Gastrointestinal Tract - Postnatal: Difference between revisions
mNo edit summary |
|||
| (38 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
==Introduction== | ==Introduction== | ||
[[Image:Newborn.jpg|thumb|Newborn infant]] | |||
This page is an introduction to postnatal gastrointestinal tract development. This nutritionally involves a change from prenatal placental vascular nutrition to postnatal oral colostrum/milk enteral nutrition (enteral = nutritient delivery as fluid into the gastrointestinal tract). Also look at the topic of Milk in relationship to neonatal nutrition. The postnatal gastrointestinal tract development is also about increased activity of the tract and associated organs as well as the populating with intestinal flora in the tract. This is also the pathway for initial passive immunity through absorption of maternal immunoglobulin from breast milk. | This page is an introduction to postnatal gastrointestinal tract development. This nutritionally involves a change from prenatal placental vascular nutrition to postnatal oral colostrum/milk enteral nutrition (enteral = nutritient delivery as fluid into the gastrointestinal tract). Also look at the topic of Milk in relationship to neonatal nutrition. The postnatal gastrointestinal tract development is also about increased activity of the tract and associated organs as well as the populating with intestinal flora in the tract. This is also the pathway for initial passive immunity through absorption of maternal immunoglobulin from breast milk. | ||
These notes should be read in conjunction with the related page on | These notes should be read in conjunction with the related page on {{Milk}} and an understanding of prenatal [[Gastrointestinal Tract Development]]. | ||
:{{Gastrointestinal Tract Links}} | |||
== Some Recent Findings == | == Some Recent Findings == | ||
| Line 8: | Line 12: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* | * '''Review - Physiology of the Neonatal Gastrointestinal System Relevant to the Disposition of Orally Administered Medications'''{{#pmid:30567878|PMID30567878}} "A thorough knowledge of the newborn (age, birth to 1 month postpartum) infant's gastrointestinal tract (GIT) is critical to the evaluation of the absorption, distribution, metabolism, and excretion (ADME) of orally administered drugs in this population. Developmental changes in the GIT during the newborn period are important for nutrient uptake as well as the disposition of orally administered medications. Some aspects of gastrointestinal function do not mature until driven by increased dietary complexity and nutritional demands later in the postnatal period. The functionalities present at birth, and subsequent maturation, can also impact the ADME parameters of orally administered compounds. This review will examine some specific contributors to the ADME processes in human neonates, as well as what is currently understood about the drivers for their maturation. Key species differences will be highlighted, with a focus on laboratory animals used in juvenile toxicity studies. Because of the gaps and inconsistencies in our knowledge, we will also highlight areas where additional study is warranted to better inform the appropriate use of medicines specifically intended for neonates." | ||
* '''Restricting microbial exposure in early life negates the immune benefits associated with gut colonization in environments of high microbial diversity'''{{#pmid:22216092|PMID22216092}} "Acquisition of the intestinal microbiota in early life corresponds with the development of the mucosal immune system. Recent work on caesarean-delivered infants revealed that early microbial composition is influenced by birthing method and environment. Furthermore, we have confirmed that early-life environment strongly influences both the adult gut microbiota and development of the gut immune system. Here, we address the impact of limiting microbial exposure after initial colonization on the development of adult gut immunity." | |||
* '''Gram negative bacteria are associated with the early stages of necrotizing enterocolitis.'''{{#pmid:21445365|PMID21445365}} "In a mouse model of the disease "Citrobacter, Klebsiella, and Tatumella are associated with Necrotizing enterocolitis (NEC). Differential colonic bacteria were identified despite the lack of inflammatory mediator elevation traditionally associated with NEC. This suggests a temporal relationship between bacteria and inflammatory mediators such that alterations in gut microbiota are associated with early NEC, while inflammatory mediator elevation is associated with advanced NEC." | |||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! More recent papers | |||
