Cardiovascular System - Ventricular Septal Defects: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 29: | Line 29: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
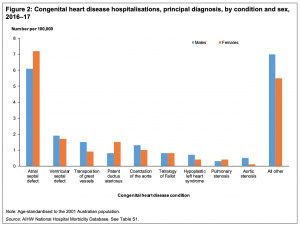
* '''Australia - Congenital heart disease hospitalisations, principal diagnosis, by condition and sex, 2016–17'''<ref>Australian Institute of Health and Welfare 2019. Congenital heart disease in Australia. Cat. no. [https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/congenital-heart-disease-in-australia/contents/table-of-contents CDK 14]. Canberra: AIHW.</ref> "In 2016–17, there were around 4,900 hospitalisations in Australia where congenital heart disease was the principal diagnosis—a rate of 20 hospitalisations per 100,000 population. The highest rate of hospitalisation for a specific form of congenital heart disease was for {{atrial septal defects}} (6.6 hospitalisations per 100,000 population), followed by {{ventricular septal defect}} (1.8), {{transposition of the great vessels}} (1.2), {{patent ductus arteriosus}} and {{coarctation of the aorta}}." | |||
* '''A new anatomic approach of the ventricular septal defect in the interruption of the aortic arch'''{{#pmid:30525196|PMID30525196}} "The aim of this study was to analyse the anatomy of the ventricular septal defect (VSD) in heart specimens with interruption of the aortic arch (IAA) in order to explore the hypothesis of different embryologic mechanisms for the different anatomic types of IAA. We examined 42 human heart specimens, 25 with IAA as the main disease with concordant atrioventricular and ventriculo-arterial connections and two distinct great arteries, and 17 hearts with IAA associated with other malformations [six common arterial trunk (CAT), five double-outlet right ventricle (DORV), three transposition of the great arteries (TGA), three atrioventricular septal defect (AVSD)]. The interruption was classified according to Celoria and Patton. ...These results reinforce the hypothesis that different pathogenic mechanisms are responsible for the two types of IAA, and the inclusion of IAA type B in the group of neural crest defects. Conversely, IAA type A could be due to overlapping mechanisms: flow-related defect (coarctation-like) and neural crest contribution." | * '''A new anatomic approach of the ventricular septal defect in the interruption of the aortic arch'''{{#pmid:30525196|PMID30525196}} "The aim of this study was to analyse the anatomy of the ventricular septal defect (VSD) in heart specimens with interruption of the aortic arch (IAA) in order to explore the hypothesis of different embryologic mechanisms for the different anatomic types of IAA. We examined 42 human heart specimens, 25 with IAA as the main disease with concordant atrioventricular and ventriculo-arterial connections and two distinct great arteries, and 17 hearts with IAA associated with other malformations [six common arterial trunk (CAT), five double-outlet right ventricle (DORV), three transposition of the great arteries (TGA), three atrioventricular septal defect (AVSD)]. The interruption was classified according to Celoria and Patton. ...These results reinforce the hypothesis that different pathogenic mechanisms are responsible for the two types of IAA, and the inclusion of IAA type B in the group of neural crest defects. Conversely, IAA type A could be due to overlapping mechanisms: flow-related defect (coarctation-like) and neural crest contribution." | ||
Latest revision as of 10:58, 14 November 2019
| Embryology - 23 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
LA88.4 Ventricular Septal Defect
| ICD-11 |
|---|
LA88.4 Ventricular septal defect
LA88.40 Trabecular muscular ventricular septal defect | LA88.41 Perimembranous central ventricular septal defect | LA88.42 Ventricular septal defect haemodynamically insignificant |
| ICD-11 Structural developmental anomalies of the circulatory system (draft) |
|---|
| ICD-11 Beta Draft - NOT FINAL, updated on a daily basis, It is not approved by WHO, NOT TO BE USED for CODING except for agreed FIELD TRIALS.
20 Developmental Anomalies - Structural Developmental Anomalies Beta coding and tree structure for "structural developmental anomalies" within this section are shown in the table below. |
| Structural developmental anomalies of the circulatory system |
|
| CD-11 Beta Draft - NOT FINAL, updated on a daily basis, It is not approved by WHO, NOT TO BE USED for CODING except for agreed FIELD TRIALS.
|
Introduction
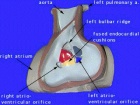
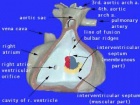
The ventricular septal defect (VSD) is one of the common forms of congenital cardiovascular anomaly, occurring in nearly 50% of all infants with a congenital heart defect. Usually occurs in the membranous (perimembranous) rather than muscular interventricular septum, and is more frequent in males that females. This defect can also contribute to outflow tract (OFT) malformations. See also the recent cardiac development online review section - Normal as opposed to abnormal ventricular septation.
