Endocrine - Adrenal Development
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The developing adrenal gland has both an interesting origin and an intruiging fetal role. Furthermore recent studies suggest that the adrenal cortex share a common embryonic origin with the early gonad. The adrenal gland and placenta also act in synergy, and the notes endocrine placenta should also be read.
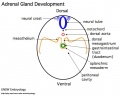
The 2 adrenal glands (suprarenal gland, glandulæ suprarenales) are named by their anatomical postion sitting above the 2 kidneys (renal). The 2 main parts of the adrenals have different embryonic origins. The inside core adrenal medulla is neural crest in origin. Mesenchyme surrounding these cells differentiates to form a fetal cortex. This fetal cortex is later replaced by the adult cortex. The outside adrenal cortex is derived from mesothelium and can be further divided into 3 distinct layers (zona reticularis, zona fasiculata, zona glomerulosa) each with distinct hormonal functions.
During fetal development, adrenal hormones are involved with production of precursor for placental estrogen production, ovary and brain development, the maturation of the lung and other developing systems.
| Historic Embryology - Adrenal |
|---|
| 1912 Suprarenal Bodies | 1914 Suprarenal Organs | 1920 Adrenal | 1940 Adrenal | 1957 Adrenal |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Adrenal Embryology
|
| Older papers |
|---|
|
Adrenal Movies
|
| ||||||
| Cartoon showing migration of neural crest cells from original location to form the fetal medulla cells. |
Surface rendering of human fetal adrenal glands in the third trimester (week 30 GA week 32). |
Adrenal Overview
- Richly vascularized - arterioles passing through cortex, capillaries from cortex to medulla, portal-like circulation
- Fetal Cortex - produces a steroid precursor (DEA), converted by placenta into estrogen
- Adult Medulla - produces adrenalin (epinephrine), noradrenaline (norepinephrine)
- Fetal adrenal hormones - influence lung maturation
Cortical Hormones
(steroids) Cortisol, Aldosterone, Dehydroepiandrosterone
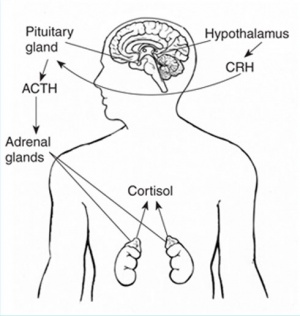
