Cardiovascular System - Spleen Development
| Embryology - 25 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
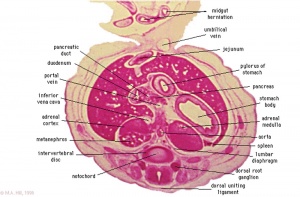
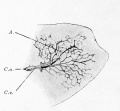
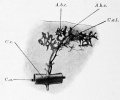
The spleen is located on the left side of the abdomen and has an initial embryonic role in blood formation, and later in immune function and red blood cell recycling. The spleen's haematopoietic function (blood cell formation) is lost with embryo development and lymphoid precursor cells migrate into the developing organ. The mesoderm (mesenchymal progenitors) within the dorsal mesogastrium forms the precursors of nearly all mature spleen stromal cells. This mesoderm form a long strip of cells adjacent to the forming stomach above the developing pancreas. Vascularization of the spleen arises initially branches from the dorsal aorta, then the celiac trunk's largest branch.
The human spleen arises in week 5 within the dorsal mesentery as proliferating mesenchyme overlying the dorsal pancreatic endoderm. Cells required for its hemopoietic function arise from the yolk sac wall and near dorsal aorta. The spleen generates both red and white cells in the second trimester.
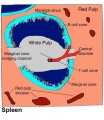
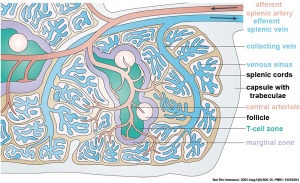
Note there are significant differences between human spleen structure and the usual mouse and rat models. White pulp components, marginal zone is absent, and red pulp sinusoid circulation differs:[1]
- mouse - circulation is closed, afferent arterial blood to the red pulp sinusoids, and to the marginal zone sinus.
- humans - circulation is open, no connection from capillaries to sinuses in the red pulp.
Mouse models have though shown that as the splenic white pulp ages it looses its structural organization, perhaps contributing to the impaired immune system function seen with age.[2]
| Historic Embryology |
|
The Malpighian bodies or Malpighian bodies of the spleen is the historic name for spleen white pulp or splenic lymphoid nodules. Named after Marcello Malpighi (1628 – 1694) an Italian biologist and physician. A spleen lymphoid region, organized as lymphoid sheaths with both T-cell and B-cell compartments, around the branching arterial vessels (resembles lymph node structure).
The term should not be confused with the renal structure, a Malpighian corpuscle (renal corpuscle). |
For all website content related to the spleen see the Category:Spleen
| Immune Links: immune | blood | spleen | thymus | lymphatic | lymph node | Antibody | Med Lecture - Lymphatic Structure | Med Practical | Immune Movies | vaccination | bacterial infection | Abnormalities | Category:Immune | ||
|
| Historic Spleen papers | ||
|---|---|---|
1908 Spleen | 1921 Spleen Hematopoietic Activity | 1936 Spleen Mesothelium |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Spleen Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Development Overview
The table below shows development during teh embryonic and fetal periods.
| Feature | ||
|---|---|---|
| Carnegie stage 14 to 17
appears as a bulge in the dorsal mesogastrium. Mesothelium pseudostratified. | ||
| Mesothelium (pseudostratified} replaced with high columnar cells and then low columnar cells. | ||
| Basement membrane present after this stage. | ||
| Hematopoietic cells detected. | ||
| Spleen is now apparent. Mesenchymal cells differentiated from cells in dorsal mesogastrium. Sinus and hilus formation after this stage. | ||
| Arteries and veins parallel entries at this stage. | ||
| Feature | ||
| reticulum cells (alpha-smooth muscle actin-positive) scattered around the arterioles | ||
| reticulum cells increase in number and began to form a reticular framework. Accumulation of T and B lymphocytes within this framework, and a primitive white pulp was observed around the arterioles. | ||
| reticular framework antigenic diversity, and T and B lymphocytes were segregated in the framework. T lymphocytes were sorted into the alpha-SMA-positive reticular framework, and the periarteriolar lymphoid sheath (PALS) was formed around the arteriole. B lymphocytes aggregated in eccentric portions to the PALS and formed the lymph follicle (LF, reticular framework alpha-SMA-negative). | ||
| marginal zone appears in reticular framework around the white pulp. | ||
| Human embryonic data[7] and fetal data [8] Links: spleen | Kyoto Collection | human timeline | ||
Embryonic Timeline
| Feature | ||
|---|---|---|
| Carnegie stage 14 to 17
appears as a bulge in the dorsal mesogastrium. Mesothelium pseudostratified. | ||
| Mesothelium (pseudostratified} replaced with high columnar cells and then low columnar cells. | ||
| Basement membrane present after this stage. | ||
| Hematopoietic cells detected. | ||
| Spleen is now apparent. Mesenchymal cells differentiated from cells in dorsal mesogastrium. Sinus and hilus formation after this stage. | ||
| Arteries and veins parallel entries at this stage. | ||
| Human data.[7] Links: spleen | Kyoto Collection | Timeline human development | ||
| Spleen Development Timeline | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||
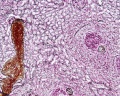
| D4 Dorsal Mesogastrium (Carnegie stage 13) |
D4 Dorsal Mesogastrium (stage 13)
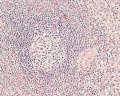
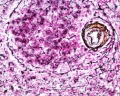
F1 Developing Human Spleen (stage 22)
F2 Developing Human Spleen (stage 22)
F3 Developing Human Spleen (stage 22)
Fetal Timeline
Fetal data from study from week 15 (GA 17) to week 38 (GA 40).[8]
- week 15 (17 GA) - alpha-smooth muscle actin (alpha-SMA)-positive reticulum cells scattered around the arterioles.