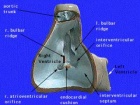
Perimembranous defects are located close to the aortic and tricuspid valves and adjacent to atrioventricular conduction bundle.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Ventricular Septal Defect |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Anatomy
|
|
|
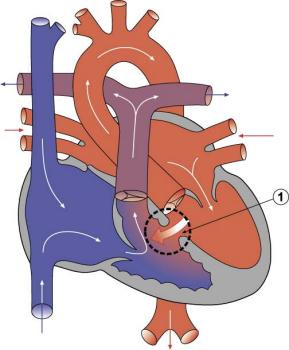
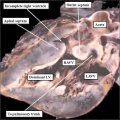
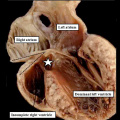
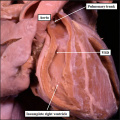
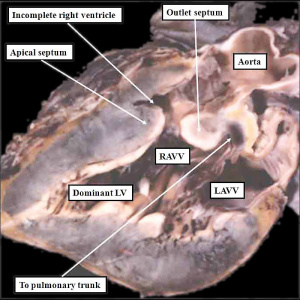
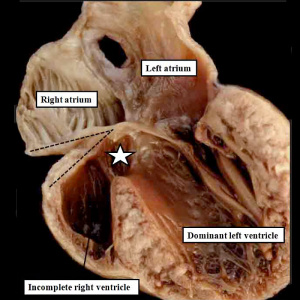
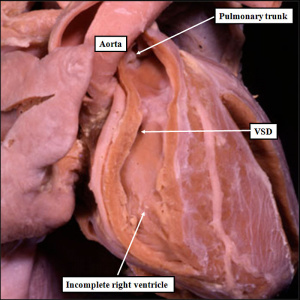
| The image shows a congenitally malformed heart in which both the right and left atrioventricular valves (RAVV, LAVV) are connected with the dominant left ventricle. The right ventricle is incomplete, and is supplied through a ventricular septal defect. Note that, in this heart, the aorta arises from the incomplete right ventricle, and the pulmonary trunk from the dominant left ventricle. This is the arrangement usually described as “transposition”, but better accounted for in terms of discordant ventriculo-arterial connections. | The image shows the lesion known as classical tricuspid atresia. There has been failure of expansion of the atrioventricular junctions, so that the floor of the right is separated from the roof of the right ventricle by the right atrioventricular groove (dashed black lines). Only the dominant left ventricle has an inlet, with the blood entering the incomplete right ventricle through the ventricular septal defect (star). | The image shows a heart dissected to reveal the structure of the incomplete right ventricle. In this heart, as in most example of tricuspid atresia, it gives rise to the pulmonary trunk. The ventricular septal defect (VSD) is restrictive in this heart.
Images from Anderson (2016)[5] |
- Anderson (2016) Human Heart Ventricular Septal Defect
History
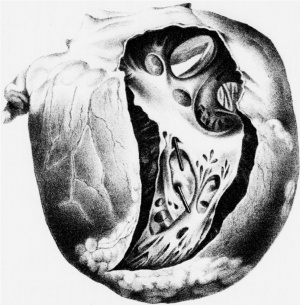 Le Gros Clark (1847)[6] Heart autopsy drawing.
|
The first western clinical description of ventricular septal defects was made by Henri Roger in 1879[7], which later became known as maladie de Roger.