|- | |||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | |||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Postnatal+Gastrointestinal+Tract ''Postnatal Gastrointestinal Tract''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Postnatal+gut+colonization ''Postnatal Gut Colonization''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=necrotizing+enterocolitis ''necrotizing enterocolitis''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Meconium ''Meconium''] | |||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
|} | |||
==Small Intestine Length== | |||
Small intestine growth in length prenatally is initially linear during the first half pregnancy (to 32 cm CRL), followed by rapid growth in the last 15 weeks doubling the overall length. | |||
Postnatally, growth continues rapidly but after 1 year slows again to a linear increase to adulthood.{{#pmid:1752463|PMID1752463}} | |||
{| | |||
|+ '''Small Intestine Length''' | |||
|-bgcolor="CEDFF2" | |||
| width=200px|'''Age''' (weeks gestational age) | |||
| width=200px|'''Average Length''' (cm) | |||
|- | |||
| 20 | |||
| 125 | |||
|- | |||
| 30 | |||
| 200 | |||
|- | |||
| term | |||
| 275 | |||
|-bgcolor="F5FAFF" | |||
| 1 year postnatal | |||
| 380 | |||
|-bgcolor="F5FAFF" | |||
| 5 years | |||
| 450 | |||
|-bgcolor="F5FAFF" | |||
| 10 years | |||
| 500 | |||
|-bgcolor="F5FAFF" | |||
| 20 years | |||
| 575 | |||
|- | |||
| colspan=2|Table data from 8 published reports of necropsy measurement of 1010 guts.{{#pmid:1752463|PMID1752463}} | |||
|} | |} | ||
:'''Links:''' [[Gastrointestinal_Tract_-_Intestine_Development|Intestine Development]] | |||
==Lipid Signalling== | ==Lipid Signalling== | ||
Lipids present in the intestine leads to a reduction in nutrient intake. Recent research has shown that lipids present in the intestine can also regulate endogenous nutrient production. | Lipids present in the intestine leads to a reduction in nutrient intake. Recent research has shown that lipids present in the intestine can also regulate endogenous nutrient production.{{#pmid:18401341|PMID18401341}} | ||
Signalling pathway: | '''Signalling pathway:''' presence of ingested lipids - intestinal lipid sensors - signal to the brain - liver - reduction in endogenous glucose production | ||
==Insulin-like Growth Factors== | |||
Some evidence to suggest that in preterm infants IGFBP-2 and IGF-II present in breast milk may have an important role in their early development. | |||
* insulin-like growth factors (IGFs) and IGF binding proteins (IGFBPs) | |||
==Gut Microorganism Population== | |||
The normal newborn gastrointestinal tract contains little if any microorganisms (commensal intestinal microbiota, microbiota, flora, microflora). | |||
Postnatally, the tract has to be populated by microorganisms, which are mainly anaerobic bacteria and then aerobic bacteria, but may also include yeast and fungi. The foregut comparatively has few microorganisms when compared to the midgut and hindgut. | |||
There are several infectious pathogens that can populate the postnatal gut leading to a number of different diseases: Escherichia coli (enterotoxigenic), Shigella (a gram-negative, non-spore forming rod-shaped bacteria infectious through poor hygeine and ingestion, fecal–oral contamination. More? Dysentery), Vibrio cholerae and Listeria. | |||
Antibiotics - Treatment of other neonatal infections systemically with antibiotics can alter the bacterial population. | |||
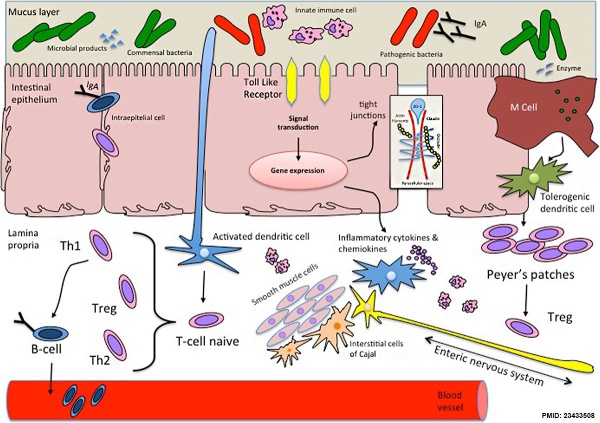
[[File:Intestinal_function_and_microbiota_01.jpg|alt=cartoon Intestinal function and microbiota|600px]] | |||
== | Cartoon showing relationship between microbiota and intestinal function{{#pmid:23433508|PMID23433508}} | ||
:'''Links:''' [[Immune_System_Development|Immune System]] | [[Abnormal_Development_-_Bacterial_Infection|Bacterial Infection]] | [http://www.ncbi.nlm.nih.gov/books/NBK7670 Medical Microbiology - Microbiology of the Gastrointestinal Tract] | |||
==Meconium== | |||
As introduced in fetal development, meconium is formed from gut and associated organ secretions as well as cells and debris from the swallowed amniotic fluid. Meconium accumulates during the fetal period in the large intestine (bowel). It can be described as being a generally dark colour (green black) , sticky and odourless. | |||