- week 18 to 21 (20 - 23 GA) - alpha-SMA-positive reticulum cells increase in number and began to form a reticular framework. An accumulation of T and B lymphocytes occurred within the framework, and a primitive white pulp was observed around the arterioles.
- week 22 (24 GA) - antigenic diversity of the reticular framework was observed, and T and B lymphocytes were segregated in the framework. T lymphocytes were sorted into the alpha-SMA-positive reticular framework, and the periarteriolar lymphoid sheath (PALS) was formed around the arteriole. B lymphocytes aggregated in eccentric portions to the PALS and formed the lymph follicle (LF). The reticular framework of the LF was alpha-SMA-negative.
- week 24 (26 GA) - marginal zone appeared in the alpha-SMA-positive reticular framework around the white pulp.
(Note - weeks above are approximate post-conception PC corrected from gestational age GA[8])
Reticular framework of white pulp and marginal zone.[8]
- "The antigenic heterogeneity of the reticular framework of the white pulp (WP) and marginal zone (MZ) is well documented in the human adult spleen. The ontogeny of the WP and MZ of human fetal spleens was examined with special reference to the heterogeneity of the reticular framework. In the spleen of the 17th gestational week (gw), alpha-smooth muscle actin (alpha-SMA)-positive reticulum cells were scattered around the arterioles. From the 20th to 23rd gw, alpha-SMA-positive reticulum cells increased in number and began to form a reticular framework. An accumulation of T and B lymphocytes occurred within the framework, and a primitive WP was observed around the arterioles. At the 24th gw, antigenic diversity of the reticular framework was observed, and T and B lymphocytes were segregated in the framework. T lymphocytes were sorted into the alpha-SMA-positive reticular framework, and the periarteriolar lymphoid sheath (PALS) was formed around the arteriole. B lymphocytes aggregated in eccentric portions to the PALS and formed the lymph follicle (LF). The reticular framework of the LF was alpha-SMA-negative. MZ appeared in the alpha-SMA-positive reticular framework around the WP at the 26th gw. The PALS, LF, and MZ developed with gestational time. The reticular framework of the PALS, LF, and MZ is thus heterogeneous in the fetal spleen, and the development of the heterogeneity is related to the ontogeny of the PALS, LF, and MZ."
Spleen Development Movies
| <html5media height="450" width="300">File:Lesser sac 01.mp4</html5media> | This animation shows the development of the lesser sac associated with the rotation of the stomach and growth of the liver.
This cross-sectional view of the abdomen viewed from above, with dorsal (back) top and ventral (front) bottom of animation. Later the retroperitoneal position of the developing kidneys is also shown either side of the dorsal (thoracic) aorta.
Legend
|
|
| Gastrointestinal Tract Movies | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
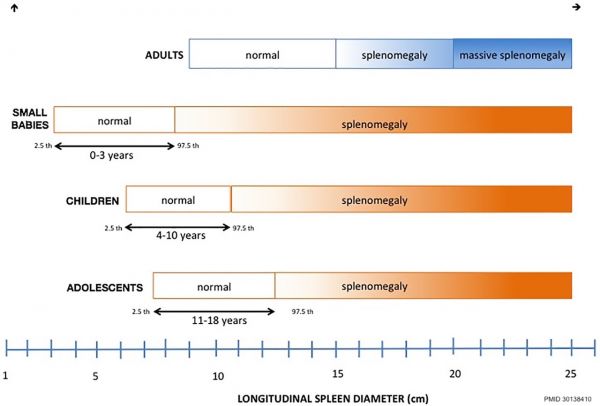
Childhood Spleen
The graph below compares normal and abnormal growth of the spleen in childhood with similar changes in the adult.[12]
Adult Spleen
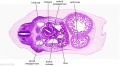
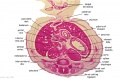
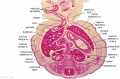
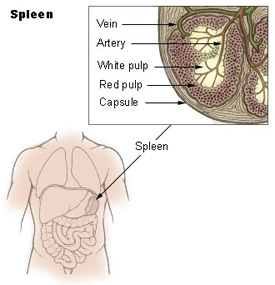
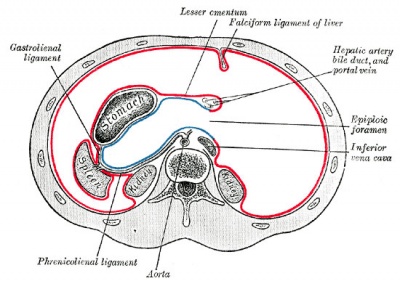
Anatomy
Adult Spleen and ligamentous attachments.
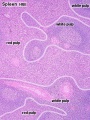
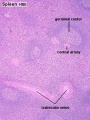
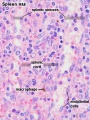
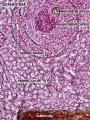
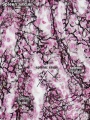
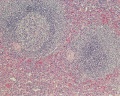
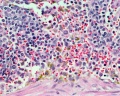
Histology
| Spleen Development: SH Lecture Spleen | SH Adult Histology | Overview Red and White Pulp | Overview Red and White Pulp | Cords and Sinuses | Reticular Fibre overview | Reticular Fibre detail | unlabeled red and white pulp | unlabeled red pulp and macrophages | unlabeled white pulp germinal centre | unlabeled reticular fibre | unlabeled white pulp reticular | unlabeled red pulp reticular | Structure cartoon | Cartoon and stain | Category:Spleen | Histology Stains | Immune System Development |
Note that a recent comparative review [13] describes structural differences between the human spleen and that of rodents (rats and mice). Rodents have an additional marginal zone, between white and red pulp, forming a well-delineated B-cell compartment.
