Category:Spermatozoa: Difference between revisions
From Embryology
mNo edit summary |
mNo edit summary |
||
| Line 1: | Line 1: | ||
This page lists {{Embryology}} pages and media related to spermatozoa development. | This page lists {{Embryology}} pages and media related to spermatozoa (male gamete) development. | ||
:'''Links:''' [[Spermatozoa Development]] | :'''Links:''' [[Spermatozoa Development]] | ||
{{Spermatozoa Terms}} | |||
[[Category:Primordial Germ Cell]] | [[Category:Primordial Germ Cell]] | ||
Revision as of 16:01, 1 November 2014
This page lists Embryology pages and media related to spermatozoa (male gamete) development.
- Links: Spermatozoa Development
Note there are additional glossaries associated with genital, spermatozoa, oocyte and renal.
- acroplaxome - structure forms the acrosome plate with intermediate filament bundles of the marginal ring at the leading edge of the acrosome. The sub-acrosomal layer located in the developing spermatozoa head perinuclear region, located between the inner acrosomal membrane and the nuclear envelope. The other part of the perinuclear region is the post-acrosomal sheath (PAS) at the post-acrosomal region.
- acrosome - Cap-shaped cellular structure formed from the golgi apparatus and contains enzymes to dissolve the oocyte (egg) zona pellucida for fertilisation.
- acrosome compaction - Acrosome reshaping process in final stages of spermatogenesis (spermatid to spermatozoa).
- acrosome reaction - Chemical change within the spermatozoa following binding to the zona pellucida, only acrosome reacted spermatozoa have an ability to fuse with oocytes.
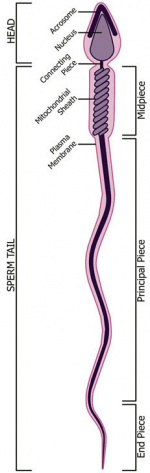
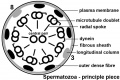
- annulus - Cytoskeletal (septin) structure located between the midpiece and principal piece regions of the tail, thought to form a diffusion barrier between these two domains. PMID 20042538
- asthenozoospermia - (asthenospermia) Term for reduced sperm motility and can be the cause of male infertility.
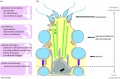
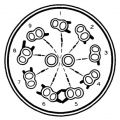
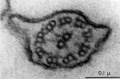
- axoneme - (axonema) The basic structure in cilia and eukaryotic flagella and in the spermatozoa tail, consisting of parallel microtubules in a characteristic "9 + 2" pattern. This pattern describes 9 outer microtubule doublets (pairs) surrounding 2 central singlet microtubules, in humans 50 μm long. The motor protein dynenin move the outer microtubules with respect to the central pair, bending the cilia and generating motility. Note that prokaryotic bacteria have a similar process (flagellum) that uses an entirely different mechanism for motility.
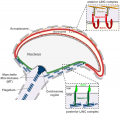
- blood-testis barrier - (BTB) Formed by tight junctions, basal ectoplasmic specializations, desmosome-like junctions and gap junctions between adjacent sertoli cells near the basement membrane of the seminiferous epithelium. image - BTB | image - tight junction
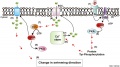
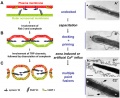
- capacitation - term describing the process by which spermaozoa become capable of fertilizing an oocyte, requires membrane changes, removal of surface glycoproteins and increased motility.
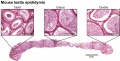
- caput - proximal head of the epididymis, epithelium with stereocilia, involved in absorbing fluid to concentrate spermatozoa. Underlying smooth muscle aids movement. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- CatSper - cationic (Ca2+) channel of spermatozoa, progesterone activated involved in hyperactivation, acrosome reaction, and possibly chemotaxis.
- cauda - distal tail of the epididymis, region with a thin epithelium and the greatest quantity of smooth muscle. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- centriole - a microtubule organising centre. First required for axoneme formation (distal centriole) that is lost and a second for pronuclei formation (proximal) following fertilisation. Rodents loose both and only have maternal centrioles.
