Lecture - Early Vascular Development
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This lecture is an introduction to the events in early embryonic development that relate to mesoderm and early cardiovascular development. Texts frequently separate heart development from vascular development in order to simplify their descriptions of cardiovascular development, although the two are functionally and embryonically connected.
Note that later in the course, the late development of the heart and vascular changes will be further discussed. The complexity of septation, cardiac outflow separation, remodelling of the peripheral vasculature, and the pre- to post-natal changes may also contribute to the relatively large proportion of birth defects associated with this system. These events of vascular development are covered in a later lecture.
It is important to note also that we are just beginning to understand vascular development which involves the careful orchestration of a variety of moleculular mechanisms. Development does appear to be an independent mechanism preceding both skeletal and smooth muscle development and using different regulatory mechanisms. In the next few years, there are certain to be new molecules identified as well as an understanding and appreciation of new roles for known molecules.
- Vasculogenesis - formation of new blood vessels assemble from individual precursor cells.
- Angiogenesis - sprouting of new vessels occurs from pre-existing vessels.
Lecture Objectives
- Understanding of mesoderm development
- Understanding of heart tube formation and early development
- Understanding of early blood vessel and blood development
- Brief understanding of vascular growth and regression
- Brief understanding of vascular growth factors
Lecture Resources
| Movies | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| References | |
|---|---|
| Hill, M.A. (2020). UNSW Embryology (20th ed.) Retrieved April 25, 2024, from https://embryology.med.unsw.edu.au |
|
| Moore, K.L., Persaud, T.V.N. & Torchia, M.G. (2015). The developing human: clinically oriented embryology (10th ed.). Philadelphia: Saunders. | The following chapter links only work with a UNSW connection. |
| Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R. & Francis-West, P.H. (2009). Larsen's human embryology (4th ed.). New York; Edinburgh: Churchill Livingstone. | The following chapter links only work with a UNSW UNSW Library subscription
|
| ECHO360 Recording |
|---|
|
Links only work with currently enrolled UNSW students. Lecture 7 - Rich Media Playback | Vodcast Playback | Podcast Playback |
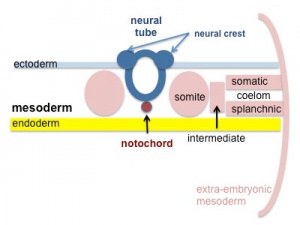
Development Overview
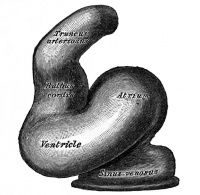
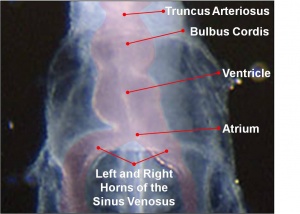
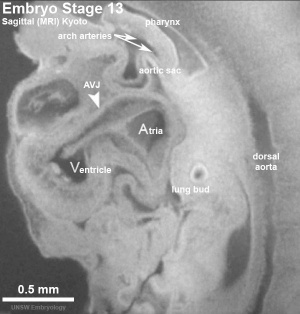
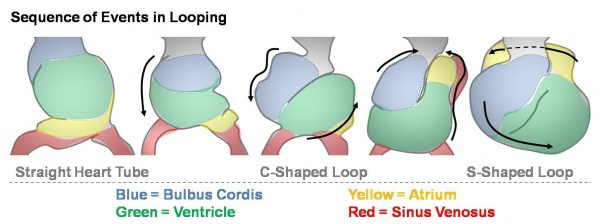
The heart develops from cardiogenic mesoderm that originally lies above the cranial end of the developing neural tube. Enlargement of the cranial neural fold brings this region ventrally to its correct anatomical position. The original paired cardiac tubes fuse, with the "ventricular" primordia initially lying above the "atria". Growth of the cardiac tube flexes it into an "S-shape" tube, rotating the "ventricles" downward and pushing the "atria" upward.
This is then followed by septation, a complex process which converts this simple tube into a four chambered heart and covered in a later lecture and lab. A key part of this process is the separation of cardiac outflow (truncus arteriosus) into a separate pulmonary and aortic arch outflow. During embryonic development there is extensive remodelling of the initially right and left symmetrical cardiovascular system and a contribution from the neural crest to some vessels.
