Book - Text-Book of Embryology 10
| Embryology - 18 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
- Contents: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The development of the vascular system
The blood vessels constitute such an extensive and complex system that it is obviously beyond the scope of this book to consider the entire system in detail. Consequently attention must be directed only to the development of the main channels, including the heart, and to the principles of vessel formation.
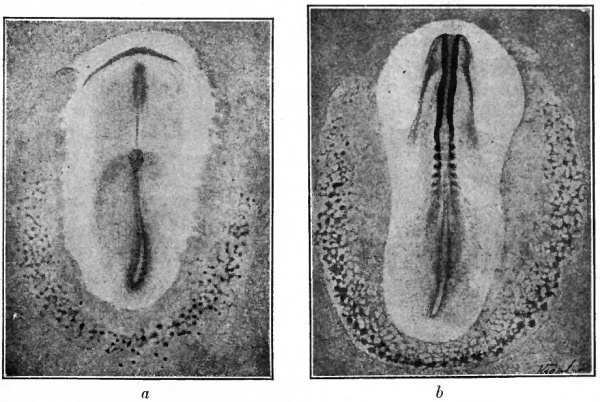
Fig. 156. Surface views of chick blastoderms. Ruckert, Hertwig.
- a, Blastoderm with primitive streak and head process; showing blood islands (dark spots in crescent-shaped area in lower part of figure).
- b, Blastoderm with 6 pairs of primitive segments. Reticulated appearance is due to blood islands (dark spots) and to developing vessels, the entire reticulated area being the area vasculosa.
The formation of blood vessels in all the higher vertebrates including mammals begins in the opaque area of the blastoderm (area opaca) while the germ layers still lie flat. Toward the end of the first day of incubation in the chick, about the time the primitive streak reaches the height of its development, the peripheral part of the area opaca caudal and lateral to the primitive streak presents a mottled appearance (Fig. 1560). This indicates the beginning of the area vasculosa, which subsequently extends forward in the peripheral portion of the opaque area, lateral to the developing body, and becomes reticulated in appearance (Fig. 156^).
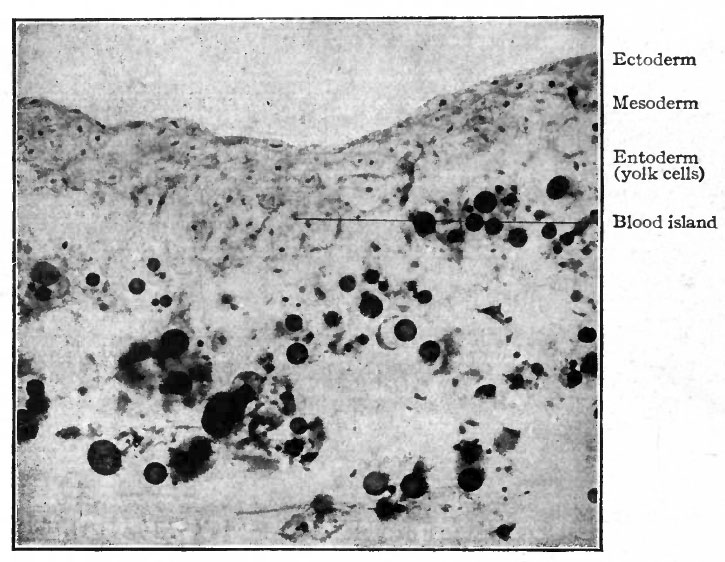
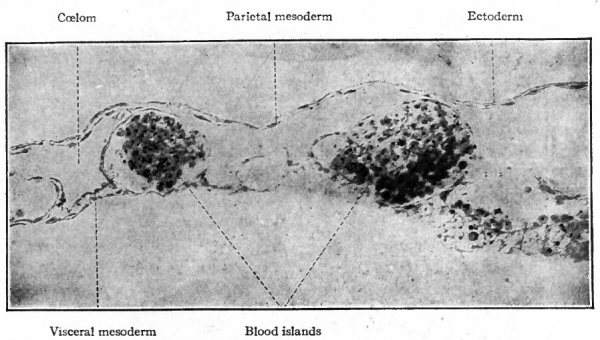
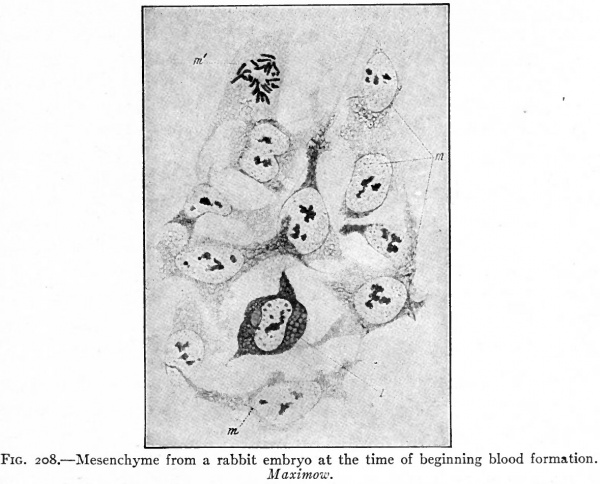
Sections of the blastoderm show that the mottled surface appearance is due to clusters of cells amidst the mesoderm, known as blood islands (Fig. 157). These are composed of rounded cells which have developed from the branched mesodermal (mesenchymal) cells, and are situated in close apposition to the entoderm. Subsequently, when the coelom appears in this region, they lie in the visceral, or splanchnic, layer of mesoderm (Fig. 158).
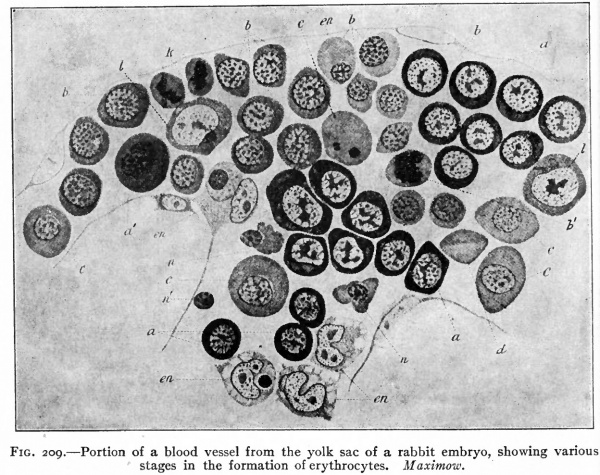
The early changes that occur in the blood islands are important as regards both developing vessels and blood cells. The superficial cells of an island are transformed into flat cells placed edge to edge which surround the remaining rounded cells. The flat cells constitute the endothelium of a primitive blood space, while the cells within the space comprise primitive blood cells (Fig. 158). These early spaces in the area vasculosa join one another and become continuous to form a net-work, or plexus, of channels to which is due the reticulated appearance referred to above (Fig. 1566). This is known as the vitelline plexus. The groups of primitive blood cells within the channels will be considered in detail in a subsequent section (page 236).
Fig. 158. Section of blastoderm of chick of 42 hours' incubation. Photograph. The cells of the blood islands are differentiated into primitive blood cells and the endothelium of the vessels.
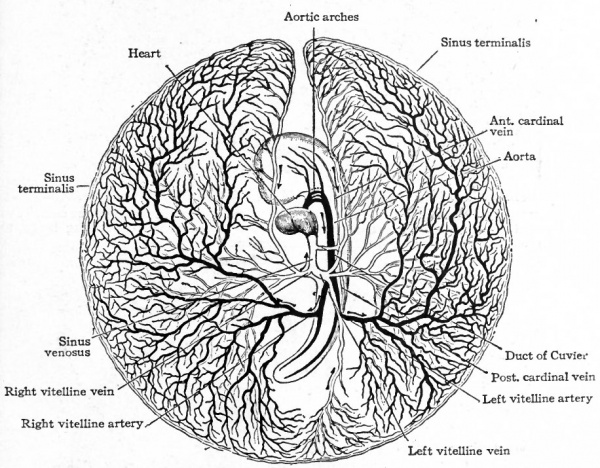
During the second day of incubation in the chick the peripheral channels of the vascular area unite to form a vessel the sinus terminalis which is continuous around the border except at the head end of the embryo (Fig. 159). At the same time the vascularization of the visceral layer of mesoderm gradually extends through the clear area of the blastoderm (area pellucida) toward and finally into the embryonic body. Reaching the region just lateral to the notocord, the vessels unite longitudinally in the embryo to form a continuous channel, the primitive aorta, which thus constitutes a natural selvage to the vascular area on each side of the blastoderm (Fig. 159). Some of the channels of the vitelline plexus increase in size and coalesce to form a large trunk which is a branch of the primitive aorta on each side and leads off into the smaller vessels in the peripheral part of the vascular area. This trunk is known as the vitelline, or omphalomesenteric, artery and is at first located near the caudal end of the embryo. When circulation is established through contractions of the heart it carries blood from the aorta to the surface of the yolk sac (Fig. 159). Other channels of the vitelline plexus nearer the head end of the embryo likewise form a large trunk, the vitelline, or omphalomesenteric, vein which collects the blood from the surface of the yolk sac and conveys it to the heart (Fig. 159).
So long as the germ layers lie flat the two primitive aortae remain separate, but with the ventral flexion and fusion of the germ layers to form the tubular body the aortae fuse into a single medial vessel, the dorsal aorta, except in the cervical region where the two original vessels persist as the dorsal aortic roots. The proximal ends of the vitelline arteries also fuse into a single trunk, the two vitelline veins, however, remaining separate. In each branchial arch on each side a vessel develops which joins with the corresponding dorsal aortic root. These vessels the aortic arches arise from single vessel on each side ventral to the pharynx which is known as the ventral aortic root. The two ventral aortic roots arise from a single medial vessel, the aortic trunk, or truncus arteriosus, which in turn is a continuatioi of the early tubular heart.
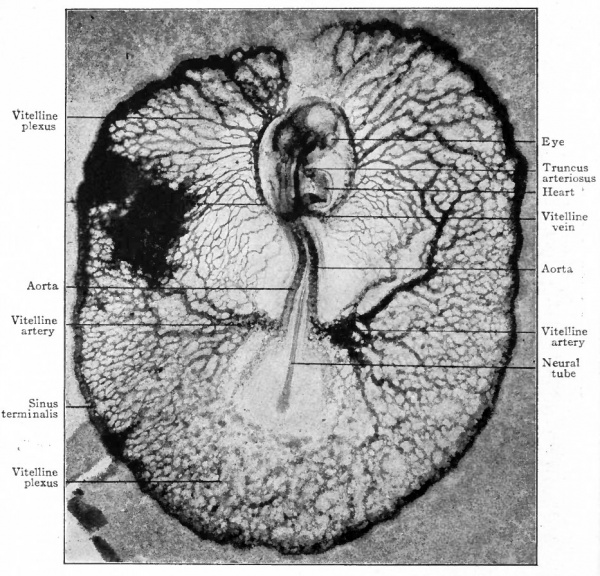
Fig. 159. Dorsal surface view of chick embryo with 18 segments, including the area vasculosa. Photograph, X 15. The blood vessels were injected with India ink, the dark blotch in the upper left corner indicating some ink which escaped during the injection.
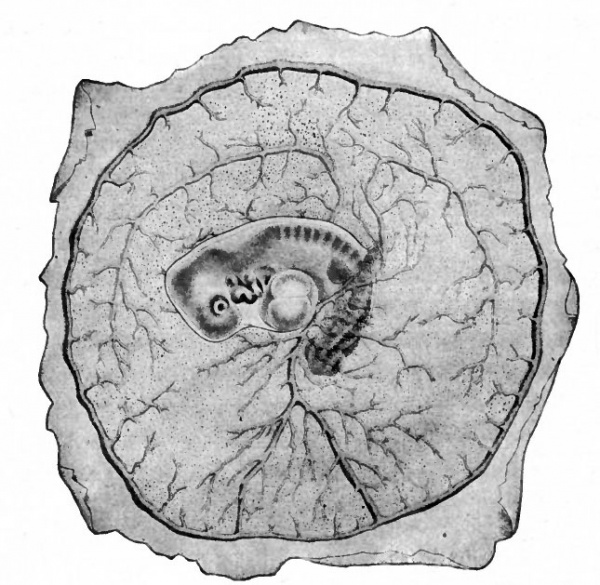
The heart, having developed and become a contractile organ in the meantime, receives the blood in its caudal end through the vitelline veins and ejects it from its cephalic end through the aortic trunk. The blood then passes through the aortic arches to the dorsal aorta whence it is distributed to the vitelline plexus by the vitelline arteries. The blood is collected by tributaries of the vitelline veins and carried to the heart. Thus the vitelline (yolk) circulation is completed (Fig. 160). From this time on, the area vasculosa gradually enlarges, as the germ layers extend farther and farther around the yolk, until it eventually surrounds the whole yolk mass. In mammals, as in the chick, the vascular rudiments develop first in the extraembryonic portion of the mesoderm as clusters of cells which give the area opaca a mottled appearance on surface view. This soon changes to a reticulated appearance as the cell clusters give rise to primitive blood spaces which join one another to form a plexus of channels. This plexus gradually extends across the area pellucida toward the embryo and terminates in a natural selvage as the primitive aorta on each side of the median line. The vitelline arteries and veins are formed out of the plexus and, with the heart, aortic arches and dorsal aorta as in the chick, constitute the vitelline circulatory system (Fig. 161). The vascular area in some mammals gradually enlarges until it embraces the "entire yolk sac (Fig. 162).
Fig. 160. Diagram of the vitelline (yolk) circulation of a chick embryo at the end of the third day of incubation. Ventral view. Balfour.
Fig. 161. Surface view of area vasculosa of a rabbit embryo of 11 days. van Beneden and Julin. The vessel around the border is the sinus terminalis; the two large vessels above the embryo are the vitelline (omphalomesenteric) veins ; the two large vessels converging below the embryo are the vitelline (omphalomesenteric) arteries.
Fig. 162. Human embryo of 3.2 mm. His. The arrows indicate the direction of the blood current.
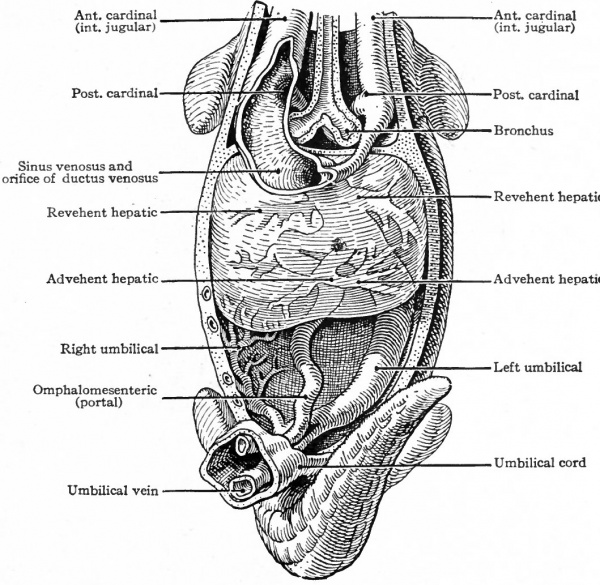
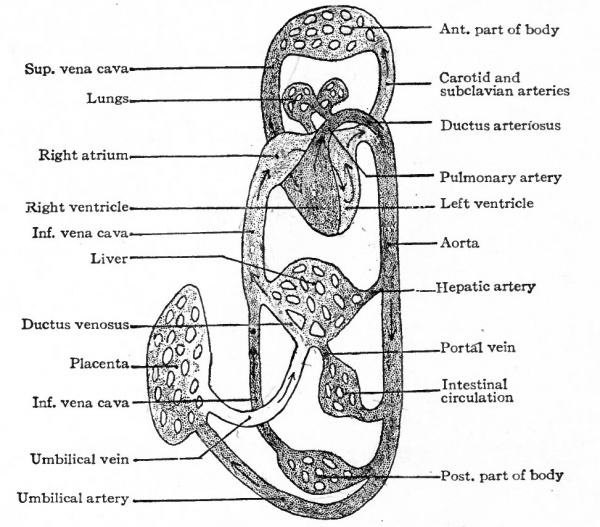
It is seen from the foregoing account that the earliest circulation is associated with the yolk sac. In animals below the mammals, where a large amount of yolk is present in the sac, the vitelline circulation is of prime importance in supplying the growing embryo with nutritive materials. In mammals the vitelline circulatory system develops as extensively as in the lower forms but, since little yolk is present, does not assume the same important role of carrying food supply; yet the portions of the vessels inside the embryo, viz. : the heart, aortic arches, aorta, the proximal part of the vitelline artery, and the vitelline veins, form parts of the permanent vascular system. In reptiles and birds a second set olyessels develops in connection with the allantois and serves to carry away the waste products of the body and deposit them in that sac-like structure. Two arteries, one on each side, arise as branches of the dorsal aorta near its caudal end and pass out of the body along with the allantoic duct to ramify upon the surface of the allantois. These are the umbilical, or allantoic, arteries. The blood is collected and carried back by the umbilical veins which pass along the 'allantoic duct to the body and then forward, one on each side, through the somatic layer of mesoderm to join the ducts of Cuvier. The duct of Cuvier, formed on each side by the junction of the anterior and posterior cardinal veins, which will be considered in a subsequent section, pour their blood into the sinus venosus. This venous trunk is formed by the junction of the ducts of Cuvier with -the vitelline veins and empties directly into the heart.
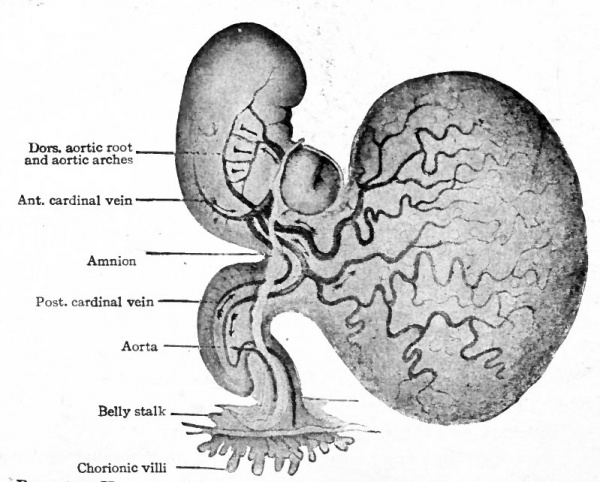
Fig. 163. Diagram of the umbilical vessels in the belly stalk and chorion. Kollmann's Atlas.
