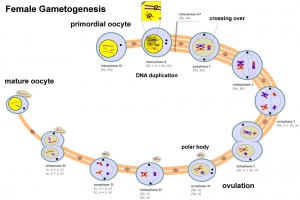
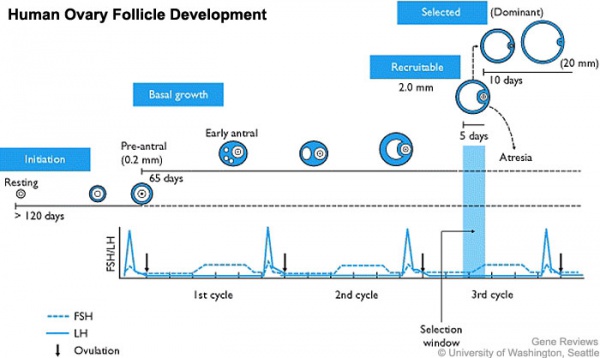
Female - Oogenesis
The ovaries have two functions.
- produce the female gametes or oocytes
- produce female hormones, estrogen and progesterone (secondary sex characteristics, menstrual cycle)
In an adult human female the development of a primordial follicle containing an oocyte to a preovulatory follicle takes in excess of 120 days.
Human Follicle Development |
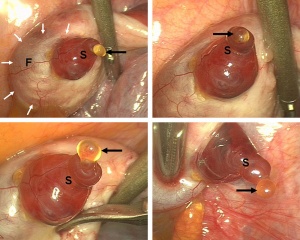
Human Ovulation |
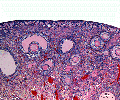
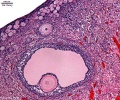
Human ovary follicle development
- Ovarian Follicle Stages: primordial follicle - primary follicle - secondary follicle - tertiary follicle - preovulatory follicle
Follicle cells (support cells) Theca cells (produce hormone)
| Ovulation Movie | |
|---|---|
| <html5media height="300" width="370">File:Follicle_001.mp4</html5media> | <html5media height="270" width="320">File:Ovulation 001.mp4</html5media> |
Meiosis Differences
Male Meiosis
Female Meiosis
Polar Bodies
| Meiosis Polar Body Movie | |||
|---|---|---|---|
| <html5media height="400" width="800">File:Oocyte_Meiosis_01.mp4</html5media>
Click Here to play on mobile device This movie shows labelled DNA (blue) of the mouse oocyte in meiosis 1 segregating DNA to the first polar body. The fluorescence image shows labelled actin (green) that lies directly under the plasma membrane of the oocyte and polar body.
|
Fertilization
Gamete formation, menstrual cycle and fertilisation will also be covered in detail in this week's Laboratory. Fertilization is the complete process resulting in the fusion of haploid gametes, egg and sperm, to form the diploid zygote. The recent development of aided fertilization is described as in vitro fertilization (in vitro = "in glass", outside the body, IVF). Clinically, all these aided fertilization techniques are grouped as Assisted Reproductive Technologies or ART.
- Oogenesis - 1 gamete produced/meiosis + 3 polar bodies, meiosis is slow, 1 egg produced and released at ovulation
- Spermatogenesis - 4 gametes produced/meiosis, meiosis is fast, 200-600 million sperm released at ejaculation
| Fertilization Movies | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <html5media height="500" width="640">File:Human fertilization 01.mp4</html5media> | |||||||||||||||
|
|
|
| ||||||||||||
Fertilization Site
- Fertilization resulting in embryo development usually occurs in first 1/3 of uterine tube (oviduct, Fallopian tube)
- The majority of fertilized oocytes do not go on to form an embryo
- Fertilization can also occur outside uterine tube associated with Assisted Reproductive Technology (IVF, GIFT, ZIFT...) and ectopic pregnancy
- Oocyte ovulation - release from the ovary with associated cells, into peritoneal cavity, uterine tube fimbria then into uterine tube (oviduct, uterine horn, fallopian tube) and epithelial cilia mediated movement.
