Endocrine - Parathyroid Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (7 intermediate revisions by the same user not shown) | |||
| Line 10: | Line 10: | ||
Interestingly, the inferior parathyroid originates from the third pharyngeal pouch and the superior arises from the fourth pharyngeal pouch, the adult anatomical position is the opposite of the pharyngeal rostro-caudal order. This occurs due to the third pharyngeal pouch also giving rise to the thymus, the superior pair descend along with the thymus. | Interestingly, the inferior parathyroid originates from the third pharyngeal pouch and the superior arises from the fourth pharyngeal pouch, the adult anatomical position is the opposite of the pharyngeal rostro-caudal order. This occurs due to the third pharyngeal pouch also giving rise to the thymus, the superior pair descend along with the thymus. | ||
Parathyroid hormone (PTH) regulates calcium and phosphate levels in conjunction with parafollicular cells of the {{thyroid}} gland (calcitonin) and Vitamin D (dietary or synthesized in the skin). | Parathyroid hormone (PTH) regulates calcium and phosphate levels in conjunction with parafollicular cells of the {{thyroid}} gland (calcitonin) and {{Vitamin D}} (dietary or synthesized in the skin). | ||
| Line 34: | Line 34: | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Parathyroid+Embryology ''Parathyroid Embryology''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Parathyroid+Embryology ''Parathyroid Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=pharyngeal+pouch+Embryology ''pharyngeal pouch Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Parathyroid+hormone ''Parathyroid hormone''] | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! Older papers | ! Older papers | ||
|- | |- | ||
| | | {{Older papers}} | ||
* '''Multiple roles for HOXA3 in regulating thymus and parathyroid differentiation and morphogenesis in mouse'''{{#pmid:25249461|PMID25249461}} "Previous studies have shown that the Hoxa3 null mutant lacks third pharyngeal pouch derivatives, the thymus and parathyroids by E18.5, and organ-specific markers are absent or downregulated during initial organogenesis. Our current analysis of the Hoxa3 null mutant shows that organ-specific domains did undergo initial patterning, but the location and timing of key regional markers within the pouch, including Tbx1, Bmp4 and Fgf8, were altered. Expression of the parathyroid marker Gcm2 was initiated but was quickly downregulated and differentiation failed; by contrast, thymus markers were delayed but achieved normal levels, concurrent with complete loss through apoptosis. To determine the cell type-specific roles of Hoxa3 in third pharyngeal pouch development, we analyzed tissue-specific mutants using endoderm and/or neural crest cell (NCC)-specific Cre drivers. Simultaneous deletion with both drivers resulted in athymia at E18.5, similar to the null. By contrast, the individual tissue-specific Hoxa3 deletions resulted in small, ectopic thymi, although each had a unique phenotype. Hoxa3 was primarily required in NCCs for morphogenesis. In endoderm, Hoxa3 temporally regulated initiation of the thymus program and was required in a cell-autonomous manner for parathyroid differentiation. Furthermore, Hoxa3 was required for survival of third pharyngeal pouch-derived organs, but expression in either tissue was sufficient for this function." {{HOX}} | * '''Multiple roles for HOXA3 in regulating thymus and parathyroid differentiation and morphogenesis in mouse'''{{#pmid:25249461|PMID25249461}} "Previous studies have shown that the Hoxa3 null mutant lacks third pharyngeal pouch derivatives, the thymus and parathyroids by E18.5, and organ-specific markers are absent or downregulated during initial organogenesis. Our current analysis of the Hoxa3 null mutant shows that organ-specific domains did undergo initial patterning, but the location and timing of key regional markers within the pouch, including Tbx1, Bmp4 and Fgf8, were altered. Expression of the parathyroid marker Gcm2 was initiated but was quickly downregulated and differentiation failed; by contrast, thymus markers were delayed but achieved normal levels, concurrent with complete loss through apoptosis. To determine the cell type-specific roles of Hoxa3 in third pharyngeal pouch development, we analyzed tissue-specific mutants using endoderm and/or neural crest cell (NCC)-specific Cre drivers. Simultaneous deletion with both drivers resulted in athymia at E18.5, similar to the null. By contrast, the individual tissue-specific Hoxa3 deletions resulted in small, ectopic thymi, although each had a unique phenotype. Hoxa3 was primarily required in NCCs for morphogenesis. In endoderm, Hoxa3 temporally regulated initiation of the thymus program and was required in a cell-autonomous manner for parathyroid differentiation. Furthermore, Hoxa3 was required for survival of third pharyngeal pouch-derived organs, but expression in either tissue was sufficient for this function." {{HOX}} | ||