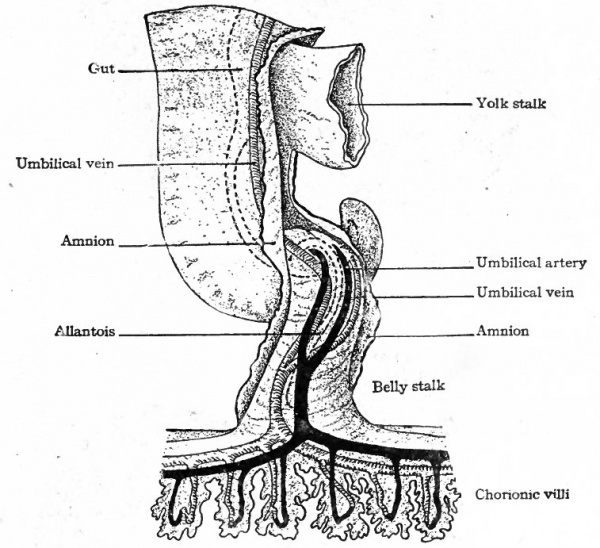
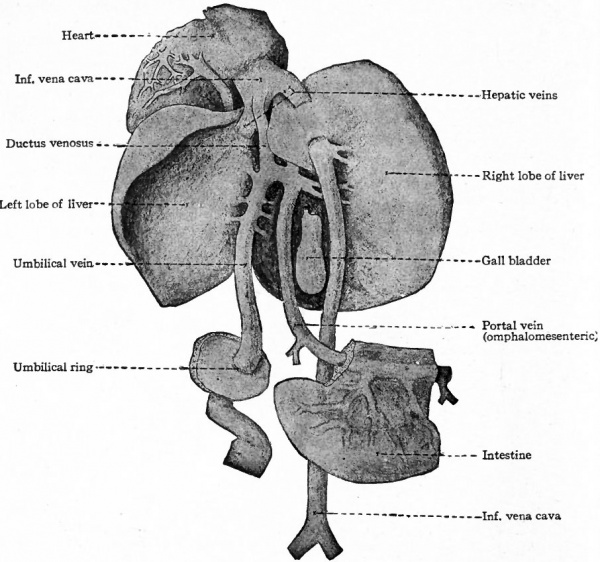
In mammals in general the allantois is a rudimentary structure incapable of receiving the total waste of the embryo. The umbilical (allantoic) vessels develop, however, as in reptiles and birds but become associatec through the belly stalk with the placenta which establishes communication between the embryo and the mother (Fig. 163). The vessels within the embryo are at first disposed in the same manner as in the lower forms, the umbilical arteries arising from the caudal portion of the aorta and the umbilical veins passing forward in the ventro-lateral body wall to join the ducts of Cuvier. With the formation of the umbilical cord the two umbilical veins within this structure fuse into a single vessel (Fig. 164). The later changes in the umbilical veins are most conveniently considered subsequently. In mammals in general the umbilical (allantoic) circulatory system performs a two-fold function. The blood carries to the placenta the waste products of the embryo for deposition in the maternal circulation, the waste in the lower forms (reptiles and birds) being deposited in the allantois. The blood carries from the placenta the food materials derived from the maternal circulation, the food in the lower forms being taken from the yolk sac and conveyed to the embryo by the vitelline vessels.
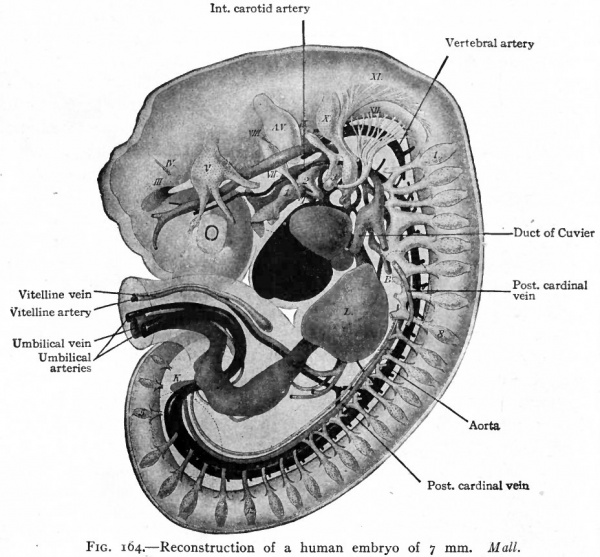
Fig. 164. Reconstruction of a human embryo of 7 mm. Mall.
- Arteries represented in black. A.V., Auditory vesicle; B, bronchus; L, liver; K, anlage o kidney; T, thyreoid gland; III-XII, cranial nerve roots; 1, 2, 3, 4, branchial grooves; 1, 8, 12, 5 (on spinal nerve roots), 1st and 8th cervical, 12th dorsal, 5th lumbar spinal nerve respectively. Dotted outlines represent limb buds.
Principles of Vasculogenesis
Upon the thesis that tissues in general must receive materials which they build up into their own substances and must discharge the products of their activities, the vascular channels of the body can be considered as structural expressions of this functional necessity. For instance, a muscle which acts must receive materials to compensate it for its loss and must discharge the waste products that result from its action, and the blood vessels are peculiarly adapted to these functions. The lymph vessels, too, similar in structure to the blood vessels, although efferent relative to the tissues, play their part in conveying the products of metabolism.
Much controversy has arisen over the actual genesis, or origin, of blood vessels and lymphatics, and as yet the opposing views have not been reconciled. In brief there are two views: One that with a few exceptions every vessel in the body develops as a sprout from another vessel, that is, the endothelium arises from preexisting endothelium by proliferation of its own cells; the other that vessels in general arise in situ, that is, the lumen of a vessel represents an intercellular tissue space^or several such spaces, whose bordering cells have been transformed into the characteristic endothelial cells, and as a corollary, the continuity of a given vessel results from the union of such spaces. According to the latter view, the whole vascular system represents intercellular tissue spaces which, with their lining of flattened cells, have united to form a set of continuous channels.
In the case of either view it is recognized that the first vessels appear in the opaque area of the blastoderm. Here the blood islands originate as clusters of cells amidst the mesoderm, differentiating from mesenchymal elements in close approximation to the entoderm (Fig. 157). The superficial cells of the clusters are then transformed into flat cells placed edge to edge to form the endothelial wall of a primitive blood space. These blood spaces join one another and thus form a net-work of channels. From this point in development the two views diverge.
The evidence adduced in favor of either theory is too great in volume to set down here. The advocates of the theory of sprouting of the endothelium lay stress upon the evidence of injected specimens. By injecting developing blood vessels at successive stages it is found that the vascular field gradually becomes larger, and the inference is that the individual channels are extending farther and farther from the focus of origin through proliferation and migration of the endothelial elements. This method, of course, would demonstrate vessels only so far as the lumina are continuous. Solid cords of cells which extend beyond the field of injection are interpreted as cords of endothelial cells which subsequently acquire lumina and become capillary tubes. If this theory is correct then the vascularization of the area pellucida and of the embryonic body would be effected through true outgrowths of the original endothelium of the opaque area. Possible exceptions to this, as noted above, are the rudiments of the heart, the aorta and the cardinal veins which arise in situ as do the first vascular rudiments. Observations upon growing vessels in living embryos, in which strands of cells were seen to extend from the endothelium already present, have also been accepted as evidence in favor of this view.
The evidence afforded by injected specimens has been attacked by those who believe in the in situ origin of vessels, on the ground that the injection shows only vessels with continuous lumina and does not prove the nonexistence of isolated vascular rudiments beyond the field of injection. It is claimed that the vascular field becomes more extensive through the gradual addition of such isolated spaces to the channels already continuous, in the same manner that the primitive blood spaces unite to form a network, and the claim is supported by demonstration of these spaces in the mesenchymal tissue with every gradation between the bordering flattened cells (endothelium) and the branching irregular mesenchymal cells. The actual formation of intercellular spaces with flat bordering cells and their union with vascular channels have been observed in the living chick blastoderm. Experimental evidence has also been brought to bear in favor of the view that vessels arise in situ. The area opaca was entirely removed from the chick blastoderm before any vascular rudiments had appeared in the area pellucida and the blastoderm was then allowed to develop further; it was found that vascular rudiments appeared both in the area pellucida and embryonic body with practically the same disposition as in the normal embryo.
The concept that the vascular channels are structural expressions of the functional necessity of carrying nutritive materials to the tissues and waste products away from them leads to consideration of such factors as may be involved in the formation of vessels; that is, factors that would cause plastic cells, like those of the mesenchyme in which the earliest and simplest vessels appear, to change in character and rearrange themselves to form capillary tubes. In a mass of mesenchymal tissue, in which there is a resemblance to a sponge with the cellular elements representing the parenchyma of the sponge and the intercellular tissue spaces the interstices, the products of cell activity naturally accumulate in the intercellular spaces. Incident to this accumulation, pressure would be exerted upon the cells bordering the spaces. Seeking outlet from the confines of the spaces, the waste products would move, or flow, and cause friction against the cells past which they flow. Similarly, pressure and friction would result from the movement of nutritive materials to and through the tissue. The plastic mesenchymal cells, reacting to these mechanical influences, would tend to become flat, and the continued operatic of the factors would result in a smooth- walled tube in which the movement of fluid is greatly facilitated.
The reaction of the irregular mesenchymal cells to the mechanical influences of pressure and friction is, of course, the crux of the question. It has been shown experimentally that cells of this type do react to mechanical stimuli. Smooth non-irritating foreign bodies have been imbedded in the loose connective tissue of an animal and the cells in contact therewith became flat and formed a mosaic apparently identical with simple squamous epithelium or endothelium. In the growth of mesenchymal tissue outside of the body (in vitro) it has been observed that the cells flatten against foreign substances which may be present.
In the embryo it has been observed that where blood vessels disappear, which they do in certain regions, the endothelium does not degenerate but that the cells assume irregular branching forms. This would indicate that endothelium comprises merely modified mesenchymal cells and that upon removal of the factors incident to the pressure and friction of blood flow the cells reassume the indifferent character of mesenchyme, thus reverting to the mesenchymal type. It militates, therefore, against the view that endothelium is a specific tissue.
It is generally recognized, whether or not the endothelium originates in situ, that a capillary network precedes the formation of larger vessels. For instance, the vitelline plexus of capillaries (p. 186) antedates any of the larger vitelline vessels which later carry blood to and from the embryo. The establishment of vascular trunks in this plexus of small vessels seems to be dependent upon the same mechanical factors that were considered as operative in the origin of vessels; viz.: pressure and friction. If the volume of blood that flows through a given capillary network at a given rate is increased the flow will naturally follow the channels that offer the least resistance, and these channels will increase in size sufficiently to accommodate the greater volume. A few channels, or perhaps even only one, will form the most direct course, and the angles in the course will be still further reduced as the blood stream impinges upon the walls of the vessels. In this manner a large vessel, or main vascular trunk, is established and the remaining smaller vessels constitute its branches or tributaries. A rather crude analogy would be the draining of a swamp in which a small rivulet, once gaming slight supremacy over its fellows, would gradually cut its way deeper into the soil and pursue a straighter course, with the result that the other rivulets would flow into it as the main channel.
The concept that the main vascular trunks are preceded by a capillary plexus, out of which they develop in response to certain mechanical stimuli, offers a simple explanation of the numerous variations found in the vascular system. In the incipient stages of the larger vessels but slight influences, due to variations in the development of surrounding structures, would be sufficient to deflect their courses and cause them to occupy positions which do not accord with the normal. So far as the thickened walls of the larger vascular channels are concerned, they may be regarded as structural adaptations to the functions they perform. For example, the large amount of elastic tissue in the wall of the aorta and other large arteries tends to maintain a uniform diameter in these vessels against the force exerted by the blood expelled from the heart at each contraction.
The Heart
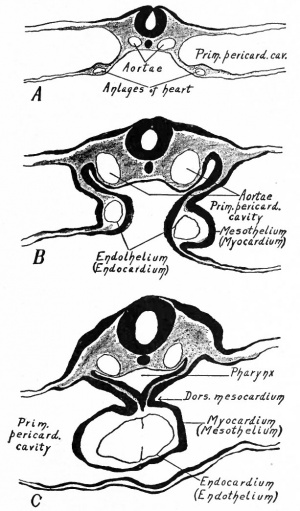
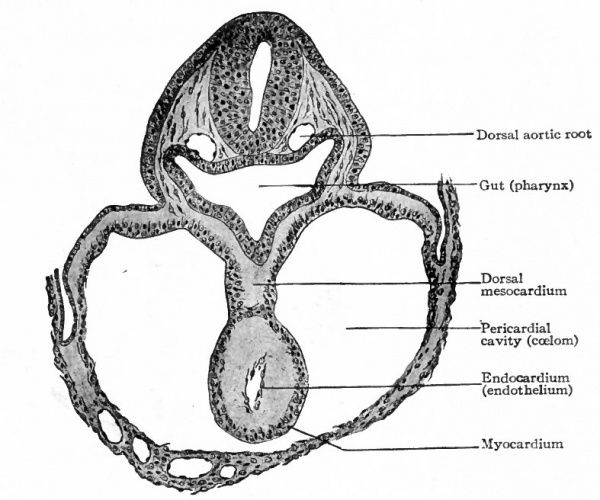
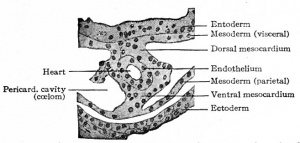
The heart has a peculiar origin in that it arises as two separate parts or anlagen which unite secondarily. In the chick, for example, it appears during the first day of incubation, at a time when the germ layers are still flat. The ccelom in the cephalic region becomes dilated to form the so-called primitive pericardial cavity (parietal cavity), and at the same time a space appears on each side, not far from the medial line, in the mesodermal layer of the splanchnopleure (Fig. 165). These spaces at first are filled with a gelatinous substance in which lie a few isolated cells. These cells then take on the appearance of endothelium and line the cavities, and the mesothelium in this vicinity is changed into a distinct, thickened layer of cells. Now by a bending ventrally of the splanchnopleure the cavities or vessels are carried toward the mid ventral line (Fig. 165). The bending continues until the entoderm of each side meets and fuses with that of the opposite side, thus closing in a flat cavity the fore-gut. The entoderm ventral to the cavity breaks away and allows the medial walls of the two endothelial tubes to come in contact. These walls then break away and the tubes are united in the midventral line to form a single tube (Fig. 165), which extends longitudinally for some distance in the cervical region of the embryo. The mesothelial layers of opposite sides meet dorsal and ventral to the endothelial tube, forming the dorsal and ventral mesocardium (Fig. 165). In the meantime the cephalic end of the tube has united with the arterial system, and the caudal end with the venous system ; and in a short time the dorsal and ventral mesocardia disappear and leave the heart suspended by its two ends in the primitive pericardial cavity. The conditions at this point may be summarized thus: The heart is a double-walled tube the inner wall composed of endothelium and destined to become the endocardium, the outer wall of a thicker mesothelial layer and destined to become the myocardium the two walls separated by a considerable space. The organ hangs, as it were, in the primitive pericardial cavity (ccelom), connected at its cephalic end with the ventral aortic trunk and at its caudal end with the omphalomesenteric veins.
In all mammals thus far studied the principle of development in the earlier stages is essentially the same as in the chick. The double origin of the heart is even more marked because of the relatively late closure, of the fore-gut. There are no observations on the origin of the heart in human embryos, but it is reasonable to assume that it has the same double origin as in other Mammals, although in embryos of 2 to 3 mm. the organ has already become a single tube (Figs. 166 and 167). At this stage the tube is somewhat coiled.
Fig. 166. Transverse section of a human embryo of 2.69 mm. von Spee, Kollmann's Atlas.
Fig. 167. Ventral view of reconstruction of human embryo of 2.15 mm. His. The ventral body wall has been removed. The vessels (in black) at the sides of the duct of the liver are the omphalomesenteric veins.
While the double origin of the heart is characteristic of all amniotic Vertebrates (Reptiles, Birds, Mammals), in all the lower forms the organ arises as a single anlage. In the region of the fore-gut the two halves of the ccelom are separated by a ventral mesentery which extends from the gut to the ventral body wall, and which is composed of two layers of mesothelium with a small amount of mesenchyme between them. In the mesenchyme a cavity appears and is lined by a single layer of flat (endothelial) cells. This cavity extends longitudinally for some distance in the cervical region and with its endothelial and mesothelial walls constitutes the simple cylindrical heart. On the dorsal side it is connected with the gut by a portion of the mesentery which is called the dorsal mesocardium; on the ventral side it is connected with the ventral body wall by the ventral mesocardium (Fig. 168). Thus the heart is primarily a single structure. The difference between the two types of development is not a fundamental one but simply depends upon the difference in the germ layers. In the lower forms the germ layers are closed in ventrally from the beginning, and the heart appears in a medial position. In the higher forms the germ layers for a time remain spread out upon the surface of the yolk or yolk sac, and the heart begins to develop before they close in on the ventral side of the embryo. Consequently the heart arises in two parts which are carried ventrally by the germ layers and unite secondarily.
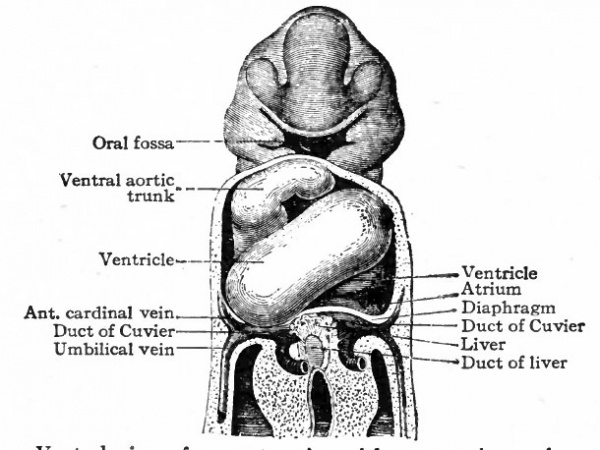
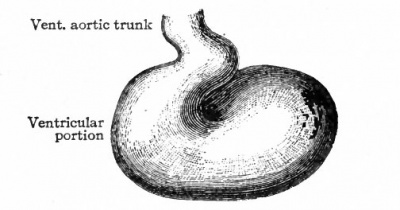
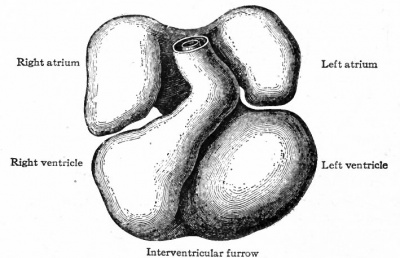
The further development of the heart consists of various changes in the shape of the tube and in the structure of its walls. At the same time the dilatation of the ccelom (primitive pericardial cavity) in the cervical region is of importance in affording room for the heart to grow. In the chick, for example, the tube begins, toward the end of the first day of incubation, to bend to the right; during the second day it continues to bend and assumes an irregular S-shape. This bending process has not been observed in human embryos, but other Mammals show the same process as the chick. In a human embryo of 2.15 mm. the S-shaped heart is present (Fig. 167). The venous end, into which the omphalomesenteric veins open, is situated somewhat to the left, extends cranially a snort distance and then passes over into the ventricular portion. The latter turns ventrally and extends obliquely across to the right side, then bends dorsally and cranially to join the aortic bjulb which in turn joins the ventral aortic trunk in the medial line. The endothelial tube, which is still separated from the muscular wall by a considerable space, becomes somewhat constricted at its junction with the aortic bulb to form the so-called f return Halleri. During these changes the heart as a whole increases in diameter, especially the ventricular portion. Gradually the venous end of the heart moves cranially and in embryos of 4.2 mm. lies in the same transverse plane as the ventricular portion. The latter lies transversely across the body (Fig. 169). At the same time two e vagina tions appear on the venous end, which represent the anlagen of the atria. In embryos of about 5 mm. further changes have occurred, which are represented in Fig. 170. The two atrial anlagen are larger than in the preceding stage and surround, to a certain extent, the proximal end of the aortic trunk. As they enlarge still more in later stages, they come in contact, their medial walls almost entirely disappear, and they form a single chamber. The ventricular portion of the heart becomes separated into a right and a left part by the interventricular furrow (Fig. 1 70) ; the right part is the anlage of the right ventricle, the left part, of the left ventricle. At the same time the atrial portion has moved still farther cranially so that it lies to the cranial side of the ventricular portion. The venous and arterial ends of the heart have thus reversed their original relative positions. At this point it should be noted that the atrial end of the heart is connected with the large venous trunk formed by the union of the omphalomesenteric veins and the ducts of Cuvier the sinus venosus.
Fig. 169. Ventral view heart of human embryo of 4.2 mm.
|
Fig. 170. Ventral view of heart of human embryo of 5 mm. His. |
During the changes in the heart as a whole, certain changes also occur in the endothelial and muscular walls. The walls of the atria are composed of compact plates of muscle with the endothelium closely investing the inner surface. The walls of the ventricular portion, on the other hand, become thicker and are composed of an outer compact layer of muscle and an inner layer made up of trabeculae which are closely invested by the endothelium.