- connecting piece - linkage between the spermatozoa head and the midpiece of the tail. PMID 22767409
- corpus - elongated body of the epididymis, This has an intermediate thickness of epithelium and thicker smooth muscle layer than caput. Epididymis three main parts : caput (head), corpus (body), cauda (tail).
- cytoplasmic bridges - Transient cytoplasm connections between spermatids arising from one spermatogonium due to incomplete cytokinesis.
- diploid - (Greek, di = double + ploion = vessel) Having two sets of chromosomes, the normal state for all cells other than the gametes.
- end piece - Last portion of the spermatozoa tail region.
- epididymis - testis tubular structure connecting the efferent ducts to the ductus deferent and functions for the storage and maturation of spermatozoa. Epididymis three main parts : caput (head), corpus (body), cauda (tail). PMID27307387
- fibrous sheath - cytoskeletal structure surrounding the axoneme and outer dense fibers, defining the extent of the principal piece region.
- haploid - (Greek, haploos = single) Having a single set of chromosomes as in mature germ/sex cells (oocyte, spermatozoa) following reductive cell division by meiosis. Normally cells are diploid, containing 2 sets of chromosomes.
- interstitial cell - (Leydig cell) Male gonad (testis) cell which secrete the androgen testosterone, beginning in the fetus.
- interstitium - testis developmental region (space between testis cords) that generates Leydig cells and other less well characterized cell types.
- Johnsen score - a clinical score (1-10) for assessing spermatogenesis in a human testicular biopsy. Named after the author of the original article. PMID 5527187
- Leydig cell - (interstitial cell) Male gonad (testis) cell that secrete the androgen testosterone, beginning in the fetus. Fetal Leydig cells develop from coelomic epithelium and undifferentiated perivascular cells in the gonad–mesonephros border region. Adult Leydig cells appear after birth from stem/progenitor cells among peritubular and peri-vascular cells. Leydig cells were first histologically identified in 1850 by Franz von Leydig (1821 - 1908) a German scientist.
- meiosis - The cell division that occurs only in production of germ cells where there is a reduction in the number of chromosomes (diploid to haploid) which is the basis of sexual reproduction. All other non-germ cells in the body divide by mitosis.
- midpiece - (middle piece) spermatozoa tail initial segment of axoneme surrounded outer dense fibres then by mitochondria. Next in the tail is the principal piece then finally the end piece.
- mitosis - The normal division of all cells, except germ cells, where chromosome number is maintained (diploid). In germ cell division (oocyte, spermatozoa) meiosis is a modified form of this division resulting in reduction in genetic content (haploid). Mitosis, division of the nucleus, is followed by cytokinesis the division of the cell cytoplasm and the cytoplasmic contents. cytokinesis overlaps with telophase.
- outer dense fibres - (ODF, outer dense fibers) cytoskeletal structures that surround the axoneme in the middle piece and principal piece of the spermatozoa tail.
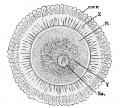
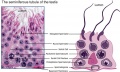
- primary spermatocyte - arranged in the seminiferous tubule wall deep (luminal) to the spermatogonia. These large cells enter the prophase of the first meiotic division. (More? meiosis)
- principal piece - Spermatozoa tail segment containing the plasma membrane calcium channels (CatSper1 and CatSper2) required for hyperactivation of motility. Region is partially separated from the midpiece by a barrier called the annulus.
- sertoli cells - (sustentacular cell) These cells are the spermatozoa supporting cells, nutritional and mechanical, as well as forming a blood-testis barrier. The cell cytoplasm spans all layers of the seminiferous tubule. The cells are named after Enrico Sertoli (1842 - 1910), and italian physiologist and histologist.
- sperm annulus - (Jensen's ring; Latin, annulus = ring) A region of the mammalian sperm flagellum connecting the midpiece and the principal piece. The annulus is a septin-based structure formed from SEPT1, 4, 6, 7 and 12. Septins are polymerizing GTPases that can act as a scaffold forming hetero-oligomeric filaments required for cytokinesis and other cell cycle roles.