* In fetal development, meconium is formed from gut and associated organ secretions as well as cells and debris from the swallowed amniotic fluid. | |||
* Meconium accumulates during the fetal period in the large intestine (bowel). | |||
* It can be described as being a generally dark colour (green black) , sticky and odourless. | |||
Normally this meconium is defaecated (passed) postnatally over the first 48 hours and then transitional stools from day 4. | |||
Abnormally this meconium is defaecated in utero, due to oxygen deprivation and other stresses, causing: | |||
* '''meconium stained liquor''' (damaging {{placenta}} and placental blood vessels) | |||
* {{meconium aspiration syndrome}} (MAS) (damaging {{respiratory}} development) | |||
==Abnormalities== | |||
===Meconium Aspiration Syndrome=== | |||
* Premature discharge into the amniotic sac can lead to mixing with amniotic fluid and be reswallowed by the fetus. | |||
* This is meconium aspiration syndrome and can damage both the developing lungs and placental vessels. | |||
* Absence or delayed passage of meconium may indicate conditions associated with meconium plugs or more seriously, Hirshsprung's disease (aganglionic colon, megacolon). | |||
* Delayed conversion to transitional stools may indicate a feeding issue. | |||
===Infections=== | |||
There are several infectious pathogens that can populate the postnatal gut leading to a number of different diseases: | |||
* Escherichia coli (enterotoxigenic) | |||
* Shigella a gram-negative, non-spore forming rod-shaped bacteria infectious through poor hygeine and ingestion, fecal–oral contamination. (More? Dysentery) | |||
* Vibrio cholerae | |||
* Listeria | |||
:'''Links:''' [[Abnormal_Development_-_Bacterial_Infection|Bacterial Infection]] | |||
===Necrotizing Enterocolitis=== | |||
* (NEC) is a disease affecting infants born prematurely (mortality rate of 15-30%) | |||
* up to 40% of afflicted premature infants require intestinal resection | |||
* usually occurs in the second week of life after the initiation of enteral feeds | |||
* pathogenesis is multifactorial | |||
* appears to involve an overreactive response of the immune system to an insult. | |||
* increased intestinal permeability, bacterial translocation, and sepsis. | |||
{| | |||
| [[File:Neonate_necrotizing_enterocolitis_bacteria_colonizing_intestinal_tissue.jpg|400px]] | |||
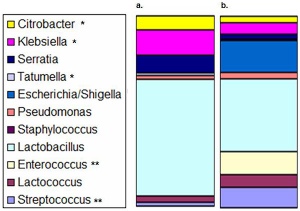
| [[File:Mouse - analysis of colonic microbiota.jpg|300px]] | |||
|- | |||
| Neonate (human) necrotizing enterocolitis bacteria colonizing intestinal tissue.{{#pmid:21486476|PMID21486476}} | |||
| Mouse model analysis of colonic microbiota.{{#pmid:21445365|PMID21445365}} Mice with NEC (a) are compared to mice without NEC (b). <nowiki>*</nowiki> indicates statistically significantly more in mice with NEC. <nowiki>**</nowiki> indicates statistically significantly more in mice without NEC. | |||
|} | |||
:'''Links:''' [http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002133/ PubMed Health] | [http://www.nlm.nih.gov/medlineplus/ency/article/001148.htm MedlinePlus] | |||
==References== | ==References== | ||
<references/> | <references/> | ||
===Reviews=== | |||
{{#pmid:30567878}} | |||
{{#pmid:22983847}} | |||
{{#pmid:15156063}} | |||
===Articles=== | |||
{{#pmid:17594176}} | |||
{{#pmid:12496227}} | |||
{{#pmid:2330231}} | |||
===Search Pubmed=== | ===Search Pubmed=== | ||
'''Search Pubmed Now:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=postnatal%20gastrointestinal%20development postnatal | '''Search Pubmed Now:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=postnatal%20gastrointestinal%20development postnatal gastrointestinal development] | ||
==External Links== | |||
{{External Links}} | |||
{{Glossary}} | |||
{{ | {{Footer}} | ||
[[Category:Gastrointestinal Tract]] | [[Category:Gastrointestinal Tract]] | ||
Revision as of 23:27, 1 June 2019
Introduction
This page is an introduction to postnatal gastrointestinal tract development. This nutritionally involves a change from prenatal placental vascular nutrition to postnatal oral colostrum/milk enteral nutrition (enteral = nutritient delivery as fluid into the gastrointestinal tract). Also look at the topic of Milk in relationship to neonatal nutrition. The postnatal gastrointestinal tract development is also about increased activity of the tract and associated organs as well as the populating with intestinal flora in the tract. This is also the pathway for initial passive immunity through absorption of maternal immunoglobulin from breast milk.