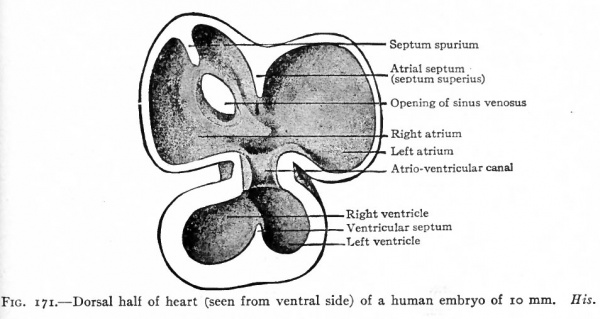
Fig. 171. Dorsal half of heart (seen from ventral side) of a human embryo of 10 mm. His.
Everywhere the endothelium is closely applied to the inner surface of the myocardium, the space which originally existed between the endothelium and mesothelium being obliterated.
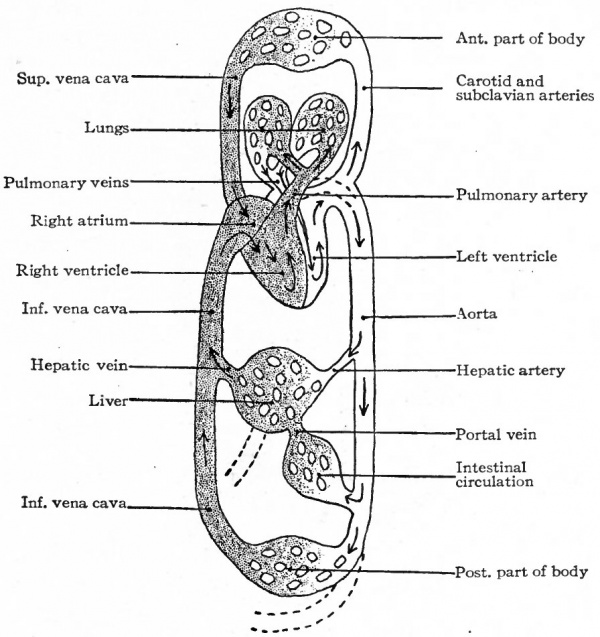
The embryonic heart in Mammals in the earlier stages resembles that of the adult in the lower Vertebrates (Fishes). The atrial portion receives the blood from the body veins and conveys it to the ventricular portion which in turn sends it out through the arteries to the body. The circulation is a single one. This condition changes during the foetal life of Mammals with the development of the lungs. The same transition occurs in the ascending scale of development in the vertebrate series in those forms in which gill breathing is replaced by lung breathing. The change consists of a division of the heart and circulation, so that the single circulation becomes a double circulation. In other words, the heart, is so divided that the lung (pulmonary) circulation is separated from the general circulation of the body. This division first appears in the Dipnoi (Lung Fishes) and Amphibians in which gill breathing stops and lung breathing begins, although here the division is not complete. In Reptiles the division is complete except for a small direct communication between the ventricles.
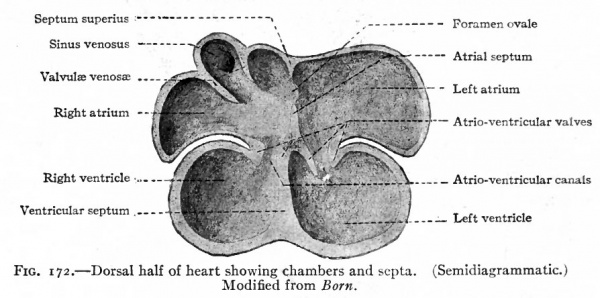
Fig. 171 represents the dorsal half of the heart at a stage when all the chambers are in open communication, and shows the conditions in a single circulation but with the beginning of a separation. The atria are rather thin-walled chambers, the ventricles have relatively thick walls. Between the atrial and ventricular portion is a canal the atrio-ventricular canal which affords a free passage for the blood. From the cephalic side of the atrial portion a ridge projects into the cavity. This ridge represents a remnant of the original medial walls of the two atria and marks the beginning of the future atrial septum. The opening of the sinus venosus is seen on the dorsal wall of the right atrium. Primarily both atria communicated directly with the sinus venosus,but in the course of development the opening of the latter migrated to the right and at this stage is found in the wall of the right atrium. The opening is guarded, as it were, by a lateral and a medial fold the significance of which will be described later. The vetricular portion also shows a ridge projecting from the caudal side, which corresponds to the interventricular groove and represents the beginning of the ventricular septum.
Fig. 172. Dorsal half of heart showing chambers and septa. (Semidiagrammatic.) Modified from Born.
The Septa
The further changes are largely concerned with the separation of the heart into right and left sides, and with the development of the valves. The atria become separated by the further growth on the cephalic side, of the ridge which has already been mentioned and which is known as the septum superius (Figs. 171 and 172). This septum grows across the cavity of the atria until it almost reaches the atrio-ventricular canal, forming the septum atriorum. A portion of the septum then breaks away, leaving the two atria still in communication. This secondary opening is the foramen ovale which persists throughout foetal life, but closes soon after birth. The atrio-ventricular canal also becomes divided into two passages by a ridge from the dorsal wall and one from the ventral wall uniting with each other and finally with the septum atriorum (Fig. 172). Thus the two atria would be completely separated if it were not for the foramen ovale.
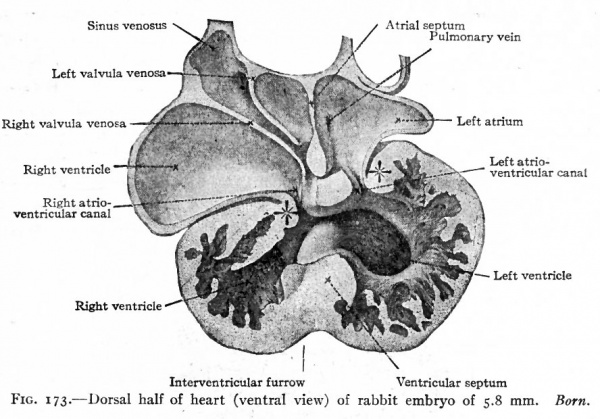
Fig. 173. Dorsal half of heart (ventral view) of rabbit embryo of 5.8 mm. Born.
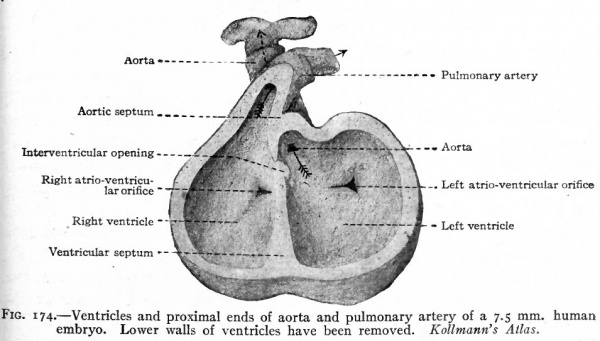
Fig. 174. Ventricles and proximal ends of aorta and pulmonary artery of a 7.5 mm human embryo. Lower walls of ventricles have been removed. Kollmann's Atlas.
During the separation of the atria, a division of the ventricular portion of the heart also occurs. On the caudal side of the ventricular portion a septum appears and gradually grows across the cavity forming the septum ventriculorum (Figs. 171 and 172). This septum is situated nearer the right side and is indicated on the outer surface by a groove which becomes the sulcus longitudinalis anterior and posterior. The dorsal edge of this septum finally fuses with the septum dividing the atrio-ventricular canal, but for a time its ventral edge remains free, leaving an opening between the two ventricles (Figs. 173 and 174).
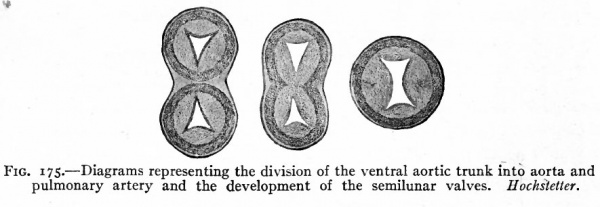
This opening then becomes closed in connection with the division of the aortic bulb and ventral aortic trunk. On the inner surface of the aortic trunk, at a point where the branches which form the pulmonary arteries arise, two ridges appear, grow across the lumen and fuse with each other, thus dividing the vessel into two channels. This partition the septum aorticum (Fig. 175) gradually grows toward the heart through the aortic bulb and finally unites with the ventral edge of the ventricular septum, thus closing the opening between the two ventricles. Corresponding with the edges of the septum aorticum, a groove appears on each side of the aortic trunk and gradually grows deeper and extends toward the heart, until finally the trunk and aortic bulb are split longitudinally into two distinct vessels, one of which is connected with the right ventricle and becomes the pulmonary artery, the other with the left ventricle and becomes the proximal part of the aortic arch (Fig. 174). The result of the formation of these various septa is the division of the entire heart into two sides. The atrium and ventricle of each side are in communication through the atrio- ventricular foramen, the two sides are in communication only by the foramen ovale which is but a temporary opening.
Fig. 175. Diagrams representing the division of the ventral aortic trunk into aorta and pulmonary artery and the development of the semilunar valves. Hochsietter.
After the opening of the sinus venosus is shifted to the right atrium, the left atrium for a short period has no vessels opening into it. As soon, however, as the pulmonary veins develop, they form a permanent union with the left atrium (Fig. 173). At first two veins arise from each lung, which unite to form a single vessel on each side; the two single vessels then unite to form a common trunk which opens into the left atrium on the cephalic side. As development proceeds, the wall of the single trunk is gradually absorbed in the wall of the atrium, until the single vessel from each side opens separately. Absorption continuing, all four veins, two from each lung finally open separately. This is the condition usually found in the adult. A partial failure in the absorption may leave one, two, or three vessels opening into the atrium. Such variations are not infrequently met with in the pulmonary veins.
The Valves
If all the passageways between the different chambers of the heart and the large vascular trunks were to remain free and clear, there would be nothing to prevent the blood from flowing contrary to its proper course. Consequently five sets of valves develop in relation to these orifices, and are so arranged that they direct the blood in a certain definite direction. These appear (a) at the openings of the large venous trunks into the right atrium, (b) at the opening between the right atrium and right ventricle, (c) at the opening between the left atrium and left ventricle, (d) at the opening between right ventricle and pulmonary artery and (e) at the opening between the left ventricle and aorta. No valves develop at the openings of the pulmonary veins into the left atrium.
(a) The sinus venosus (which is formed by the union of the large body veins) opens into the right atrium on its cranial side, as has already been mentioned (p. 201). By a process of absorption, similar to that in the case of the pulmonary veins, the wall of the sinus is taken up into the wall of the atrium. The result is that the vena cava superior, vena cava inferior, and sinus coronarius (a remnant of the left duct of Cuvier) open separately into the atrium. As the sinus is absorbed, its wall forms two ridges on the inner surface of the atrium, one situated at the right of the opening and one at the left (Figs. 172 and 173). These two ridges valvulce venosce are united at their cranial ends with the septum spurium (Fig. 171), a ridge projecting from the cephalic wall of the atrium. The septum spurium probably has a tendency to draw the two valves together and prevent the blood from flowing back into the veins. The left valve and the septum spurium later atrophy to a certain extent and probably unite with the septum atriorum to form part of the limbus fossce ovalis (Vieussenii) . The right valve is the larger and in addition to its assistance in preventing a backward flow of blood into the veins, it also serves to direct the flow toward the foramen o\;ale. As the veins come to open separately, the cephalic part of the right valve disappears; the greater part of the remainder becomes the valvula Deuce cavce inferioris (Eustachii) and during fcetal life directs the blood toward the foramen ovale. In the adult it becomes a structure of variable size. A small part of the remainder of the right valve forms the valvula sinus coronarii (Thebesii) which guards the opening of the coronary sinus.
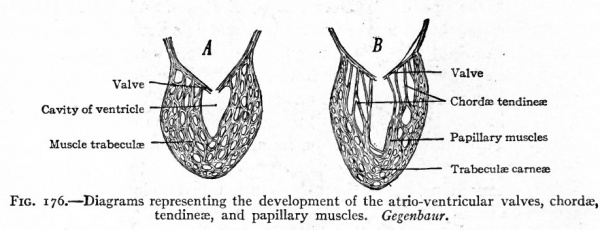
(b) and (c) The valves between the atrium and ventricle on each side develop for the most part from the walls of the triangular atrio-ventricular opening (ostium atrio-ventriculare) . Elevations or folds appear on the rims of the openings and project into the cavities of the ventricles where they become attached to the muscle trabeculae of the ventricle walls (Figs. 176 and 177). On the right side three of these folds appear, and develop into the valvula tricuspidalis which guards the right atrio-ventricular orifice. On the left side only two folds appear, and these become the valvula biscuspidalis (mitralis) which guards the left atrio-ventricular orifice. These valves, which are at first muscular, soon change into dense connective tissue. The muscle trabeculae to which they are attached also undergo marked changes. Some become condensed at the ends which are attached to the valves into slender tendinous cords the chorda tendinece, while at their opposite ends they remain muscular as the Mm. papillares; others remain muscular and lie in transverse planes in the ventricles, or fuse with the more compact part of the muscular wall, or form irregular, anastomosing bands and constitute the Irabecula carnea (Fig. 176).
Fig. 176. Diagrams representing the development of the atrio-ventricular valves, chordae, tendineas, and papillary muscles. Gegenbaur.
(d) and (e) The valves of the pulmonary artery and aorta develop at the point where originally the endothelial tube was constricted to form the f return Halleri (p. 200) where the ventricular portion of the heart joined the aortic bulb. Before the aortic trunk and bulb are divided into the aortic arch and pulmonary artery, four protuberances appear in the lumen (Fig. 213). The septum aorticum then divides the two which are opposite so that each vessel receives three (Fig. 175). These then become concave on the side away from the heart, in a manner which has not been fully determined, and at the same time enlarge so that they close the lumen. Those in the pulmonary artery are known as the valvula semilunares arteria pulmonalis, those in the aorta as the valvula semilunares aorta.
Changes after Birth
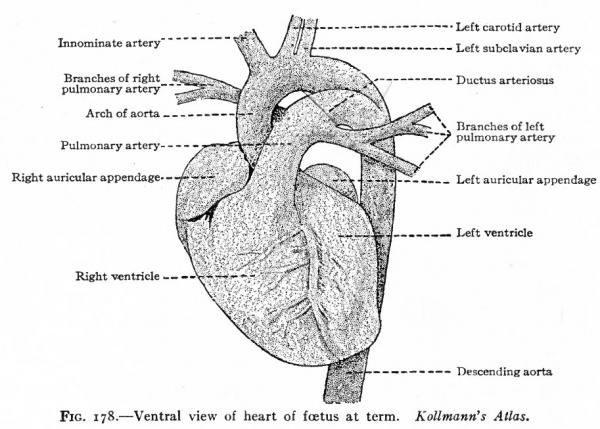
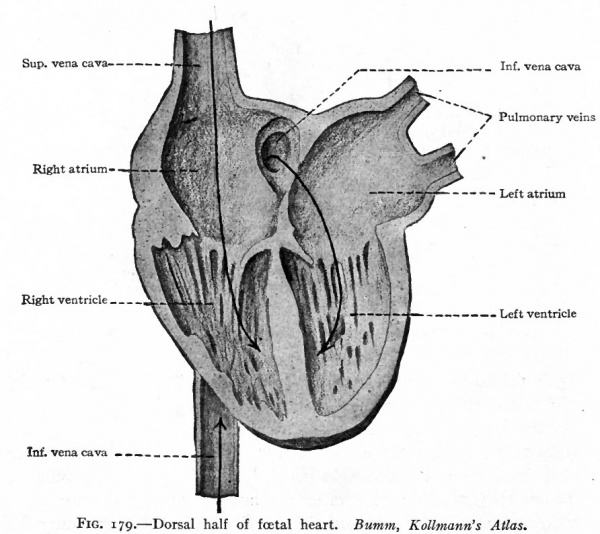
The migratory changes of the heart from its original position in the cervical region to its final position in the thorax will be considered in connection with the development of the pericardium (Chap. XIV). With the exception of the septum atriorum, the heart acquires during fcetal life practically the form and structure characteristic of the adult (Fig. 178). So long as the individual continues to grow, the heart, generally speaking, increases in size accordingly. This increase takes place by intussusception in the endocardium and myocardium. At the time of birth the two atria are in communication through the foramen ovale which is simply an orifice in the atrial septum (Fig. 179). Thus the blood which is brought to the right atrium by the body veins is allowed to pass directly into the left atrium, thence to the left ventricle, and thence is forced out to the body again through the aorta. A certain amount of blood also passes from the right atrium into the right ventricle and thence into the pulmonary artery; but this blood does not enter the lungs but passes directly into the aorta through the ductus arteriosus (Fig. 178). After birth the lungs begin to function and the placental blood is cut off, so that the right atrium receives venous blood only and the left arterial blood only. If the foramen ovale were to persist it would allow a mingling of venous and arterial blood. Consequently the foramen ovale closes soon after birth and the two currents of blood are completely separated. At the same time the ductus arteriosus atrophies and becomes the ligamentum arteriosum. Consequently there is no direct communication between the pulmonary artery and aorta.
Fig. 177. Transverse section of pig embryo of 14 mm. Photograph.
Fig. 178. Ventral view of heart of foetus at term. Kollmann's Atlas.
Fig. 179. Dorsal half of foetal heart. Bumm, Kollmann's Atlas.
Certain features of development have an important bearing on the theories regarding the physiology of the heart, particularly on the theory that the heart is an automatic organ. Whether the theory that the heart beats automatically, i.e., independently of stimuli from the nervous system, is true or not, it is a fact that in the embryo it begins to beat before any nerve cells appear in it and before any nerve fibers are connected with it. At least no technic has yet been devised by which it is possible to demonstrate nerve cells in, or fibers connected with it, at the time when it begins to perform its characteristic function. And, furthermore, at the time when the heart begins to beat, no heart muscle cells are developed. This last fact seems to indicate an inherent contractility in the mesothelial cells which form the anlage of the myocardium.
The Arteries
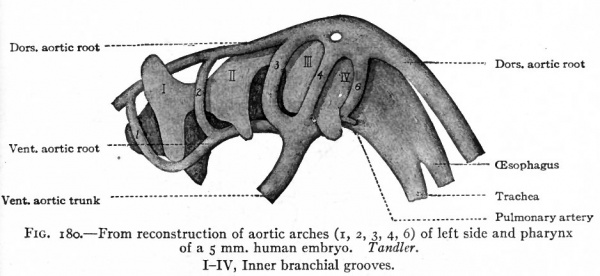
The simplest condition of the arterial system, following the establishment of the vitelline and allantoic circulation (p. 189 and p. 191), is as follows: The single ventral aortic trunk is given off from the cephalic end of the heart. This is a short vessel, soon dividing into the two vejntral aortic roots which pass forward beneath the pharynx (Fig. 180). Each ventral aortic root gives rise to branches which pass dorsally, one in each branchial arch, as the aortic arches to unite in a common stem along the dorsal wall of the pharynx. This common stem is the dorsal aortic root (Fig. 1 80) which fuses with its fellow of the opposite side in the middorsal line to form the dorsal aorta. The single dorsal aorta, situated ventral to the notochord, extends from the cervical region to the caudal end of the embryo. Somewhat caudal to the middle of the embryo a branch of the aorta passes ventrally through the mesentery as the vitelline artery which enters the umbilical cord (Fig. 164). Still farther caudally the paired umbilical (allantoic) arteries are given off from the aorta and pass out into the umbilical cord (Fig. 164).
Fig. 180. From reconstruction of aortic arches (i, 2, 3, 4, 6) of left side and pharynx of a 5 mm human embryo. Tandler. I-IV, Inner branchial grooves.