His description was based upon 6 acyanotic patients and autopsy finding of a child with ventricular septal defect. |
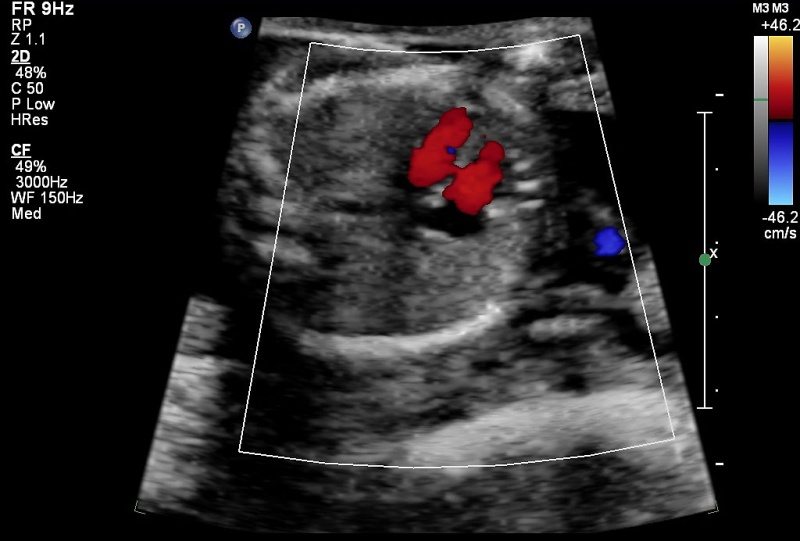
Ultrasound
There is a defect in the ventricular septum adjacent to the atrioventricular valves. Blood flow is seen across the defect on Colour Doppler imaging.
Pulmonary Changes
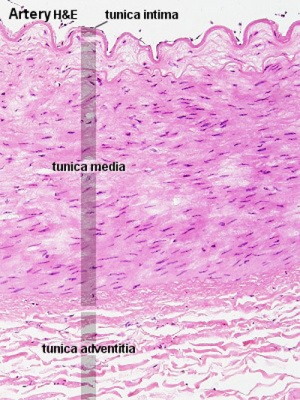
Heath-Edwards classification
(Heath-Edward grade) A pathological grading system for pulmonary artery structural changes that occur with congenital cardiac septal defects. The classification is named after the two original paper authors Donald HEATH and Jessee EDWARDS[8] and grades from I to VI with increasing severity of the arterial changes.
- Grade I - hypertrophy of the media of small muscular arteries and arterioles.
- Grade II - intimal cellular proliferation in addition to medial hypertrophy.
- Grade III - advanced medial thickening with hypertrophy and hyperplasia including progressive intimal proliferation and concentric fibrosis. Results in an obliteration of the arterioles and small arteries.
- Grade IV - "plexiform lesions" of the muscular pulmonary arteries and arterioles with a plexiform network of capillary-like channels within a dilated segment.
- Grade V - complex plexiform, angiomatous and cavernous lesions and hyalinization of intimal fibrosis.
- Grade VI - necrotizing arteritis.
Movies
|
| ||||||||||
|
| ||||||||||
|
|
|
Clinical Classification
Number of parameters are considered when classifying:
- Size of the defect.
- Location of the defect.
- Number of defects.
- Presence or absence of a ventricular septal aneurysm.
Cardiovascular Abnormalities
Heart defects and preterm birth are the most common causes of neonatal and infant death. The long-term development of the heart combined with extensive remodelling and post-natal changes in circulation lead to an abundance of abnormalities associated with this system.
A UK study literature showed that preterm infants have more than twice as many cardiovascular malformations (5.1 / 1000 term infants and 12.5 / 1000 preterm infants) as do infants born at term and that 16% of all infants with cardiovascular malformations are preterm. (0.4% of live births occur at greater than 28 weeks of gestation, 0.9% at 28 to 31 weeks, and 6% at 32 to 36 weeks. Overall, 7.3% of live-born infants are preterm)[9]
"Baltimore-Washington Infant Study data on live-born cases and controls (1981-1989) was reanalyzed for potential environmental and genetic risk-factor associations in complete atrioventricular septal defects AVSD (n = 213), with separate comparisons to the atrial (n = 75) and the ventricular (n = 32) forms of partial AVSD. ...Maternal diabetes constituted a potentially preventable risk factor for the most severe, complete form of AVSD."[10]
In addition, there are in several congenital abnormalities that exist in adults (bicuspid aortic valve, mitral valve prolapse, and partial anomalous pulmonary venous connection) which may not be clinically recognized.
References
- ↑ Australian Institute of Health and Welfare 2019. Congenital heart disease in Australia. Cat. no. CDK 14. Canberra: AIHW.
- ↑ Mostefa Kara M, Houyel L & Bonnet D. (2019). A new anatomic approach of the ventricular septal defect in the interruption of the aortic arch. J. Anat. , 234, 193-200. PMID: 30525196 DOI.
- ↑ Fraser CD, Zhou X, Palepu S, Lui C, Suarez-Pierre A, Crawford TC, Magruder JT, Jacobs ML, Cameron DE, Hibino N & Vricella LA. (2018). Tricuspid Valve Detachment in Ventricular Septal Defect Closure Does Not Impact Valve Function. Ann. Thorac. Surg. , , . PMID: 29625102 DOI.