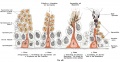
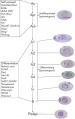
- spermatogenesis - (Greek, genesis = origin, creation, generation) The term used to describe the process of diploid spermatagonia division and differentiation to form haploid spermatazoa within the testis (male gonad). The process includes the following cellular changes: meiosis, reoorganization of DNA, reduction in DNA content, reorganization of cellular organelles, morphological changes (cell shape). The final process of change in cell shape is also called spermiogenesis.
- spermatogenesis - (Greek, genesis = origin, creation, generation) The maturation process of the already haploid spermatazoa into the mature sperm shape and organization. This process involves reorganization of cellular organelles (endoplasmic reticulum, golgi apparatus, mitochondria), cytoskeletal changes (microtubule organization) and morphological changes (cell shape, acrosome and tail formation).
- spermatogonia - The cells located in the seminiferous tubule adjacent to the basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
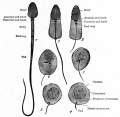
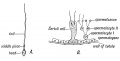
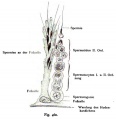
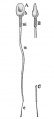
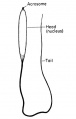
- spermatozoa head - Following spermiogenesis, the first region of the spermatozoa containing the haploid nucleus and acrosome. In humans, it is a flattened structure (5 µm long by 3 µm wide) with the posterior part of nuclear membrane forming the basal plate region. The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and spermatozoa tail).
- spermatozoa neck - Following spermiogenesis, the second region of the spermatozoa attached to basal plate, transverse oriented centriole, contains nine segmented columns of fibrous material, continue as outer dense fibres in tail. In humans, it forms a short structure (1 µm). The human spermatozoa is about 60 µm long, actively motile and divided into 3 main regions (head, neck and tail).
- spermatozoa tail - Following spermiogenesis, the third region of the spermatozoa that has a head, neck and tail). The tail is also divided into 3 structural regions a middle piece, a principal piece and an end piece. In humans: the middle piece (5 µm long) is formed by axonema and dense fibres surrounded by mitochondria; the principal piece (45 µm long) fibrous sheath interconnected by regularly spaced circumferential hoops; the final end piece (5 µm long) has an axonema surrounded by small amount of cytoplasm and plasma membrane.
- spermatogonial stem cells - (SSCs) The spermatagonia cells located beside the seminiferous tubule basal membrane that either divide and separate to renew the stem cell population, or they divide and stay together as a pair (|Apr spermatogonia) connected by an intercellular cytoplasmic bridge to differentiate and eventually form spermatazoa.
- spermatozoon - singular form of of spermatozoa.
- sperm protein 56 - A component of the spermatozoa acrosomal matrix released to the sperm surface during capacitation.
- teratospermia - Clinical term for a spermatozoa with abnormal morphology (small, large, defects in the head, tail, and/or mid-piece) present in the semen or ejaculate.
- testis cords - developmental structure that give rise to the adult seminiferous tubules, the other developmental region is the interstitium.
- vasectomy - Clinical term for ligation of the scrotal portion of the ductus deferens.
See also: Spermatozoa Terms collapse table
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Subcategories
This category has the following 2 subcategories, out of 2 total.
Pages in category 'Spermatozoa'
The following 68 pages are in this category, out of 68 total.