- Human splenic secondary follicles three zones - germinal centre, mantle zone and superficial zone.
Molecular
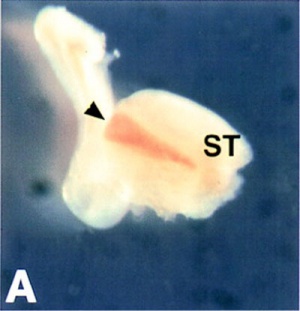
Hox11
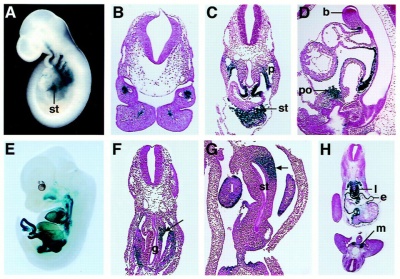
The spleen of wild-type (A) embryos is observed as a brown stripe next to the stomach (black arrowhead).[14]
Mouse spleen formation commences normally at E11.5 and Hox 11 gene expression was previously shown as essential for cell survival during spleen development.[15][16]
Capsulin
A subpopulation of splanchnic mesoderm cells in mice expresses this basic helix-loop-helix (bHLH) transcription factor early in spleen organogenesis. This transcription factor is also expressed in mesenchymal cells that encapsulate the epithelial primordia of internal organs.[17]
Capsulin also known as Epicardin, Podocyte-Expressed 1, POD1, TRANSCRIPTION FACTOR 21; TCF21
- Links: OMIM
Bapx1
Mouse Model
Human spleen microanatomy: why mice do not suffice.[13] (text below edited from paper summary)
- mouse, rat and human - spleen consists of the white pulp embedded in the red pulp.
- white pulp - T and B lymphocytes form accumulations, the periarteriolar lymphatic sheaths and the follicles, located around intermediate-sized arterial vessels, the central arteries.
- red pulp - reticular connective tissue containing all types of blood cells.
- mouse and rat - an additional B-cell compartment, the marginal zone, between white and red pulp.
- marginal zone absent in human spleen.
- Human splenic secondary follicles comprise three zones: a germinal centre, a mantle zone and a superficial zone. In humans, arterioles and sheathed capillaries in the red pulp are surrounded by lymphocytes, especially by B cells.
- Human sheathed capillaries are related to the splenic ellipsoids of most other vertebrates.
- Such vessels are lacking in rats or mice, which form an evolutionary exception. Capillary sheaths are composed of endothelial cells, pericytes, special stromal sheath cells, macrophages and B lymphocytes.
- Human spleens most probably host a totally open circulation system, as connections from capillaries to sinuses were not found in the red pulp.
- Three stromal cell types of different phenotype and location occur in the human white pulp.
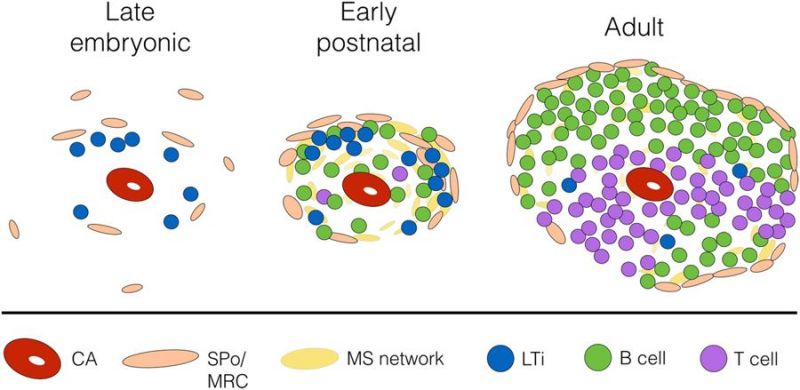
Proposed interactions between spleen organiser cells and LTi/B cells which leads to marginal zone remodelling and adult white pulp formation. (text below modified from figure legend[18]) | ||
| Late embryogenesis (E16.5), CD4+IL-7Rα+ lymphoid tissue inducer (LTi) cells can be observed between the central arteriole (CA) and presumptive MAdCAM-1+VE-Cadherin+ spleen organiser (SPo) cells. | After birth LTi continue to co-localise with SPo (MAdCAM-1+CD31+), which form a primitive marginal zone network surrounding the CD31+ central arterioles. This postnatal developmental stage coincides with B cell migration into the spleen, and both LTi and B cells contribute to lymphotoxin signalling which upregulates MAdCAM-1 expression in the marginal zone, and initiates vascular reorganisation of the Flk-1+ephrinB2+ marginal sinus (MS) network that extends through the white pulp. | Adult MAdCAM-1+CD31+ cells are localised in the marginal zone and represent mature marginal zone reticular cells (MRC). |
Abnormalities
Congenital absence of the spleen is usually accompanied by complex cardiac malformations, malposition and maldevelopment of the abdominal organs, and abnormal lobation of the lungs. There are a range of other spleen anatomical development abnormalities, some of which have no effect and others are very rare, including asplenia, polysplenia, hyposplenia, lobulation of spleen, accessory spleens, accessory splenic nodules, wandering spleen, splenogonadal and splenopancreatic fusion, splenic cysts, and cavernous haemangioma of the spleen.