| Line 68: | Line 68: | ||
* chondrocyte differentiation{{#pmid:19076361|PMID19076361}} | * chondrocyte differentiation{{#pmid:19076361|PMID19076361}} | ||
==Molecular== | |||
===Sonic Hedgehog=== | |||
Signalling by {{SHH}} has been shown to restrict the expression of Gcm2 and controls the position of the developing parathyroids.{{#pmid:21349263|PMID21349263}} | |||
=== Glial Cells Missing=== | |||
The transcription factor, Glial Cells Missing, Drosophila, Homolog Of, 2 (GCM2) is a key to parathyroid molecular development.{{#pmid:17382312|PMID17382312}}{{#pmid:30390809|PMID30390809}} | |||
:'''Links:''' [https://www.omim.org/entry/603716 OMIM GCM2] | |||
==Abnormalities== | ==Abnormalities== | ||
| Line 92: | Line 105: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:30390809}} | |||
{{#pmid:18660669}} | {{#pmid:18660669}} | ||
| Line 98: | Line 113: | ||
{{#pmid:10912527}} | {{#pmid:10912527}} | ||
===Articles=== | ===Articles=== | ||
{{#pmid:21203493}} | |||
{{#pmid:17382312}} | |||
===Search PubMed=== | ===Search PubMed=== | ||
Revision as of 23:21, 20 May 2019
| Embryology - 23 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
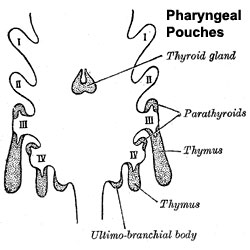
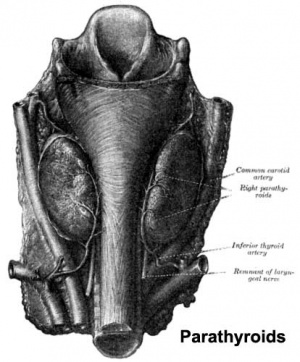
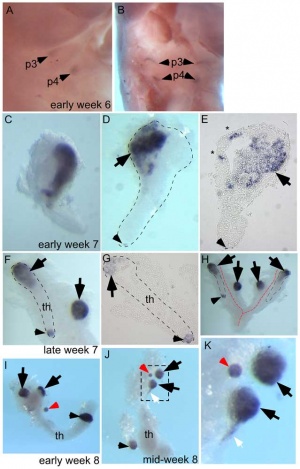
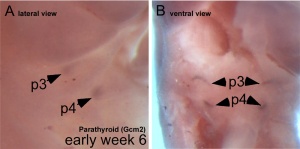
The parathyroid gland appears in the adult as a pair of inferior and a pair of superior "bumps" on the beside the (dorsal) thyroid (hence the name, "para"). The embryonic origin of this gland is from the endoderm of the third and fourth pharyngeal pouches, and could also have ectoderm and neural crest contributions. This developmental process also generates multiple small parathyroid clusters in addition to the main parathyroid glands.[1]
At 6 weeks a diverticulum elongates from the pouch, initially hollow and then solidifynig with cell proliferation.
Interestingly, the inferior parathyroid originates from the third pharyngeal pouch and the superior arises from the fourth pharyngeal pouch, the adult anatomical position is the opposite of the pharyngeal rostro-caudal order. This occurs due to the third pharyngeal pouch also giving rise to the thymus, the superior pair descend along with the thymus.
Parathyroid hormone (PTH) regulates calcium and phosphate levels in conjunction with parafollicular cells of the thyroid gland (calcitonin) and Vitamin D (dietary or synthesized in the skin).
The fetal parathyroids appear functional as they respond to calcium levels. The fetal calcium levels also higher than maternal levels.
Historically, see also the 1938 paper on the fate of the ultimobranchial body within the human thyroid gland.[2]
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Parathyroid Embryology | pharyngeal pouch Embryology | Parathyroid hormone
|
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Development Overview
- Endoderm - third and fourth pharyngeal pouches, could also have ectoderm and neural crest
- 3rd Pharyngeal Pouch - inferior parathyroid, initially descends with thymus
- 4th Pharyngeal Pouch - superior parathyroid
- Week 6 - diverticulum elongate, hollow then solid, dorsal cell proliferation
- Fetal parathyroids - respond to calcium levels, fetal calcium levels higher than maternal
Parathyroid Hormone
(PTH, parathormone or parathyrin) A polypeptide (84 amino acids) hormone which increases the concentration of calcium ions in the blood. Its actions oppose the hormone calcitonin from the parafollicular cells (C cells) of the thyroid gland, which decrease calcium. Acts through the parathyroid hormone receptor in bone, kidney and gastrointestinal tract.
- stimulate osteoclasts - degrade bone matrix, releasing calcium
- increase calcium gastrointestinal tract absorption
(PTHrP) Originally identified in the clinical syndrome humoral hypercalcemia of malignancy. It's developmental role is that of a regulatory protein expressed during the formation of many organs.
- mammary gland development - epithelial-mesenchymal interactions[6]
- chondrocyte differentiation[7]
Molecular
Sonic Hedgehog
Signalling by SHH has been shown to restrict the expression of Gcm2 and controls the position of the developing parathyroids.[8]
Glial Cells Missing
The transcription factor, Glial Cells Missing, Drosophila, Homolog Of, 2 (GCM2) is a key to parathyroid molecular development.[9][10]
- Links: OMIM GCM2
Abnormalities
Hyperparathyroidism
Postnatal
- Postnatal adult ageing increased parathyroid hormone plasma levels are associated with cognitive decline and dementia.