- Spermatozoa ejaculation - deposited in vagina, movement of tail to "swim" in uterine secretions through cervix, uterine body and into uterine tube, have approximately 24-48h to fertilize oocyte.
Prior to the fertilization process commencing both the gametes complete of a number of biological processes.
- Oocyte Meiosis - completes Meiosis 1 and commences Meiosis 2 (arrests at Metaphase II).
- Spermatozoa Capacitation - following release (ejaculation) and mixing with other glandular secretions, activates motility and acrosome preparation.
- Migration - both oocyte and spermatozoa.
Endocrinology - Diagram of the comparative anatomy of the male and female reproductive tracts
| Gamete Movement Movies |
|---|
| Oocyte Motility
<html5media height="500" width="640">File:Bovine uterine tube oocyte transport 1.mp4</html5media> |
| Spermatozoa Motility
<html5media height="520" width="512">File:Spermatozoa_motility_01.mp4</html5media> |
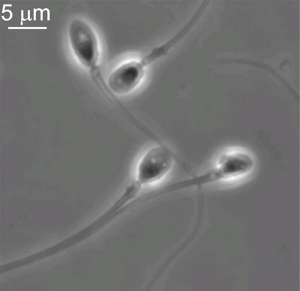
Fertilization - Male
Spermatozoa: Ejaculation - Capacitation - Spermatozoa motility - Chemotaxis - Binding to zona pellucida - Acrosome reaction - Membrane fusion
Ejaculation
- about 3.5 ml, containing 200 - 600 million spermatozoa
- by volume less than 10 % spermatozoa
- accessory glands contribute majority of volume (60 % seminal vesicle, 10 % bulbourethral, 30 % prostate)
Male Infertility
- Oligospermia (Low Sperm Count) - less than 20 million sperm after 72 hour abstinence from sex
- Azoospermia (Absent Sperm) - blockage of duct network
- Immotile Cilia Syndrome - lack of sperm motility
Capacitation
- spermatozoa activation process - removal of glycoprotein coat and seminal proteins and alteration of sperm mitochondria
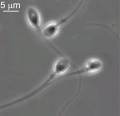
Spermatozoa motility
- tail of spermatozoa provide movement by microtubules
- energy for this movement is provided by mitochondria in tail initial segment
Chemotaxis
- oocyte cumulus cells release progesterone (may also be other oocyte and follicular fluid factors)
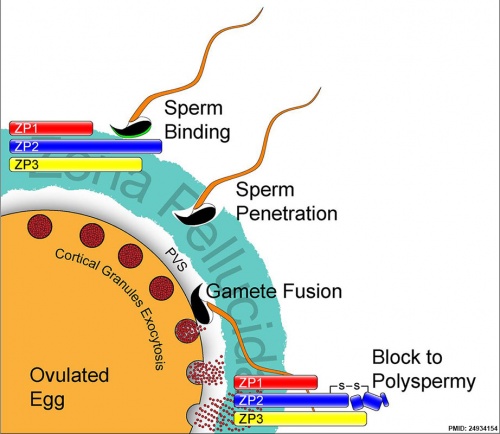
Spermatozoa Binding
- Zona pellucida protein ZP3 acts as receptor for spermatozoa binding (species specific)
Acrosome Reaction
- exocytosis of acrosome contents (calcium mediated) MBoC - Figure 20-31. The acrosome reaction that occurs when a mammalian sperm fertilizes an egg
- enzymes to digest the zona pellucida
- exposes sperm surface proteins to bind ZP2
Membrane fusion