LB22 Structural developmental anomalies of spleen
ICD-11 LB22 Structural developmental anomalies of spleen
LB22.0 Congenital asplenia
LB22.0 Congenital asplenia - A condition caused by failure of the spleen to develop during the antenatal period. This condition may present with pneumococcal sepsis. Confirmation is through verification of no spleen by imaging.
Can be due to left/right patterning abnormality or failure of early spleen differentiation.
LB22.1 Polysplenia
LB22.1 Polysplenia - A condition caused by the development of supernumerary spleens during the antenatal period. This condition may present with cardiac defects, anomalies in abdominal organs, or may be asymptomatic.
Clinically no significant efects in most patients, occur as single or multiple and generally found in autopsy or as an an incidental finding, occurs in approximately 10% to 15% of individuals.[19] Thought to occur due to a failure of primordia fusion within the dorsal mesogastrium.
LB22.2 Ectopic spleen
A very rare abnormality where the spleen can be found anatomically located in a range of places in the abdominal or thoracic cavity.
Splenogonadal Fusion
Rare resulting from abnormal fusion of the splenic and gonadal primordia during prenatal development. On the left side and more common in male and adhesion to the gonad, epididymis or ductus deferens and then follows the caudal descent with the gonad. Failure of complete descent can also result in associated intraabdominal cryptorchism.
Two classifications:
- continuous - orthotopic spleen connects to the gonad with a cord of fibrous or splenic tissue.
- discontinuous - no connection between the orthotopic spleen and gonad.
(More? testis)
Wandering Spleen
Connexin-43 involved with abnormal spleen development (cardiac and lung also).
References
- ↑ Steiniger B, Bette M & Schwarzbach H. (2011). The open microcirculation in human spleens: a three-dimensional approach. J. Histochem. Cytochem. , 59, 639-48. PMID: 21525186 DOI.
- ↑ Masters AR, Jellison ER, Puddington L, Khanna KM & Haynes L. (2018). Attrition of T Cell Zone Fibroblastic Reticular Cell Number and Function in Aged Spleens. Immunohorizons , 2, 155-163. PMID: 30706058 DOI.
- ↑ Mebius RE & Kraal G. (2005). Structure and function of the spleen. Nat. Rev. Immunol. , 5, 606-16. PMID: 16056254 DOI.
- ↑ Steiniger BS, Wilhelmi V, Berthold M, Guthe M & Lobachev O. (2018). Locating human splenic capillary sheaths in virtual reality. Sci Rep , 8, 15720. PMID: 30356180 DOI.
- ↑ Golub R, Tan J, Watanabe T & Brendolan A. (2018). Origin and Immunological Functions of Spleen Stromal Cells. Trends Immunol. , 39, 503-514. PMID: 29567327 DOI.
- ↑ Barlow-Anacker AJ, Fu M, Erickson CS, Bertocchini F & Gosain A. (2017). Neural Crest Cells Contribute an Astrocyte-like Glial Population to the Spleen. Sci Rep , 7, 45645. PMID: 28349968 DOI.
- ↑ 7.0 7.1 7.2 7.3 Endo A, Ueno S, Yamada S, Uwabe C & Takakuwa T. (2015). Morphogenesis of the spleen during the human embryonic period. Anat Rec (Hoboken) , 298, 820-6. PMID: 25403423 DOI.
- ↑ 8.0 8.1 8.2 8.3 8.4 Satoh T, Sakurai E, Tada H & Masuda T. (2009). Ontogeny of reticular framework of white pulp and marginal zone in human spleen: immunohistochemical studies of fetal spleens from the 17th to 40th week of gestation. Cell Tissue Res. , 336, 287-97. PMID: 19255788 DOI.
- ↑ Asayesh A, Sharpe J, Watson RP, Hecksher-Sørensen J, Hastie ND, Hill RE & Ahlgren U. (2006). Spleen versus pancreas: strict control of organ interrelationship revealed by analyses of Bapx1-/- mice. Genes Dev. , 20, 2208-13. PMID: 16912273 DOI.
- ↑ Steiniger B, Ulfig N, Risse M & Barth PJ. (2007). Fetal and early post-natal development of the human spleen: from primordial arterial B cell lobules to a non-segmented organ. Histochem. Cell Biol. , 128, 205-15. PMID: 17624541 DOI.
- ↑ Drayton DL, Liao S, Mounzer RH & Ruddle NH. (2006). Lymphoid organ development: from ontogeny to neogenesis. Nat. Immunol. , 7, 344-53. PMID: 16550197 DOI.
- ↑ Pelizzo G, Guazzotti M, Klersy C, Nakib G, Costanzo F, Andreatta E, Bassotti G & Calcaterra V. (2018). Spleen size evaluation in children: Time to define splenomegaly for pediatric surgeons and pediatricians. PLoS ONE , 13, e0202741. PMID: 30138410 DOI.
- ↑ 13.0 13.1 Steiniger BS. (2015). Human spleen microanatomy: why mice do not suffice. Immunology , 145, 334-46. PMID: 25827019 DOI.
- ↑ 14.0 14.1 Lettice LA, Purdie LA, Carlson GJ, Kilanowski F, Dorin J & Hill RE. (1999). The mouse bagpipe gene controls development of axial skeleton, skull, and spleen. Proc. Natl. Acad. Sci. U.S.A. , 96, 9695-700. PMID: 10449756
- ↑ Roberts CW, Shutter JR & Korsmeyer SJ. (1994). Hox11 controls the genesis of the spleen. Nature , 368, 747-9. PMID: 7908720 DOI.