These notes should be read in conjunction with the related page on milk and an understanding of prenatal Gastrointestinal Tract Development.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Postnatal Gastrointestinal Tract | Postnatal Gut Colonization | necrotizing enterocolitis | Meconium |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page. |
Small Intestine Length
Small intestine growth in length prenatally is initially linear during the first half pregnancy (to 32 cm CRL), followed by rapid growth in the last 15 weeks doubling the overall length.
Postnatally, growth continues rapidly but after 1 year slows again to a linear increase to adulthood.[4]
| Age (weeks gestational age) | Average Length (cm) |
| 20 | 125 |
| 30 | 200 |
| term | 275 |
| 1 year postnatal | 380 |
| 5 years | 450 |
| 10 years | 500 |
| 20 years | 575 |
| Table data from 8 published reports of necropsy measurement of 1010 guts.[4] | |
- Links: Intestine Development
Lipid Signalling
Lipids present in the intestine leads to a reduction in nutrient intake. Recent research has shown that lipids present in the intestine can also regulate endogenous nutrient production.[5]
Signalling pathway: presence of ingested lipids - intestinal lipid sensors - signal to the brain - liver - reduction in endogenous glucose production
Insulin-like Growth Factors
Some evidence to suggest that in preterm infants IGFBP-2 and IGF-II present in breast milk may have an important role in their early development.
- insulin-like growth factors (IGFs) and IGF binding proteins (IGFBPs)
Gut Microorganism Population
The normal newborn gastrointestinal tract contains little if any microorganisms (commensal intestinal microbiota, microbiota, flora, microflora).
Postnatally, the tract has to be populated by microorganisms, which are mainly anaerobic bacteria and then aerobic bacteria, but may also include yeast and fungi. The foregut comparatively has few microorganisms when compared to the midgut and hindgut.
There are several infectious pathogens that can populate the postnatal gut leading to a number of different diseases: Escherichia coli (enterotoxigenic), Shigella (a gram-negative, non-spore forming rod-shaped bacteria infectious through poor hygeine and ingestion, fecal–oral contamination. More? Dysentery), Vibrio cholerae and Listeria.
Antibiotics - Treatment of other neonatal infections systemically with antibiotics can alter the bacterial population.
Cartoon showing relationship between microbiota and intestinal function[6]
- Links: Immune System | Bacterial Infection | Medical Microbiology - Microbiology of the Gastrointestinal Tract
Meconium
As introduced in fetal development, meconium is formed from gut and associated organ secretions as well as cells and debris from the swallowed amniotic fluid. Meconium accumulates during the fetal period in the large intestine (bowel). It can be described as being a generally dark colour (green black) , sticky and odourless.
- In fetal development, meconium is formed from gut and associated organ secretions as well as cells and debris from the swallowed amniotic fluid.
- Meconium accumulates during the fetal period in the large intestine (bowel).
- It can be described as being a generally dark colour (green black) , sticky and odourless.
Normally this meconium is defaecated (passed) postnatally over the first 48 hours and then transitional stools from day 4.
Abnormally this meconium is defaecated in utero, due to oxygen deprivation and other stresses, causing:
- meconium stained liquor (damaging placenta and placental blood vessels)
- meconium aspiration syndrome (MAS) (damaging respiratory development)
Abnormalities
Meconium Aspiration Syndrome
- Premature discharge into the amniotic sac can lead to mixing with amniotic fluid and be reswallowed by the fetus.
- This is meconium aspiration syndrome and can damage both the developing lungs and placental vessels.
- Absence or delayed passage of meconium may indicate conditions associated with meconium plugs or more seriously, Hirshsprung's disease (aganglionic colon, megacolon).
- Delayed conversion to transitional stools may indicate a feeding issue.