- zona glomerulosa - regulated by renin-angiotensin-aldosterone system controlled by the juxtaglomerular apparatus of the kidney.
- zona fasciculata - regulated by hypothalamo-pituitary axis with the release of CRH and ACTH respectively.
Medullary Hormones
(amino acid derivatives) Epinephrine, Norepinephrine
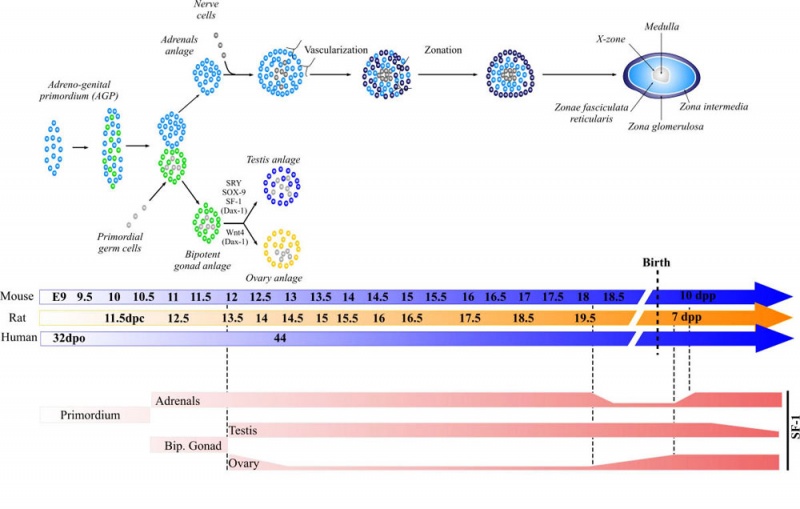
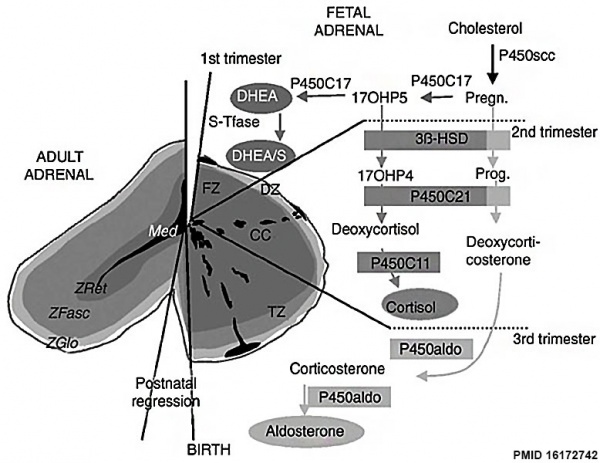
Adrenal Development
| Adrenal gland development and steroid hormone synthesis | ||
|---|---|---|
| ||
Overview cartoon of changes in adrenal gland structure and steroid hormone syntesis[8]
|
Adrenal Cortex
Adrenal Medulla
|
|
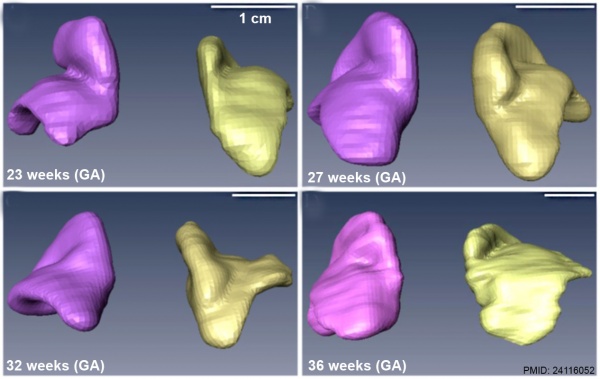
|
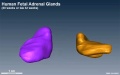
| Human fetal adrenal gland morphology and size.[3] |
Development Overview
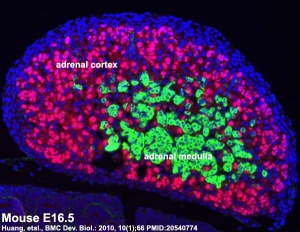
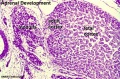
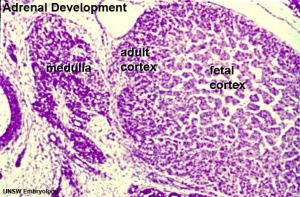
Medulla - Neural crest cells migrate toward the coelomic cavity wall and form the adrenal medulla. These chromaffin (chromaphil) cells originally named because of their staining (yellow) with chromium salts.
Cortex - Week 4 celomic epithelium (mesothelium) cells proliferate initially forming small buds that separate from the epithelium. Week 6 these now mesenchymal cells first form the fetal adrenal cortex which will be later replaced by the adult cortex.
Adrenal Cortex
|
|
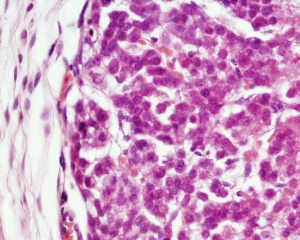
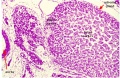
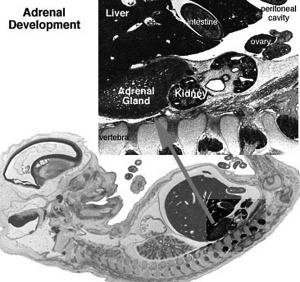
| Human Embryo (8 weeks, stage 22) adrenal gland showing the fetal and permanent adrenal cortex. Note that the medulla of the adrenal gland is not yet encapsulated by the cortex. | Human Fetus (10 week, 40mm, parasagittal section) shows location of the developing adrenal gland. The spongy appearance at the centre of the adrenal is the degenerating fetal cortex. The dense region around the outside of the adrenal is the developing adult cortex.) |
Week 4 - coelomic epithelium (mesothelium) cells proliferate initially forming small buds that separate from the epithelium.
Week 6 - these now mesenchymal cells surrounding the developing medulla cells differentiate first form the fetal adrenal cortex which will be later replaced by the adult cortex.