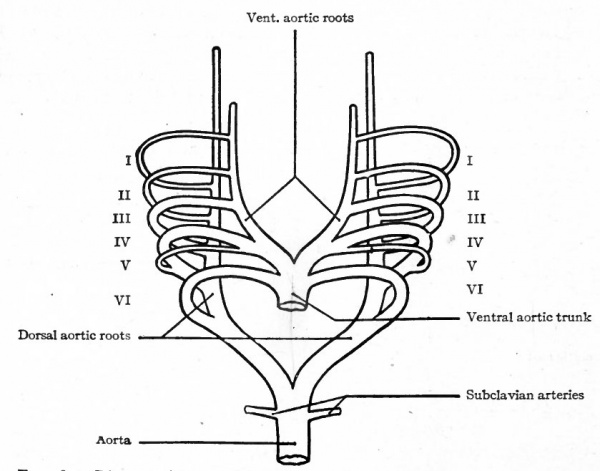
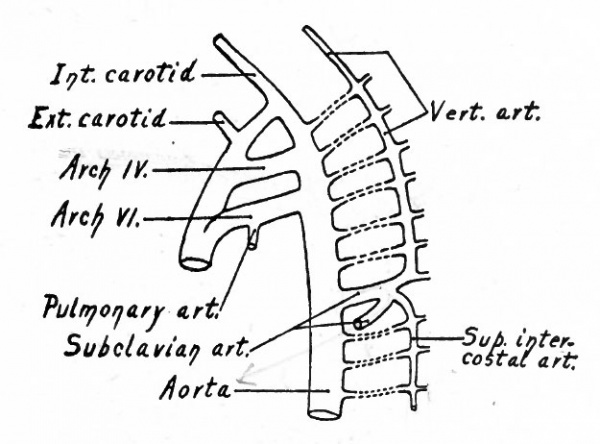
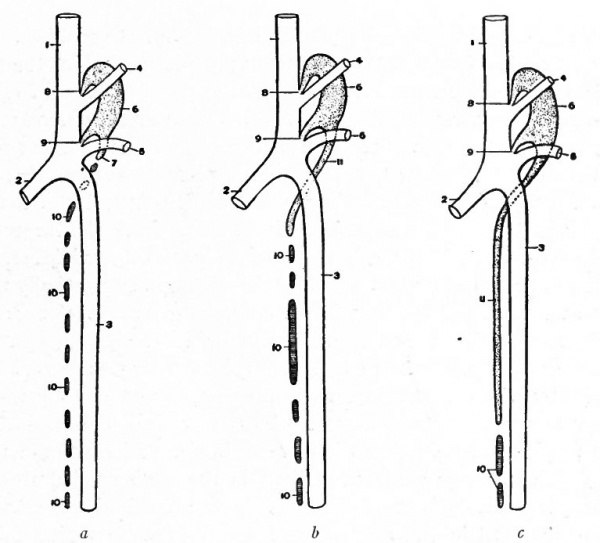
The conditions which exist at this stage in the region of the aortic arches in mammalian embryos are indicative of the conditions which persist as a whole or in part throughout life in the lowest Vertebrates. The changes which occur in Mammals, however, are profound and the adult condition bears no resemblance to the embryonic. Yet certain features in the adult are intelligible only from a knowledge of their development. In the human embryo ,ix aortic arches appear on each side. The first, second, third, and fourth pass through the corresponding branchial arches. The fifth arch, which is merely a loop from the fourth, seems to pass through the fourth branchial arch. The sixth aortic arch passes through the region behind the fourth branchial. All these arches are present in embryos of 5 mm. (Fig. 180). In Fishes and larval Amphibians, where the branchial arches develop into the gills, the aortic arches are broken up into capillary networks which ramify in the gills, and the ventral aortic root becomes the afferent vessel, the dorsal aortic roots the efferent vessels. In the higher Vertebrates and in man the aortic arches begin, at a very early period, to undergo changes; some disappear and others become portions of the large arterial trunks which leave the heart. In connection with the following description, constant reference to Figs. 181 and 182 will assist the student in understanding <he changes.
Fig. 181. Diagram of the aortic arches of a Mammal. Modified from Hochstetter.
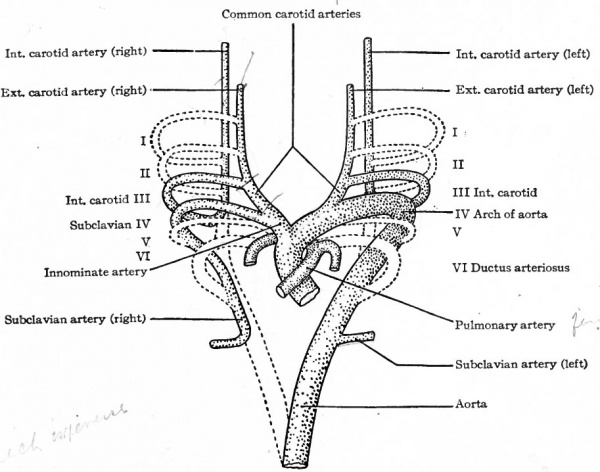
The first and second arches soon atrophy and disappear. The third arch on each side becomes the proximal part of the internal carotid artery, while the continuation of the dorsal aortic root, cranially to the third arch, becomes its more distal part. The continuation of the ventral aortic root cranially to the third, arch, becomes L the proximal artery, while the portion of the ventral aortic root between the third and fourth arches becomes the common carotid artery. The portion of the dorsal aortic root between the third and fourth arches disappears. The fourth aortic arch on the left side enlarges and becomes the arch of the aorta (arcus aorta) which is then continued caudally through the left dorsal aortic root into the dorsal aorta. On the right side, the fourth arch becomes the proximal part of the subclavian artery. Since the third, foutth, fifth, and 'sixth arches really leave the ventral aortic trunk as a single vessel, it will be seen that these changes bring it about that the common carotid and subclavian on the right side arise by a common stem, the innominate artery, which in turn is a branch of the arch of the aorta. On the left side, for the same reason, the common carotid is a branch of the arch of the aorta. The fifth aortic arch from the beginning is rudimentary and disappears very early. The sixth arch on each side undergoes wide changes. A branch from each enters the corresponding lung. On the right side the portion of the sixth arch between the branch which enters the lung and the dorsal aortic root disappears, as does also that portion of the right dorsal aortic root between the subclavian artery and the original bifurcation of the dorsal aorta. On the left side, however, that portion of the sixth arch between the branch which enters the lung and the dorsal aortic root persists until birth as the ductus arteriosus (Botalli). This conveys the, blood from the right ventricle to the aorta until the lungs become functional (Fig. 178); it then atrophies and becomes the ligamentum arteriosum. In the meantime the septum aorticum has divided the original ventral aortic trunk into two vessels (see p. 204); one of the vessels communicates with the left ventricle and is the proximal part of the arch of the aorta, the other communicates with the right ventricle and becomes the large pulmonary artery (fig. 174).
Fig. 182. Diagram representing the changes in the aortic arches of a Mammal. Compare with Fig. 181. Modified from Hochstetter.
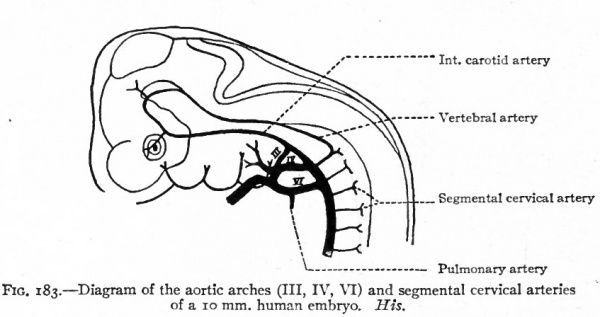
Fig. 183. Diagram of the aortic arches (III, IV, VI) and segmental cervical arteries of a 10 mm human embryo. His.
In human embryos of 10 mm. the dorsal aortic root on each side gives off several lateral branches the segmental cervical vessels (Fig. 183). The first of these (first cervical, suboccipital), which arises nearly opposite the fourth aortic arch, is a companion, as it were, to the hypoglossal nerve, and J sends a branch cranially which unites with its fellow of the opposite side inside the skull to form the basilar artery. The basilar artery again bifurcates and each branch unites with the corresponding internal carotid by means of the circulus arteriosus (Fig. 185). The other segmental cervical vessels arise from the aortic root at intervals, the eighth arising near the point of bifurcation of the aorta. In a short time a longitudinal anastomosis appears between these segmental arteries, which extends as far as the seventh (Fig. 184). The proximal ends of the first six disappear, and the longitudinal vessel forms the vertebral artery which then opens into the aortic root through the seventh segmental artery, and which is continued cranially as the basilar artery (Fig. 185). The seventh (it is held by some to be the sixth) segmental artery becomes the subclavian, and consequently the vertebral opens into the subclavian, as in the adult (Fig. 184). But it should be borne in mind that the right subclavian artery is more than equivalent to the left, since the proximal part of the former is made up of the fourth aortic arch and a part of the aortic root (see Figs. 181 and 182). Furthermore, changes occur in the position of the heart during development, which alter the relations of the vessels. The heart migrates from its original position in the cervical region into the thorax, and this produces an elongation of the carotid arteries and an apparent shortening of the arch of the aorta; consequently the subclavian artery on the left side arises relatively nearer the heart.
Fig. 184. Diagram illustrating the formation of the vertebral and superior intercostal arteries. The broken lines represent the portions of the original segmental vessels that disappear. Modified from Hochstetter.
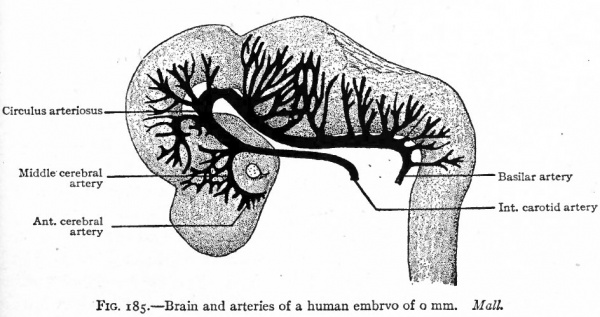
Fig. 185. Brain and arteries of a human embryo of 6 mm(?). Mall.
The arteries of the brain arise as branches of the internal carotid and circulus arteriosus. The anterior cerebral artery and the middle cerebral artery arise primarily from a common stem which in turn is a branch of the most cranial part of the internal carotid (Figs. 185 and 186). The posterior cerebral artery arises as a branch of the circulus arteriosus (Fig. 185).
Fig. 186. Brain, arteries and veins of a human embryo of 33 mm. Mall
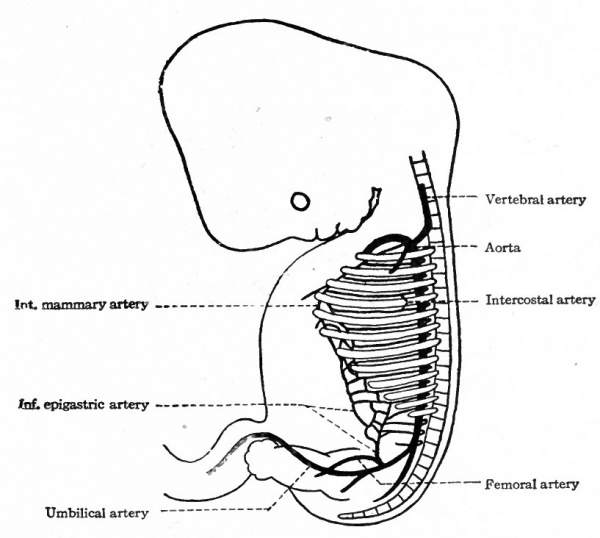
From the point of its bifurcation to its caudal end the aorta gives off paired, segmental branches which accompany the segmental nerves. The last (eighth) cervical branch and the first two thoracic branches undergo longitudinal anastomoses, similar to those between the first seven cervical, to form the superior intercostal artery (A. intercostalis suprema) which opens into the subclavian (Fig. 184). The other thoracic branches persist as the intercostal arteries; the lumbar branches persist as the lumbar arteries. At the same time anastomoses are formed between the distal ends of the intercostal and lumbar arteries in the ventro-lateral region of the body wall, which give rise, on the one hand, to the internal mammary artery and, on the other hand, to the inferior epigastric artery. Of these two the former opens into the subclavian, the latter into the external iliac. By a further anastomosis the distal ends of the internal mammary and inferior epigastric are joined, thus forming a continuous vessel from the subclavian to the external iliac (Fig. 187). It is interesting to note that while originally all the lateral branches of the aorta are arranged segmentally, many of them lose their segmental character and are replaced or supplemented by longitudinal vessels.
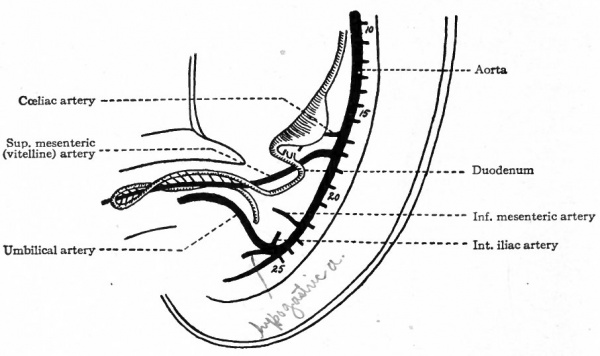
In addition to the dorsal segmental branches of the aorta, which have been described, other branches develop which carry blood to the viscera. A number of these, or possibly all, are also primarily segmental vessels, although they lose every trace of their segmental character during development. The first of the visceral branches to appear is the omphalomesenteric artery which arises from the ventral side of the aorta and which has been mentioned in connection with the vitelline circulation. Originally it passes out through the mesentery and follows the yolk stalk to ramify on the surface of the yolk sac. But since the yolk sac is of slight importance, the distal part of the artery soon disappears, while the proximal part becomes the superior mesenteric artery (Fig. 188). The cceliac artery arises from the ventral side of the aorta a short distance cranially to the omphalomesenteric (Fig. 1 88) and gives rise in turn to the gastric, hepatic and splenic arteries. The inferior mesenteric artery also arises from the ventral side of the aorta some distance caudal to the omphalomesenteric (Fig. 188). In the early stages these visceral arteries arise relatively much farther cranially than in the adult. During development they gradually migrate caudally to their normal positions.
Fig. 187. Diagram of human embryo of 13 mm. Showing the mode of development of the internal mammary and inferior epigastric arteries. Mall.
Other branches of the aorta develop in connection with the urinary and genital organs. Several lateral branches supply the mesonephroi, but when the latter atrophy and disappear the vessels also disappear. A periaortic plexus of vessels, with many branches from the aorta, supplies the developing kidneys until these organs reach their definitive position, when one of the branches on each side enlarges to become the renal artery. The developing genital glands are likewise supplied by several branches from the aorta. Later the majority of these vessels disappear, one pair only persisting as the internal spermatic arteries which differ in accordance with the sex of the individual. In both sexes they are at first very short; in the female, as the ovaries move farther into the pelvic region, they become considerably elongated to form the ovarian arteries; in the male, with the descent of the testes, they become very much elongated to form the testicular arteries.
Fig. 188. Diagram of the visceral arteries in a human embryo of 12.5 mm. Numerals indicate segmental arteries. Tandler.
The fifth (or fourth?) pair of segmental lumbar arteries primarily gives rise to the vessels which supply the lower extremities, viz., the iliac arteries. These then would be serially homologous to the subclavians. But certain changes occur in this region, which are due to the relations of the umbilical arteries. The latter, as has already been noted, arise as paired branches of the aorta in the lumbar region, pass ventrally through the genital cord (Chap. XV) and then follow the allantois (urachus) to the umbilical cord.
During foetal life they carry all the blood that passes to the placenta. At an early period a branch from each iliac artery anastomoses with the corresponding umbilical, and the portion of the umbilical artery between the aorta and the anastomosis then disappears. This makes the umbilical artery a branch of the iliac; and the blood then passes from the aorta into the proximal part of the liiac which becomes the common iliac artery of the adult. At birth, when the umbilical cord is cut, the umbilical arteries no longer carry blood to the placenta, and their intraembryonic portions, often called the hypogastric arteries, persist only in part; their proximal ends persist as the superior vesical arteries, while the portions which accompanied the urachus degenerate to form the lateral umbilical ligaments.
So far as a complete history of the growth of the arteries of the extremities is concerned, knowledge is lacking. The facts of comparative anatomy and the anomalies which occur in the human body have led to certain conclusions which have been largely confirmed by embryological observations; but much more work on the development of the arteries is yet necessary to complete their history. The extremities represent outgrowths from several segments oft the body, the nerve supply is derived from several segments, and the limb buds are likewise primarily supplied by plexuses of vessels arising from several branches of the aorta. In the upper extremity the subclavian, which represents the seventh cervical branch of the aortic root, is the single vessel which eventually develops out of the original plexus. In the lower extremity the common iliac, which represents the fifth lumbar branch of the aorta, is the single vessel which develops out of the plexus supplying the lower limb bud.
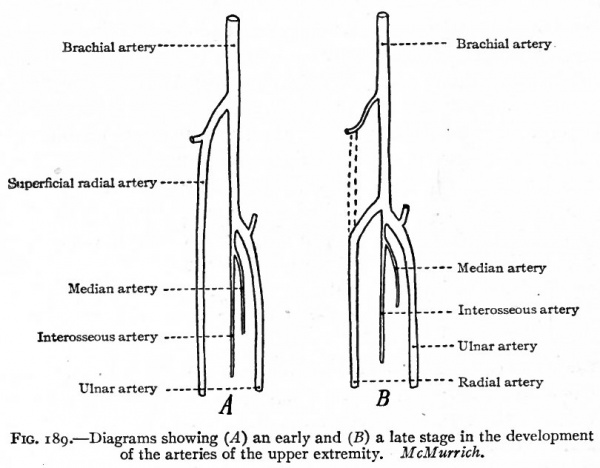
In the upper extremity the subclavian grows as a single vessel to the wrist and then divides into branches corresponding to the fingers. In the forearm it lies between the radius and ulna. In a short time a branch is given off just distal to the elbow and accompanies the median nerve. As this branch increases, the original vessel in the forearm diminishes to form the volar interosseous artery; and at the same time the branch unites again with the lower end of the interosseous, takes up the digital branches and becomes the chief vessel of the forearm at this stage, forming the median artery. Later, however, it diminishes in size as another vessel develops, the ulnar artery, which arises a short distance proximal to the origin of the median and, passing along the ulnar side of the forearm, unites with the median to form the superficial volar arch. From the artery of the arm, which is called the brachial artery, a branch develops about the middle and extends distally along the radial side of the forearm. A little later another branch grows out from the brachial just proximally to the origin of the ulnar and extends across to, and anastomoses with, the first branch. Then the portion of the first branch between its point of origin and the anastomosis atrophies, leaving only a small vessel which goes to the biceps muscle. The second branch and the remaining part of the first branch together form the radial artery (Fig. 189) (McMurrich).
Fig. 189. Diagrams showing (A) an early and (B) a late stage in the development of the arteries of the upper extremity. McMurrich.