- ↑ Lee H, Cho JY & Kim GJ. (2011). Complete Repair of Coarctation of the Aorta and a Ventricular Septal Defect in a 1,480 g Low Birth Weight Neonate. Korean J Thorac Cardiovasc Surg , 44, 183-5. PMID: 22263148 DOI.
- ↑ Anderson RH. Teratogenecity in the setting of cardiac development and maldevelopment. (2016)
- ↑ Clark Fle G. (1847). Case of cyanosis, with a description of the appearances presented on dissection, illustrated by the preparation, and a drawing, of the heart. Med Chir Trans , 30, 112.2-120. PMID: 20895864
- ↑ Allwork SP. (1979). Maladie du Roger 1879: a new translation for the centenary. Am. Heart J. , 98, 307-11. PMID: 382810
- ↑ HEATH D & EDWARDS JE. (1958). The pathology of hypertensive pulmonary vascular disease; a description of six grades of structural changes in the pulmonary arteries with special reference to congenital cardiac septal defects. Circulation , 18, 533-47. PMID: 13573570
- ↑ Tanner K, Sabrine N & Wren C. (2005). Cardiovascular malformations among preterm infants. Pediatrics , 116, e833-8. PMID: 16322141 DOI.
- ↑ Loffredo CA, Hirata J, Wilson PD, Ferencz C & Lurie IW. (2001). Atrioventricular septal defects: possible etiologic differences between complete and partial defects. Teratology , 63, 87-93. PMID: 11241431 <87::AID-TERA1014>3.0.CO;2-5 DOI.
Reviews
Ikai A. (2018). Surgical strategies for pulmonary atresia with ventricular septal defect associated with major aortopulmonary collateral arteries. Gen Thorac Cardiovasc Surg , 66, 390-397. PMID: 29802565 DOI.
Penny DJ & Vick GW. (2011). Ventricular septal defect. Lancet , 377, 1103-12. PMID: 21349577 DOI.
Articles
Ghosh S, Sridhar A, Solomon N & Sivaprakasham M. (2018). Transcatheter closure of ventricular septal defect in aortic valve prolapse and aortic regurgitation. Indian Heart J , 70, 528-532. PMID: 30170648 DOI.
Nishioka M, Fuchigami T, Akashige T & Nagata N. (2018). [Successful Staged Surgical Management for Double Outlet Right Ventricle with Ebstein's Anomaly and Aortic Coarctation;Report of a Case]. Kyobu Geka , 71, 615-621. PMID: 30185760
Lee H, Cho JY & Kim GJ. (2011). Complete Repair of Coarctation of the Aorta and a Ventricular Septal Defect in a 1,480 g Low Birth Weight Neonate. Korean J Thorac Cardiovasc Surg , 44, 183-5. PMID: 22263148 DOI.
Callaghan MA, O'Hare B & Casey W. (2012). What other anomalies? Failure to wean post ventricular septal defect repair secondary to anomalous origin of the left coronary artery from the pulmonary artery. Paediatr Anaesth , 22, 487-9. PMID: 22229525 DOI.
Bian C, Ma J, Wang J, Xu G, Jiang J, Yao S & Liu Y. (2011). Perimembranous ventricular septal defect with aneurysm: two options for transcatheter closure. Tex Heart Inst J , 38, 528-32. PMID: 22163127
Fried R, Falkovsky G, Newburger J, Gorchakova AI, Rabinovitch M, Gordonova MI, Fyler D, Reid L & Burakovsky V. (1986). Pulmonary arterial changes in patients with ventricular septal defects and severe pulmonary hypertension. Pediatr Cardiol , 7, 147-54. PMID: 3808993 DOI.
Haworth SG. (1986). Pulmonary vascular bed in children with complete atrioventricular septal defect: relation between structural and hemodynamic abnormalities. Am. J. Cardiol. , 57, 833-9. PMID: 2938464
Hoffmeister HM, Fischbach H & Hoffmeister HE. (1981). Pulmonary arterial changes and hemodynamic parameters in isolated ventricular septal defect. Thorac Cardiovasc Surg , 29, 355-8. PMID: 6179217 DOI.
Search Pubmed
Search Pubmed: Ventricular Septal Defect
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Terms
| Cardiovascular Terms |
|---|
Cardiovascular System Development See also Heart terms, Immune terms and Blood terms.
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 23) Embryology Cardiovascular System - Ventricular Septal Defects. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Ventricular_Septal_Defects
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G