Infections
There are several infectious pathogens that can populate the postnatal gut leading to a number of different diseases:
- Escherichia coli (enterotoxigenic)
- Shigella a gram-negative, non-spore forming rod-shaped bacteria infectious through poor hygeine and ingestion, fecal–oral contamination. (More? Dysentery)
- Vibrio cholerae
- Listeria
- Links: Bacterial Infection
Necrotizing Enterocolitis
- (NEC) is a disease affecting infants born prematurely (mortality rate of 15-30%)
- up to 40% of afflicted premature infants require intestinal resection
- usually occurs in the second week of life after the initiation of enteral feeds
- pathogenesis is multifactorial
- appears to involve an overreactive response of the immune system to an insult.
- increased intestinal permeability, bacterial translocation, and sepsis.
|
|
| Neonate (human) necrotizing enterocolitis bacteria colonizing intestinal tissue.[7] | Mouse model analysis of colonic microbiota.[3] Mice with NEC (a) are compared to mice without NEC (b). * indicates statistically significantly more in mice with NEC. ** indicates statistically significantly more in mice without NEC. |
- Links: PubMed Health | MedlinePlus
References
- ↑ Neal-Kluever A, Fisher J, Grylack L, Kakiuchi-Kiyota S & Halpern W. (2019). Physiology of the Neonatal Gastrointestinal System Relevant to the Disposition of Orally Administered Medications. Drug Metab. Dispos. , 47, 296-313. PMID: 30567878 DOI.
- ↑ Mulder IE, Schmidt B, Lewis M, Delday M, Stokes CR, Bailey M, Aminov RI, Gill BP, Pluske JR, Mayer CD & Kelly D. (2011). Restricting microbial exposure in early life negates the immune benefits associated with gut colonization in environments of high microbial diversity. PLoS ONE , 6, e28279. PMID: 22216092 DOI.
- ↑ 3.0 3.1 Carlisle EM, Poroyko V, Caplan MS, Alverdy JA & Liu D. (2011). Gram negative bacteria are associated with the early stages of necrotizing enterocolitis. PLoS ONE , 6, e18084. PMID: 21445365 DOI.
- ↑ 4.0 4.1 Weaver LT, Austin S & Cole TJ. (1991). Small intestinal length: a factor essential for gut adaptation. Gut , 32, 1321-3. PMID: 1752463
- ↑ Wang PY, Caspi L, Lam CK, Chari M, Li X, Light PE, Gutierrez-Juarez R, Ang M, Schwartz GJ & Lam TK. (2008). Upper intestinal lipids trigger a gut-brain-liver axis to regulate glucose production. Nature , 452, 1012-6. PMID: 18401341 DOI.
- ↑ Di Mauro A, Neu J, Riezzo G, Raimondi F, Martinelli D, Francavilla R & Indrio F. (2013). Gastrointestinal function development and microbiota. Ital J Pediatr , 39, 15. PMID: 23433508 DOI.
- ↑ Smith B, Bodé S, Petersen BL, Jensen TK, Pipper C, Kloppenborg J, Boyé M, Krogfelt KA & Mølbak L. (2011). Community analysis of bacteria colonizing intestinal tissue of neonates with necrotizing enterocolitis. BMC Microbiol. , 11, 73. PMID: 21486476 DOI.
Reviews
Neal-Kluever A, Fisher J, Grylack L, Kakiuchi-Kiyota S & Halpern W. (2019). Physiology of the Neonatal Gastrointestinal System Relevant to the Disposition of Orally Administered Medications. Drug Metab. Dispos. , 47, 296-313. PMID: 30567878 DOI.
Jacobi SK & Odle J. (2012). Nutritional factors influencing intestinal health of the neonate. Adv Nutr , 3, 687-96. PMID: 22983847 DOI.
Hao WL & Lee YK. (2004). Microflora of the gastrointestinal tract: a review. Methods Mol. Biol. , 268, 491-502. PMID: 15156063 DOI.
Articles
Palmer C, Bik EM, DiGiulio DB, Relman DA & Brown PO. (2007). Development of the human infant intestinal microbiota. PLoS Biol. , 5, e177. PMID: 17594176 DOI.
van Elburg RM, Fetter WP, Bunkers CM & Heymans HS. (2003). Intestinal permeability in relation to birth weight and gestational and postnatal age. Arch. Dis. Child. Fetal Neonatal Ed. , 88, F52-5. PMID: 12496227
Wiswell TE, Tuggle JM & Turner BS. (1990). Meconium aspiration syndrome: have we made a difference?. Pediatrics , 85, 715-21. PMID: 2330231
Search Pubmed
Search Pubmed Now: postnatal gastrointestinal development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 19) Embryology Gastrointestinal Tract - Postnatal. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Gastrointestinal_Tract_-_Postnatal
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G