- between spermatozoa and oocyte cell membranes, allows sperm nuclei passage into egg cytoplasm
- membrane fusion also initiates oocyte processes to block polyspermy
Fertilization - Oocyte
Oocyte: Membrane depolarization - Cortical reaction - Meiosis 2 completion
Membrane Depolarization
- caused by spermatozoa membrane fusion, acts as primary block to polyspermy (fertilisation by more than one spermatozoa)
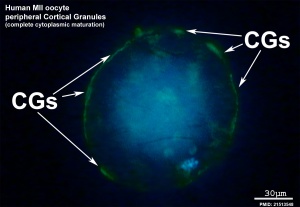
Cortical Reaction
- Inositol triphosphate (IP3) pathway elevates intracellular calcium, exocytosis of cortical granules
- enzyme alters ZP2 so it will no longer bind sperm plasma membrane
- MBoC - Figure 20-32. How the cortical reaction in a mouse egg is thought to prevent additional sperm from entering the egg
Meiosis 2
- completion of 2nd meiotic division
- forms second polar body (third polar body may be formed by meiotic division of the first polar body)
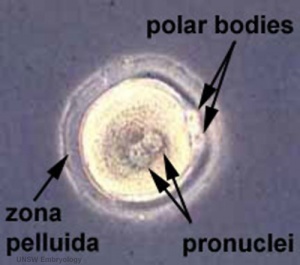
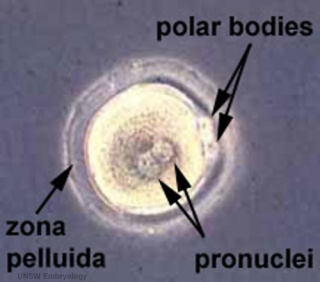
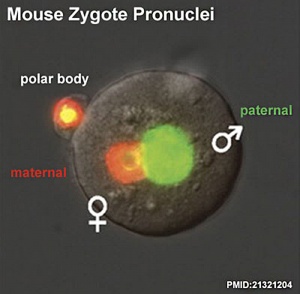
Formation of the Zygote
| Early Zygotes | |
|---|---|
|
|
| Human Zygote | Mouse Zygote |
- Pronuclei - Male and Female haploid nuclei approach each other and nuclear membranes break down
- chromosomal pairing, DNA replicates, first mitotic division
- Sperm contributes - centriole which organizes mitotic spindle
- Oocyte contributes - mitochondria (maternally inherited)
Sex Determination
- based upon whether an X or Y carrying sperm has fertilized the egg, should be 1.0 sex ratio.
- actually 1.05, 105 males for every 100 females, some studies show more males 2+ days after ovulation.
- cell totipotent (equivalent to a stem cell, can form any tissue of the body)
Men - Y Chromosome
- Y Chromosome carries Sry gene, protein product activates pathway for male gonad (covered in genital development)
Women - X Chromosome
- Gene dosage, one X chromosome in each female embryo cell has to be inactivated
- process is apparently random and therefore 50% of cells have father's X, 50% have mother's X
- Note that because men only have 1 X chromosome, if abnormal, this leads to X-linked diseases more common in male that female where bothe X's need to be abnormal.
Abnormalities
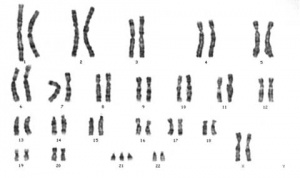
- The most common chromosome abnormality is aneuploidy, the gain or loss of whole chromosomes.
- Caused by meiotic nondisjunction, the failure of chromosomes to correctly separate homologues during meiosis I or sister chromatids during meiosis II.