Week 8 to 9 - fetal adrenal cortex synthesizes cortisol and is maximal at 8-9 weeks post conception (wpc) under the regulation of ACTH (also stimulates androstenedione and testosterone secretion).[9]
Adult cortex - mesothelium mesenchyme encloses fetal cortex.
Late Fetal Period - differentiates to form cortical zones.
Birth - zona glomerulosa, zona fasiculata present.
Year 3 - zona reticularis present.
Fetal Cortex
Fetal adrenal cortical growth involves several cellular processes: hypertrophy, hyperplasia, apoptosis, and migration.
In the second and third trimesters a steroid precursor dehydroepiandrosterone (DHEA) and sulphated derivative (DHEAS) which is converted by placenta into estrogen.
Three functional zones:
- Fetal zone - throughout gestation expresses enzymes required for DHEA-S synthesis.
- Transitional zone - initially identical to the fetal zone but later (after 25-30 weeks) expresses enzymes that suggest glucocorticoid synthesis.
- Definitive zone - after 22-24 weeks expresses enzymes that suggest mineralocorticoid synthesis.
Neonatal
- human males produce high levels of DHEA) and DHEAS
- decline within a few months of birth
- due to regression of the adrenal fetal zone
Adult
- zona reticularis (ZR) source for production of DHEA and DHEAS
Adult Cortex
Early Adult Cortex (week 12)
- Reticularis - narrow band, many small cells and capillaries androgens. source for production of DHEA and DHEAS
- Fasiculata - high lipid content, pale foamy cells cortisol, corticosterone, cortisone.
- Glomerulosa - small cells, cords or oval groups, aldosterone.
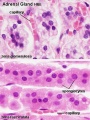
Species Difference
- rat - zona glomerulosa and zona fasciculata separated by an undifferentiated zone (ZU, or Zona Intermedia)
- mouse - no undifferentiated zone separation.
- capsule mesenchyme cells have properties of adrenocortical stem/progenitor cells.
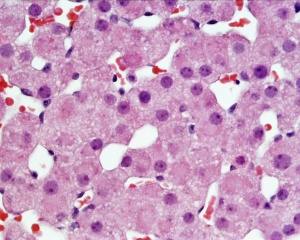
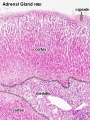
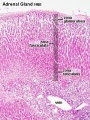
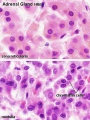
Adult Histology
- Adrenal Histology: Cortex and Medulla | Unlabelled Overview | Cortical Zones | Zona Glomerulosa and Fasciculata | Zona Glomerulosa | Zona Fasciculata | Zona Reticularis and Medulla | Zona Reticularis | Medulla | Fetal Cortex | Developing Adult Cortex | BGD - Endocrine Histology | Histology Stains | Adrenal Development
Molecular
Steroidogenic Factor 1
Adrenal and gonad steroidogenic factor 1 (SF-1) expression in different species [10]
- 53 kDa protein called Ad4BP (Adrenal 4 Binding Protein) or SF-1 (Steroidogenic Factor 1)
- classical DNA-binding domain (DBD) characterized by two Cys2-Cys2 zinc fingers in the N-terminal region
- SF-1 binds DNA as a monomer
- high homology with the drosophila Ftz-F1 transcription factor that controls fushi tarazu homeotic gene expression
Steroidogenic Factor 1 Mutation Effects
| Organism | Human | ||
| Genotype | SF-1 -/- | SF-1 +/- | SF-1 -/+ or SF-1 -/- |
| Adrenal | Agenesis | Histological defects
Hyporesponse to stress Compensatory growth defects |
Insufficiency (agenesis or dysgenesis) |
| Testis | Agenesis
Sex reversal |
Sex reversal | |
| Ovary | Agenesis | Normal | |
| Ventro-Medial Hypothalamus | Agenesis
Obesity caused by absence of the VMH (8 weeks) |
||
| Pituitary | Defects of gonadotrope cells | ||
Table modified from review.[10]
SoxE
Sry-box (Sox) 8, and Sox10 are expressed in the neural crest and in neural crest cells migrating to the adrenal gland.[11]
DAX1
CYP17
Abnormalities
Congenital Adrenal Hyperplasia
The adrenal abnormality of congenital adrenal hyperplasia (CAH) is a family of inherited disorders of adrenal steroidogenesis enzymes which impairs cortisol production by the adrenal cortex.
