Book - Manual of Human Embryology 17-7
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Grosser O. Lewis FT. and McMurrich JP. The Development of the Digestive Tract and of the Organs of Respiration. (1912) chapter 17, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Liver
Early Development of the Entodermal Portion
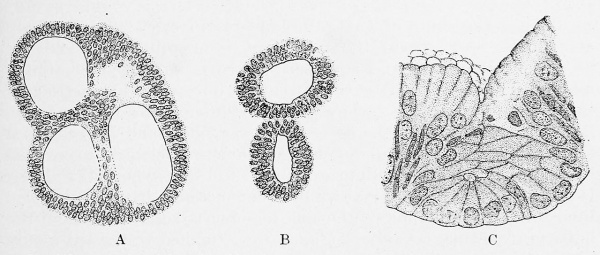
In the embryo of 2.5 mm which Thompson described (Fig. 236, p. 311), the liver is in a very early stage of development. It is a median ventral outgrowth of the entodermal tube, with thick walls which inclose a cavity in wide communication with the alimentary canal (Thompson, 1908). The latter presents a groove-like ventral border, but the liver appears as a well-defined cul-de-sac, the external form of which is shown in Fig. 288, A and B. Brachet (1896) and Swaen (1897) have described the liver as arising from a longitudinal groove.
In the slightly older Bremer embryo (4 mm), the median hepatic diverticulum is still present, but there has been an extensive proliferation of the cells in its anterior and ventral walls. The proliferating cells have formed irregular masses and anastomosing cords. In places the nuclei are peripheral, so that there is a lightly staining protoplasmic core, but no lumen is present in these outgrowths. Ventral and lateral views of a model of the liver in this embryo are shown in Fig. 288, C and D. (A small nodular outgrowth of the digestive tube, shown in the figure, and a similar structure beyond the lower limit of the model, are not connected with the liver.) The 4.9 mm embryo described by Ingalls (Fig. 239, p. 314) is considerably older. The anastomising cords of hepatic cells have formed a large crescentic mass, Fig. 289, A, with wings extending backward on either side of the intestinal tube. This mass connects with an outpocketing of the intestine, which corresponds with the original hepatic diverticulum. Distally the diverticulum shows a rounded subdivision, or outgrowth, which gives rise to the gall-bladder and cystic duct. Toward the intestine, in the angle between the diverticulum and the duodenum, the ventral pancreas has developed.
In an embryo of 7.5 mm the liver is much larger (Fig. 240, p. 316). The crescentic mass of anastomosing trabecular joins the elongated diverticulum by a short solid stem, the hepatic duct (Fig. 289, B). The diverticulum has become tubular, thus giving rise to the common bile-duct (ductus choledochus) . Distally the gall-bladder and cystic duct are represented by a cylindrical prolongation of the diverticulum, which is nearly solid.
Fig. 288. A and B, ventral and lateral views of the liver of an embryo with 23 pairs of somites (2.5 mm.), from a model by Peter Thompson. X 55 (?) diam. C and D, ventral and lateral views of the liver of a 4 mm. embryo, from a model by J. L. Bremer. X 135 diam. Div. hep., hepatic diverticulum; Tr., hepatic trabecular.
Before mammalian embryos had been satisfactorily studied, it was known that in the chick the liver arises from two outgrowths of the intestine, and Bischoff (1845), Remak (1855), and Kolliker (1861) believed that this would prove generally true for mammals. Although His (1SS5) found only single outgrowths in human embryos of 2-3 mm., Felix (1892) concluded that two are present. In addition to the " cranial hepatic duct " or diverticulum he found, in a single specimen, a " caudal groove " which he believed to be analogous with the posterior hepatic outgrowth in the chick. The embryo which he studied had been injured in the hepatic region. According to Felix, the caudal groove in man produces the gall-bladder and cystic duct, together with some of the hepatic trabeculae, and it fuses with the cranial portion. Swaen (1897), in an embryo of 3.8 mm., found the liver represented by a longitudinal groove, which fomis a cul-de-sac anteriorly and becomes gradually lower until it disappears posteriorly. Other writers have described the cul-de-sac, or hepatic diverticulum, as consisting of an anterior pars hepatica, which gives rise to the trabecular, and a posterior pars cystica, which produces the gall-bladder (cf. Hammar, 1897). Thompson, following Maurer (1906), applies these terms to portions of the diverticulum shown in Fig. 288, A and B. Geraudel (1907), without describing young embryos, concludes that the bile-ducts and the hepatic trabeculae are genetically independent; the former arise from the entoderm, and the latter from the mesoderm. This has been discussed and denied by Debeyre (1910). A bilaterally paired origin of the liver has been described in some vertebrates, but has not been found in man.
Certain investigators, following His (1885), recognize an early compact stage in the development of the liver, in which there are epithelial masses instead of anastomosing trabecular. It has been discussed whether the trabecular arise by epithelial outgrowths or by the breaking up of the solid masses through the invasion of mesenchymal tissue. The liver of the Bremer embryo is in the compact stage.
Fig. 289. — A, lateral view of the liver and pancreas of a 4.9 mm. embryo, from a model by N. W. Ingalls. X 65 diam. B, similar view of a model in which the hepatic trabecular are not included, from a 7.5 mm. embryo. Modelled by F. W. Thyng. X 50 diam. D. chol., common bile-duct ; D. cyst., cystic duct ; D. hep., hepatic duct ; Div. hep., hepatic diverticulum ; Ga., stomach ; Pane, d., dorsal pancreas ; Pane, v., ventral pancreas ; Tr., trabecular ; Ves. fel. (vesica fellea), gall-bladder ; x, aberrant duct.