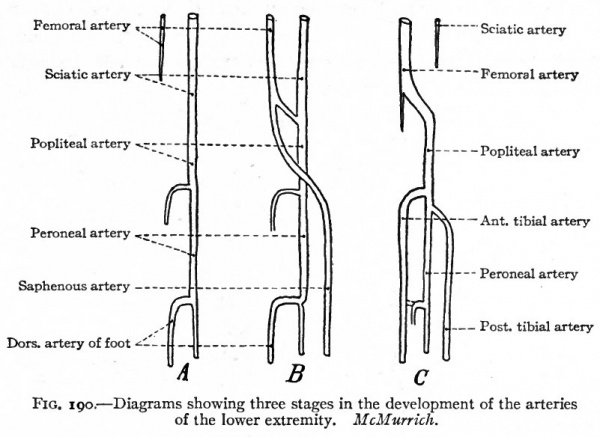
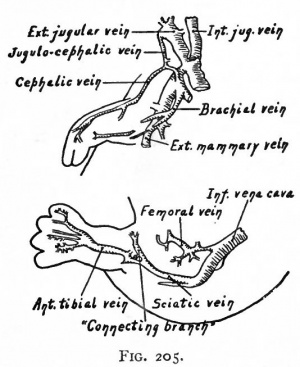
In the lower extremity the primary artery is a continuation of the common iliac which, in turn, is a branch of the aorta. This primary vessel, the sciatic artery, passes distally as far as the ankle. Below the knee it gives off a short branch which corresponds to the proximal part of the anterior tibial artery. Just above the ankle it gives off another branch which corresponds to the distal part of the anterior tibial. As will be seen, these two parts join at a later period to form a continuous vessel. At this early stage the external iliac artery is but a small branch of the common iliac; but it gradually increases in size, extends farther distally in the thigh as the femoral artery and unites with the sciatic near the knee. Just proximal to its union with the sciatic it gives off a branch which extends distally along the inner side of the leg to the plantar surface of the foot, where it gives off the digital branches. This vessel is the saphenous artery in the embryo, and disappears in part during further development. From this time on, the femoral and its direct continuation, the popliteal, increase in size; and at the same time the sciatic loses its primary connection and becomes much reduced to form the inferior gluteal artery. The direct continuation of the sciatic in the leg, which is now the direct continuation of the popliteal, becomes reduced to form the peroneal artery. The branch of the original sciatic, which was given off just below the knee, unites with the branch which was given off just above the ankle to form a continuous vessel, the anterior tibial artery. A new branch arises from the proximal portion of the peroneal, extends down the back of the leg, and unites with the distal part of the embryonic saphenous to form the posterior tibial artery. The proximal part of the saphenous then atrophies, leaving but one of the small genu branches of the popliteal (Fig. 190) (McMurrich).
Fig. 190. Diagrams showing three stages in the development of the arteries of the lower extremity. McMurrich.
The Veins
The changes which occur during the development of the venous system are so complicated, and in some cases so varied, that the scope of this book permits only a brief outline of the growth of the more important of the venous trunks.
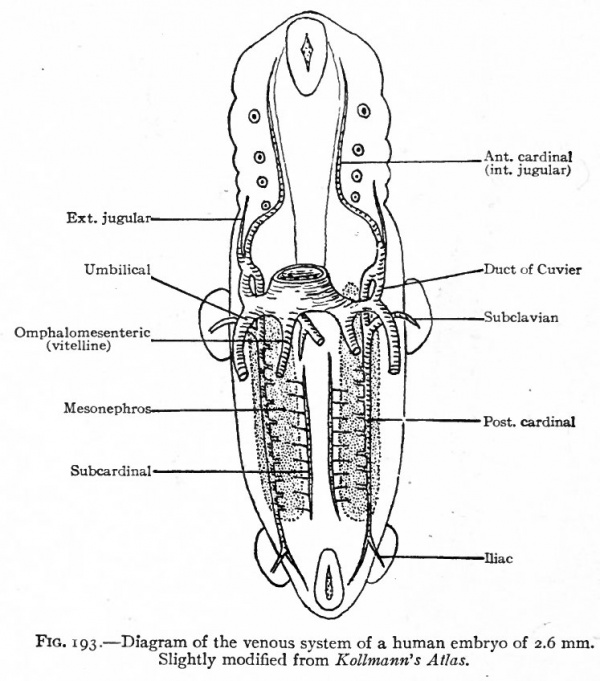
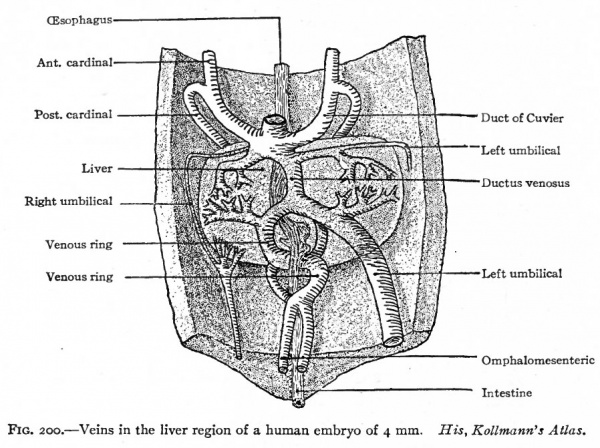
Corresponding to the arterial system, the first veins to appear are the omphalomesenteric veins. These vessels, which carry blood from the yolk sac to the heart, arise in the area vasculosa, enter the embryonic body at the sides of the yolk stalk, pass cranially along the intestinal tract, and join the caudal end of the heart (Figs. 160, 162, 164 and 193). Next in point of time to appear are the umbilical veins which carry back to the heart the blood which has been carried to the placenta by the umbilical arteries. These also are paired veins within the embryo, although they form a single trunk in the umbilical cord. They extend cranially on each side through the ventrolateral part of the body wall and join the duct of Cuvier (see below) in the septum transversum (Figs. 163, 164 and 193). Very soon after the appearance of the umbilical veins two other longitudinal vessels develop, one on each side of the aorta. In the cervical region they lie dorsal to the branchia arches and are called the anterior cardinal veins (Figs. 162 and 193). The more caudal parts of the vessels are situated in the region of the developing mesonephros and are called the posterior cardinal veins (Figs. 162 and 193). At a point about opposite the heart the anterior and posterior cardinals on each side unite to form a single vessel, the duct ofCuvier, which turns medially through the septum transversum and opens into the sinus venosus (Figs. 162 and 178). Thus three primary sets of veins are formed at a very early stage of development: (i) The omphalomesenteric veins; (2) the umbilical veins; (3) the cardinal veins.
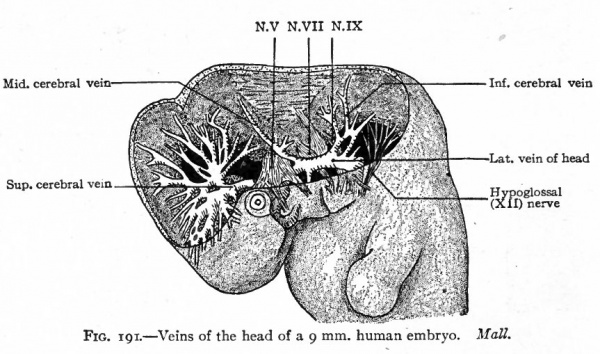
Fig. 191. Veins of the head of a 9 mm. human embryo. Mall.
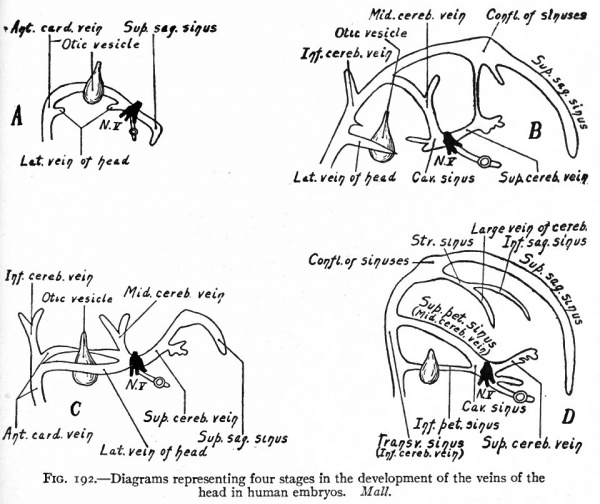
The veins of the head and neck regions are derivatives of the anterior cardinals. The proximal parts of these vessels are present in embryos of 3.2 mm.; later they extend cranially along the ventro-lateral surface of the brain on the medial side of the roots of the cranial nerves. The position relative to the nerves is only temporary, however, for collaterals arising from the veins pass to the lateral side of the nerves and enlarge to form the main channels. The original channels atrophy except in the region of the trigeminal nerves where they still remain on the medial side of the nerves as the forerunners of the cavernous sinuses. The vessel thus formed laterally to the cranial nerves (except the trigeminal) on each side of the brain is known as the lateral vein of the head (vena lateralis capitis) (Fig. 191.) The blood is collected from the brain region by small vessels which unite to form three main stems; one of these, the superior cerebral vein, opens into the cranial end of the cavernous sinus; another, the middle cerebral vein, opens into the opposite end of the cavernous sinus; and the third, the inferior cerebral vein^ opens into the lateral vein of the head behind the ear vesicle (Figs. 191 and 186). The branches of the superior cerebral vein extend over the cerebral hemispheres and unite with their fellows of the opposite side to form the superior sagittal sinus which lies in the medial line (Figs. 186 and 192). The superior sagittal sinus is at first naturally drained by the superior cerebral veins; but later, as the cerebral hemispheres enlarge and extend farther toward the mid-brain region, it is carried back and joins the middle cerebral vein; still later, for the same reason, it joins the inferior cerebral vein (Fig. 192, A and B). During these later changes the connection between the superior sagittal sinus and the superior cerebral vein is lost (Fig. 192). The middle cerebral vein becomes the superior petrosal sinus which forms a communication between the cavernous sinus and transverse sinus. The transverse sinus represents the channel between the superior sagittal sinus and the cranial end of the cardinal vein; or in other words, its cranial portion represents the connection between the superior sagittal sinus and the inferior cerebral vein while its caudal portion represents the inferior cerebral vein itself (Fig. 192, compare C and D). The caudal end of the superior sagittal sinus becomes dilated to form the confluence of the sinuses (confluens sinuum). From the latter a new vessel grows out to form the straight sinus, and a further growth from the straight sinus forms the large vein of the cerebrum (vein of Galen). The inferior sagittal sinus also represents a new outgrowth at the point of junction of the large vein of the cerebrum and inferior sagittal sinus (Fig. 192, D). During the course of development the lateral vein of the head gradually atrophies and finally disappears, and the inferior petrosal sinus probably represents a new formation which extends from the cavernous sinus to the transverse sinus (Fig. 192, C and D). At the point where the inferior petrosal joins the transverse sinus the latter passes out of the skull through the jugular foramen to become the internal jugular vein (anterior cardinal). (Mall.)
Fig. 192. Diagrams representing four stages in the development of the veins of the head in human embryos. Mall.
Fig. 193. Diagram of the venous system of a human embryo of 2.6 mm. Slightly modified from Kollmann's Atlas.
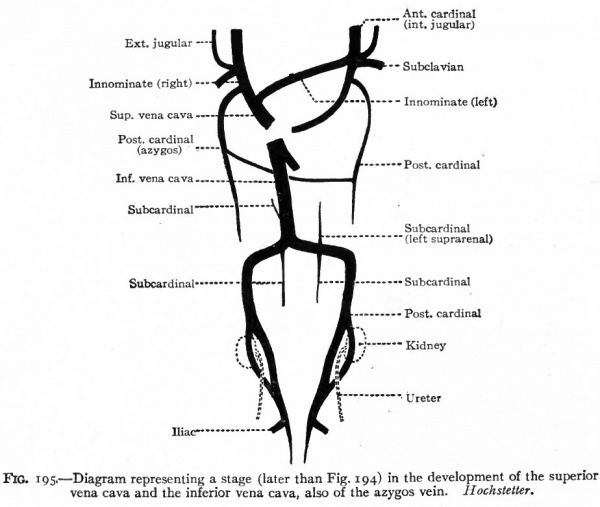
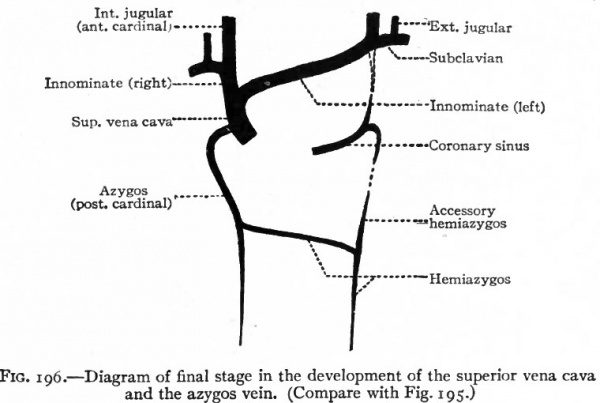
As stated in a preceding paragraph, the anterior cardinal veins extend from the ducts of Cuvier to the head region, passing to the dorsal side of the branchial arches. They are at first paired and symmetrical, but, since the heart is situated in the cervical region, are comparatively short and receive blood from the cervical region through segmental branches which belong only to the most cranial of the cervical segments. The other segmented cervical I veins, including the subdavian veins, open at first into the posterior cardinals (Fig. 193). Later, however, as the heart recedes into the thorax the anterior cardinal veins are elongated and the segmental cervical veins, including the subclavians, come to open into them (Fig. 195). The bilateral symmetry is then broken by an anastomosing vessel which extends obliquely across from a point on the left cardinal about opposite the subclavian to a point nearer the heart on the right subclavian (Figs. 194, B, and 195). The portion of the left cardinal cranial to the subclavian becomes the left internal jugular vein which communciates with the intracranial sinuses. The anastomosis itself becomes the left innominate vein. The portion of the left cardinal between the subclavian and the duct of Cuvier, the duct of Cuvier itself, and the left horn of the sinus venosus together form the coronary sinus (Fig. 196). On the right side the more distal part of the cardinal becomes the internal jugular vein; the portion between the subclavian and the anastomosis (left innominate vein) becomes the right innominate vein ; and the common stem formed by the latter and the left innominate constitutes the superior vena cava which opens into the right atrium (see p. 205) . The external jugular vein on each side appears later than the superior cardinal as an independent vessel which comes to lie parallel to the internal jugular and opens into it near the subclavian. The opening, however, shifts to the subclavian, where it is usually found in the adult (Figs. 195 and 196).
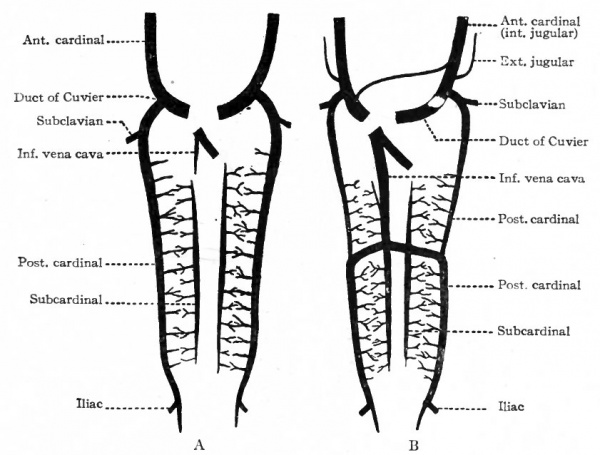
Fig. 194. Diagrams of two stages in the development of the anterior and posterior cardinal veins, the subcardinal veins (revehent veins of the primitive kidney), and the inferior vena cava. The small branches of the cardinals and subcardinals ramify in the primitive kidneys (mesonephroi). Slightly modified from Hochstetter.
The changes which occur in the posterior cardinal veins are very extensive and result in conditions which bear but little resemblance to those in the earlier stages. In connection with these changes the development of the inferior vena cava must be considered. The posterior cardinal veins appear very early as paired, bilaterally symmetrical vessels which extend from the duct of Cuvier to the tail region and are situated ventro-lateral to the aorta
Fig.195, Diagram representing a stage (later than Fig. 194) in the development of the superior vena cava and the inferior vena cava, also of the azygos vein. Hochstetter.
(Fig. 193). From the first they receive blood from the body wall through segmental branches, and as the primitive kidneys (mesonephroi) develop they receive blood from them also, as well as from the mesentery. They return practically all the blood from the region of the body situated caudal to the heart, just as the anterior cardinals return the blood from the region of the body situated cranial to the heart. In other words, the two sets of cardinal veins are the body veins par excellence during the earlier stages of development. While the anterior set persists for the most part as permanent vessels and increases with the development of the body, the posterior set undergoes regressive changes, its function being taken by a new vessel the inferior vena cava.
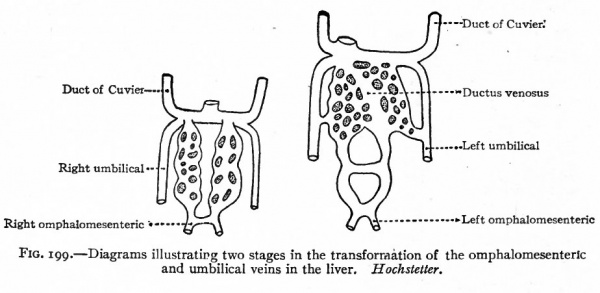
Not long after the appearance of the posterior cardinals, another pair of longitudinal veins appears in the medial part of the mesonephroi. They increase in size as the mesonephroi increase and receive blood from the latter. They also communicate with the cardinals by means of transverse channels which, however, are later broken up as the mesonephroi become more complicated in structure. These vessels are known as the subcardinal veins, or revehent veins of the primitive kidneys (Fig. 194, A). After they have attained a considerable size, a large anastomosis is formed between them ventral to the aorta and just caudal to the omphalomesenteric (superior mesenteric) artery (Tig. "194, B). In the meantime, a branch of the ductus venosus (see p. 229) grows caudally through the dorsal part of the liver and the mesentery, and joins the right subcardinal vein a short distance cranial to the above mentioned anastomosis (Fig. 194, A and B). This branch forms the proximal part of the inferior vena cava. At the same time, also, each subcardinal forms a direct connection with the corresponding cardinal at a point opposite the first anastomosis; consequently the inferior vena cava, the subcardinals and the cardinals are all in direct communication (Fig. 194, B). Thus two ways are formed by which the blood may return to the heart: It may return via the cardinals and ducts of Cuvier, and via the inferior vena cava.
Fig. 196. Diagram of final stage in the development of the superior vena cava and the azygos vein. (Compare with Fig. 195.)
It is obvious that while these conditions exist, that is, while the mesonephros is functional, and blood is carried to it by the cardinal veins and from it by the subcardinal veins, there is a true renal portal system. The blood from the body walls and lower extremities is collected by the segmental vessels and poured into the cardinal veins and is then distributed in the mesonephros by smaller channels or sinusoids (Minot), whence it is collected and carried off by the subcardinal veins. This passage of blood through purely venous channels simulates the conditions in the liver where there is a true hepatic portal system.
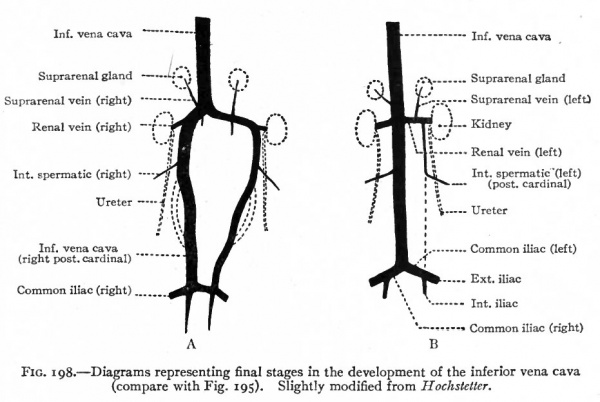
Frcm this time on, the changes are largely regressions in the cardinal and subcardinal systems, corresponding to the atrophy of the mesonephroi, and rapid increase in the vena cava and its branches. The cranial end of each cardinal becomes smaller; the left loses its connection with both the vena cava and the duct of Cuvier, the right its connection with the vena cava only (Fig. ig6j. Subsequent changes in these parts of the cardinals will be considered in the following paragraph. For a time the caudal ends of the two cardinals are of equal importance. Later, however, the right becomes larger, while the left atrophies. The right thus becomes a direct continuation and really a part of the vena cava (Figs. 195 and 198). This is brought about, of course, by the original anastomosis between the vena cava and the subcardinal and cardinal. On the left side the anastomosis persists simply as the proximal part of the renal vein (Fig. 198); on the right side the renal vein is a new structure which develops after the kidney has attained practically its final position, and opens into the vena cava secondarily. The inferior vena cava itself is a composite vessel derived from four different anlagen. i. The part which extends from the ductus venosus to the right subcardinal is of independent origin. 2. A short portion is derived from a part of the right subcardinal. 3. Another short portion is derived from the cross-anastomosis between the subcardinals and cardinals. 4. The caudal end is a derivative of the caudal part of the right cardinal (compare Figs. 194, 195, 198.)