- ↑ Dear TN, Colledge WH, Carlton MB, Lavenir I, Larson T, Smith AJ, Warren AJ, Evans MJ, Sofroniew MV & Rabbitts TH. (1995). The Hox11 gene is essential for cell survival during spleen development. Development , 121, 2909-15. PMID: 7555717
- ↑ Lu J, Chang P, Richardson JA, Gan L, Weiler H & Olson EN. (2000). The basic helix-loop-helix transcription factor capsulin controls spleen organogenesis. Proc. Natl. Acad. Sci. U.S.A. , 97, 9525-30. PMID: 10944221
- ↑ 18.0 18.1 Tan JKH & Watanabe T. (2018). Determinants of postnatal spleen tissue regeneration and organogenesis. NPJ Regen Med , 3, 1. PMID: 29367882 DOI.
- ↑ Wadham BM, Adams PB & Johnson MA. (1981). Incidence and location of accessory spleens. N. Engl. J. Med. , 304, 1111. PMID: 7207579
Reviews
Tan JKH & Watanabe T. (2018). Determinants of postnatal spleen tissue regeneration and organogenesis. NPJ Regen Med , 3, 1. PMID: 29367882 DOI.
Varga I, Babala J & Kachlik D. (2018). Anatomic variations of the spleen: current state of terminology, classification, and embryological background. Surg Radiol Anat , 40, 21-29. PMID: 28631052 DOI.
Cesta MF. (2006). Normal structure, function, and histology of the spleen. Toxicol Pathol , 34, 455-65. PMID: 17067939 DOI.
Drayton DL, Liao S, Mounzer RH & Ruddle NH. (2006). Lymphoid organ development: from ontogeny to neogenesis. Nat. Immunol. , 7, 344-53. PMID: 16550197 DOI.
Shapiro-Shelef M & Calame K. (2005). Regulation of plasma-cell development. Nat. Rev. Immunol. , 5, 230-42. PMID: 15738953 DOI.
Straub RH. (2004). Complexity of the bi-directional neuroimmune junction in the spleen. Trends Pharmacol. Sci. , 25, 640-6. PMID: 15530642 DOI.
Balliu PR, Bregante J, Pérez-Velasco MC, Fiol M, Galiana C, Herrera M & Mulet J. (2004). Splenic haemorrhage in a newborn as the first manifestation of wandering spleen syndrome. J. Pediatr. Surg. , 39, 240-2. PMID: 14966753
Chadburn A. (2000). The spleen: anatomy and anatomical function. Semin. Hematol. , 37, 13-21. PMID: 10676919
Lane PA. (1995). The spleen in children. Curr. Opin. Pediatr. , 7, 36-41. PMID: 7728201
Articles
Xu SY, Sun K, Xie HY, Zhou L, Zheng SS & Wang W. (2017). Accessory spleen located in the right parietal peritoneum: The first case report. Medicine (Baltimore) , 96, e7957. PMID: 28930831 DOI.
Search PubMed
Search NCBI Bookshelf: Spleen Development
Search PubMed: Search August 2006 "Spleen Development" 13,401 reference articles of which 450 were reviews.
Search term = Spleen Development | Spleen Abnormalities
Additional Images
Historic
Fig. 519. Total mount of an injected spleen embryo pig 12 cm
Terms
- adenoid - (Greek " +-oeides = in form of) in the form of a gland, glandular; the pharyngeal tonsil.
- afferent lymph - vessel carrying lymph towards a node containing antigen-presenting cells, antigen, effector and memory T cells, and regulatory T cells.
- acquired immune deficiency syndrome - (AIDS) note this is now better described as "advanced HIV disease", decrease in the number of CD4 T cells. (More? Immunobiology - AIDS)
- anastomose - joining of two tubes or structures together.
- Antibody mediated immunity - the immune function of plasma cells (active B lymphocytes) secreting antibody which binds antigen.
- antibodies - mammals have five classes (IgA, IgD, IgE, IgG, and IgM)
- antigen - any substance that is recognised by the immune system and stimulates antibody production.
- appendix - is a gut-associated lymphoid tissue (GALT) located at the beginning of the colon. The anatomy is as a finger-like structure that arises from the cecum. The length (2.5-13 cm) is longer in both infants and children and also has more abundant lymphatic tissue in early life. The wall structure is similar to the small intestine (though with no villi), nor plicae circularis. Lymph nodules surround the lumen of the gastrointestinal tract and extend from the mucosa into the submucosa.
- B cell - (B-cell, B lymphocyte) historically named after a structure called the bursa of Fabricius in birds, a source of antibody-producing lymphocytes. These immune cells develop in the bone marrow. (More? Electron micrographs of nonactivate and activated lymphocytes)
- blood - liquid connective tissue containing cells of the lymphatic system see Cardiovascular terms
- B lymphocyte - (B cell, B-cell)
- BALT - (Bronchus Associated Lymphoid Tissue) immune tissue associated with the respiratory tract.
- band cell - (band neutrophil or stab cell) immature neutrophil seen in bone marrow smear, a cell undergoing granulopoiesis, derived from a metamyelocyte, and leading to a mature granulocyte. Also occasionally seen in circulating blood.
- bone marrow sinusoid - endothelial cells and no supporting cells vascular space supplied by arteriole and capillary vessels, interconnected by inter-sinusoidal capillaries, spanning throughout the bone marrow. Radially distributed around the draining central sinus (about 100 µm in diameter). Bone marrow sinusoids are unique and are not comparable with regular veins.