A
B
F
H
M
P
- Paper - A study of the function of the epididymis 1 (1929)
- Paper - Cytology of the human spermatozoon
- Paper - Electron microscopy of the sperm tail - results obtained with a new fixative
- Paper - Studies in the physiology of spermatozoa
- Paper - The duration of life of the spermatozoa in the human uterine tube
- Paper - The mammalian spermatozoon
- Template:Primary spermatocyte
- Template:Primordial germ cell
- Template:Pronuclei
- Template:Pronucleus
R
S
- Sertoli cell
- Template:Sperm
- Template:Spermatogenesis
- Template:Spermatogonia
- Template:Spermatogonial stem cell
- Template:Spermatogonium
- Template:Spermatozoa
- Spermatozoa Chemotaxis
- Spermatozoa Development
- Spermatozoa Meiosis Movie 1
- Spermatozoa Structure Movie
- Template:Spermatozoa Terms
- Template:Spermatozoa Terms collapse table
- Template:Spermatozoon
- Template:Spermiogenesis
- Template:SSC
Media in category 'Spermatozoa'
The following 150 files are in this category, out of 150 total.
- Adult hermaphrodite gonad arm.jpg 800 × 377; 66 KB
- Azoospermia.jpg 768 × 554; 77 KB
- Bailey004.jpg 364 × 1,013; 40 KB
- Bailey005.jpg 747 × 1,050; 138 KB
- Bailey006.jpg 354 × 1,113; 131 KB
- Bailey007.jpg 772 × 803; 138 KB
- Bailey008.jpg 838 × 815; 89 KB
- Bailey009.jpg 774 × 766; 68 KB
- Bailey012.jpg 946 × 530; 73 KB
- Bailey013.jpg 866 × 896; 197 KB
- BurgosFawcett1955 fig11.jpg 1,453 × 2,015; 528 KB
- BurgosFawcett1955 fig13.jpg 1,460 × 2,049; 501 KB
- BurgosFawcett1955 fig14.jpg 1,456 × 1,965; 381 KB
- BurgosFawcett1955 text-fig01.jpg 1,280 × 1,137; 143 KB
- Cat spermatozoa bound to oocyte zona pellucida.jpg 1,000 × 917; 161 KB
- Cytomegalovirus infected spermatozoa EM01.jpg 990 × 991; 204 KB
- Cytomegalovirus infected spermatozoa.jpg 1,000 × 1,260; 324 KB
- Cytomegalovirus virions EM.jpg 911 × 987; 212 KB
- Detection and Localisation of HPV in Sperms.png 600 × 238; 288 KB
- Disomic XY spermatozoa.jpg 393 × 440; 7 KB
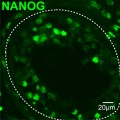
- Dog- spermatozoa NANOG expression.jpg 800 × 691; 109 KB
- Fawcett1975 fig31.jpg 1,280 × 403; 128 KB
- Fawcett1975 fig34.jpg 1,280 × 1,746; 506 KB
- Frazer002 bw600.jpg 600 × 501; 45 KB
- Hamster fused oocyte and spermatozoa.jpg 888 × 405; 98 KB
- Hamster oocyte and spermatozoa.jpg 883 × 836; 266 KB
- Hilfer1990 Fig01.jpg 461 × 1,000; 28 KB
- Human fertilization movie 1 frame 01.jpg 600 × 409; 27 KB
- Human fertilization movie 1 frame 02.jpg 600 × 409; 27 KB
- Human fertilization movie 1 frame 03.jpg 600 × 409; 26 KB
- Human fertilization movie 1 frame 04.jpg 600 × 409; 24 KB
- Human fertilization movie 1 frame 05.jpg 600 × 409; 25 KB
- Human fertilization movie 1 frame 06.jpg 600 × 409; 25 KB
- Human fertilization movie 1 frame 07.jpg 600 × 409; 24 KB
- Human fertilization movie 1 frame 08.jpg 600 × 409; 25 KB