Relation between the Entodermal Portion and the Blood vessels
Von Baer (1828) described the hepatic outgrowths in the chick as arising in close relation with the veins. Similarly Janosik (1887) observed that "in birds, through constant ramification, new outgrowths form, which grow into the lumen of the omphalomesenteric vein, pushing the endothelium before them." But in a young human embryo he failed to find such an intimate relation between the liver and the veins. Swaen, however (in 1897), states that in human embryos "the cavities of these veins have probably been invaded by the hepatic trabeculae and transformed into vascular ramifications and capillary networks." Bremer (1906) described the liver shown in Fig. 288, C and D, as follows: "The liver cords are found to be growing into mesenchyma at a level ventral to the vitelline (or omphalomesenteric) veins; in this same niesenchyma, however, we find branches of the veins ramifying and forming plexuses, and in certain places these plexuses come into intimate relation with the liver cords." With further growth the places of contact rapidly increase. As the trabecule ramify, new branches of the venous plexus extend between and around them, so that the cords of liver-cells are closely invested with endothelium. The process, therefore, is not a simple invasion of the lumen of the veins by the trabecular As the right and left wings of the liver extend dorsally, they encounter respectively the main trunks of the right and left omphalomesenteric veins, which are passing from the intestine to the heart. The hepatic trabecular surround these veins. The left omphalomesenteric vein soon loses its identity within the hepatic plexus, but the right vein remains as a distinct channel. These relations are shown in the diagram, Fig. 290, A, from an embryo similar to Ingalls' specimen. (For a reconstruction of these veins, see Ingalls, 1908.) The further development of the large hepatic vessels may be considered briefly, since details are supplied in Chapter XVIII. In an embryo of 6.5 mm., Fig. 290, B, the two omphalomesenteric veins have produced a single vessel, which winds behind the intestine to enter the liver, the entering portion being a persistent part of the right vein. This is the portal vein of later stages. Within the liver the right omphalomesenteric vein can be followed continuously, and the left through a plexiform network, to the superior surface. Here a single vessel, the hepatic vein, conveys the blood to the heart. The hepatic vein is essentially the persistent outlet of the right omphalomesenteric vein.
Fig. 290. — Semi-diagrammatic reconstructions of the veins of the liver (ventral views). (After Mall. A, embryo of 4.5 mm.; B, 6.5 mm.; C, 7 mm. d. v., ductus venosus; H, liver; Int., intestine; r. u., recessus umbilicalis; v. h., hepatic vein; v. o-m. d., v. o-m. s., right and left omphalomesenteric veins; v. p., portal vein; v. u., umbilical vein; v. u. d., v. u. s., right and left umbilical veins.
Thus it will be seen that by intercrescence with the hepatic trabecule, the original omphalomesenteric veins have been resolved -into an afferent portal vein, which empties into a network of branches, and an efferent hepatic vein, which drains these branches. This purely venous type of circulation has long been described as a portal circulation. (In the liver it is the hepatic portal system; in the Wolffian body, the renal portal, etc.) It has also been described as a sinusoidal circulation (Minot, 1900; Lewis, 1904).
In addition to the blood from the intestines, received through the portal vein, the liver very early receives blood from the placenta, through the left umbilical vein. The pair of umbilical veins at first pass to the heart without entering the liver, but in some 4 mm. embryos the left umbilical vein already sends out branches which join the hepatic plexus. In the 6.5 mm. embryo, Fig. 290, B, one of these branches has become the chief outlet for the placental blood. At first the umbilical vein merges in the general plexus, but later it forms a large channel across the inferior portion of the liver (Fig. 290, C). Although it is a left vein, it gradually moves toward the median line, and the gall-bladder, which is morphologically median, is found on the right.
After birth, the portion of the umbilical vein extending from the umbilicus to the liver becomes reduced to a fibrous cord, — the round ligament (lig. teres hepatis). In the adult, a large branch of the portal vein within the liver extends toward this ligament and ends blindly. Rex (1888) has described this blind ending, as an appendage of the left branch of the portal vein, and named it the recessus umbilicalis. Mall (1906) has applied this term to a portion of the embryonic vessels, as indicated in Fig. 290, C. The vessel which appears as a continuation of the umbilical vein, passing from the portal to the hepatic (in later stages to the vena cava inferior), is the ductus venosus. Since the ductus venosus and the umbilical vein appear on the under surface of the liver, they will be further described with the surface markings.
As stated by Toldt and Zuckerkandl (1875), the capillaries of the liver in early embryonic stages are considerably wider than later, both absolutely and as compared with the glandular parts which they surround. Minot (1892), in describing the embryonic mammalian liver, noted that the "blood-channels are very large," and Schenk (1896) referred to the "lacunar vascularization" in the liver of batrachians. In 1900 Minot proposed the term sinusoids, in distinction from capillaries, for wide endothelial tubes fitted closely against the cells of the organ in which they are developed; those in the adult liver, which have become narrower, he distinguished as capUliform sinusoids. The intimate relation between the hepatic cells and the endothelium has lon<>' been known. Thus Hering (1871) recorded that "the secreting cells of the liver exhibit a peculiar arrangement, whereby there exists a much closer and more extensive contact between them and the capillaries than in other glands." In describing this relation in the embryo, the perivascular tissue will be considered first, and then the cells which occur within it.
Perivascular Tissue
The gross relation between the bloodvessels and the hepatic trabeculae in a 9.4 mm. embryo is shown in the model, Fig. 308, p. 432. Along the upper surface of the model the liver is seen in transverse section. The histological features of such a section are shown in Fig. 291. In many places the endothelium has become separated from the hepatic trabeculae, thus producing a perivascular space. The space is bridged by slender protoplasmic processes of the endothelial cells. These processes, together with the peripheral protoplasm of the endothelial cells, are directly transformed into connective-tissue fibres of a peculiar sort. Kon (1908) observed the transformation in embryos of four and five months, but Mollier (1909) declares that it begins much earlier, since the fibres are clearly present at 30 mm. 15 As compared with the adult, Maresch (1905) finds that the fibres in embryonic livers are poorly developed, and that "not until birth can an abundant supporting tissue be demonstrated." The nature of the delicate felt-work of perivascular fibres found in the adult has been discussed by Mall (1896) as follows: " Kupffer considers them to be elastic, while Ewald and Kiihne consider them white fibrous. The fact that they are digested by pancreatin and yield but little gelatine when boiled excludes both views; and, since they seem to be identical with the reticulum of lymphatic glands, spleen, and mucous membrane, I shall retain for them the name reticulum." Usually they are regarded as delicate strands of white fibrous tissue. Apart from the nuclei of the endothelium, no nuclei are found in relation with the fibres, as noted by His (1860) and abundantly confirmed.
Fig. 291. — Section of the liver of a 7.5 mm. embryo (Harvard Collection, Series 256). X 120 diam. BZ., blood-forming cells; C. hep., cells of the hepatic trabeculae; Corp., nucleated blood-corpuscles; Endo., endothelium; V., branch of the portal vein.
- Mall found the network of fibrils in a pig embryo of 20 mm. Amer. Journ. of Anat., vol i, p. 354, 1902.