Enzymes most commonly affected: 21-hydroxylase (21-OH), 11beta-hydroxylase, 3beta-hydroxysteroid dehydrogenase.
Enzymes less commonly affected: 17 alpha-hydroxylase/17,20-lyase and cholesterol desmolase.
Classical CAH - androgen excess leads newborn females with external genital ambiguity and postnatal progressive virilization in both sexes.
| Congenital Adrenal Hyperplasia | |||
|---|---|---|---|
| Type | Enzyme Deficiency | Female | Male |
| classic virilizing adrenal hyperplasia | 21-hydroxylase, 11-beta-hydroxylase, or 3-beta-hydroxysteroid dehydrogenase |
ambiguous genitalia at birth - complete or partial fusion of the labioscrotal folds and a phallic urethra to clitoral enlargement (clitoromegaly), partial fusion of the labioscrotal folds, or both | normal genitalia, present at age 1-4 weeks with salt wasting (classic salt-wasting adrenal hyperplasia) |
| simple virilizing adrenal hyperplasia | mild 21-hydroxylase | identified later in childhood because of precocious pubic hair, clitoral enlargement (clitoromegaly), or both, often accompanied by accelerated growth and skeletal maturation | early genital development (pubic hair and/or phallic enlargement) accelerated growth and skeletal maturation |
| nonclassic adrenal hyperplasia | milder deficiencies of 21-hydroxylase or 3-beta-hydroxysteroid dehydrogenase |
present at puberty or adult with infrequent menstruation (oligomenorrhea), abnormal hair growth (hirsutism), and/or infertility | |
| 17-hydroxylase deficiency syndrome | 17-hydroxylase deficiency or 3-beta-hydroxysteroid dehydrogenase |
rare, phenotypically female at birth do not develop breasts or menstruate in adolescence and may have hypertension | steroidogenic acute regulatory (StAR) deficiency have ambiguous genitalia or female genitalia, at puberty may lack breast development and may have hypertension |
| This is a complex steroidogenic abnormality, and the above table of clinical descriptions are provided only a guide. | |||
- Links: genital abnormalities
Pheochromocytomas
(PCC) Catecholamine-producing (neuro)endocrine tumor located in the adrenal medulla. Similar catecholamine-producing tumors outside the adrenal gland are called paragangliomas (PGL).
Cushing's Syndrome
(hypercortisolism) A relatively rare metabolic hormonal disorder caused by prolonged exposure of the body’s tissues to high levels of the adrenal hormone cortisol, most commonly affects adults aged between 20 to 50 and also the obese with type 2 diabetes.
Adrenocortical Tumour
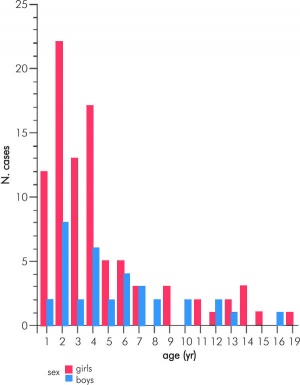
Adrenocortical tumours (ACT) can occur at all ages and have a bimodal distribution with peaks of incidence at about 5 years of age and again at 40 to 50 years of age. Clinically, a routine hormonal profile for suspected patients includes measurements of serum (8am, 11pm) cortisol, testosterone, DHEA-S, androstenedione, 17-hydroxyprogesterone, aldosterone, and plasma renin activity.[12]
References
- ↑ Heese S, Hammer K, Möllers M, Köster HA, Falkenberg MK, Eveslage M, Braun J, Oelmeier de Murcia K, Klockenbusch W & Schmitz R. (2018). Adrenal gland size in growth restricted fetuses. J Perinat Med , , . PMID: 29543592 DOI.
- ↑ Furlan A, Dyachuk V, Kastriti ME, Calvo-Enrique L, Abdo H, Hadjab S, Chontorotzea T, Akkuratova N, Usoskin D, Kamenev D, Petersen J, Sunadome K, Memic F, Marklund U, Fried K, Topilko P, Lallemend F, Kharchenko PV, Ernfors P & Adameyko I. (2017). Multipotent peripheral glial cells generate neuroendocrine cells of the adrenal medulla. Science , 357, . PMID: 28684471 DOI.