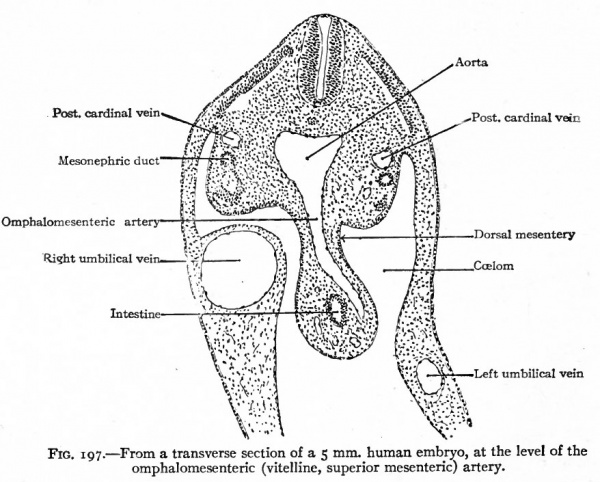
Fig. 197. From a transverse section of a 5 mm. human embryo, at the level of the omphalomesenteric (vitelline, superior mesenteric) artery.
Before the caudal end of the left cardinal vein atrophies, an interesting and important change occurs in the relations of the ureters and cardinals. Primarily the cardinal veins develop to the ventral side of the ureters. But later a collateral of each cardinal develops to the dorsal side of the ureter. These join the cardinal cranial and caudal to the ureter. In other words, a venous loop is formed around the ureter (Fig. 195). The ventral arm of the loop then atrophies and disappears, leaving the dorsal arm as the direct part of the cardinal vein. On the right side, where the cardinal persists as a portion of the vena cava, the latter vessel comes to lie ventral to the ureter (Fig. 198, A). On the left side the cardinal atrophies, leaving only the portion cranial to the loop as the proximal end of the internal spermatic (testicular or ovarian ) vein (Fig. 198, B). Since on the left side the original anastomosis between the subcardinals and cardinals persists as the renal vein, the left internal spermatic is a branch of the renal. The right internal spermatic vein probably represents a branch of the vena cava which is independent of the cardinal.
Fig. 198. Diagrams representing final stages in the development of the inferior vena cava (compare with Fig. 195). Slightly modified from Hochstetter.
In the cat embryo the venous loop around the ureter is much more extensive than in the other forms. The dorsal arm of the loop, named the supracardinal vein, extends from the iliac vein to the original anastomosis between the subcardinals and cardinals. In the course of further development the supracardinals approach each other and finally fuse, forming a large single vessel which becomes the portion of vena cava caudal to the renal veins. In this event the portions of both cardinals forming the ventral arms of the venous loops atrophy and disappear.
Near the caudal end of each cardinal vein a branch arises which receives the blood from the corresponding lower extremity. Then a transverse anastomosis appears between the two cardinals at this point (Fig. 198, A). Since the portion of the left cardinal caudal to the renal vein atrophies, the anastomosis itself constitutes the left common iliac vein (Fig. 198, B). The right common iliac is, of course, the original branch of the right cardinal. As the iliacs enlarge they form the two great branches of the vena cava.
Fig. 199. Diagrams illustrating two stages in the transformation of the omphalomesenteric and umbilical veins in the liver. Hochstetter.
With the atrophy of the mesonephroi, the subcardinal veins diminish in size and finally disappear for the greater part. The part of the right subcardinal cranial to the point of junction with the vena cava disappears entirely. The portion of the left subcardinal cranial to the anastomosis between the two subcardinals becomes much reduced in size, but persists as the left suprarenal vein. The left suprarenal vein is thus a branch of the left renal vein, since the latter represents the anastomosis itself (Figs. 194, 195, 198). The right suprarenal vein probably does not represent a persistent right subcardinal, but is a new vessel opening into the vena cava. The portion of each subcardinal caudal to the anastomosis probably disappears entirely, but this has not been definitely determined.
The observations on the development of the azygos veins in the human embryo are only fragmentary. In the rabbit the portions of the posterior cardinal veins immediately cranial to the anastomosis between the sub-cardinals and cardinals, that is, just cranial to the renal veins, disappear. The more cranial portion of the right cardinal persists as the azygos vein which receives the intercostal (segmental) branches and opens into the superior vena cava. An oblique anastomosis is formed, dorsal to the aorta, between the two cardinals (Fig. 195). This anastomosis and the portion of the left cardinal caudal to it together form the hemiazygos vein. The portion of the left cardinal cranial to the anastomosis loses its connection with the duct of Cuvier (or coronary sinus) and becomes the accessory hemiazygos vein (Fig. 196). The ascending lumbar veins, which join the azygos and hemiazygos, probably do not represent persistent parts of the caudal ends of the cardinals, but are formed by longitudinal anastomoses between the original segmental lumbar veins.
The changes which occur in the region of the liver are of much importance and result in conditions which bear no resemblance to the primary ones. As has already been noted, the omphalomesenteric veins enter the body at the umbilicus, pass cranially along the intestine and open into the caudal end of the heart. The umbilical veins, which appear soon after, enter the body at the umbilicus and pass cranially, one on each side, in the ventro-lateral part of the body wall; at the level of the heart they turn mesially through the septum transversum and join the corresponding omphalomesenteric veins to form a common trunk on each side, into which the duct of Cuvier then opens (Fig. 193). When the liver grows out as an evagination from the intestine, it comes in contact with the proximal ends of the omphalomesenteric veins and, as it enlarges, breaks them up into numerous smaller channels (Fig. 199).
The blood then, instead of having a direct channel, is forced to flow through these smaller channels which have been termed sinusoids. When the liver has attained a considerable size a more direct and definite channel is formed, which extends through the substance of the liver from the proximal end of the right omphalomesenteric vein obliquely caudally to the left omphalomesenteric vein. This newly formed channel is the ductus venosus (Figs. 199 and 200). In the meantime, three transverse anastomoses develop between the omphalomesenteric veins just caudal to the liver. The middle one is dorsal to the intestine, the other two ventral, so that the intestine is surrounded by two venous loops or rings (Figs. 199 and 200). At the same time a cross-anastomosis develops between the left umbilical vein, which is primarily the smaller, and the corresponding omphalomesenteric. This anastomosis joins the omphalomesenteric at about the point where the latter joins the ductus venosus, so that it seems to be a continuation of the ductus venosus. A similar cross-anastomosis also develops between the right umbilical and right omphalomesenteric (Figs. 199 and 200). Thus the blood that is brought in from the placenta by the umbilical veins may pass through the liver. Then the portion of each umbilical between the anastomosis and the duct of Cuvier atrophies and disappears (Fig. 200). The remaining portion of the left umbilical, which was originally the smaller, gradually increases in size and finally carries all the blood from the placenta. The right umbilical, on the other hand, loses its connection with the liver and persists only as a small vein in the body wall, which opens into the left umbilical vein near the umbilical cord (Fig. 201). Thus there is the peculiar phenomenon of a vessel carrying blood in different directions at different periods of its history. During the course of development of the septum transversum and diaphragm the left umbilical is withdrawn from the body wall and passes directly from the umbilicus to the ventral side of the liver. During fcetal life it conveys all the blood from the placenta to the liver. A part of the blood is distributed in the liver, a part is carried directly to the inferior vena cava by the ductus venosus (Fig. 202). After birth' the placental blood is cut off and the umbilical vein degenerates to form the round ligament of the liver.
Fig. 200. Veins in the liver region of a human embryo of 4 mm. His, Kollmann's Atlas.
The venous rings around the intestine also undergo marked changes. The right side of the most caudal and the left side of the most cranial disappear; the remaining vessel finally loses its connection with the ductus venosus and becomes the portal vein (Figs. 199, 200, 201 and 202). The portal vein is thus a derivative of the omphalomesenterics. After birth, when the placental blood is cut off, blood is distributed in the liver by branches of the portal vein, which represent the advehent hepatic veins; it is collected again by branches which unite to form the revehent hepatic veins, or hepatic veins proper, and the latter open into the inferior vena cava. The advehent and revehent hepatic veins are formed by the enlargement of some of the original sinusoids (Figs. 199 and 201).
Fig. 201. Veins in the liver region of a human embryo of 10 mm. Kollmann's Atlas.
++++++++++++++++++++++++++++++++++
Observations on the development of the veins in the extremities of human embryos are so fragmentary that it seems advisable to make use of the work that has been done on the rabbit. In the upper extremity the first vein to develop is the primary ulnar vein which begins in the radial (cranial) side of the extremity near its proximal end, extends distally along the radial border, thence proximally along the ulnar (caudal) border, and opens into the anterior cardinal vein (internal jugular) near the duct of Cuvier (Fig. 203). This condition is present in rabbit embryos of thirteen days. A little later a second vessel, the cephalic vein, appears as a branch of the external jugular, extends along the radial side of the extremity and becomes connected with the digital veins (Fig. 204). When the digital veins are taken up by the cephalic, the distal portion of the primitive ulnar undergoes regression. These changes have taken place in rabbit embryos of fifteen days, and for a short period the cephalic vein is the chief vessel of the extremity. The primitive ulnar vein, however, develops more rapidly than the cephalic and,
Fig. 202. Veins of the liver (seen from below) of a human foetus at term Kollmann's Atlas.
++++++++++++++++++++++++++++++++++
with its branches, soon becomes the chief vessel; the portion in the forearm gives rise to either the ulnar or basilic vein; the portion in the arm becomes the brachial vein which then passes over into the axillary, and the latter in turn passes over into the subclavian. The cephalic vein of the embryo persists as the cephalic of the adult, and, during the period when it forms the chief vessel of the extremity, a branch arises from it which becomes the radial vein. Primarily the cephalic vein opens into the external jugular, but later a new connection is formed with the axillary, while the original connection persists as the j ugulocephalic (Fig. 205).
In a rabbit embryo of ten and one-half days a vein follows the border of the lower extremity all the way round, connecting on the cranial side with the umbilical and on the caudal side with the posterior cardinal. This is the primitive fibular vein, and from its course is homologous with the primitive ulnar vein of the upper extremity (Fig. 203). From this time on, however, the course of development in the lower extremity differs from that in the upper. The connection of the fibular vein with the umbilical is soon lost. In older embryos (fifteen days) two branches of the fibular vein have appeared; one of these, the anterior tibial vein, begins on the embryo of 14 days (n mm.), dorsum of the foot and extends diagonally proximally, to open into the fibular in the caudal border; the other, the so-called connecting branch, begins as twigs in the abdominal wall and tibial side of the extremity and opens into the fibular just proximal to the opening of the anterior tibial (Fig. 204). Later the distal part of the primitive fibular is broken up by the differentiation of the digits (toes) and disappears almost up to the point of junction with the anterior tibial. The latter enlarges and receives the digital branches, and appears as a continuation of the proximal part of the primitive fibular. The anterior tibial and primitive fibular together thus constitute the sciatic vein (Fig. 205). Another vessel appears in embryos of fifteen days, which represents the beginning of the femoral vein and opens into the cardinal, cranial to the opening of the sciatic (Fig. 205). From this time on the femoral, with its branches, enlarges at the expense of the other veins and becomes the principal vein of the lower extremity. In the human embryo the femoral anastomoses with the sciatic near the knee and the proximal portion of the sciatic then atrophies, the distal portion persisting as the small sephenous vein. The
Fig. 203. Diagram of the veins in the extremities of a rabbit embryo of 14 days (11 mm). Modified from Lewis
| Fig. 204. Diagram of the veins in the extremities of a rabbit embryo of 14 days and 18 hours (14.5 mm.). Modified from Lewis. | Fig. 205. Diagram of the veins in the extremities of a rabbit embryo of 17 days (21 mm.). Modified from Lewis. |
Fig. 206. Diagram illustrating the foetal circulation. Compare with Fig. 207. Modified from Kollmann. The shading represents the relative impurity of the blood in different regions, the darkest shading representing the most impure blood large saphenous vein and the posterior tibial vein possibly are derivatives of the femoral, but this question has not been settled.
Changes in the Circulation at Birth
During fcetal life the course of the blood is adapted to the placental circulation, since the placenta is the only means by which the blood is purified and from which the foetus derives its nutriment. The pure blood from the placenta passes through the umbilical vein to the liver; there a part of it is distributed to the liver by some of the advehent veins, is collected again by the revehent veins and poured into the inferior vena cava; a part passes directly to the vena cava through the ductus venosus. At this point the blood acquires some impurity from the stream brought in by the vena cava itself and the portal vein. The slightly impure blood then flows into the right atrium, is directed by the Eustachian valve through the foramen ovale into the left atrium, thence flows into the left ventricle and is forced out into the aorta. A part of the blood flows on through the aorta, a part is carried to the upper extremities and head and neck regions by the subclavian and carotid arteries. The latter part, then becoming impure, is carried back to the right atrium by the subclavian and jugular veins and superior vena cava; from the right atrium the greater portion flows into the right ventricle and thence is forced out into the large pulmonary artery. But since the lungs are non-functional, this blood passes through the ductus arteriosus to join the stream in the aorta. The blood received by the more cranial portion of the foetus is but slightly impure, for the impure blood from the ductus arteriosus joins the aortic stream distal to the origin of tlie subclavian and carotid arteries. This accounts for the fact that the more cranial portion of the body generally is better developed than the more caudal portion. It is well to note here that the liver receives purer blood than any other part of the body, and this is undoubtedly correlated with the relatively enormous size of that organ in the foetus. The rather impure blood which starts through the dorsal aorta is in part distributed to the viscera, body walls, and lower extremities by the visceral and segmental arteries, and thence is collected by the branches of the portal vein and inferior vena cava to be returned as impure blood to the umbilical current at the liver; in part it is carried by the umbilical arteries to the placenta, there to be purified and collected by the branches of the umbilical vein (see Fig. 206).
Fig. 207. Diagram illustrating the circulation in the adult. Compare with Fig; 206. The shading represents the relative impurity of the blood, the white being the purest blood.
At birth, when the placental circulation is cut off, the proximal end of the umbilical vein atrophies to form the round ligament of the liver; the ductus venosus also atrophies and becomes merely a connective-tissue cord in the liver. The hepatic portal circulation is still maintained by the portal vein. The foramen ovale is closed and the impure blood from the inferior vena cava as well as that from the superior, passes from the right atrium into the right ventricle and thence is forced out through the pulmonary artery to the lungs, which at this time become functional, and is returned to the left atrium by the pulmonary veins. The ductus arteriosus atrophies to form the ligamentum arteriosum. From the left atrium the pure blood flows into the left ventricle, thence is forced out through the aorta and its branches to all parts of the body. At the same time the more distal portions of the umbilical arteries in the embryo atrophy to form the lateral umbilical ligaments, their proximal portions persisting as the superior vesical arteries (see Fig. 207).
Haemopoiesis Histogenesis of the Blood Cells.
Two sharply contrasting views are held regarding the origin and genetic relationships of the different kinds of blood cells. The one view, expressed in the monophyletic theory, holds that there is differentiated out of the mesenchyme a certain type of cells the primitive blood cells, or haemoblasts and that from this single type all the cells of the blood arise through processes of development along divergent lines. The other view, expressed in the polyphyletic theory, holds that while the blood cells are of mesenchymal origin the red cells and white cells have a dual origin, each type arising from its own mother-cells; and further that perhaps each kind of white cells arises from a distinct parent-cell. The recent extensive studies of the problem have yielded evidence that turns the balance at present in favor of the monophyletic theory, and the following account is based in the main upon these studies, particularly those of Maximow on the rabbit and Dantschakoff on the chick.
The sites of blood formation, or haemopoiesis, are (i) the area opaca (yolk sac), (2) the body mesenchyme, including the endothelium of the early blood-vessels, (3) the liver and spleen, (4) bone marrow, and (5) the lymph glands. These various structures are functional at successive periods of development of the embryo, but overlap to a certain extent, the marrow and lymph glands being probably the only foci of origin of blood cells in the adult. In the area opaca blood-cell development is initiated in the formation of the blood islands. Some of the mesenchymal cells become less irregular in shape by retraction of their protoplasmic processes and isolation from the general syncytium. They assume amoeboid properties and the cytoplasm aquires a distinctly basophilic character (Fig. 208). These then represent primitive blood cells, or hcemoUasts. Maximow has given them the name primitive lymphocytes, or lymphoblasts , regarding them as the common ancestors of all the blood cells. Clusters of these cells constitute the blood islands which are involved in the development of the primitive blood spaces, the superficial cells being transformed into endothelium (see p. 186) and the central cells remaining as primitive lymphocytes. Other primitive lymphocytes also differentiate in the mesenchyme outside of the blood spaces, afterward probably entering the vessels by virtue of their amoeboid properties.
Fig. 208. Mesenchyme from a rabbit embryo at the time of beginning blood formation.
Maximow.
m, Ordinary mesenchyme cells; m', mesenchyme cell in mitosis; /, primitive Wood cell
(primitive lymphocyte).
++++++++++++++++++++++++++++++++++
There is a view that both the blood cells and the endothelium of blood vessels arise from certain mesam&boid cells of entodermal origin, which are insinuated between the entoderm and mesoderm but are not in the strict sense constituents of the latter, and which collectively have been called the | angioUast. While the mesamceboid cells are probably identical with the j primitive lymphocytes, the idea that they constitute a set of specific rudiments of entodermal origin, from which both blood cells and endothelium arise, has not been generally accepted. The view, however, is not discordant with the monophyletic concept of the origin of blood cells.
Fig. 209. Portion of a blood vessel from the yolk sac of a rabbit embryo, showing various
stages in the formation of erythrocytes. Maximow. fl, megaloblasts; a', megaloblast in mitosis; b, normoblasts; b', normoblast in mitosis; c, erythro
blasts; d, erythrocyte, not yet discoid; en, endothelium; /, primitive lymphocytes;
k, normoblast recently divided; n, shrunken erythroblasts (?); n' ', extruded nucleus.
++++++++++++++++++++++++++++++++++
The primitive lymphocytes (of Maximow), constituting the parent stem from which all the blood cells arise according to the monophyletic theory, specialize at first in two general directions. In one direction the specialization leads toward the erythrocytes, or red blood corpuscles, and in the other toward the leucocyte, or white blood cell, series including the myelocytes. In the former case the lymphocytes become modified in that the cytoplasm becomes less basophilic and acquires a trace of haemoglobin; the nuclei become somewhat eccentric, the chromatin network a little denser and the nucleoli less conspicuous. While these changes are in progress the cells multiply by mitosis. The resulting cells are termed megaloblasts (Fig. 209, a). These continue to multiply by mitosis, the cytoplasm acquiring more haemoglobin and the nuclei becoming more dense, the resulting cells being somewhat smaller and known as normoblasts (Fig. 209, b) . The normoblasts, still dividing by mitosis, acquire still more haemoglobin and become erythroblasts (Fig. 209, c). These lose their nuclei and thus become erythrocytes, the definitive red blood corpuscles. The manner in which the nuclei are lost is a matter of dispute. Some claim it is absorbed (karyolysis) ; others claim it is extruded (karyorrhexis) (Fig. 210); recently the observation has been made that the nucleus with a small amount of surrounding cytoplasm escapes from the cell in a manner resembling constriction.
In the specialization leading to the white blood cell series, the parent stem cell (primitive lymphocyte) proliferates by mitosis and undergoes certain divergent changes in its nucleus and cytoplasm which yield the characters of the various kinds of leucocytes. Some of the cells become polymorphonuclear and acquire neutrophile granules to become neutrophile leucocytes; others acquire acidophile granules as acidophiles; still others, basophile granules as basophiles. The large mononuclear leucocytes, with the transitional forms having the horseshoe-shaped nuclei, possibly represent but slightly modified primitive lymphocytes. The definitive lymphocytes are probably derived from the primitive by division and but slight changes in character. Thus the various forms of white blood cells would not represent different stages in a series but divergent lines of specialization from a parent stem.