Perivascular spaces, bounded on one side by the capillary wall and on the other directly by hepatic cells, with here and there a connective-tissue fibre stretched across, were described by Biesiadecki in 1867. He concluded that these spaces were preformed and not due to imperfect fixation. Hering (1871) recognized that, although the spaces are increased by shrinkage, they indicate a structural peculiarity. MacGillavry (1864) had injected these spaces through the lymphatic vessels, and also through the bileducts, but they do not normally open into either. They are interfibrillar tissue spaces, and are quite distinct from lymphatic vessels.
Blood-forming Cells
In the liver of the 7.5 mm. embryo, in addition to the flattened nuclei of the endothelium and the round, coarsely granular nuclei of the hepatic trabecular, there is a third group consisting of small, darkly staining, and densely granular forms, occasionally somewhat indented, situated between the endothelium and the trabecular (Fig. 291). Frequently they occur in groups. The protoplasm surrounding them, in this specimen, is scarcely perceptible.
Toldt and Zuekerkandl (1875) designated these as round cells, in contrast with the cuboidal cells of the trabecule, and described them as follows : " These cells are distinctly round, variable in size, sharply outlined, very finery granular, and clear; they never contain fat droplets, even when such are present in considerable quantity in the cuboidal cells, nor do pigment granules occur in them. The relatively large nucleus is generally distinctly outlined, yet not with so refractive a contour as in the cuboidal cells. It is of quite homogeneous consistency. Nucleoli are almost always visible, even two or three in a nucleus." These cells were observed in a 10 weeks embryo and in all the older ones examined. " Toward the end of embryonic life the number of the round cells, as compared with the cuboidal cells, strikingly decreases, but they are still present at birth, situated either singly in the wall of the gland tubes, or in groups by themselves, surrounded only by a capillary mesh." " After birth their number diminishes very rapidly, and even in the first weeks they seem to disappear entirely." Toldt and Zuekerkandl mistook the round cells for young hepatic cells. The fact that they were not dislodged from the liver after the veins had been thoroughly washed out with salt solution confirmed their opinion that they were not the developing blood-corpuscles described by Kolliker. Kolliker (1846), from a study of one human and several sheep embryos, had concluded that ' ' as the liver develops, the multiplication of corpuscles ceases elsewhere in the blood, and in its place, probably because all of the blood of the umbilical vein now flows into the liver, an aotive formation of blood-cells occurs in the hepatic vessels." Kolliker 's communication is followed by a letter from E. H. Weber, who states that in the liver of the frog the corpuscles develop outside of the vessels, to which they may gain entrance by local absorption of the vessel wall. In 1874 Neumann described the corpuscles as occurring in nests between the hepatic cells in human embryos of 8-9 months, and he thought that they arose endogenously in the protoplasm of certain elongated cells (presumably endothelial). Schmidt (1892) states that they arise through karyokinetic division of the endothelial cells and multiply by mitosis. Van der Stricht (1892) concluded that the cells described by Toldt and Zuckerkandl were young red blood-corpuscles. He observed that the capillary wall becomes discontinuous at the places where these cells appear to be imbedded in the trabecule, and therefore he considered that they came from the circulating blood.
In the perivascular spaces of the embryonic liver, not only are red corpuscles produced, but also leucocytes and giant cells such as are characteristic of red bone marrow. They are described in connection with the blood (Chapter XVIII). In early stages these cells are abundant, but, according to Nattan-Larrier (1904), after the fifth month giant cells and basophilic myelocytes are very rare, and at birth the nucleated red corpuscles remain almost exclusively. Lobenhoffer (1908) states that as blood formation in the liver diminishes, the capillary recesses become fewer and smaller, disappearing in the eighth month, but at birth, in almost every field, he found one or two blood-forming groups between the hepatic cells. He agrees with Schmidt that "the cells of the capillaries are capable of forming blood elements." It is probable that the primitive blood-cells seen in the liver of the 7.5 mm. embryo are derived from the endothelium, but their possible origin from cells of the circulating blood must be considered (see Chapter XVIII).
In the adult, as shown by Kupffer (1876 and 1899), the endothelium is so perforated that its cells have become stellate. Mollier (1909) considers that the stellate condition is associated with blood formation and is most highly developed in the embryo. He believes that the endothelial cells and the blood-corpuscles both arise from a reticular syncytium, and, as the formation of corpuscles ceases, the syncytium becomes a closed endothelium with perivascular fibres. This change occurs first along the capillaries which are to become the main branches of the portal vein.
The Gall-bladder, Ductus Cysticus and Ductus Choledochus
The solid stage of the gall bladder, which occurs regularly in young human embryos, is presumably acquired with the elongation of the round diverticulum, such as is found at 4.9 mm. (Fig. 289, A). Both the gall-bladder and the common bile-duct have been recorded as solid in an embryo of 6.8 mm. (Piper, 1900) and in another of 6.75 mm. (Keibel and Elze, 1908). These are the youngest stages in which the solid condition has been observed. At 7.5 mm. (Fig. 289, B) there is a lumen in the common bile-duct, but the gall-bladder is impervious. Near the hepatic duct the lumen is subdivded, appearing in cross sections as two or three minute pores. At 16 mm. there are irregular subdivisions near the hepatic duct, and the distal part of the gall-bladder is solid, but in both the cystic duct and the common bile duct the lumen is single and well defined. Occasionally the common bile-duct has a double lumen in the midst of its course, as was noted at 14.5 and at 22.8 mm. (Fig. 292, B). In these cases the two cavities unite in a single lumen both proximally and distally.
As the gall-bladder expands it may present "intra-epithelial cvsts," as recorded by Keibel and Elze at 18 mm, or the lumen may be bridged by epithelial strands, as in Fig. 292, A (29 mm.). At this stage the greater part of the gall-bladder has a clear-cut round lumen. Its wall shows two rows of oval nuclei, with mitotic figures in the inner row (22.8 mm.). In a 42 mm. embryo the tapering proximal part of the gall-bladder presents several rounded outpocketings, resembling the intestinal diverticula, sometimes sectioned so as to appear detached from the main tube. Similar pockets are apparently more definite and abundant in the sheep and pig than in man. The lining of the gall-bladder in a 78 mm. specimen shows numerous well-defined folds, such as are characteristic of all later stages. The development of the folds in the cystic duct, constituting the spiral valve, has apparently not been studied.
Fig. 292. A section of the gall-bladder of a 29 mm embryo (Harvard Collection, Series 914). X 180 diam. B section of the common bile-duct of a 22.8 mm embryo (Harvard Collection, Series 871). X 180 diam. C epithelium of the gall-bladder, two weeks after birth. X 580 diam.