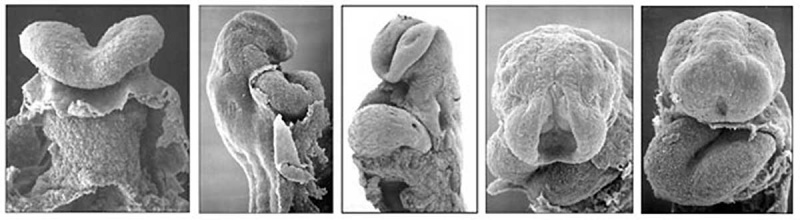
|
| The Human Heart from day 10 to 25 (scanning electron micrograph) |
Timecourse
Vasculogenesis and Angiogenesis
| Vasculogenesis | Angiogenesis |
|---|---|
| formation of new blood vessels (endothelium from mesoderm) |
formation of blood vessels from pre-existing vessels (occurs in development and adult) |
- Begins week 3 in extraembryonic mesoderm and then embryonic splanchnic mesoderm
- Begins as the formation of blood islands
- Earliest islands - yolk sac, connecting stalk and chorion (Area vasculosa)
- Growth factors stimulate growth and development - Vascular Endothelial Growth Factor (VEGF) and Placental Growth Factor (PlGF, PGF)
- Growing blood vessels follow a gradient generated by target tissues/regions of Vascular Endothelial Growth Factor (VEGF) to establish a vascular bed. Recent findings suggest that Notch signaling acts as an inhibitor for this system, preventing sprouting of blood vessels. Notch is a transmembrane receptor protein involved in regulating cell differentiation in many developing systems.
- PIGF is also a VEGF released from the placental trophoblast cells.
- angioblasts form clusters called "blood islands"
- blood islands extend and fuse together to form a primordial vascular network
Blood islands
- Blood islands contain cells (haemangioblasts) which are capable of differentiating into 2 populations of cells
- Vascular precursors (angioblasts) - form endothelial cells
- Blood cell precursors (haemocytoblasts)
- These angioblasts migrate, coalesce into cords and form a lumen. This process of vessel formation is called vasculogenesis and is dominant in very early embryogenesis e.g. formation of the dorsal aorta
- Sprouting from pre-existing vessels is called angiogenesis e.g. brain is an organ which is vascularized by this process
- Note: the vascular tree undergoes constant remodeling as the embryo grows.
Blood vessel lumen formation
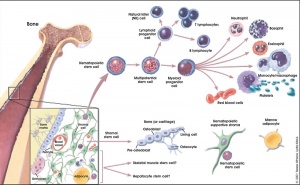
Blood formation
Red blood cells
Blood stem cells
Hematopoietic and stromal cell differentiation (adult)
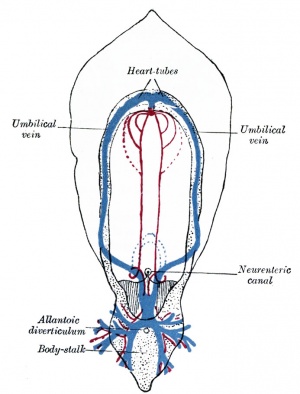
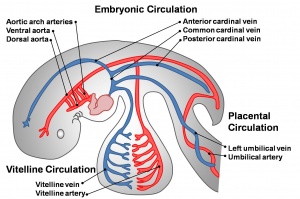
Early vascular systems
- one vascular system with 3 components - vitelline, embryonic (system) and placental
- each component has own system of artery and vein
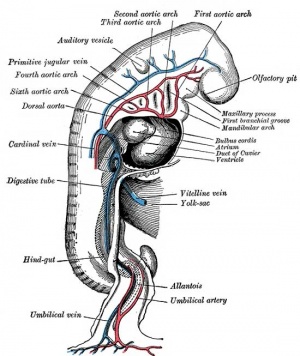
Vitelline blood vessels
- Angioblasts form a network of vessels over the yolk sac and connecting stalk
