Cardiovascular System - Heart Valve Development: Difference between revisions
No edit summary |
|||
| Line 17: | Line 17: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Twist1 directly regulates genes that promote cell proliferation and migration in developing heart valves'''<ref><pubmed>22242143</pubmed></ref> "Twist1, a basic helix-loop-helix transcription factor, is expressed in mesenchymal precursor populations during embryogenesis and in metastatic cancer cells. In the developing heart, Twist1 is highly expressed in endocardial cushion (ECC) valve mesenchymal cells and is down regulated during valve differentiation and remodeling. Previous studies demonstrated that Twist1 promotes cell proliferation, migration, and expression of primitive extracellular matrix (ECM) molecules in ECC mesenchymal cells. Furthermore, Twist1 expression is induced in human pediatric and adult diseased heart valves." | |||
* '''Hemodynamic patterning of the avian atrioventricular valve''' <ref><pubmed>21181939</pubmed></ref> "In this study, we develop an innovative approach to rigorously quantify the evolving hemodynamic environment of the atrioventricular (AV) canal of avian embryos. Ultrasound generated velocity profiles were imported into Micro-Computed Tomography generated anatomically precise cardiac geometries between Hamburger-Hamilton (HH) stages 17 and 30. Computational fluid dynamic simulations were then conducted and iterated until results mimicked in vivo observations. Blood flow in tubular hearts (HH17) was laminar with parallel streamlines, but strong vortices developed simultaneous with expansion of the cushions and septal walls. For all investigated stages, highest wall shear stresses (WSS) are localized to AV canal valve-forming regions. Peak WSS increased from 19.34 dynes/cm(2) at HH17 to 287.18 dynes/cm(2) at HH30, but spatiotemporally averaged WSS became 3.62 dynes/cm(2) for HH17 to 9.11 dynes/cm(2) for HH30. Hemodynamic changes often preceded and correlated with morphological changes. These results establish a quantitative baseline supporting future hemodynamic analyses and interpretations." | * '''Hemodynamic patterning of the avian atrioventricular valve''' <ref><pubmed>21181939</pubmed></ref> "In this study, we develop an innovative approach to rigorously quantify the evolving hemodynamic environment of the atrioventricular (AV) canal of avian embryos. Ultrasound generated velocity profiles were imported into Micro-Computed Tomography generated anatomically precise cardiac geometries between Hamburger-Hamilton (HH) stages 17 and 30. Computational fluid dynamic simulations were then conducted and iterated until results mimicked in vivo observations. Blood flow in tubular hearts (HH17) was laminar with parallel streamlines, but strong vortices developed simultaneous with expansion of the cushions and septal walls. For all investigated stages, highest wall shear stresses (WSS) are localized to AV canal valve-forming regions. Peak WSS increased from 19.34 dynes/cm(2) at HH17 to 287.18 dynes/cm(2) at HH30, but spatiotemporally averaged WSS became 3.62 dynes/cm(2) for HH17 to 9.11 dynes/cm(2) for HH30. Hemodynamic changes often preceded and correlated with morphological changes. These results establish a quantitative baseline supporting future hemodynamic analyses and interpretations." | ||
|} | |} | ||
Revision as of 01:07, 8 June 2012
Introduction
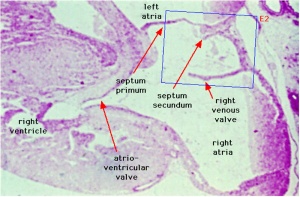
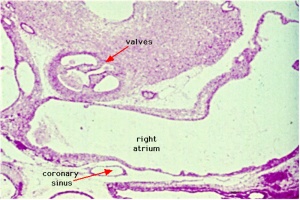
The heart valves form between the atria and ventricles (mitral valve, tricuspid valve) and between the atria and blood vessels (aortic valve, pulmonary valve). The cardiac cushions in the atrioventricular (AV) canal contain cells that are the primordia of the cardiac valves. The atrioventricular valves are attached to papillary muscles by chordae tendineae.
Mitral valve also called the "bicuspid valve".
Some Recent Findings
|
Textbooks
- Human Embryology (2nd ed.) Larson Ch7 p151-188 Heart
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Ch14: p304-349
- Before we Are Born (5th ed.) Moore and Persaud Ch12; p241-254
- Essentials of Human Embryology Larson Ch7 p97-122 Heart
- Human Embryology Fitzgerald and Fitzgerald Ch13-17: p77-111
Molecular
Scleraxis (Scx) - basic helix–loop–helix transcription factor expressed in the progenitors and cells of all tendon tissues (mouse).[3]
Periostin - regulates lineage commitment of valve precursor cells (chicken).[4]
Gata4 and Gata6
Tbx5
Abnormalities
Noonan syndrome
An autosomal dominant single-gene cause of congenital heart disease. Patients also have proportionate short stature, facial abnormalities, and an increased risk of myeloproliferative disease. About half the patients have mutations in PTPN11, encoding the protein tyrosine phosphatase SHP2. A recent study in mice has identified PTPN11 acting in endocardium to enhance endocardial-mesenchymal transformation.[5]
References
Reviews
<pubmed>20809794</pubmed> <pubmed>20201901</pubmed> <pubmed>14567955</pubmed> <pubmed>12768658</pubmed>
Articles
<pubmed>17549728</pubmed> <pubmed>16914500</pubmed>
Search PubMed
Search Pubmed: heart valve development | heart valve morphogenesis | Valvulogenesis
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 24) Embryology Cardiovascular System - Heart Valve Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Heart_Valve_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G