The epithelium of the gall-bladder of a child two weeks old, born prematurely at the seventh month, is simple and columnar, with distinct cell walls ending in terminal bars (Fig. 292. C). A broad clear border, or top plate, with radial striation, such as Virchow (1857) described in the gall-bladder of adults and children, could not be detected. The cells contain oval. pale, vesicular nuclei, together with more elongated and darkly staining forms, apparently due to compression. The dark nuclei may be scattered among the others or may form considerable gronps. Occasionally at the bottom of the depressions between the folds, a pearl-like group of cells is seen (Fig. 292, C), suggesting the buds of the intestine.
The development of the " glands " of the bile-ducts has not been studied einbryoiogically. These structures are generally considered to be epithelial pockets rather than true mucous glands. They formerly attracted much attention, culminating in a thorough study of them by Riess in 1863, for it had been supposed by Henle (1861) that these glands were the source of bile, and that the hepatic trabecule produced sugar. Riess states that they are most numerous in the hepatic duct, less numerous in the upper part of the common bile-duct and lower part of the cystic duct, and entirely absent from the lower part of the common bile-duct and upper part of the cystic duet; probably there are none in the gall-bladder. The largest of them are branched tubes with rounded terminations; the small ones are simple pockets, which give place, in the smaller branches of the hepatic duct, to rounded diverticula and swellings. Riess has noted that the glandular appendages of the bile-ducts are much less developed in children than in adults, and " in the earlier embryonic life they are perhaps wholly lacking." The outer coats of the gall-bladder and cystic duct develop as follows : At 7.5 mm the epithelium is surrounded by a layer of mesenchyma, and the entire structure is so imbedded in the under surface of the liver that it causes only a slight swelling of the peritoneal surface. Above and on the sides the mesenchyma is in direct relation with the hepatic trabecular, and it receives a few prolongations of the venous capillaries. Below it is covered by the peritoneal epithelium except on the left, where that layer is reflected to the abdominal walls in connection with the falciform ligament. In later stages the gall-bladder is separated from the hepatic trabecular on either side, and is attached to the liver only along its upper surface.
At 16 mm the mesenchyma surrounding the gall-bladder is still undifferentiated, but at 22.8 mm it forms two broad concentric zones, of which the inner is darker and more compact than the outer. At 29 mm. certain cells in the peripheral part of the dark zone form a third layer, which is thin and somewhat interrupted. As seen in later stages these cells are myoblasts, so that at 29 mm all three layers of the adult gall-bladder are indicated. These are the mucosa, muscularis, and serosa. The layers become gradually less distinct toward the hepatic duct.
The vessels and nerves of the gall-bladder are branches of those seen at 10 mm. near the pyloric end of the stomach (Fig. 274, C, p. 370). Of these the hepatic artery is of special interest.
The Hepatic Artery
At 10 mm the hepatic branch of the coeliac artery can be followed to the hepatic duct. Later it extends along the hepatic and cystic ducts, but as the cystic branches develop first, the hepatic artery appears primarily as the artery of the gall-bladder. Thus, at 22.8 mm, the main stem lies in a wing-like fold of the tunica serosa of the gall-bladder, and other branches follow the attached border of the gall-bladder, lying close to the hepatic trabecular They connect with a capillary plexus in the mesenchyina, which empties at various points into the venous network among the adjacent trabecular The cystic vein of the adult, which conveys the blood from the gall-bladder to the main trunk of the portal vein, is a later formation. Therefore three stages may be recognized in the development of the blood-vessels of the gall-bladder: in the first, the capillaries from the portal network extend into the mesenchyma around the gall-bladder; in the second, they are joined by the arterial capillaries and become efferent vessels; in the third, a single efferent vein, extending along the cystic duct and emptying into the portal trunk, is developed from the capillary system.
After the cystic branch of the hepatic artery has become established, mesenchyma develops around the hepatic duct and its ramifications, and branches of the hepatic artery appear in this mesenchyma. They form capillary plexuses, especially around the branches of the hepatic duct, and the blood passes from these capillaries into the subdivisions of the portal vein found among the adjacent trabecular veins comparable with the cystic vein, which collect the blood from the arterial capillaries and convey it to the main branches of the portal vein, have been described within the liver of the adult, but according to Mall (1906) they do not exist.
Certain branches of the hepatic artery reach the surface of the liver and ramify in the capsule. They either empty into the portal network beneath the capsule, or are drained by " branches of the hepatic vein which come to the surface of the liver and spread out between the meshes of the arterial plexus " (Mall).
The Hepatic Duct
At 9.4 mm. the hepatic duct is a short stem connecting the great mass of hepatic trabecular with the common bile-duct (Fig. 308, p. 432). It is solid, or nearly so, in this specimen, but in a 10 mm. embryo it contains a lumen. The nuclei are crowded so that the duct stains deeply and contrasts sharply with the trabecular Where it joins the trabecular the transition is so abrupt that it has led to the erroneous opinion that the two tissues are of different origin.
Although the hepatic duct in man is a single stem, there are certain mammals in which there are several ducts which pass from the trabecular to the cystic duct, or, in some species, to the gallbladder (cf. Rex, 1888). Rudimentary additional ducts are common in human embryos. They may join the hepatic, cystic, or common bile-duct, but usually they occur very near the junction of the three. Thus, at 7.5 mm. (Fig. 289, B), on either side at this junction, there is a solid knob which does not quite reach the trabecular In the same position there is a single outgrowth in the 9.4 mm. embryo and also in the 10 mm. specimen. An aberrant duct with a lumen empties into the proximal end of the cystic duct at 14.5 mm, and the same embryo shows a detached nodule of epithelium beside the common bile-duct. A detached nodule containing a lumen was found at 16 mm These structures may represent outgrowths of the original diverticulum which have contributed to the formation of the mass of trabecule and are now degenerating, or they may be abortive secondary ducts which have never reached the trabecular.