- Human fertilization movie 1 frame 09.jpg 600 × 409; 24 KB
- Human fertilization movie 1 frame 10.jpg 600 × 409; 25 KB
- Human sperm pathologies EM01.jpg 761 × 759; 148 KB
- Human sperm pathology EM02.jpg 800 × 256; 22 KB
- Human spermatid electron micrograph.jpg 619 × 918; 206 KB
- Human spermatid EM01.jpg 1,000 × 762; 162 KB
- Human spermatid EM02.jpg 1,000 × 762; 186 KB
- Human spermatozoa acrosomal protein SP-10.jpg 1,100 × 1,189; 239 KB
- Human spermatozoa chemotaxis labeled model.jpg 1,260 × 699; 158 KB
- Human spermatozoa chemotaxis model.jpg 1,260 × 699; 132 KB
- Human spermatozoa nucleus EM01.jpg 600 × 476; 27 KB
- Human spermatozoa nucleus EM02.jpg 597 × 476; 52 KB
- Human spermatozoa nucleus EM03.jpg 600 × 475; 44 KB
- Human spermatozoa phospholipase C zeta.jpg 1,000 × 571; 119 KB
- Human testis NANOG expression.jpg 1,000 × 328; 77 KB
- Human- spermatozoa NANOG expression 01.jpg 798 × 797; 79 KB
- Human- spermatozoa NANOG expression.jpg 1,000 × 333; 77 KB
- Human- vacuolated spermatozoa.jpg 1,000 × 801; 77 KB
- Human-spermatozoa 01.jpg 1,000 × 805; 90 KB
- Human-spermatozoa 01a.jpg 800 × 644; 66 KB
- Human-spermatozoa 01b.jpg 600 × 483; 43 KB
- Human-spermatozoa 01c.jpg 400 × 322; 22 KB
- Human-spermatozoa EM01.jpg 1,000 × 204; 26 KB
- Human-spermatozoa.jpg 600 × 581; 19 KB
- Keibel Mall 004.jpg 195 × 1,080; 42 KB
- Keith1921 fig011.jpg 1,200 × 614; 76 KB
- Kollmann006.jpg 494 × 680; 21 KB
- Kollmann007.jpg 956 × 582; 45 KB
- Kollmann008.jpg 641 × 577; 61 KB
- Kollmann009.jpg 572 × 552; 24 KB
- Kollmann010.jpg 550 × 541; 19 KB
- Kollmann011.jpg 550 × 541; 28 KB
- Kollmann013.jpg 732 × 626; 95 KB
- Kollmann458.jpg 1,000 × 520; 120 KB
- Kollmann459.jpg 1,000 × 385; 50 KB
- Kollmann460.jpg 551 × 569; 40 KB
- Meiotic prophase I stages.jpg 1,000 × 341; 66 KB
- Minot1897 fig001.jpg 293 × 644; 14 KB
- Minot1897 fig002.jpg 828 × 746; 162 KB
- Model capacitation-induced acrosome docking to sperm membrane.jpg 600 × 489; 73 KB
- Mouse oocyte fertilization 01.jpg 675 × 494; 58 KB
- Mouse spermatogenesis stage cartoon.jpg 1,020 × 800; 298 KB
- Mouse spermatogonial self-renewal.jpg 500 × 794; 49 KB
- Mouse spermatozoa cartoon.jpg 231 × 729; 26 KB
- Mouse spermatozoa mito movie icon.jpg 495 × 495; 43 KB
- Mouse spermatozoa mitochondria 01.jpg 831 × 1,280; 141 KB
- Mouse spermiogenesis 01.jpg 1,200 × 299; 48 KB
- Mouse spermiogenesis model.png 600 × 571; 448 KB
- Mouse- epididymis histology.jpg 751 × 383; 82 KB
- Mouse- gonadal supporting cell development.jpg 1,000 × 588; 74 KB
- Mouse- seminiferous tubule histology.jpg 715 × 427; 76 KB
- Mouse- spermatozoa EM and diagram.jpg 729 × 407; 49 KB
- Mouse- spermatozoa NANOG expression.jpg 800 × 512; 111 KB
- Mouse- zona pellucida 01.jpg 800 × 430; 83 KB
- Mouse- zona pellucida 02.jpg 700 × 688; 95 KB
- Mouse- zona pellucida 03.jpg 1,000 × 345; 64 KB