- cecum - (caecum, Latin, caecus = "blind") within the gastrointestinal tract a pouch that connects the ileum with the ascending colon of the large intestine.
- cell - has a specific cell biology definition, but is often used instead of "lymphocyte" when describing B and T cells.
- cell-mediated immunity - the immune function of T lymphocytes. (More? Immunobiology - T Cell-Mediated Immunity)
- central tolerance - in thymus mediated by cortical epithelial cells, medullary epithelial cells and thymic dendritic cells, involves deletion of self reactive thymocytes (T cell) (see [https://www.ncbi.nlm.nih.gov/pubmed/30476234 PMID30476234).
- "clockface" - a term used to describe the appearance of plasma cell nuclei due to the clumping of the chromatin at the nucleus periphery. More clearly seen in tissue plasma cells that the bone marrow smear, where they are sometimes confused with the basophilic erythroblasts. Image - plasma cell
- CD - (cluster of differentiation) identifies immunological surface markers on cells. Positive (+) generally means that the substance is expressed/identified, while negative (-) means that it is missing/not identified.
- CD4+ - (T helper cells) refers to T lymphocytes that express CD4 (cluster of differentiation 4, a glycoprotein of the immunoglobulin superfamily) on their surface, associated with helper/inducer function. These cells can be infected by human immunodeficiency virus (HIV).
- CD4/CD8 ratio - clinical measurement of different immune cell types (ratios between 1.5 to 2.5 are considered normal). Viral infections such as HIV, cytomegalovirus, Epstein-Barr virus, and influenza virus, associated with an inversion of the ratio.
- CD8+ - (cytotoxic T cells) refers to T lymphocytes that express CD8 (glycoprotein of the immunoglobulin superfamily) on their surface, associated with cytotoxic/suppressor activity.
- "clockface" - a term used to describe the appearance of plasma cell nuclei due to the clumping of the chromatin at the nucleus periphery. More clearly seen in tissue plasma cells that the bone marrow smear, where they are sometimes confused with the basophilic erythroblasts.
- cords of Billroth - spleen cellular columns located in red pulp. surrounded by splenic sinusoids. Cords contain reticular cells, macrophages, lymphocytes, plasma cells and erythrocytes.
- cortex - outer layer, used in association with medulla (innner layer or core) a general description that can be applied to describing an organ with a layered structure.
- cortical Thymic Epithelial Cell - (cTEC, types I - IV) support and antigen presenting cells located in the cortex regions of the thymus required for positive and negative selection of maturing T cells. See also medullary epithelial cell.
- crypt - (tonsil crypt) tonsil squamous epithelium infold, with intraepithelial passages containing non-epithelial cells. Functions include: intimate contact between immune response effector cells, facilitate transport of antigens, synthesise secretory components, and contain a pool of immunoglobulins. PMID 7559106
- dendritic cell - (DC, antigen-presenting cell, APC) cells that present antigens and induce a primary immune response in resting naïve T lymphocytes. Originate from the same common progenitor as monocytes (PMID 20193011). In 2011 Ralph M. Steinman received half the Nobel Prize half of the award to to Ralph M. Steinman for his discovery of the dendritic cell and its role in adaptive immunity.
- Effector cells - the immune functioning (active) B and T lymphocytes.
- Efferent lymph - vessel carrying lymph away from a node.
- fibroblastic reticular cell - (FRC) specialized myofibroblasts that form the structural mesenchymal network "sponge" within lymphoid tissue that regulate immune cell migration, activation, and survival. Immune T cells, B cells, dendritic cells (DCs), plasma cells and macrophages move and interact.
- follicular dendritic cell - (FDC) in B cell follicles of secondary lymphoid organs, cells interspersed within the stromal cell network function: Primary - help B cells to cluster. Secondary - in GC long-term retention of intact antigen and support B cell survival.
- GALT - Gut Associated Lymphatic Tissue consisting of Peyer’s patches, isolated lymphoid follicles and mesenteric lymph nodes.
- germinal centre - (GC) centre of B cell follicles of secondary lymphoid organs, where antigen-activated B-cell clones expand and undergo immunoglobulin gene hypermutation and selection.
- haemopoiesis (hemopoiesis) formation of blood cells.
- Hassall's corpuscles - (Hassall's body, thymic corpuscle) Epithelial reticular cells located in the thymic medulla. Named after Arthur Hill Hassall (1817-1894) a British physician and chemist.
- high endothelial venule - (HEV) the specialised post-capillary venous region that enables blood lymphocytes and pre-dendritic cells to enter a lymph node. The endothelial cells express ligands that bind lymphocytes, aiding their adhesion and subsequent transmigration into the lymph node. With inflammation, monocytes and NK cells can also enter here.
- humoral immune response - production of antibody by plasma cells derived from B lymphocytes (B cells).
- IEL - Intraepithelial Lymphocyte are T lymphocytes located in the gastrointestinal tract epithelium. Natural IELs (previously ‘type b’ IELs) acquire activated phenotype during development in the thymus in the presence of self antigens. Induced IELs (previously ‘type a’ IELs) progeny of conventional T cells activated post-thymically in response to peripheral antigens.
- IgA - the main class of antibody released at mucosal surfaces and in secretions (saliva, tears, milk, and respiratory and intestinal secretions) and the most abundantly produced antibody (70%). PMID 22566964
- IgD - the immunoglobulin B cell starts to produce as a cell-surface molecule after leaving the bone marrow.