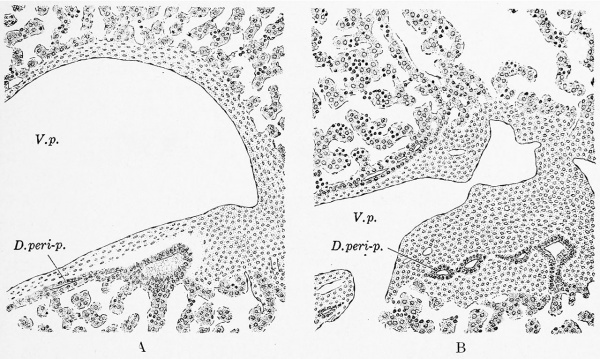
Within the liver of 9-10 mm. embryos the branches of the hepatic duct usually cannot be traced far, but there is marked variation in this respect. Frequently among the hepatic trabecular one or more very short ducts may be found which certainly do not connect with other ducts. They may blend with the hepatic trabecular at one or both ends. These detached ducts are lined with regular cuboidal or columnar epithelium and may show a clear-cut lumen. Such ducts were noted in embryos measuring 8, 9.4, and 10.2 mm., and, according to Elze (1907), in those of 7 and 11 mm. Lewis (1903) found similar detached cysts in the liver of a 12 mm. pig, and considered them to be cut off from the secondary hepatic ducts. Whether they are detached portions of the hepatic ducts is questionable. They may arise in situ by a transformation of the cells of the hepatic trabecular The Periportal Ducts.— In an embryo of 22.8 mm. (Fig. 293, A) the spread of the bile-ducts along the main branches of the portal vein has begun. The trabecular form cords extending along the surface of the periportal mesenchyma, and in them a lumen is formed. In places the cells on the mesenchymal side of the lumen are distinctly flatter than those toward the portal capillaries. As seen in the figure, the trabecular connect freely with these ducts. In a later stage (29 mm, Fig. 293, B) the mesenchyma has increased, so that it surrounds the ducts which were seen forming along its surface. Their epithelium has become regularly cuboidal or columnar. On the upper side of the vein in Fig. 293, B, the ducts are in the earlier stage of development.
The periportal ducts clearly form a plexus. The larger ducts, which have become surrounded by mesenchyma, are also plexiform, although with the enlargement of the liver their anastomoses become less numerous. However, the plexiform arrangement of the main branches of the hepatic duct, which was clearly seen in a single frontal section at 29 mm., persists throughout life, as has long been known.
In the adult, Kiernan (1833) found that an injection of the left branch of the hepatic duct returns by the right duct. "From this experiment ... it appears that the right and left ducts anastomose with each other." Hering (1871) stated that there is an anastomosis of the branches of the large ducts, and that the smallest ones sometimes appear to anastomose around the vein which they accompany (as stated by Riess), "but this requires further investigation." He described the connection between these ducts and the trabecule? as formed by canals "bounded on one side by small epithelial cells and on the other by large hepatic cells." These transitional ducts, sometimes called the "canals of Hering," were observed "in a child of 3 months.
Toldt and Zuekerkandl (1875) described them in an embryo of the tenth week as follows:
- "Those hepatic trabecular which are found in the immediate vicinity of the relatively very large portal branches, almost without exception are perpendicular to the latter, so that in cross sections of a portal branch they show a radial arrangement, and in longitudinal sections they appear in parallel rows. They open into the ducts almost at right angles, and their cuboidal cells are inserted directly into the flat epithelium of the ducts."
The Bile-Capillaries
In an embryo of the fourth week Kolliker (1861) found the liver composed of solid trabecular Remak (1855) had observed a similar condition in chick embryos, and had named the solid cords of cells "hepatic cylinders." J'liisalix(1888), in describing a 10 mm embryo, states that he agrees with Kolliker that there is no lumen in the primitive hepatic cylinders. This view is generally accepted. But Toldt and Zuekerkandl (1875) described the liver of an embryo of the fourth week, in which the tubular structure appears most distinctly. "That we have to do with tubes and not with solid cords of cells can be shown both in cross sections and frequently in longitudinal sections; the lumen is always bounded by so sharp an outline that there is no question of an artificial separation of the cells. ' ' They find that in the slender tubes the lumen is bounded by three or four cells, in the larger tubes by still more.
Fig. 293. Sections showing the formation of the periportal ducts (D.peri-p.) around a branch of the portal vein (V.p.). A, from an embryo of 22.8 mm (Harvard Collection, Series 871 ); B, from an embryo of 32 mm (Harvard Collection, Series 913). X 185 diam.
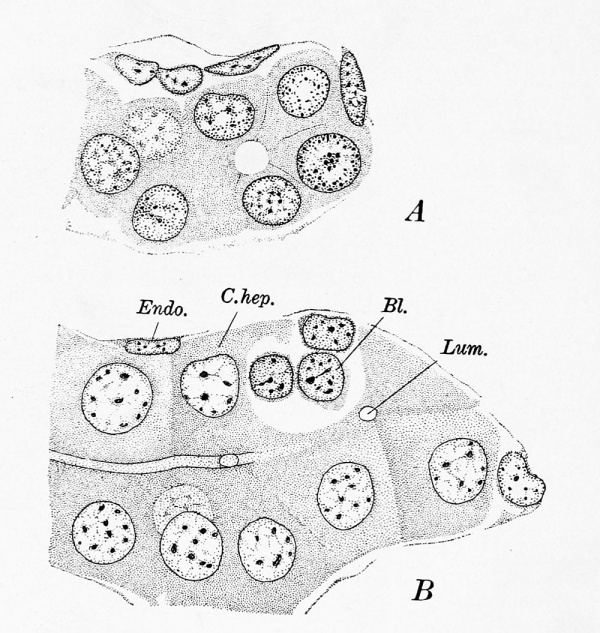
Although in a 10 mm. embryo most of the trabecular are solid, there are some, scattered irregularly through the liver, which show a very distinct lumen (Fig. 294, A). The lumen is larger than that of the future bile-capillary and lacks the characteristic cuticular border. It is bounded usually by five or six cells and ends blindly in the adjoining sections. At 29 mm. the trabecular are extensively broken up by the nests of blood-cells, but those which are least disturbed often show a lumen. At 37 mm. the lumen is more distinctly outlined, but it may still be bounded by as many as six cells. In a 44 mm embryo, bile-capillaries with cuticular borders are abundant (Fig. 294, B). They occur in all parts of the liver, but appear to be most numerous near the periportal ducts, into which some of them empty. They extend axially through the trabecula?, and where the latter branch, the capillaries branch- also. Usually they are separated from the venous endothelium by an entire hepatic cell, but, as seen in the figure, nests of blood-cells sometimes approach very close to them.