- ↑ 3.0 3.1 Zhang Z, Meng H, Hou Z, Ma J, Feng L, Lin X, Tang Y, Zhang X, Liu Q & Liu S. (2013). Fetal adrenal gland in the second half of gestation: morphometrical assessment with 3.0T post-mortem MRI. PLoS ONE , 8, e75511. PMID: 24116052 DOI.
- ↑ Ozgüner G, Sulak O & Koyuncu E. (2012). A morphometric study of suprarenal gland development in the fetal period. Surg Radiol Anat , 34, 581-7. PMID: 22430763 DOI.
- ↑ Inoue S, Cho BH, Song CH, Fujimiya M, Murakami G & Matsubara A. (2010). Migration and distribution of neural crest-derived cells in the human adrenal cortex at 9-16 weeks of gestation: an immunohistochemical study. Okajimas Folia Anat Jpn , 87, 11-6. PMID: 20715567
- ↑ Hui XG, Akahira J, Suzuki T, Nio M, Nakamura Y, Suzuki H, Rainey WE & Sasano H. (2009). Development of the human adrenal zona reticularis: morphometric and immunohistochemical studies from birth to adolescence. J. Endocrinol. , 203, 241-52. PMID: 19723922 DOI.
- ↑ Ishimoto H & Jaffe RB. (2011). Development and function of the human fetal adrenal cortex: a key component in the feto-placental unit. Endocr. Rev. , 32, 317-55. PMID: 21051591 DOI.
- ↑ Chamoux E, Otis M & Gallo-Payet N. (2005). A connection between extracellular matrix and hormonal signals during the development of the human fetal adrenal gland. Braz. J. Med. Biol. Res. , 38, 1495-503. PMID: 16172742 DOI.
- ↑ Goto M, Piper Hanley K, Marcos J, Wood PJ, Wright S, Postle AD, Cameron IT, Mason JI, Wilson DI & Hanley NA. (2006). In humans, early cortisol biosynthesis provides a mechanism to safeguard female sexual development. J. Clin. Invest. , 116, 953-60. PMID: 16585961 DOI.
- ↑ 10.0 10.1 Val P, Lefrançois-Martinez AM, Veyssière G & Martinez A. (2003). SF-1 a key player in the development and differentiation of steroidogenic tissues. Nucl. Recept. , 1, 8. PMID: 14594453 DOI.
- ↑ Reiprich S, Stolt CC, Schreiner S, Parlato R & Wegner M. (2008). SoxE proteins are differentially required in mouse adrenal gland development. Mol. Biol. Cell , 19, 1575-86. PMID: 18272785 DOI.
- ↑ 12.0 12.1 Marques-Pereira R, Delacerda L, Lacerda HM, Michalkiewicz E, Sandrini F & Sandrini R. (2006). Childhood adrenocortical tumours: a review. Hered Cancer Clin Pract , 4, 81-9. PMID: 20223012 DOI.
Online Textbooks
Endocrinology: An Integrated Approach Nussey, S.S. and Whitehead, S.A. Oxford, UK: BIOS Scientific Publishers, Ltd; 2001. 4.7. Embryology of the adrenal gland | The Adrenal Gland | Anatomical and functional zonation in the adrenal cortex
Developmental Biology (6th ed) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000. Figure 13.6. Final differentiation of a trunk neural crest cell committed to become either an adrenomedullary (chromaffin) cell or a sympathetic neuron
Molecular Biology of the Cell (4th Edn) Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter. New York: Garland Publishing; 2002. table 15-1. Some Hormone-induced Cell Responses Mediated by Cyclic AMP | Cells Can Respond Abruptly to a Gradually Increasing Concentration of an Extracellular Signal
Health Services/Technology Assessment Text (HSTAT) Bethesda (MD): National Library of Medicine (US), 2003 Oct. Adrenal Gland search Results
Search NLM Online Textbooks- "adrenal development" : Endocrinology | Molecular Biology of the Cell | The Cell- A molecular Approach
Reviews
Asby DJ, Arlt W & Hanley NA. (2009). The adrenal cortex and sexual differentiation during early human development. Rev Endocr Metab Disord , 10, 43-9. PMID: 18670886 DOI.