- IgE - bind Fc receptors (surface of mast cells in tissues and basophils in the blood) release of potent pro inflammatory molecules mediators of allergic reactions.
- IgG - the major class of immunoglobulin in the blood.
- IgM - the first class of antibody made by a developing B cell, which may switch to making other classes of antibody.
- immunodeficiency - when one or more components of the immune system is defective. (More? Immunobiology - immunodeficiency)
- immunoglobulin - (antibody, Ab) protein produced by plasma cells.
- immunosenescence - in ageing and disease, refers to a weaker immune responses producing a progressive deterioration and increased susceptibility to infectious diseases, neoplasia, and autoimmune diseases.
- innate lymphoid cells - (ILCs) subset of lymphocytes that lack antigen-specific receptors, are located in peripheral tissues and abundant at barrier surfaces, decrease in number with age. PMID 29924974
- intraepithelial lymphocyte (IEL) immune cells residing in the gastrointestinal tract epithelium. image - Intraepithelial lymphocyte differentiation
- involution - in the thymus refers to the replacement, mainly in the cortex, of cells by adipose tissue. (More? PubMed- thymus involution) | Cancer Medicine - Thymomas and Thymic Tumors)
- Kupffer cells - stellate macrophage cells located in the liver sinusoids, named after Karl Wilhelm von Kupffer (1829 - 1902) a German anatomist who originally identified these cells. (More? Liver Development)
- lacteal - term used to describe the lymphatic vessels of the small intestine.
- lamina propria - a layer of loose connective tissue found underneath an epithelium, together with the epithelium described as mucosa.
- Langerhans cell - (LC, dendritic cell) Antigen-presenting immune cell found mainly in the basal/suprabasal layers of adult skin and mucosa. Cells lie in the basal/suprabasal layers of stratified epidermal and mucosal tissues. First in the innate antiviral immune defines and can migrate to lymph nodes and induce a T cell–mediated adaptive immune response. (More? Integumentary | Immune System Development)
- Leukocyte - (Greek, lukos = clear, white) white blood cell.
- lingual - related to the tongue, as in lingual tonsil, forms part of Waldeyer’s ring.
- lymph node - connective tissue encapsulated lymphoid organ (1mm - 2cm in size), positioned in the pathway of lymph vessels. (More? Lymph Node Development)
- lymphangion - the functional unit of a lymph vessel that lies between two semilunar (half moon-shaped) valves.
- lymphangiogenesis - formation of new lymph vessels from pre-existing lymphatic structures. During embryogenesis and in adult tissues as reaction to inflammation or injury.
- M cell - (microfold cell) found in the follicle-associated epithelium of the Peyer's patch. Function to transport gut lumen organisms and particles to immune cells across the epithelial barrier.
- macrophage - a large highly motile white blood cell which engulfs foreign material (bacteria etc) and both degenerating cells and cell fragments. Differentiates from a monocyte and found in many different tissues and locations. Current theory suggests tissue macrophage is also derived from resident stem cell population in many tissues. More? Immunobiology - Defects in phagocytic cells are associated with persistence of bacterial infection)
- MALT - Mucosa Associated Lymphoid Tissue.
- medulla - inner layer or core, used in association with cortex (outer layer) a general description that can be applied to describing an organ with a layered structure.
- medullary Thymic Epithelial Cell - (mTEC, types I-VII) support and antigen presenting cells located in the medullary regions of the thymus, required for central tolerance (negative selection) of maturing T cells (PMID 11375064). See also cortical thymic epithelial cell.
- Memory Cell - effector T cell (lymphocyte)
- mesenteric lymph nodes - Part of GALT as well as being involved in gut-draining. image - mesenteric lymph nodes
- Mononuclear Phagocytic System - (MPS, Lymphoreticular System, Reticuloendothelial System, RES) Consists of circulating monocytes in the peripheral blood and non-circulating (fixed) tissue macrophages (MΦ) located in tissues and organs.
- NAVL - (naval) mnemonic to remember the neurovascular bundle components Nerve Artery Vein Lymph found travelling together within organs and tissues.
- negative selection - T cells bearing autoreactive T cell antigen receptors (TCRs) are eliminated during their development in the thymus, protects against autoimmunity.
- normoblast - seen in bone marrow smear, a developing erythroblast (red blood cell) that still retains a nucleus.
- nude mice - (nu/nu) mice which are congenitally hairless and athymic, therefore they do not reject tissue and tumor grafts.
- PALS - acronym for PeriArterial Lymphoid Sheath in the spleen white pulp.
- parenchyma - (Greek = enkeim "to pour in") cells forming the functional cells of an organ or tissue. These cells carry out the function of the organ at a cellular level, and are not the structural cells, connective tissue, extracellular matrix (stromal).
- periarterial lymphoid sheath - (PALS) in the spleen the white pulp that surrounds the central arteries. (T-lymphocytes,macrophages and plasma cells)
- pharyngeal pouch III - origin of endodermal component of the thymus (also formed from neural crest). Pharyngeal arches
- Plasma Cell - active B cell (lymphocyte) which is secreting antibody. Located in either bone marrow or peripheral lymphoid tissues, these cells have and increased cytoplasmic volume (due to increase rough endoplasmic reticulum) in comparison to the inactive (non-secreting) lymphocyte.
- primary follicle - follicle that does not contain germinal centre, secondary follicles do germinal centre.
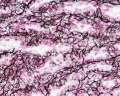
- red pulp - spleen region, organized as cell cords (splenic cords, cords of Billroth) and vascular sinuses.