Fig. 294. A hepatic trabecula containing a lumen. From a 10 mm. embryo (Harvard Collection, Series 1000). X 1065 diam. B, bile-capillaries in a 44.3 mm embryo (Harvard Collection, Series 1611). x 1065 diam. Bl., blood-corpuscle; C.hep., hepatic cell; Endo., endothelium ; Lum., lumen of a bilecapillary.
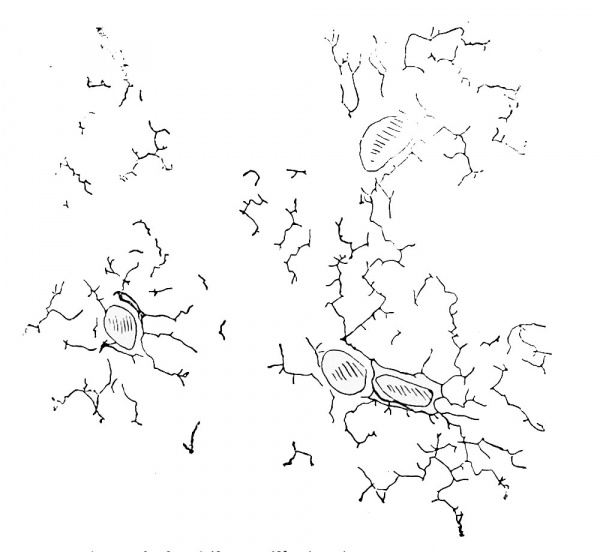
Hendrickson (1898) studied the bile-capillaries with Golgi's method, and found them extensively developed at 50 mm. (Fig. 295). In his preparations they are somewhat more abundant around the branches of the portal vein, but it could not be shown that they develop peripherally from the periportal ducts. They may arise within the trabecule as blind tubes, which later anastomose and join the ducts. In a Golgi preparation from an embryo of 100 mm., Hendrickson found numerous polygona] meshes made by the bile-capillaries. These may encircle a single cell within a thick trabecula, or they may run through trabecular which anastomose at both ends. Short lateral branches are seen in Hendrickson's preparations, but whether they represent axial capillaries, either ending blindly or passing out of the plane of section, or whether they are intercellular branches radiating from the axial capillary toward the venous endothelium, was not determined.
Fig. 295. Golgi preparation of the bile-capillaries in an embryo of 50 mm. x 53 diam. (After Hendrickson.)
The number of hepatic cells bounding a bile-capillary is greater in embryos than in the adult. The capillary of the adult is usually a minute canal in the midst of the boundary between two adjacent cells, but occasionally it is surrounded by three or four cells ; and von Biesiadecki, the discoverer of the human bile-capillaries, declared that five is the usual number, — rarely four. Von Biesiadecki studied only pathological specimens in which the bilecapillaries were distended. Hering (1871) was unable to find any capillaries bounded by five or more cells, either in the adult or at birth. At birth, however, in contrast with the adult, he found that the lumen of a bile-capillary is often surrounded by three or four hepatic cells, thus resembling the condition in certain amphibia. Toldt and Zuckerkandl (1875), who found three or four cells bounding the capillaries in an embryo of the fourth week, stated that in embryos of four to seven months the number was "four to six or even more," but they included in their count the blood-cells lodged within the hepatic trabecular Toward the end of fetal life they found usually three or four cells ; and in the fourth and fifth years, "generally only two, but occasionally three and even four." The reduction from three or four to two cells, occurring shortly after birth, is accompanied, according to Toldt and Zuckerkandl, by a "stretching" of the trabeculae, whereby they become longer and more slender, with their cells arranged in rows. They recognize the occasional persistence of three- and four-celled tubes, even after twenty years.
The Hepatic Cells
In all stages the hepatic cells are characterized by large, very round nuclei, containing a coarse chromatic network, and surrounded by abundant, densely granular protoplasm. According to Toldt and Zuckerkandl, the diameter of the nucleus at birth is generally about 9.6 /* and in the adult about 8 //<, so that the nuclei of the adult are distinctly smaller than at birth. As a whole, however, the cells apparently increase in size. They multiply by mitosis. In a 10 mm. embryo the mitotic figures are abundant throughout the trabeculae, but in later stages, according to Mall (1906), they are particularly numerous around the terminal (periportal) bile-ducts. Frequently the cell division is incomplete, so that a single cell may contain two nuclei. In early stages cell membranes are entirely lacking, and at 10 mm., although the cells readily separate from one another, the membranes are indistinct. Toldt and Zuckerkandl have noted that at five and six months the cells do not show the sharp outlines observed at birth. They find that at birth a portion of the cells, when isolated in salt solution, are irregularly cuboidal, but that most of them are somewhat elongated. The isolated cells of the adult vary in shape, but are more nearly cuboidal than at birth.
The hepatic cells always stand in close relation to the blood which contains absorbed nutriment. First the blood from the yolk-sac, then that from the placenta, and after birth the blood from the intestine passes directly to the liver and flows through its vascular network. Kolliker (1861) noted that the liver in the embryo is physiologically a very important organ, but that its significance is rather in producing chemical and morphological changes in the blood than in secreting bile. Fat appears in the hepatic cells before it is present in the subcutaneous tissue, as shown by Chipman (1902) for the rabbit. In the liver of human embryos Toldt and Zuckerkandl found fat droplets as early as the third or fourth month. Nattan-Larrier (1903) found that fat was forming in the fourth month, and that at birth certain hepatic cells were filled with large fat droplets, separated from one another by thin layers of protoplasm. In the liver of the rabbit, glycogen appears in the 22d day of gestation, six days after the formation of fat, and it increases steadily and rapidly until birth ( ( Jhipman). Apparently the time of its appearance in human embryos has not been determined.
Kolliker found that the secretion of bile begins as early as the third month. He states that "from the third to the fifth month, material like bile is found in the small intestine, and in the second half of pregnancy it occurs also in the large intestine. . . . Until the sixth month the gall-bladder contains only mucus, but after that it contains bile." Zweifel" (1875) recorded that the intestinal contents of embryos of three months respond to the ordinary tests for bile acids and pigments. Toldt and Zuckerkandl observed yellow pigment granules within the embryonic liver beginning with the fourth or fifth mouth, but the granules were limited to the epithelial cells of the ducts, together with the adjacent hepatic cells. Even at birth they found that pigment granules are infrequent, and that the cells are clearer and more transparent than those of the adult.