- Parathyroid carcinoma (cancer) is a rare malignancy, occurring with an incidence of 0.5 to 4% of all cases of primary hyperparathyroidism.
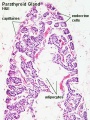
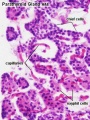
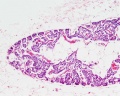
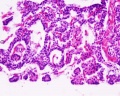
Adult Histology
References
- ↑ 1.0 1.1 1.2 1.3 Liu Z, Farley A, Chen L, Kirby BJ, Kovacs CS, Blackburn CC & Manley NR. (2010). Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions. PLoS Genet. , 6, e1001251. PMID: 21203493 DOI.
- ↑ Kingsbury BF. On the fate of the ultimobranchial body within the human thyroid gland. (1935) Anat. Rec. 61(2): 155–173.
- ↑ Bain VE, Gordon J, O'Neil JD, Ramos I, Richie ER & Manley NR. (2016). Tissue-specific roles for sonic hedgehog signaling in establishing thymus and parathyroid organ fate. Development , 143, 4027-4037. PMID: 27633995 DOI.
- ↑ Figueiredo M, Silva JC, Santos AS, Proa V, Alcobia I, Zilhão R, Cidadão A & Neves H. (2016). Notch and Hedgehog in the thymus/parathyroid common primordium: Crosstalk in organ formation. Dev. Biol. , 418, 268-82. PMID: 27544844 DOI.
- ↑ Chojnowski JL, Masuda K, Trau HA, Thomas K, Capecchi M & Manley NR. (2014). Multiple roles for HOXA3 in regulating thymus and parathyroid differentiation and morphogenesis in mouse. Development , 141, 3697-708. PMID: 25249461 DOI.
- ↑ Dunbar ME & Wysolmerski JJ. (1999). Parathyroid hormone-related protein: a developmental regulatory molecule necessary for mammary gland development. J Mammary Gland Biol Neoplasia , 4, 21-34. PMID: 10219904
- ↑ Alman BA & Wunder JS. (2008). Parathyroid hormone-related protein regulates glioma-associated oncogene transcriptional activation: lessons learned from bone development and cartilage neoplasia. Ann. N. Y. Acad. Sci. , 1144, 36-41. PMID: 19076361 DOI.
- ↑ Grevellec A, Graham A & Tucker AS. (2011). Shh signalling restricts the expression of Gcm2 and controls the position of the developing parathyroids. Dev. Biol. , 353, 194-205. PMID: 21349263 DOI.
- ↑ Liu Z, Yu S & Manley NR. (2007). Gcm2 is required for the differentiation and survival of parathyroid precursor cells in the parathyroid/thymus primordia. Dev. Biol. , 305, 333-46. PMID: 17382312 DOI.
- ↑ Peissig K, Condie BG & Manley NR. (2018). Embryology of the Parathyroid Glands. Endocrinol. Metab. Clin. North Am. , 47, 733-742. PMID: 30390809 DOI.
Reviews
Peissig K, Condie BG & Manley NR. (2018). Embryology of the Parathyroid Glands. Endocrinol. Metab. Clin. North Am. , 47, 733-742. PMID: 30390809 DOI.
Zajac JD & Danks JA. (2008). The development of the parathyroid gland: from fish to human. Curr. Opin. Nephrol. Hypertens. , 17, 353-6. PMID: 18660669 DOI.
Parvari R, Diaz GA & Hershkovitz E. (2007). Parathyroid development and the role of tubulin chaperone E. Horm. Res. , 67, 12-21. PMID: 17008776 DOI.
Jüppner H. (2000). Role of parathyroid hormone-related peptide and Indian hedgehog in skeletal development. Pediatr. Nephrol. , 14, 606-11. PMID: 10912527
Articles
Liu Z, Farley A, Chen L, Kirby BJ, Kovacs CS, Blackburn CC & Manley NR. (2010). Thymus-associated parathyroid hormone has two cellular origins with distinct endocrine and immunological functions. PLoS Genet. , 6, e1001251. PMID: 21203493 DOI.
Liu Z, Yu S & Manley NR. (2007). Gcm2 is required for the differentiation and survival of parathyroid precursor cells in the parathyroid/thymus primordia. Dev. Biol. , 305, 333-46. PMID: 17382312 DOI.
Search PubMed
Search April 2010
- Parathyroid Development - All (3523) Review (768) Free Full Text (741)
Search Pubmed: parathyroid development
Additional Images
Terms
- parathyroid hormone - (PTH, parathormone or parathyrin) A polypeptide (84 amino acids) hormone secreted by the parathyroid gland, which increases the concentration of calcium ions in the blood. Its actions oppose the hormone calcitonin from the thyroid gland parafollicular cells (C cells), which decrease calcium. Acts through the parathyroid hormone receptor located mainly in bone, kidney and gastrointestinal tract. Hormone dual role is to: stimulate osteoclasts in bone to degrade bone matrix releasing calcium; increase gastrointestinal tract absorption of calcium.
| Endocrine Terms (expand to view) |
|---|
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 23) Embryology Endocrine - Parathyroid Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Parathyroid_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G