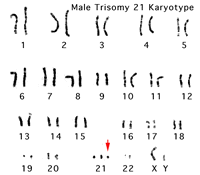
- Down Syndrome - caused by an extra copy of chromosome 21. Trisomy 21 (Down Syndrome)Maternal Age
- Chromosomal translocations occur when there is an inappropriate exchange of chromosomal material. Philadelphia chromosome
- Philadelphia chromosome - piece of Chr9 exchanged with Chr22 Generates truncated abl, overstimulates cell production, leads to chronic myelogenous leukemia
- Complete Mole - Only paternal chromosomes (no oocyte nucleus contribution)
- Partial Mole - 3 sets of chromosomes ( (triploidy) instead of the usual 2 (2 spermatozoa contribution)
UNSW Embryology Links
- spermatozoa oocyte fertilization Trisomy 21 (Down Syndrome)
| Cell Division Links: meiosis | mitosis | Lecture - Cell Division and Fertilization | spermatozoa | oocyte | fertilization | zygote | Genetics |
References
Online Textbooks
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000Figure 2.9. Summary of meiosis | fusion of egg and sperm plasma membranes
- Molecular Biology of the Cell 4th ed. Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 - IV. Internal Organization of the Cell Chapter 17. The Cell Cycle and Programmed Cell Death Programmed Cell Death | An Overview of the Cell Cycle | Figure 17-1. The cell cycle | Fertilization
- Molecular Cell Biology by Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E. New York: W. H. Freeman & Co.; c1999 Chapter 13. Regulation of the Eukaryotic Cell Cycle Regulation of the Eukaryotic Cell Cycle | Overview of the Cell Cycle and Its Control | Figure 13-2. Current model for regulation of the eukaryotic cell cycle | Movies Proposed alternative mechanisms for chromosome congression. | Centromeric attachment of microtubules. | The stages of mitosis and cytokinesis in an animal cell.
- The Cell - A Molecular Approach by Cooper, Geoffrey M. Sunderland (MA): Sinauer Associates, Inc.; c2000- IV. Cell Regulation Chapter 14. The Cell Cycle The Eukaryotic Cell Cycle | Figure 14.1. Phases of the cell cycle | Figure 14.32. Comparison of meiosis and mitosis | Figure 14.37. Meiosis of vertebrate oocytes
- HSTAT - In Vitro Fertilization As A Medical Treatment For Male or Female Infertility
- MBoC MBoC - Figure 20-18. Influence of Sry on gonad development
- Endocrinology Endocrinology - Comparative anatomy of male and female reproductive tracts
Search
- Bookshelf cell division | mitosis | meiosis | fertilization
- Pubmed cell division | mitosis | meiosis | fertilization
Reviews
<pubmed>18163464</pubmed> <pubmed>17189856</pubmed>
Terms
Note there are additional glossaries associated with genital, spermatozoa, oocyte and renal.
- acroplaxome - structure forms the acrosome plate with intermediate filament bundles of the marginal ring at the leading edge of the acrosome. The sub-acrosomal layer located in the developing spermatozoa head perinuclear region, located between the inner acrosomal membrane and the nuclear envelope. The other part of the perinuclear region is the post-acrosomal sheath (PAS) at the post-acrosomal region.
- acrosome - Cap-shaped cellular structure formed from the golgi apparatus and contains enzymes to dissolve the oocyte (egg) zona pellucida for fertilisation.
- acrosome compaction - Acrosome reshaping process in final stages of spermatogenesis (spermatid to spermatozoa).
- acrosome reaction - Chemical change within the spermatozoa following binding to the zona pellucida, only acrosome reacted spermatozoa have an ability to fuse with oocytes.
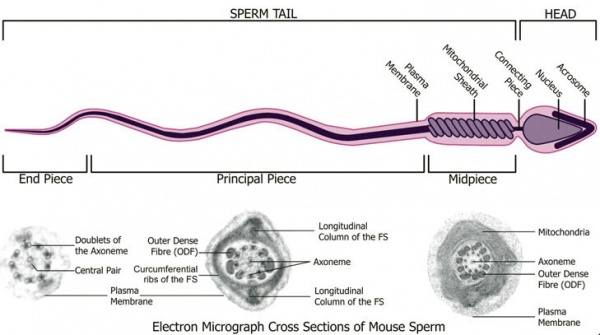
- annulus - Cytoskeletal (septin) structure located between the midpiece and principal piece regions of the tail, thought to form a diffusion barrier between these two domains. PMID 20042538
- asthenozoospermia - (asthenospermia) Term for reduced sperm motility and can be the cause of male infertility.
- axoneme - (axonema) The basic structure in cilia and eukaryotic flagella and in the spermatozoa tail, consisting of parallel microtubules in a characteristic "9 + 2" pattern. This pattern describes 9 outer microtubule doublets (pairs) surrounding 2 central singlet microtubules, in humans 50 μm long. The motor protein dynenin move the outer microtubules with respect to the central pair, bending the cilia and generating motility. Note that prokaryotic bacteria have a similar process (flagellum) that uses an entirely different mechanism for motility.