Nerves and Lymphatics
It has already been shown that the common bile-duct, cystic duct, and hepatic duct are very early surrounded by mesenchyma. Later the mesenchyma spreads along the ramifications of the portal vein, into the substance of the liver. Thus, in an embryo of the tenth week Toldt and Zuckerkandl found that the portal branches are surrounded by a considerable mass of connective tissue, and so can easily be distinguished from the thin-walled branches of the hepatic vein which are closely surrounded by the liver-cells. In the third and fourth month the difference becomes very striking. Ducts have formed at the periphery of the periportal tissue, and branches of the hepatic artery together with nerves and lymphatics have extended into it. The path by which the vessels and nerves reach the liver is shown in Fig. 274, B and C (p. 370) ; they enter at the transverse fissure or porta of the liver.
Little is known regarding the development of the nerves. Sympathetic fibres may readily be found at the entrance of the liver in embryos of 20-40 mm., but in the specimens at hand they can be traced no further than the primary division of the hepatic duct. They are associated with scattered clumps of nuclei, apparently ganglionic. In the adult the nerves form plexuses around the branches of the portal vein and hepatic ducts, and especially around the branches of the hepatic artery. In addition to the sympathetic nerves, there are fibres from the vagus, presumably entering from the pylorus.
The lymphatic vessels, which extend to the porta in a 42 mm. embryo, drain into the lower part of the thoracic duct. Later they grow into the periportal tissue, in which at birth they are conspicuously large. In the adult they extend as far as the smallest ramifications of the hepatic ducts, and terminate in the connective tissue. Herring and Simpson (1906) find that the lymphatic vessels accompany the hepatic artery and its branches, forming networks around these vessels, as well as around the branches of the portal vein and bile-ducts. There are no lymphatics among the trabecular Mall (1906) concludes that the great amount of lymph which flows from the liver is derived directly from the blood-plasma. It passes out between the stellate and endothelial cells, and flows through the perivascular reticulum to the periportal tissue, where it enters the lymphatic vessels.
There is another system of lymphatic vessels in the liver, which has not been studied embryologically. This includes the vessels which extend downward from the diaphragm, through the ligaments of the liver, to ramify in the capsule.
Lobules
The great mass of hepatic trabecular is arranged in more or less definite lobules, which were discovered by Wepfer in the liver of the pig in 1664, and which have been familiar to anatomists since the time of Malpighi (see Kiernan, 1833). The liver was compared with a bunch of grapes. According to some anatomists its lobules were appended to the extremities of the portal vein, but Kiernan agreed with those who made the hepatic vein the axial structure. In the centre of each lobule he recognized a terminal branch of the hepatic vein. Between the lobules there are intervals, which Kiernan named portal canals, filled with connective tissue containing branches of the portal vein, hepatic artery, bile-ducts, nerves, and lymphatics. Three portal canals nia}^ be expected at the periphery of a single lobule. In the pig the connective tissue filling these canals spreads around the lobules, investing them with capsules, and thus making them conspicuous. In the human adult there are normally only indications of such capsules, and at birth they are wholly lacking. The portal canals then stand as isolated "boundary stones." Kiernan recognized certain objections to describing the liver on the basis of these lobules, for he wrote, "The essential part of a gland is undoubtedly its duct; vessels it possesses in common with every other organ ; and it may be thought that in the above description too much importance is attached to the hepatic veins." Recognizing this, Brissaud and Sabourin (1888) proposed the term biliary or portal lobule for the group of trabecule centred about a portal canal, leaving hepatic lobule for the structures described by Kiernan. Others also have considered that the portal lobule is morphologically the true unit of the liver.
As stated by Mall, " In all other glands we make the duet the centre of the structural unit. From this centre often the artery and the framework radiate. In the liver everything radiates from the so-called interlobular space, — arterial and portal blood-vessels, bile-duct, lymphatics, nerves, and connective tissue. . . . Throughout ruy description I shall use (he term portal unit, structural unit, or unit for the clump of tissue which surrounds each terminal branch of the portal vein. In order to avoid confusion, I shall use the term lobule in its old sense, — as the hepatic lobule, — for after much discussion carried on during two centuries, it has become well established." Sclienk (1874) stated that the embryonic liver, in a certain stage, represents a single lobule of the adult organ. This view was adopted by Toldt and Zuckerkandl, and defined by Mall (1906). Mall states that in a 4 mm embryo "the single lobule is perfect; it is composed of a complete capillary network without an anastomosing vein through it."
Mall believes that the further development of the hepatic vessels is in accordance with the laws established by Thoma (1S93). These are:
- An acceleration of the current leads to an enlargement of the lumen of a vessel, and a slowing of the current leads to its narrowing and final disappearance.
- An increase in the blood-pressure is the cause for new formation of capillaries.
- The growth in thickness of the vessel wall depends on the tension of the wall, which in turn is dependent upon the blood-pressure and the diameter of the vessel.
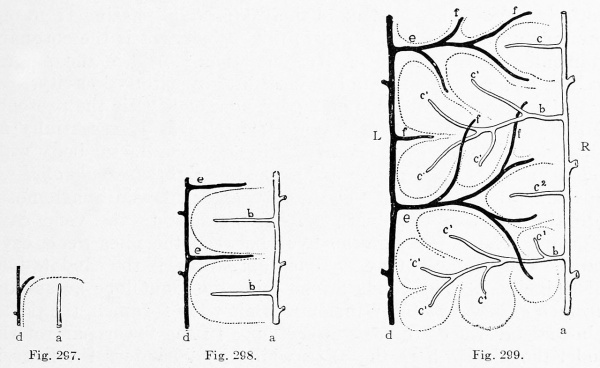
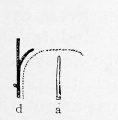
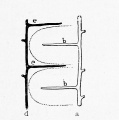
These laws apply to the liver, as shown in Fig. 290, B and C. In C a new and direct channel, the ductus venosus, has been formed across the liver, apparently by the enlargement of capillaries in which the current has been accelerated. At the same time the current becomes slower in the circuitous right omphalomesenteric vein, shown in B, which is reduced to capillaries in C. The liver then consists of two lobules, right and left respectively. The blood enters them from below, and is drained by the hepatic branches above. In an embryo of 11 mm. (Fig. 296) Mall finds that six lobules are indicated. These are obscure in the figure, since many of the enlarging capillary vessels have been drawn. It will be observed that the portal branches tend to alternate with the hepatic branches. "They are beginning to dovetail with each other." Thus the formation of a great number of lobules is suggested. The way in which this is accomplished is shown in Mall's diagrams, Figs. 297, 298, and 299. The single branches d and a in Fig. 297 become the main branches d and a in Fig. 299, and the successive orders of new branches e, f and b, c have arisen by the enlargement of capillary vessels. The lobules b, indicated by dotted outlines in Fig. 298, have become clusters of lobules in Fig. 299, and some new simple lobules, c 2 , have appeared between them.