Ferraz-de-Souza B & Achermann JC. (2008). Disorders of adrenal development. Endocr Dev , 13, 19-32. PMID: 18493131 DOI.
Hanley NA & Arlt W. (2006). The human fetal adrenal cortex and the window of sexual differentiation. Trends Endocrinol. Metab. , 17, 391-7. PMID: 17046275 DOI.
Huber K. (2006). The sympathoadrenal cell lineage: specification, diversification, and new perspectives. Dev. Biol. , 298, 335-43. PMID: 16928368 DOI.
Yanase T, Gondo S, Okabe T, Tanaka T, Shirohzu H, Fan W, Oba K, Morinaga H, Nomura M, Ohe K & Nawata H. (2006). Differentiation and regeneration of adrenal tissues: An initial step toward regeneration therapy for steroid insufficiency. Endocr. J. , 53, 449-59. PMID: 16807499
Jaffe RB, Mesiano S, Smith R, Coulter CL, Spencer SJ & Chakravorty A. (1998). The regulation and role of fetal adrenal development in human pregnancy. Endocr. Res. , 24, 919-26. PMID: 9888597
Mesiano S & Jaffe RB. (1997). Developmental and functional biology of the primate fetal adrenal cortex. Endocr. Rev. , 18, 378-403. PMID: 9183569 DOI.
Articles
Speiser PW. (2010). Growth and development: congenital adrenal hyperplasia-glucocorticoids and height. Nat Rev Endocrinol , 6, 14-5. PMID: 20010965 DOI.
Hui XG, Akahira J, Suzuki T, Nio M, Nakamura Y, Suzuki H, Rainey WE & Sasano H. (2009). Development of the human adrenal zona reticularis: morphometric and immunohistochemical studies from birth to adolescence. J. Endocrinol. , 203, 241-52. PMID: 19723922 DOI.
Val P, Martinez-Barbera JP & Swain A. (2007). Adrenal development is initiated by Cited2 and Wt1 through modulation of Sf-1 dosage. Development , 134, 2349-58. PMID: 17537799 DOI.
Goto M, Piper Hanley K, Marcos J, Wood PJ, Wright S, Postle AD, Cameron IT, Mason JI, Wilson DI & Hanley NA. (2006). In humans, early cortisol biosynthesis provides a mechanism to safeguard female sexual development. J. Clin. Invest. , 116, 953-60. PMID: 16585961 DOI.
Villa-Cuesta E & Modolell J. (2005). Mutual repression between msh and Iro-C is an essential component of the boundary between body wall and wing in Drosophila. Development , 132, 4087-96. PMID: 16093324 DOI.
Boglione L, Bondone C, Corno E, Gastaldo L, Borghi F, Gattolin A & Levi AC. (2001). The development of the suprarenal gland: surgical and anatomical considerations. Panminerva Med , 43, 33-7. PMID: 11319516
Jaffe RB, Mesiano S, Smith R, Coulter CL, Spencer SJ & Chakravorty A. (1998). The regulation and role of fetal adrenal development in human pregnancy. Endocr. Res. , 24, 919-26. PMID: 9888597
- "The rapid growth of the human fetal adrenal gland, which is primarily a reflection of the growth of the unique fetal zone, is regulated by ACTH acting indirectly to stimulate the expression of locally produced growth factors, of which IGF-II and bFGF appear to play key roles. Through most of gestation, the outer definitive zone appears to function as a reservoir of progenitor cells which move centripetally to populate the rest of the gland. At the end of pregnancy, the fetal zone undergoes senescence through an apoptotic process. Activin and TGF-beta are capable of inducing apoptosis in the fetal zone. Corticotropin-releasing hormone, which is produced by the placenta in markedly increased amounts at the end of gestation, may orchestrate a variety of processes, including direct stimulation of fetal adrenal steroidogenesis, culminating in the initiation of parturition."
Search PubMed
Search April 2010
- Adrenal Development - All (646) Review (52) Free Full Text (84)
- Congenital Adrenal Hyperplasia - All (2091) Review (211) Free Full Text (314)
Search Pubmed: adrenal development | Congenital Adrenal Hyperplasia
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Terms
| Endocrine Terms (expand to view) |
|---|
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 25) Embryology Endocrine - Adrenal Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Adrenal_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G