- Join into two main vessels, the vitteline veins (omphalomesenteric)
- Pass through vitello-intestinal duct (yolk sac stalk)
- Enter caudal end of cardiac tube
- Vitelline Arteries - arises from dorsal aorta, contribute to adult GIT arteries (fuse to become superior mesenteric artery (midgut)
- Vitelline Veins - empties into sinus venosus, contribute to the adult portal system
Embryo blood vessels
- (systemic) will form the most of the cardiovascular system
- some vessels have neural crest contribution
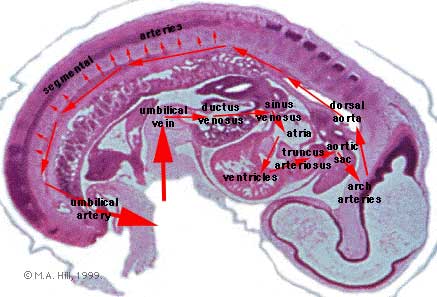
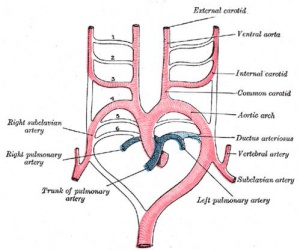
- Arterial blood flow - aortic sac → aortic arches → dorsal aorta →umbilical artery
- dorsal aorta,
- paired initially , later fuses from T4 to L4 (gives off segmental arteries)
- connect to ventral aorta via pharyngeal arches arteries.
- caudally, give rise to umbilical arteries
- laterally, give rise to intersegmental arteries
- dorsal aorta,
- Veins - 3 pairs of veins empty into the sinus venosus of the heart
- vitelline, umbilical (right and left from developing placenta enter caudal cardiac tube; only left persists)
- cardinal veins - anterior, common, posterior
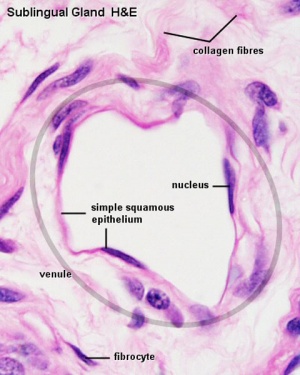
|
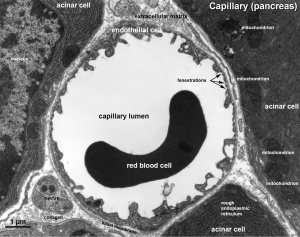
|
| Vein histology | Blood capillary (EM) |
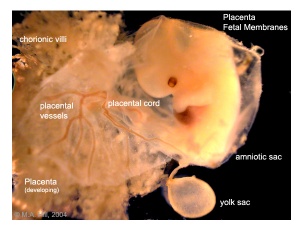
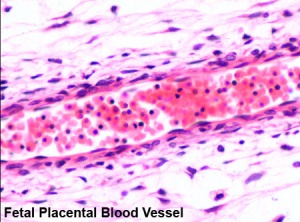
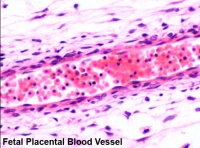
Placental blood vessels
(Placenta development covered in next lecture)
- form initially in the connecting stalk (then umbilical cord) and anastomose in chorion
- extend maternally - toward the chorionic villi
- extend embryonically - toward the sinus venosus and dorsal aorta
- Arteries - paired and carry deoxygenated blood (from dorsal aorta) and waste products to the placental villi
- Veins - paired initially then only left at end of embryonic period and carry oxygenated blood to the embryo (sinus venosus)
Blood flow through the embryo
High pressure pathway
Maternal Blood | -> umbilical vein -> liver -> anastomosis -> sinus venosus -> atria ventricles-> truncus arteriosus -> aortic sac -> aortic arches-> dorsal aorta-> pair of umbilical arteries | Maternal Blood.
Low pressure pathway
- Head - Large veins lateral to dorsal aortae. These are the superior or anterior cardinal veins. Their function is to drain the head region.
- Body - Large veins lateral to dorsal aortae. These are the inferior or posterior cardinal veins. Their function is to drain the lower part of the embryo.