- regulatory T cells - (Tregs) maintain self tolerance and suppress pathological immune responses by control of immune response to non-self antigens.
- reticular fibres - reticular cells secrete this extracellular matrix protein, composed of type III collagen.
- right lymphatic duct - drains most of the right upper quadrant. See also thoracic duct.
- secondary follicle - contain germinal centre, primary follicle does not contain germinal centre.
- sentinel lymph node - the hypothetical first lymph node or group of nodes reached by metastasizing cancer cells from a primary tumour.
- sinus - a larger vessel or space usually curved that may contain air, blood, or lymph. e.g. splenic medullary sinus, lymph node medullary sinus, sub-capsular sinus, trabecular sinus.
- sinusoid - a tiny vessel with a tortuous path and many connections to similar vessels. e.g. hepatic and bone marrow sinusoids.
- splenic capillary sheaths - in spleen around capillary endothelium and consist of three main cell types: CD271+ stromal capillary sheath cells, CD68+CD163− macrophages and recirculating B-lymphocytes. Sheaths may; 1. allow interaction among sheath macrophages and B-lymphocytes, 2. attract recirculating B-lymphocytes from the open circulation of the red pulp to start migration into white pulp follicles. 30356180
- splenic sinusoids - enlarged splenic spaces located in red pulp and surrounding cords of Billroth.
- sphingosine-1-phosphate - (S1P) sphingolipid secreted into the extracellular space establishing a gradient acting through G protein-coupled receptors to attract lymphocytes out of lymphoid organs (lymph node, thymus, spleen) into the circulation.
- stroma - (Greek = "a cover, table-cloth, bedding") tissue forming the framework/support of an organ or tissue. That is the structural cells which form connective tissue and secrete extracellular matrix, rather than the functional cells (parenchymal). All organs can therefore be functionally divided into these 2 components, stromal/parenchymal.
- Subcapsular sinus (=marginal sinus) space lying under the connective tissue capsule which receives lymph from afferent lymphatic vessels.
- T cell - (T-cell, T lymphocyte) named after thymus, where they develop, the active cell is responsible for cell-mediated immunity (killer T cells and helper T cells). Cells express T-cell receptor on surface and directly kill virally or bacterially infected cells. These cells can themselves be infected by HIV. (More? Electron micrographs of nonactivate and activated lymphocytes)
- TEC - (Thymic Epithelial Cell) thymus support and antigen presenting cells further divided anatomically and functionally into medullary TEC (mTEC, types I-VII, for central tolerance) and cortical epithelial cell (cTEC, types I-IV, positive and negative selection) populations (see PMID 28800929 PMID 30308217).
- T cell activation - (T lymphocyte activation)The activation process begins with T-cells searching for and encountering antigen-bearing dendritic cells within lymph nodes.
- thoracic duct - (TD) largest and main lymphatic vessel, drains the lower body including the extremities and abdomen. Intra-thoracic tributaries include: intercostal, mediastinal, and bronchomediastinal trunks.
- Thymic corpuscle - (Hassall's corpuscle) a mass of concentric epithelioreticular cells found in the thymus. The number present and size tend to increase with thymus age. (see classical description of Hammar, J. A. 1903 Zur Histogenese und Involution der Thymusdriise. Anat. Anz., 27: 1909 Fiinfzig Jahre Thymusforschung. Ergebn. Anat. Entwickl-gesch. 19: 1-274.)
- thymic epitheliocytes - reticular cells located in the thymus cortex that ensheathe the cortical capillaries, creating and maintain the microenvironment necessary for the development of T-lymphocytes in the cortex.
- T helper cells - (helper T-cells) (Th cells, CD4+) refers to T lymphocytes that when mature express CD4 (glycoprotein of the immunoglobulin superfamily) on their surface.
- T lymphocyte - (T cell, T-cell) regulate cell-mediated immunity.
- thymus - an immune/endocrine (thymic hormone, thymosins) organ involved in the maturation (differentiation) of T lymphocytes (T-cells).
- tonsils - lymph nodules embedded in the mucus membranes located at the back of the mouth and top of the throat. The overlying epithelium helps identify the location.
- tonsillitis - a common bacterial infection of the palatine tonsils, occurring mostly in children and young adults and can also become recurrent tonsillitis.
- vermiform appendix - see appendix, anatomical region containing gut-associated lymphoid tissue located within the gastrointestinal tract at the beginning of the colon. The anatomy is as a finger-like structure that arises from the cecum. The length (2.5-13 cm) is longer in both infants and children and also has more abundant lymphatic tissue in early life. The wall structure is similar to the small intestine (though with no villi), nor plicae circularis. Lymph nodules surround the lumen of the gastrointestinal tract and extend from the mucosa into the submucosa.
- VDJ recombination - (variable, diversity and joining gene segments) genetic recombination event that occurs in immune cell maturation in primary lymphoid organs, B cells ((bone marrow) and T cells (thymus).
- Waldeyer’s ring - ring of lymphoid tissue in the pharyngeal wall: palatine tonsils, nasopharyngeal tonsil (adenoid) and lingual tonsil. First described in 1884 by von Waldeyer-Hartz.
- white pulp - (Malpighian follicles, Malpighian bodies of the spleen, white nodules, splenic lymphoid nodules) spleen lymphoid region, organized as lymphoid sheaths with both T-cell and B-cell compartments, around the branching arterial vessels (resembles lymph node structure).
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 25) Embryology Cardiovascular System - Spleen Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Spleen_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G