- blood-testis barrier - (BTB) Formed by tight junctions, basal ectoplasmic specializations, desmosome-like junctions and gap junctions between adjacent sertoli cells near the basement membrane of the seminiferous epithelium. image - BTB | image - tight junction
- capacitation - term describing the process by which spermaozoa become capable of fertilizing an oocyte, requires membrane changes, removal of surface glycoproteins and increased motility.
- caput - proximal head of the epididymis, epithelium with stereocilia, involved in absorbing fluid to concentrate spermatozoa. Underlying smooth muscle aids movement. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- CatSper - cationic (Ca2+) channel of spermatozoa, progesterone activated involved in hyperactivation, acrosome reaction, and possibly chemotaxis.
- cauda - distal tail of the epididymis, region with a thin epithelium and the greatest quantity of smooth muscle. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- centriole - a microtubule organising centre. First required for axoneme formation (distal centriole) that is lost and a second for pronuclei formation (proximal) following fertilisation. Rodents loose both and only have maternal centrioles.
- connecting piece - linkage between the spermatozoa head and the midpiece of the tail. PMID 22767409
- corpus - elongated body of the epididymis, This has an intermediate thickness of epithelium and thicker smooth muscle layer than caput. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- cytoplasmic bridges - Transient cytoplasm connections between spermatids arising from one spermatogonium due to incomplete cytokinesis.
- diploid - (Greek, di = double + ploion = vessel) Having two sets of chromosomes, the normal state for all cells other than the gametes.
- end piece - Last portion of the spermatozoa tail region.
- epididymis - testis tubular structure connecting the efferent ducts to the ductus deferent and functions for the storage and maturation of spermatozoa. Epididymis three main parts : caput (head), corpus (body), cauda (tail). PMID27307387
- fibrous sheath - cytoskeletal structure surrounding the axoneme and outer dense fibers, defining the extent of the principal piece region.
- haploid - (Greek, haploos = single) Having a single set of chromosomes as in mature germ/sex cells (oocyte, spermatozoa) following reductive cell division by meiosis. Normally cells are diploid, containing 2 sets of chromosomes.
- interstitial cell - (Leydig cell) Male gonad (testis) cell which secrete the androgen testosterone, beginning in the fetus.
- interstitium - testis developmental region (space between testis cords) that generates Leydig cells and other less well characterized cell types.
- Johnsen score - a clinical score (1-10) for assessing spermatogenesis in a human testicular biopsy. Named after the author of the original article. PMID 5527187
- Leydig cell - (interstitial cell) Male gonad (testis) cell that secrete the androgen testosterone, beginning in the fetus. Fetal Leydig cells develop from coelomic epithelium and undifferentiated perivascular cells in the gonad–mesonephros border region. Adult Leydig cells appear after birth from stem/progenitor cells among peritubular and peri-vascular cells. Leydig cells were first histologically identified in 1850 by Franz von Leydig (1821 - 1908) a German scientist.
- meiosis - The cell division that occurs only in production of germ cells where there is a reduction in the number of chromosomes (diploid to haploid) which is the basis of sexual reproduction. All other non-germ cells in the body divide by mitosis.
- midpiece - (middle piece) spermatozoa tail initial segment of axoneme surrounded outer dense fibres then by mitochondria. Next in the tail is the principal piece then finally the end piece.
- mitosis - The normal division of all cells, except germ cells, where chromosome number is maintained (diploid). In germ cell division (oocyte, spermatozoa) meiosis is a modified form of this division resulting in reduction in genetic content (haploid). Mitosis, division of the nucleus, is followed by cytokinesis the division of the cell cytoplasm and the cytoplasmic contents. cytokinesis overlaps with telophase.
- outer dense fibres - (ODF, outer dense fibers) cytoskeletal structures that surround the axoneme in the middle piece and principal piece of the spermatozoa tail.