As mentioned before, the various blood forming organs function as such at successive stages of development of the embryo. The mesenchyme generally, both in the yolk sac and in the body, gives rise to blood cells during the earlier stages and may continue to do so until relatively late in embryonic life as has been demonstrated in the chick. It is interesting to note in this connection that in certain regions endothelial cells may also be transformed into primitive blood cells. In the earlier stages of liver development active haemopoiesis is observed in the sinusoids, probably partly from cells carried in by the blood stream and partly from primitive blood cells derived from the neighboring mesenchyme (Fig. 211). This function ceases in the liver in later embryonic life. The formation of blood cells takes place in the developing spleen but erythrocyte formation ceases after birth, although following severe haemorrhage the function may be resumed even in adult life. The formation of lymphocytes, however, goes on throughout life in the splenic corpuscles.
Fig. 210. Showing the escape of the nuclei from nucleated red blood cells. Howell.
I, 2, 3, 4, represent stages of extrusion observed in living cells; a, from circulating blood of adult cat after bleeding four times; b, from young kitten after bleeding; c, from 90 mm. cat embryo; others from marrow of adult cat. ++++++++++++++++++++++++++++++++++
Fig. 211. From the liver of a rabbit embryo, showing formation of red blood cells. Maximow. a, Megaloblasts; a', megaloblast in mitosis; b, normoblasts; c. erythroblasts; en, en', en, endotheHal cells; h, liver cells; /, primitive lymphocytes; /', primitive lymphocyte in mitosis; , nucleus being extruded from small erythroblast.
++++++++++++++++++++++++++++++++++
The lymph glands are constant sources of lymphocytes, the parent cells being the large mononuclear cells found in the germinal centers. These cells are regarded as closely allied to the primitive lymphocytes, perhaps even identical, although here in this particular environment giving rise only to the lymphocyte line.
The bone marrow is an important source of blood cells in the embryo, and in the adult under normal conditions is regarded as the only source of red corpuscles. The parent stem cells, here called myeloblasts, are recognizable in the form of large mononuclear, non-granular cells, with the general characters of primitive lymphocytes, which give rise to the red blood cells through clearly distinguishable megaloblast and normoblast stages, and to the various forms of leucocytes and lymphocytes. In addition the parent cells also give rise to certain other cells which are normally confined to the marrow, viz., the myelocytes. These are large mononuclear cells, with vesicular nuclei, the cytoplasm containing neutrophile, acidophile, or basophile granules similar to those of the leucocyte series (Fig. 212). The genetic relationships of the "giant" cells, or myeloplaxes, in the marrow are not clear. The myeloplaxes are large masses (30 to 100 micra in diameter) of homogeneous or finely granular, slightly basophilic cytoplasm containing either a single lobulated, annular nucleus (megakaryocytes, Fig. 212, meg) or many nuclei (polykaryocytes). The polykaryocytes have been considered identical with the osteoclasts, which may represent fused osteoblasts, but this relationship has not been definitely established. Both kinds of cells have been considered as derivatives of the myeloblasts, the polykaryocytes being later stages of megakaryocytes.
Fig. 212. From a section of red marrow from the femur of a young rabbit. Schafer. e, Erythrocy tes ; e r , normoblasts; e", normoblast in mitosis; /, outlines of fat cells; ^, polymorphonuclear leucocytes; m, neutroohile myelocytes; m', myelocytes in mitosis; m", eosinophile myelocytes; m'". basophile myelocytes.
++++++++++++++++++++++++++++++++++
The blood platelets are now regarded by some authors as derivatives of the megakaryocytes; pseudopodia of the latter breaking off and gaining access to the blood stream. By others they are not believed to be formed constituents of the circulating blood, but appear only after shed blood comes in contact with a foreign substance.
The accompanying table, which is a tentative graphic scheme of the monophyletic theory, will assist the student in tracing the lineage of the blood cells.
The Lymph Vascular System
A controversy has arisen over the origin of the lymph channels and their endothelium, similar to the one that arose over the genesis of the blood vessels. There are therefore two main views, viz.: (i) that the endothelium of the lymph vessels arises as sprouts from the endothelium of veins and continues to grow by proliferation and migration of its own cells, the lymphatics thus being direct derivatives of the venous channels; (2) that the lymph vessels arise in situ through enlargement and coalescence of intercellular tissue spaces, the mesenchymal cells bounding these spaces becoming flattened and rearranged to form the endothelial walls of the vessels, and, as a corollary, that the junction of the lymph vessels with the veins, which occurs at certain definite points, is a secondary matter.
Fig. 213. Diagram showing the arrangement of the lymphatic vessels in a pig embryo of 40 mm. Sabin.
Here again the scope of the work does not permit presentation in detail of the evidence adduced in favor of either of these views. The advocates of the first view have placed much dependence upon the method of injection, which, as in the case of the origin of blood vessels, has been met with the criticism that injection shows only those lymph channels with continuous lumina and leaves undetermined the field beyond the injected area (see page 194). To supplement their studies by the injection method, the investigators who maintain that lymphatic endothelium grows by sprouting of preexisting endothelium have added studies on living tissues in which the sprouting phenomena are claimed to be clearly observable. Those who maintain that the lymphatics and their endothelium arise in situ from intercellular tissue spaces and the bordering cells, argue that the same principles underlie the formation of lymphatics that determine blood-vessel development and that it has been shown experimentally that blood vessels develop in regions which have been entirely cut off from any source of endothelium except the mesenchymal cells in situ (see page 194).
According to the first view lymphatic development can be divided into two stages: (i) the formation of isolated lymph sacs, derived from veins, which become united into a system, and (2) the peripheral growth of lymph vessels which sprout from the endothelium of these sacs and spread through the body. (i) The first sacs appear, one on each side, along the jugular (anterior cardinal) veins. The branches of these veins at first form a plexus; a portion of the plexus becomes cut off from the parent stems and lies as a series of isolated spaces in the mesenchyme ; these spaces then enlarge and coalesce to form an endothelial-lined sac the jugular lymph sac or heart which afterward joins the jugular vein by a new opening (Fig. 213). A second pair of sacs the posterior lymph sacs or hearts develops in the same manner from the more caudal branches of the posterior cardinal veins (Fig. 213). Two other saclike structures develop the cisterna chyli and retroperitoneal sac the former in the region of the renal veins and the latter in the vicinity of the suprarenal bodies. Through the longitudinal fusion of the chain of sac-like structures, the axial lymphatic drainage line of the body is established (Fig. 213). The thoracic duct probably represents the fused cisterna chyli and jugular lymph hearts. The lymph hearts in the avian and mammalian embryo become relatively smaller as development proceeds until in the adult they are barely discernible as slight dilatations in the lymph vessels. The cisterna chyli, however, may persist as a clearly distinguishable dilatation at the caudal end of the thoracic duct.
(2) The peripheral lymph channels, which drain into the thoracic duct, represent outgrowths from the lymph sacs. From the jugular sacs sprouts invade the neck, head, shoulders, and finally the entire upper extremities and upper part of the body wall (Fig. 214). Similarly, from the posterior lymph hearts sprouts invade the lower extremities and lower portion of the body wall (Fig. 214). Outgrowths from the original axial drainage line invade the various visceral organs (Fig. 213). Thus the lymphatic drainage of the body is effected through outgrowths from a few primary centers which represent derivatives of the venous channels. The lymph glands are secondary foci of development along the lymph vessels (see page 249).
Fig. 215. From cross-sections of cat embryos in successive stages (<z, b, c, d) of development, in the region of the jugular lymph sac; diagrammatic but amply supported by studies of serial sections and reconstructions. Huntington.
- i, Anterior cardinal vein; 2, somatic tributary of same; 4, developing blood cells in the mesenchyme; 5, mesenchymal intercellular spaces rudiments of the jugular lymph sac; 6, rudimerits of brachio-cephalic venous anastomosis; 7, brachio-cephalic venous anastomosis; 8, haemophoric lymphatic plexus forerunner of jugular lymph sac; u, thoracic duct'approach' of jugular lymph sac; 12, rudiments of thoracic duct; 13, jugular lymph sac preparing to rejoin vein and to establish secondary connection with rudiments of thoracic duct (12) and of other systemic lymphatics (14); 15, jugular lymph sac, which has rejoined vein through permanent lymphatico-venous tap (16); 17, thoracic duct; 1 8, jugular and cephalic systemic lymphatics.
Fig. 216. From a photograph (X 600) of a cross-section through the caudal end of a chick embryo of 15 mm. Showing enlarged mesenchymal intercellular spaces as rudiments of the posterior lymph sac. West. 3, Coccygeal vein; 6, caudal muscle plate; 8, isolated enlarged intercellular spaces, the bounding cells becoming flattened; 9, lateral branch of coccygeal vein; 10, lymphatics containing collections of developing blood cells.
The view that lymphatics arise as enlarged isolated intercellular spaces in the mesenchymal tissue does not include any dispute as to the general disposition of the lymph channels in the body, but comprises a fundamentally different concept of the origin of these vessels. Upon a long and exhaustive series of observations on closely graded series of embryos of Fishes, Amphibia, Reptiles, Birds, and Mammals is based the conclusion that not only the lymph sacs but the peripheral lymphatics as well originate independently of the veins; and that the opening of the main lymphatic drainage lines into the jugular or subclavian veins near their junction, and into the inferior vena cava and renal veins (in some monkeys) , is second* arily established. The same hydrodynamic mechanical factors regarded as operative in the formation of blood vessels, viz.: pressure and friction incident to blood flow (see page 195), are considered as effective likewise in the development of lymphatics. Fundamentally, therefore, the lymph vascular system from the viewpoint of development differs in no wise from the blood vascular system.
Fig. 217. Diagrams showing three stages (a, b, c) in the development of the thoracic duct in the cat embryo, in which the jugular lymph sac has established two permanent venous connections (8 and 9). Huntington.
- i, Anterior cardinal vein; 2, duct of Cuvier; 3, posterior cardinal vein; 4, external jugularcephalic vein; 5, subclavian vein; 6, jugular lymph sac; 7, thoracic duct 'approach' of lymph sac; 8, common jugular opening of lymph sac; 9, jugulo-subclavian opening of lymph sac; 10, rudiments of thoracic duct; n, thoracic duct.
The lymph sacs, both jugular and posterior, arise through enlargement and confluence of mesenchymal intercellular spaces, the cells bounding the spaces becoming flattened and rearranged to form endothelium. In the case of the mammalian (cat) jugular sac, intercellular spaces in the region dorso-lateral to the anterior cardinal vein unite into an intricate plexus of channels which then opens into the vein (Fig. 215, a and b). Following this the components of the plexus enlarge and coalesce to form the sac, which then temporarily severs connection with the vein (Fig. 21 5, c) . Finally the sac effects a permanent connection with the vein through one or more openings which represent the lymphatico-venous communications of the adult (Fig. 215, d). In the case of the posterior sac, intercellular spaces dorsal to the posterior cardinal vein (Fig. 216, 8) first form a plexus the components of which then unite into a large endothelial-lined space which opens into the dorsal tributaries of the vein.
The thoracic duct also arises as a chain of isolated endothelial-lined spaces along the line of the aorta. These unite longitudinally into a continuous channel which joins the jugular lymph sac, thus forming the axial lymphatic drainage line of the body (Fig. 217, a, b, c). In reptilian embryos the spaces first fuse into a distinct periaortic plexus out of which the thoracic duct is established. In the avian embryo the chain of spaces follows the general line of the aorta but does not become so intimately associated with the great arterial trunk as in reptiles. In the mammalian forms rudiments of the thoracic duct follow the same general plan of development, but are associated topographically with the ventro-medial tributaries of the azygos veins. These tributaries finally become detached from the larger venous trunks, atrophy and disappear, being replaced by the thoracic duct.
On the same principles laid down for the development of the lymph sacs and thoracic duct, the peripheral lymphatics also are developed. In all the regions of the body not immediately drained by the lymph sacs or thoracic duct, mesenchymal intercellular spaces enlarge and coalesce, the cells bounding the coalesced spaces being transformed directly into endothelium; the spaces unite to form a plexus of endothelial-lined channels, and in this plexus certain channels increase in size to form the larger lymphatics which converge and eventually join the main axial drainage line. Thus the lymphatic drainage of the entire body is established.
One of the most interesting and significant phases of lymphatic development, which has been brought out through recent studies of the problem, is the role played by certain early lymph channels in conveying blood cells to the general circulation. It has been found that, in the region subsequently occupied by the lymph sacs, extensive blood cell formation (haemopoiesis) occurs prior to the formation of lymphatic rudiments. As the lymph spaces appear and unite into a plexus the developing blood cells are included within them (Fig. 215, a and Fig. 216, 10). When the lymphatic plexus joins the veins the blood cells are carried into the general circulation (Fig. 215, b). This haemophoric function of the early lymph channels is especially prominent in the case of the thoracic duct in the chick. Here extensive collections of blood cells develop in the mesenchymal tissue along the line of the aorta and become included within the rudiments of the thoracic duct and eventually, when the latter unites with the jugular lymph sac, are carried into the veins and thus enter the general circulation. After these early lymphatics, which transport blood cells and which have been defined as haemophoric lymphatics, or veno-lymphatics, fulfil their haemophoric function they are retained as permanent lymph channels in the general lymphatic organization. In a broader interpretation, the haemophoric function of certain lymph vessels during ontogeny is particularly significant in that it indicates essential and fundamental similarity of lymphatic vascular development to haemal vascular development.
The Lymph Glands
The lymph glands do not begin to develop for some time after the lymphatic vessels, since there are no indications of them in the human foetus until the latter part of the third month and none in pig embryos until thev
Fig. 218. From a section through the axilla of a human embryo of 125 mm. (4-5 months), showing an early stage of a lymph gland. Kling.
++++++++++++++++++++++++++++++++++
have reached a length of 30 mm. While it is definitely settled that lymph glands originate in very close relation with the lymphatic vessels, certain points in their later development need further study. In the axilla and groin, for example, the lymphatic vessels form a dense network in the meshes of which are masses of connective tissue. These masses become more cellular and with the surrounding vessels constitute the anlagen of lymph glands (Fig. 218). The new cells which appear in the masses are lymphocytes which may pass through the walls of the neighboring blood vessels and lodge here or may be derived directly from connective tissue (mesenchymal) cells in situ. Whatever the origin of the lymphocytes may be, they have the opportunity here to divide freely. The mass becomes still more cellular and enlarges at the expense of the lymphatic vessels which then come to form a network around the mass. This network is the marginal plexus, and it communicates freely with the neighboring lymphatic channels. Within the mass of cells blood vessels are present from the beginning, and these are destined to be the blood vessels of the lymph gland, and the point of their entrance and exit marks the hilus. Outside of the marginal plexus the connective tissue condenses to form the capsule. The gland at this stage thus consists of a central compact cellular mass, made up of connective tissue and lymphocytes, in which blood vessels ramify; a plexus of lymphatic channels around the mass which communicate with the neighboring channels; and around the whole structure a capsule of connective tissue (Fig. 218). Further development consists of the breaking up of the cell mass by
Fig. 219. Diagram illustrating a stage (later than Fig. 218) in the development of a lymph gland. Stohr.
++++++++++++++++++++++++++++++++++
iymj hatic channels and the formation of the follicles. It seems probable that branches from the marginal plexus invade the cell mass principally from an area around the hilus, thus breaking it up into smaller irregular masses or cords. At the side opposite the hilus the invading channels are less numerous, leaving larger parts of the mass which become the follicles (nodules) of the cortex. On all sides the invading channels communicate with the marginal plexus and form the so-called intermediary plexus. The gland as a whole enlarges and its peripheral part pushes outward into the surrounding tissue. Over the follicles the capsule is pushed outward, while between them it remains in place and comes to dip into the gland as the trabeculcz. The blood vessels tend to lie in the trabeculae, but a small branch probably passes to each follicle. In the follicles themselves the lymphocytes proliferate and the central part of each follicle becomes a germinal center. The connective tissue among the lymphatic vessels composing the marginal plexus becomes proportionately less as the vessels enlarge and finally exists only as strands of reticular tissue which, naturally, are covered by the endothelium ; thus the marginal plexus becomes the marginal sinus. The intermediary sinus is formed by the channels which originally invaded the cell mass. The reticular tissue is probably composed of remnants of the original connective tissue. All the channels converge at the hilus to form the efferent lymphatic vessels (Figs. 219 and 220).
Fig. 220. Diagram illustrating a late stage in the development of a lymph gland. Compare with Fig. 219. Stohr.
++++++++++++++++++++++++++++++++++
The haemolymph glands are probably developed in much the same manner as the lymph glands except that in the former the sinuses are filled with red blood cells.
The first lymph glands to develop are those in the axilla, in the inguinal region, in the neck, and in the base of the mesentery. These are the so-called primary glands and develop during fcetal life. They are of constant occurrence in these regions, but vary in number in different individuals. The secondary lymph glands are those in the bend of the elbow, in the popliteal space, in the mesentery, and around the aorta. Some of these develop during foetal life and some later. While lymph glands are of constant occurrence in some regions throughout life, the number may vary at different times in any region; and there may also be variations in different individuals. Glands may be called into existence at any time during life, in almost any region, as the result of exceptional activity of some organ, or in pathological conditions. Such structures are known as tertiary lymph glands.
The origin of the lymph (plasma) itself is probably extremely complex. At one time it was considered as the result of nitration from the blood plasma through the capillary walls. If lymph originates in this way the nitration is selective, for the chemical composition of the lymph differs from that of the blood plasma. In all probability the lymph plasma consists of blood plasma which has escaped through the vessel walls plus the products of cell activity in the tissues.
The Spleen
Since the spleen is generally considered as a lymphatic organ and since recent researches have shown that its structure is quite comparable to that of the lymph glands, it seems advisable to consider it under the head of lymphatic organs. Its ultimate origin is not yet settled and the details of its later development are still obscure. The same difficulties are met with as in the case of the origin and development of blood cells, for it is known that the spleen plays a part in the formation of the blood cells. Its structure differs from that of the lymph glands chiefly in that it possesses no distinct lymphatic sinuses; but it does possess lymph follicles (splenic corpuscles) and densely cellular cords (pulp cords) which are separated by cavernous blood vessels (cavernous veins).
For some time the spleen was considered as a derivative primarily of the mesenchyme in the region of the dorsal mesogastrium. More recently, however, investigators have taken the view that it arises partly, or possibly entirely, from the mesothelium (coelomic epithelium) of the dorsal mesogastrium. In human embryos during the fifth week the anlage of the spleen appears as an elevation on the left (dorsal) side of the mesogastrium (Fig. 221). This elevation is produced by a local thickening and vascularization of the mesenchyme, accompanied by a thickening of the mesothelium which covers it; and, furthermore, the mesothelium is not so distinctly marked off from the mesenchyme as in other regions. Cells from the mesothelium then migrate into the subjacent mesenchyme and the latter becomes much more cellular (Fig. 222). The migration is brief, and in embryos of about forty-two days has ceased, and the mesothelium is again 1 reduced to a single layer of cells. The elevation becomes larger and projects ; into the body cavity. At first it is attached to the mesentery (mesogastrium) by a broad, thick base, but as development proceeds the attachment if becomes relatively smaller and finally forms only a narrow band of tissue 'j through which the blood vessels (splenic artery and vein) pass.
Further development of the substance of the spleen consists of the breaking up of the cellular mesenchymal tissue by blood vessels and the formation of the splenic corpuscles. The connective tissue trabeculce, as well as the jfj capsule of the spleen are derived from the original mesenchymal tissue. The blood vessels become dilated in parts of their course to form the cavernous vessels (cavernous veins) which are separated by the pulp cords. The connective (reticular) tissue of the pulp cords is a derivative of the mesenchyme, as are also the various types of cells in the cords. The adventitia of the walls of some of the small arteries becomes infiltrated with lymphocytes to form the splenic corpuscles (lymph follicles).
Fig. 221. From transverse section through stomach region of a 14 pig embryo. Photograph.
