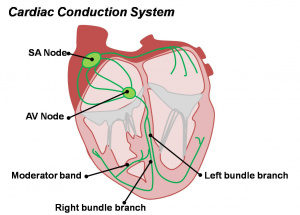
Detailed Cardiac - Sinus Node
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Detailed Cardiac: Systemic Venous Sinus | Pulmonary Vein | Superior Interatrial Fold | Atrioventricular Cushions | Atrioventricular Canal | Interventricular Communication | Subpulmonary Infundibulum | Arterial Roots | Intrapericardial Arterial Trunks | Extrapericardial Arterial Channels | Sinus Node | Atrioventricular Conduction Axis |
Appearance and Remodelling of the Sinus Node
Introduction
The sinoatrial node (SAN) is the primary pacemaker of the heart that initially develops from part of pro-pacemaking embryonic venous pole that expresses both Hcn4 and the transcriptional factor Shox2.[1] The chicken embryo has been used as a model of SAN development, where early canonical Wnts expression within the right lateral plate mesoderm just posterior of the heart field results in development of these cells.[2]
The adult node is a mixture of about 10,000 cells distinct myocytes located at the entry of the right superior caval vein (SVC) into the right atrium.
| Cardiovascular Links: cardiovascular | Heart Tutorial | Lecture - Early Vascular | Lecture - Heart | Movies | 2016 Cardiac Review | heart | coronary circulation | heart valve | heart rate | Circulation | blood | blood vessel | blood vessel histology | heart histology | Lymphatic | ductus venosus | spleen | Stage 22 | cardiovascular abnormalities | OMIM | 2012 ECHO Meeting | Category:Cardiovascular | ||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Sinus Node Development <pubmed limit=5>Sinus Node Development</pubmed> |
Molecular
Wnt
A secreted glycoprotein patterning switch with different roles in different tissues and signaling has generally been divided into the canonical and non-canonical pathways. The name was derived from two drosophila phenotypes wingless and int and the gene was first defined as a protooncogene, int1.
Canonical Pathway
- Wnt binds the surface receptor Frizzled (Fz) and LRP5/6 receptor complex
- Induces the stabilization of beta-catenin (through the DIX and PDZ domains of Dishevelled and other factors including Axin, glycogen synthase kinase 3 and casein kinase 1)
- Beta-catenin translocates into the nucleus
- Beta-catenin complexes with members of the LEF/TCF family of transcription factors.
- Transcriptional induction of target genes.
- Beta-catenin is then exported from the nucleus and degraded via the proteosomal machinery.
- Links: Wnt | Wnt Canonical Pathway
Hcn4
Hyperpolarization-Activated Cyclic Nucleotide-Gated Potassium Channel 4 (HCN4) gene located at 15q24.1, the 1,203 amino acid protein contains 6 putative transmembrane segments, a pore region, and a cyclic nucleotide-binding domain.
- Links: [OMIM HCN4
Shox2
Short Stature Homeobox 2 (SHOX2) gene located at 3q25.32, the protein acts as a homeobox transcription factor.
- mouse og12 gene - developmental sinus venosus (aorta), female genitalia, diencephalon, mes- and myelencephalon, nasal capsula, palate, eyelid, and limbs
- Links: OMIM SHOX2
ISL1
ISL Lim Homeobox 1 (ISL1) gene located at 5q11.1 is a member of the LIM/homeodomain family of transcription factors that binds to the enhancer region of the insulin gene. Functions in survival, proliferation, and function of pacemaker cells throughout development.[5]
- Links: OMIM ISL1
Abnormalities
Sinus node dysfunction (SND) or sick sinus syndrome (SSS) can occur in the elderly.
References
Reviews
<pubmed>26023305</pubmed> <pubmed>24672485</pubmed> <pubmed></pubmed> <pubmed></pubmed>
Articles
<pubmed>27291063</pubmed> <pubmed>26682210</pubmed> <pubmed>26611337</pubmed> <pubmed>26835096</pubmed>
Cite this page: Hill, M.A. (2024, April 24) Embryology Detailed Cardiac - Sinus Node. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Detailed_Cardiac_-_Sinus_Node
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G