Blood vessel remodeling
Early vascular development is laterally symmetrical (paired left and right). With embryo development this scheme is extensively remodelled leading to an asymmetric adult system in the body.
Complex balance between Stimulators and Inhibitors of Angiogenisis.
- Links: Blood Vessel Development
Vascular Endothelial Growth Factor (VEGF)
- belongs to the platelet derive growth factor (PDGF) family.
- required for early stages of blood vessel patterning.
- required later for endothelial cell maintenance in tissues.
- autocrine VEGF loop from endothelial cell secretion involved in vascular growth.
- 4 protein isoforms generated from a single gene.
VEGF protein family - VEGF (or VEGF-A), VEGF-B, VEGF-C, VEGF-D and placental growth factor (PGF),
VEGF receptors - VEGFR-1, -2 and -3.
- Cells expressing the receptors are directed in their growth.
- Note that there are other growth factor families (FGF, Tie, TGF-β, netrins, semaphorins) that can also influence vessel growth.
- Some angiogenic factors also involved in organ development (liver).
Heart Development
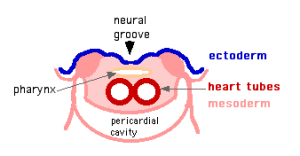 MH - Later development of the heart (septation) will be covered in another lecture.
MH - Later development of the heart (septation) will be covered in another lecture.
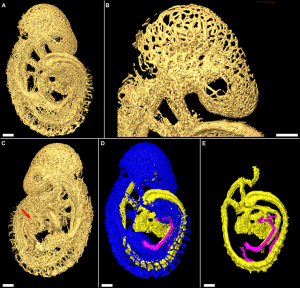
Mouse Model
| Mouse E9.5 heart (stage 10) | Mouse E8.5 - 14.5 heart (external) | Mouse E8.5 - 14.5 heart (internal) | 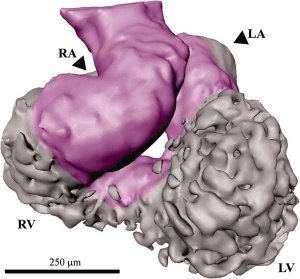
|

Early Human Heart Development
Heart layers
Heart looping
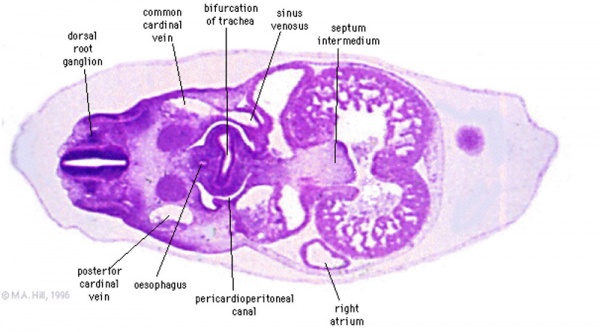
Transverse section- Heart is 2 tubes that fuse in the midline anterior to pharynx. The pericardial cavity can be imagined as the top of the "horseshoe" of the intraembryonic coelom. (where the arms become the pleural cavity and the ends fuse anteriorly to form a single peritoneal cavity). This view shows the initial positioning of the ventricles above the atria. The ventricles are rotated into their correct anatomical position by the growth of the heart tube, bending into an "S" shape. Initially...
Heart neural crest
Data from: Chan WY, Cheung CS, Yung KM, Copp AJ. Chan WY, Cheung CS, Yung KM, Copp AJ. Cardiac neural crest of the mouse embryo: axial level of origin, migratory pathway and cell autonomy of the splotch (Sp2H) mutant effect. Development. 2004 Jul;131(14):3367-79. PMID: 15226254
Embryonic heart rate
Internet LinksEmbryo Images Unit: Embryo Images Online Early Cell Populations (cardiogenic section) | Cardiovascular Development | Week 3 Development | Week 4 Development | Heart Chambers and Outflow Tract | Atrioventricular Septation | Outflow Tract Septation | Ventricular Septation | Atrial Septation | Atrial Walls Aortic Arch Vessels | Changes at Birth ReferencesOnline Textbooks
Terms
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||