It is generally recognized that during foetal life the spleen is a hematopoietic organ, that is, both leucocytes and nucleated red blood cells ai.e produced within it. Normally, the formation of erythrocytes stops at or soon after birth. In severe anaemia or in pernicious anaemia in postnatal life, however, the presence of dividing nucleated red blood cells suggests a return to embryonic conditions. The reticular tissue constitutes the source of these nucleated forms (erythroblasts) . It has also been suggested that the spleen acts as a destroyer of worn-out erythrocytes, for in many cases apparent remnants of the latter have been observed within the cytoplasm of the "spleen cells." The lymphocytes proliferate to a certain extent in the splenic corpuscles, and in that way, at least, the spleen serves as a base of supply for leucocytes. There is a possible suggestion that the first leucocytes of the spleen have their origin in the mesenchymal cells of the spleen anlage. This would be in accord with the observations which indicate that leucocytes are derived from indifferent mesenchyme cells.
Fig. 222. From section through dorsal mesogastrium (anlage of spleen) of a chick embryo of 3 days and 21 hours incubation. Tonkofl.
++++++++++++++++++++++++++++++++++
Glomus Coccygeum.
The coccygeal skein (coccygeal gland) was originally considered as belonging to the same category as the suprarenal glands, but the latest researches have indicated that its cells do not possess the characteristic chromamn reaction and that it belongs rather to the category of lymph glands. It develops ventral to the apex of the coccyx in relation with branches of the middle sacral artery.
Although the thymus gland becomes a lymphatic structure it is primarily derived from the epithelium (entoderm) of the branchial grooves and will be considered in connection with the development of the alimentary tract (Chap. XII). The tonsils also will be considered in the same connection.
Anomalies
Anomalies of the Heart
Acardia
The malformation known as acardia occurs in the case of twins that have but one chorion. The so-called acardiac condition does not necessarily imply the absence of the heart in the affected twin, for the latter may develop to a considerable degree and possess a functionating heart. On the other hand, the affected twin may be only an amorphous mass of tissue which derives its total blood supply through the agency of the stronger twin's heart. Or there may be any intermediate form between these two extremes. The point is that the acardiac monster (acardiacus) derives its blood wholly or in part through the agency of the stronger heart. A further discussion of acardiac monsters and their possible explanation will be found in Chap. XX.
Double Heart
But one or two cases of a double heart in a single human foetus have been recorded. In some of the lower forms (chick) it occurs more frequently. The explanation is probably to be found in the double origin of the heart in Amniotes (p. 196).
Anomalous Position of the Heart
Congenital anomalies in the position of the heart are rare. Dextrocardia (heart on the right side) is almost invariably associated with changes in the position of the viscera (see transposition of the viscera, page 304) . In the condition known as ectopia cordis, the heart, with the pericardium, protrudes through a cleft in the ventral wall of the thorax, the cleft being probably due to an imperfect fusion of the two sides of the body wall in that particular region.
Anomalies of the Septa
The most frequent anomaly in the atrial septum is the persistence of the foramen ovale. The entire foramen may remain patent, or, as is more frequently the case, a smaller opening may persist between the ventral (anterior) border of the foramen and the valve of the latter (p. 203).
The atrial septum may be wholly lacking, but this always occurs in conjunction with other defects. It sometimes happens that the primary atrial septum (septum superius), which grows from the cephalic side of the common chamber, fails to fuse with the septum of the atrio-ventricular aperture (p. 203 and Fig. 171).
Defects in the ventricular septum occur less frequently than in the atrial septum. It may happen that the cephalic (upper) border of the ventricular septum fails to fuse with the septum which divides the aortic trunk and bulb into the aorta and pulmonary artery. This affects the cephalic (upper) part of the septum sometimes called the pars membranacea (p. 204 and Fig. 174); and since the defect is situated near the opening of the aorta it brings about the so-called "origin of the aorta from both ventricles." Stenosis of the pulmonary artery usually accompanies this condition. Rarely is there a deficiency in the caudal (lower) part of the ventricular septum. Complete absence of the ventricular septum may occur, and along with it also an absence of the atrial septum, so that the heart is simply two-chambered; or the single ventricle may open into two atria. The causes of these defects ] are obscure.
Anomalies of the Valves
There may be congenital variations in the j size and number of the atrio-ventricular valves, depending upon abnormal position, fusion, or division of the pad-like masses from which the valves ! develop (p. 206).
There may be also a greater or lesser number of semilunar valves in the | aorta and pulmonary artery. This irregularity can probably be referred back to an atypical division of the aortic trunk and bulb, and a corresponding \ atypical division of the protuberances which give rise to the valves (p.. 206). Variations in the valves may or may not be accompanied by functional dis- i turbances. The congenital diminution in the number of valves should be distinguished from the acquired, where chronic endocarditis may cause a fusion.
Anomalies of the Large Vascular Trunks
Anomalies of the Arteries
There may be a transposition of the aorta \ and pulmonary artery. This results from an anomalous division of the aortic trunk and bulb. The partition develops in such a way as to put the aorta in communication with the right ventricle, and the pulmonary artery with 1 the j left ventricle (p. 204). Or the aorta and pulmonary artery may remain in \ direct communication on account of an imperfect development of the partition. Rarely the two vessels remain as a common stem.
Congenital stenosis (constriction) of the pulmonary artery may occur, j accompanied by an increase in the size of the aorta, possibly due to an unequal j division of the aortic trunk and bulb. After birth little or no blood can pass \ to the lungs, and the result is a general damming (stasis) of the venous blood ! with marked cyanosis. This is at least one explanation of the so-called "blue babies." Less frequently there is a stenosis of the proximal end of the aorta, with excessive size of the pulmonary artery, also due to an unequal division of the aortic trunk and bulb (p. 204) . These stenoses are usually, though not always, accompanied by defects in the ventricular septum.
Persistence of the ductus arteriosus may occur without any other defect; ;i but usually the persistence is associated with anomalous conditions of the aorta and pulmonary artery.
Occasionally the arch of the aorta is found on the right side. This condition is due to the persistence of the fourth aortic arch on the right side instead of the corresponding arch on the left side; this is the normal condition in Birds. Rarely both fourth aortic arches persist, which results in a double arch of the aorta the normal condition in Reptiles. (Compare Figs. 181 and 182.)
The dorsal aorta, particularly the abdominal part, is occasionally found to consist of two parallel, imperfectly separated vessels a condition known as double aorta. This anomaly is due to an imperfect fusion of the two primitive aortae (p. 187 and Fig. 165).
Numerous variations are met with in the larger branches of the aorta,, many of which are explained by referring them to embryonic conditions. Especially noteworthy are the branches from the arch of the aorta, since their development is so closely associated with the changes in the aortic arches. The normal arrangement passing from the heart, is innominate artery, left common carotid artery, left subclavin artery (see Fig. 182).
1. All these branches may be collected into a single trunk a condition characteristic of the horse.
2. Two branches may arise from the arch, (a) The left common carotid unites with the innominate, and the left subclavian arises separately. This is the normal arrangement among the apes, and is probably the most common variation in man. (b) Very rarely there are two innominate arteries, each formed by the union of a common carotid and subclavian a condition characteristic of Birds.
3. Three branches may arise from the arch but in a manner differing from the normal. Each subclavian arises separately and the two common carotids are united into a single vessel. This arrangement is found in some of the Cetacea.
4. Four vessels may arise from the arch, (a) These are, in order, innominate, left common carotid, left vertebral, left subclavian. (b) Or the order may be right common carotid, left common carotid, left subclavian, right subclavian. In this case the proximal part of the right subclavian represents the portion of the right dorsal aortic root just cranial to the bifurcation; the fourth arch on the right side disappears, (c) Or very rarely the order may be right subclavian, right common carotid, left common carotid, left subclavian.
5. Five branches of the arch are rare. In order they are right subclavian, right vertebral, right common carotid, left common carotid, left subclavian.
6. Very rarely there are six branches of the arch; right subclavian, right vertebral, right .common carotid, left common carotid, left vertebral, left subclavian.
Anomalies of the Veins
The two pulmonary veins on each side, more frequently those on the left side, many unite into a common trunk before opening into the atrium. This variation is probably due to the fact that the absorption of the originally single pulmonary trunk into the wall of the atrium does not proceed far enough to cause all four of the pulmonary veins to open separately (see p. 205) . The upper (more cephalic) vein on the right side may open into the superior vena cava; or the upper vein on the left side may open into the left innominate vein. A possible explanation for this is that the pulmonary veins are formed after the heart and other vessels have developed to a considerable degree, and some of them may unite with the other vessels instead of with the atrium.
Occasionally two superior vena cava are met with. In this case the right opens into the right atrium in the normal position; the left opens into the right atrium through the coronary sinus which naturally is much enlarged. This condition represents a persistence of the proximal end of the left anterior cardinal vein and the left duct of Cuvier, and is the normal arrangement in many of the lower Vertebrates. Even with two venae cavae there may be a small anastomosing branch in the position of the left innominate vein, which represents the normal structure in the Marsupials (see Figs. 194 and 195 and p. 223). There are a few cases on record of a single left superior vena cava.
The inferior vena cava is also subject to variations which represent the abnormal persistence of certain embryonic vessels. Perhaps the most striking of these variations is the condition known as double inferior vena cava. There may be two parallel vessels, of equal or unequal size, which unite at or above the level of the renal veins. This condition is to be explained by the persistence of parts of both posterior cardinal veins. It is met with not infrequently among the lower Mammals, especially the Marsupials (see Figs. 195 and 198).
Rarely the inferior vena cava opens into the superior, and in this case the hepatic veins open directly into the right atrium. This anomaly probably represents a failure of the absorption of the sinus venosus into the wall of the atrium (p. 205).
A left renal vein may open into the left common iliac, which condition represents a persistence of the more caudal part of the left posterior cardinal (Fig. 198). This anomaly is rare.
The azygos vein occasionally presents variations which are due to anomalous development. All the intercostal veins on the left side may be collected into a vessel which opens into the left innominate vein. There may be a single median azygos vein; or there may be a transposition of the azygos vein. It may be on the left side and open into the coronary sinus (normal conditions in the sheep and a few other Mammals). The latter condition represents a persistence of the more cephalic part of the left posterior cardinal vein (see Figs. 195 and 106).
Space does not permit a discussion of the great number of congenital variations that occur in the smaller blood vessels, both arteries and veins. The student is referred, however, to the more extensive text-books of anatomy.
- Next: Muscular
References for Further Study
BORN, G.: Beitrage zur Entwicklungsgeschichte des Saugetierherzens. Archiv f. mik. Anat. Bd. XXXIII, 1899.
BREMER, J. L.: The Origin of the Renal Artery in Mammals and Its Anomalies. Am. Jour, of Anat., Vol. XVIII, 1915.
CLARK, E. R.: Further Observations on Living Growing Lymphatics; their Relation to Mesenchymal Cells. Am. Jour, of Anat., Vol. XIII 1911.
CLARKE, W. C.: Experimental Mesothelium. Anat. Record, Vol. VIII, 1914.
DANTSCHAKOFF, W.: Untersuchungen iiber die Entwicklung des Blutes und Bindegewebes bei den Vogeln. Anat. Hefte, Bd. XXXVII, 1908.
DANCHAKOFF, V.: Origin of the Blood Cells. Development of the Haematopoietic Organs and Regeneration of the Blood Cells from the Standpoint of the Monophyletic School. Anat. Record, Vol. X, No. 5, 1916.
DANCHAKOFF, VERA: Cell Potentialities and Differential Factors in Relation to Erythropoiesis. Am. Jour, of Anat., Vol. XXIV, 1918.
ETERNOD, A. C. F.: Premiers stades de la circulation sanguine dans 1'ceuf et embryon humain. Anat. Anz., Bd. XV, 1899.
EVANS, H. M.: On the Earliest Blood Vessels in the Anterior Limb Buds of Birds and their Relation to the Primary Subclavian Artery. Am. Jour, of Anat., Vol. IX, 1909.
His, W.: Anatomic menschlicher Embryonen. Leipzig, 1880-1885. With Atlas.
HOCHSTETTER, F.: Die Entwickelung des Blutgefasssystems. In Hertwig's Handbuch der vergleich. und experiment. Entwickelungslehre. Bd. Ill, Teil II, 1901. Contains also extensive bibliography.
HOWELL, W. H.: The Life History of the Formed Elements of the Blood, Especially the Red Blood-corpuscles. Journal of Morph., Vol. IV, 1890.
HUNTINGTON, G. S., and McCLURE, C. F. W.: Development of Postcava and Tributaries in the Domestic Cat. Am. Jour, of Anat., Vol. VI, 1907.
J HUNTINGTON, G. S.: The Phylogenetic Relations of the Lymphatic and Blood Vascular Systems in Vetebrates. Anat. Record, Vol. IV, 1910.
HUNTINGTON, G. S.: The Genetic Principles of the Development of the Systemic Lymphatic Vessels in the Mammalian Embryo. Anat. Record, Vol. IV, 1910.
HUNTINGTON, G. S.: The Development of the Lymphatic System in Reptiles. Anat. Record, Vol. V, 1911.
HUNTINGTON, G. S.: The Anatomy and Development of the Systemic Lymphatic Vessels in the Domestic Cat. Memoirs of the Wistar Institute of Anatomy and Biology, No. i, 1911.
HUNTINGTON, G. S.: The Development of the Mammalian Jugular Lymph Sac, of the Tributary Primitive Ulnar Lymphatic and the Thoracic Ducts from the Viewpoint of recent Investigations of Lymphatic Ontogeny, Am. Jour, of Anat., Vol. XVI, No. 3, 1914.
HUNTINGTON, GEORGE S.: The Morphology of the Pulmonary Artery in the Mammalia. Anat. Record, Vol. XVII, 1919.
KLING, C. A.: Studien iiber die Entwicklung der Lymphdriisen beim Menschen. Archvo f. mik. Anat., Ed. LXIII, 1904.
KOLLMANN, J.: Handatlas der Entwickelungsgeschichte des Menschen, Bd. II, 1907.
LEHMAN, H.: On the Embryonic History of the Aortic Arches in Mammals. Anat. Am., Bd. XXVI, 1905.
LEWIS, F. T.: The Development of the Vena Cava Inferior. Am. Jour, of Anat., Vol. I, 1902.
LEWIS, F. T.: The Development of the Veins in the Limbs of Rabbit Embryos. Am. Jour, of Anat. Vol. V, 1906.
MALL, F. P.: Development of the Internal Mammary and Deep Epigastric Arteries in Man. Johns Hopkins Hosp. Bull., 1898.
MALL, F. P.: On the Development of the Blood Vessels of the Brain in the Human Embryo. Am. Jour, of Anat., Vol. IV, 1905.
MALL, F. P.: On the Development of the Human Heart. Am. Jour, of Anat., Vol. XIII, 1912.
MAXIMOW, A.: Die Friihesten Entwicklungsstadien der Blut- und Bindegewebszellen beim Saugetierembryo, bis zum Anfang der Blutbildung in der Leber. Arch. f. mik. Anat., Bd. LXXIII, 1909.
MAXIMOW, A.: Lymphozyt als gemeinsame Stammzelle der verschiedenen Blutelemente in der embryonalen Entwicklung und im postfetalen Leber der Saugetiere. Folia Hdmatolog., Bd. VIII, 1909.
MAXIMOW, A.: Die embryonale Histogenese des Knochenmarks der Saugetiere. Arch.f. mik. Anat., Bd. LXXVI, 1910.
McCLURE, C. F. W.: The Development of the Lymphatic System in Fishes with Especial Reference to its Development in the Trout. Memoirs of the Wistar Institute of Anatomy and Biology, No. 4, 1915.
McCLURE, C. F. W., and SILVESTER, C. F.: A Comparative Study of theLymphatico- Venous Communications in Adult Mammals. Anat. Record, Vol. Ill, 1909.
MILLER, A. M.: Histogenesis and Morphogenesis of the Thoracic Duct in the Chick; Development of Blood Cells and their Passage to the Blood Stream via the Thoracic Duct. Am. Jour, of Anat., Vol. XV, 1913.
MINOT, C. S.: On a Hitherto Unrecognized Form of Blood Circulation without Capillaries in the Organs of Vertebrata. Proc. Boston Soc. Nat. Hist., Vol. XXIX, 1900.
REAGAN, F. P.: Experimental Studies on the Origin of Vascular Endothelium and of Erythrocytes. Am. Jour, of Anat., Vol. XXI, 1917.
ROSE, C.: Zur Entwickelungsgeschichte des Saugetierherzens. Morph. Jahrbuch, Bd. XV, 1889.
RUCKERT, J., and MOLLIER, S.: Die erste Entstehung der Gefasse und des Blutes bei Wirbeltiere. In Hertwig's Handbuch der vergleich und experiment. Entwickelungslehre, Bd. I, Teil I, 1906. Contains also extensive bibliography.
SABIN, F. R. : On the Origin of the Lymphatic System from the Veins and the Development of the Lymph Hearts and Thoracic Duct in the Pig. Am. Jour, of Anat., Vol. I, 1902.
SABIN, F. R.: The Origin and Development of the Lymphatic System. The Johns Hopkins Hospital Reports Monographs, New Series, No. 5, 1913.
SALA, L.: Svilluppo dei cuori linfatici e dei dotti toracici nelP embrione di polio. Ricerche fatte nel laboratorio de anatomia normale della R. Universita di Roma, Vol. VII, 1900.
SCAMMON, R. E., and NORRIS, E. H.: On the Time of the Post-natal Obliteration- of the Foetal Blood-passages (Foramen ovale, Ductus arteriosus, Ductus Venosus). Anat. Record, Vol. XV, 1918.
SCHULTE, H. VON W.: Early Stages of Vasculogenesis in the Cat (Felis domestica) with Especial Reference to the Mesenchymal Origin of Endothelium. Memoirs of the Wistar Institute of Anatomy and Biology, No. 3, 1914.
SCHULTE, H. VON W.: The Fusion of the Cardiac Anlages and the Formation of the Cardiac Loop in the Cat (Felis domestica). Am. Jour, of Anat., Vol. XX, 1916.
SENIOR, H. D.: The Development of the Arteries of the Human Lower Extremity. Am. Jour, of Anat., Vol. XXV, 1919.
STOCKARD, CHAS. R.: The Origin of Blood and Vascular Endothelium in Embryos without a Circulation of the Blood and in the Normal Embryo. Am. Jour.] of Anat. t Vol. XVIII, No. 2, 1915.
STOERK, O.: Uber die Chromreaktion der Glandula coccygea und die Beziehung dieser Druse zum Nervus sympthathicus. Arch. f. mik. Anat., Bd. LXIX, 1906.
STOHR, P.: Uber die Entwicklung der Darmlymphknotchen und uber die Riickbildung von Darmdriisen. Arch. f. mik. Anat., Bd. LI, 1898.
STREETER, GEORGE L.: The Development of the Venous Sinuses of the Dura Mater in the Human Embryo. Am. Jour, of Anat., Vol. XVIII, 1915.
TANDLER, J.: Zur Entwickelungsgeschichte der menschlichen Darmarterien. Anat. Heft, Bd. XXIII, 1903.
TONKOFF, W.: Die Entwickelung der Milz bei den Amnioten. Archil), f. mik. Anat., Bd. LVI, 1900.
WEIDENREICH, F.: Die Morphologic der Blutzellen und ihre Beziehungen zu einander. Anat. Record, Vol. IV, 1910.
WEST, R.: The Origin and Early Development of the Posterior Lymph Heart in the Chick. Am. Jour, of Anpt., Vol. XVII, 1915.
WRIGHT, J. H.: The Origin and Nature of the Blood Plates. Boston Med. and Surg. Jour., Vol. CLIV, 1906.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Text-Book of Embryology: Germ cells | Maturation | Fertilization | Amphioxus | Frog | Chick | Mammalian | External body form | Connective tissues and skeletal | Vascular | Muscular | Alimentary tube and organs | Respiratory | Coelom, Diaphragm and Mesenteries | Urogenital | Integumentary | Nervous System | Special Sense | Foetal Membranes | Teratogenesis | Figures
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 18) Embryology Book - Text-Book of Embryology 10. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Book_-_Text-Book_of_Embryology_10
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G