- primary spermatocyte - arranged in the seminiferous tubule wall deep (luminal) to the spermatogonia. These large cells enter the prophase of the first meiotic division. (More? meiosis)
- principal piece - Spermatozoa tail segment containing the plasma membrane calcium channels (CatSper1 and CatSper2) required for hyperactivation of motility. Region is partially separated from the midpiece by a barrier called the annulus.
- sertoli cells - (sustentacular cell) These cells are the spermatozoa supporting cells, nutritional and mechanical, as well as forming a blood-testis barrier. The cell cytoplasm spans all layers of the seminiferous tubule. The cells are named after Enrico Sertoli (1842 - 1910), and italian physiologist and histologist.
- sperm annulus - (Jensen's ring; Latin, annulus = ring) A region of the mammalian sperm flagellum connecting the midpiece and the principal piece. The annulus is a septin-based structure formed from SEPT1, 4, 6, 7 and 12. Septins are polymerizing GTPases that can act as a scaffold forming hetero-oligomeric filaments required for cytokinesis and other cell cycle roles.
- spermatogenesis - (Greek, genesis = origin, creation, generation) The term used to describe the process of diploid spermatagonia division and differentiation to form haploid spermatazoa within the testis (male gonad). The process includes the following cellular changes: meiosis, reoorganization of DNA, reduction in DNA content, reorganization of cellular organelles, morphological changes (cell shape). The final process of change in cell shape is also called spermiogenesis.
- spermatogenesis - (Greek, genesis = origin, creation, generation) The maturation process of the already haploid spermatazoa into the mature sperm shape and organization. This process involves reorganization of cellular organelles (endoplasmic reticulum, golgi apparatus, mitochondria), cytoskeletal changes (microtubule organization) and morphological changes (cell shape, acrosome and tail formation).
- spermatogonia - The cells located in the seminiferous tubule adjacent to the basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
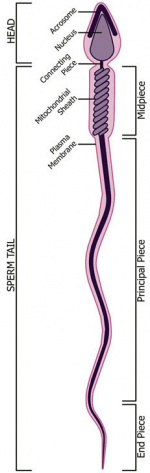
- spermatozoa head - Following spermiogenesis, the first region of the spermatozoa containing the haploid nucleus and acrosome. In humans, it is a flattened structure (5 µm long by 3 µm wide) with the posterior part of nuclear membrane forming the basal plate region. The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and spermatozoa tail).
- spermatozoa neck - Following spermiogenesis, the second region of the spermatozoa attached to basal plate, transverse oriented centriole, contains nine segmented columns of fibrous material, continue as outer dense fibres in tail. In humans, it forms a short structure (1 µm). The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and tail).
- spermatozoa tail - Following spermiogenesis, the third region of the spermatozoa that has a head, neck and tail). The tail is also divided into 3 structural regions a middle piece, a principal piece and an end piece. In humans: the middle piece (5 µm long) is formed by axonema and dense fibres surrounded by mitochondria; the principal piece (45 µm long) fibrous sheath interconnected by regularly spaced circumferential hoops; the final end piece (5 µm long) has an axonema surrounded by small amount of cytoplasm and plasma membrane.
- spermatogonial stem cells - (SSCs) The spermatagonia cells located beside the seminiferous tubule basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (|Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
- spermatozoon - singular form of of spermatozoa.
- sperm protein 56 - A component of the spermatozoa acrosomal matrix released to the sperm surface during capacitation.
- teratospermia - Clinical term for a spermatozoa with abnormal morphology (small, large, defects in the head, tail, and/or mid-piece) present in the semen or ejaculate.
- testis cords - developmental structure that give rise to the adult seminiferous tubules, the other developmental region is the interstitium.
- vasectomy - Clinical term for ligation of the scrotal portion of the ductus deferens.
See also: Spermatozoa Terms collapse table
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- McGraw-Hill Animation comparing Mitosis and Meiosis
- Salmon Lab Mitosis Movies
Cite this page: Hill, M.A. (2024, April 25) Embryology Lecture - Fertilization. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Lecture_-_Fertilization
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G