- Mouse-fertilization 01.jpg 600 × 593; 30 KB
- Mouse-fertilization 02.jpg 1,342 × 691; 118 KB
- Mouse-spermatozoa SLY protein.jpg 637 × 767; 352 KB
- Nelsen1953 fig022.jpg 1,200 × 839; 207 KB
- Pig sperm capacitation 01.jpg 1,000 × 840; 204 KB
- Pig sperm capacitation 02.jpg 600 × 504; 82 KB
- Rat- immortal germ cells are spermatogonial stem cells.jpg 459 × 1,000; 72 KB
- Rugh 019.jpg 383 × 600; 15 KB
- Seminiferous tubule cartoon.jpg 800 × 544; 92 KB
- Single human spermatozoa.jpg 1,000 × 780; 53 KB
- Spermatocyte prophase 1 stages 01.jpg 1,280 × 266; 81 KB
- Spermatogenesis androgen action cartoon.jpg 1,000 × 659; 125 KB
- Spermatogenesis cartoon 01.jpg 1,064 × 759; 142 KB
- Spermatozoa animation icon.jpg 300 × 200; 6 KB
- Spermatozoa animation.gif 300 × 200; 123 KB
- Spermatozoa animation.mov ; 113 KB
- Spermatozoa histology 001.jpg 1,280 × 1,024; 366 KB
- Spermatozoa histology 002.jpg 1,280 × 1,024; 246 KB
- Spermatozoa histology 003.jpg 1,280 × 1,024; 166 KB
- Spermatozoa mitochondria 1cell.jpg 906 × 306; 33 KB
- Spermatozoa mitochondria 2cell.jpg 906 × 306; 34 KB
- Spermatozoa mitochondria 4cell.jpg 906 × 306; 42 KB
- Spermatozoa mitochondria 8cell.jpg 906 × 306; 37 KB
- Spermatozoa mitochondria morula.jpg 906 × 306; 38 KB
- Spermatozoa mitochondria PMID23878233.gif 495 × 495; 974 KB
- Spermatozoa motility 01.mov ; 486 KB
- Spermatozoa principal piece.jpg 800 × 532; 74 KB
- Spermatozoa tail cross-section cartoon.jpg 429 × 429; 43 KB
- Spermatozoa tail EM01.jpg 932 × 613; 75 KB
- Testis histology 001.jpg 1,280 × 1,024; 574 KB
- Testis histology 002.jpg 1,280 × 1,024; 599 KB
- Testis histology 003.jpg 1,280 × 1,024; 183 KB
- Testis histology 004.jpg 1,280 × 1,024; 396 KB
- Testis histology 005.jpg 1,280 × 1,024; 266 KB
- Testis histology 006.jpg 1,280 × 1,024; 251 KB
- Testis histology 007.jpg 1,280 × 1,024; 256 KB
- Testis histology 008.jpg 1,280 × 1,024; 454 KB
- Testis histology 009.jpg 1,280 × 1,024; 339 KB
- Testis histology 010.jpg 1,280 × 1,024; 422 KB
- Testis histology 011.jpg 1,280 × 1,024; 245 KB
- Testis histology 012.jpg 1,280 × 1,024; 266 KB
- Testis histology 013.jpg 1,280 × 1,024; 418 KB
- Testis histology 014.jpg 1,280 × 1,024; 352 KB
- Testis histology 015.jpg 1,280 × 1,024; 281 KB
- Testis histology 016.jpg 1,280 × 1,024; 322 KB
- Testis histology 017.jpg 1,280 × 1,024; 283 KB
- Testis histology 018.jpg 1,280 × 1,024; 350 KB
- Testis histology 019.jpg 1,280 × 1,024; 239 KB
- Testis histology 02.jpg 246 × 481; 49 KB
- Testis histology 023.jpg 600 × 375; 35 KB
- Testis histology 1.jpg 400 × 500; 113 KB
- Testis histology 2.jpg 400 × 500; 32 KB
- Testis histology.jpg 400 × 500; 54 KB
- Testis, young H&E reproductive system, male, convoluted seminiferous tubules x10.jpg 1,280 × 1,024; 396 KB
- Y chromosome haplogroup distribution.jpg 800 × 522; 42 KB