In this way the 480,000 lobules, which according to Mall's estimate are found in the liver of an adult dog, are produced from a single lobule. Regulated by Thoma 's laws the main vascular stems develop in such a way that all the lobules are equally favored. "If fluids of different consistency are injected either into the portal or hepatic vein, all of the terminal veins fill simultaneously. "Moreover, the final branches of the portal and hepatic veins are always as far from one another as possible. "At all times this distance is half the diameter of a lobule, and since this is in the neighborhood of one millimetre, the distance is abont half a millimetre, the normal length of a capillary vessel. ' ; The lobules at birth, according to Toldt and Zuckerkandl, differ from those of the adult, as follows : " In the child there are indeed vascular territories which show a certain independence. However, since they are drained by a group of terminal branches and not by a single venous root, they are not comparable with the lobules of the adult. They correspond rather with a combination of the latter, and to a certain extent represent lobules of a higher order, from which gradually single portions will be detached." The multiplication of lobules continues long after birth, and partly divided, compound forms were recognized in the adult by Kiernan.
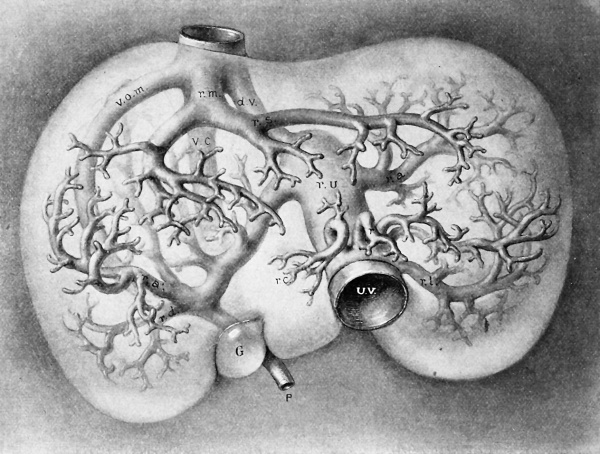
Fig. 296. Ventral view of a reconstruction of the hepatic vessels in an embryo of 11 mm. X 25 diam. (After Mall.) The principal veins are — d. v., ductus venosus; p., portal vein; r.m. and r. s., middle and left rami of the hepatic vein; r. »., recessus umbilicalis; u. v., umbilical vein; v. c, vena cava; v. o. m., omphalomesenteric vein.
Ligaments and Lobes
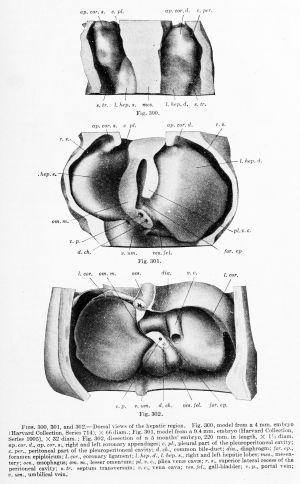
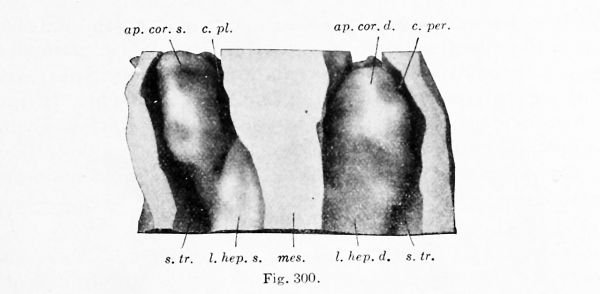
The relation of the liver of a 4 mm. embryo to the body cavity is shown in dorsal view in Fig. 300. The model shown in the figure has three vertical cut surfaces, — the body wall on either side, and the mesentery, prolonged upward as the mediastinal septum, in the centre. The ventral portion of the mesentery bulges laterally below, where the liver is growing into it, and then joins the septum transversum. The latter is the plate of tissue which forms the ventral surface of the model. Its position is so nearly vertical that it separates the pair of pleuroperitoneal cavities behind from the pericardial cavity in front. The pleuroperitoneal cavities are shown in the figure. Anteriorly they turn ventrally over the free margin of the septum transversum and empty into the median pericardial cavity.
Most embryologists. following His, state that the liver develops in the septum transversum. and as seen in median sagittal sections (Fig. 236, p. 311) this appeal's to be correct. The septum transversum. however, is early divisible into two parts, related to one another like the arms of a T. The ti'ansverse portion forms a part of the' diaphragm. The median sagittal portion is the ventral mesentery, and it is in this subdivision of the septum transversum that the liver develops. The relatively very broad attachment of the mesentery to the diaphragm forms the falciform ligament of the liver, and the lateral bulgings indicate respectively the right and left hepatic lobes.
Figs. 297, 298, and 299. Diagrams of three successive stages in the formation of lobules. (After Mall.) d, branch of the portal vein ; a, branch of the hepatic vein.
His (1880) recognized that in a -1 mm. embryo the tissue in which the liver develops is more or less independent of the septum transversum, and he named it the " Vorleber." Hertwig (190G), who states — following His — that the liver grows into the septum transversum. writes also that the liver develops in the ventral mesentery.
In the 4 mm. embryo (Fig. 300), each of the lateral lobes of the liver is prolonged upward by an irregular mass of tissue, which nearlv fills the body cavity. Each mass is attached alone,' its ventral border to the septum transversum. and thus it separates the medial pleural part of the body cavity from the lateral peritoneal part. But superiorly these cavities connect with one another and open into the pericardial cavity, as already noted. The upward prolongations of the liver may be called the right and left coronary appendages. They are the anterior portions of the "Vorleber" of His, which were described in a 4 mm. embryo as containing a plexus of blood-vessels but no network of trabecular 16 They are certainly the ventral pillars bounding the pleuroperitoneal opening, first described by Uskow (1883) and later, for human embryos, by Swaen (1897). Their relation to the dorsal pillars, which have been called the suspensory ligaments of the Wolffian body, have been discussed in Chapter XIII.
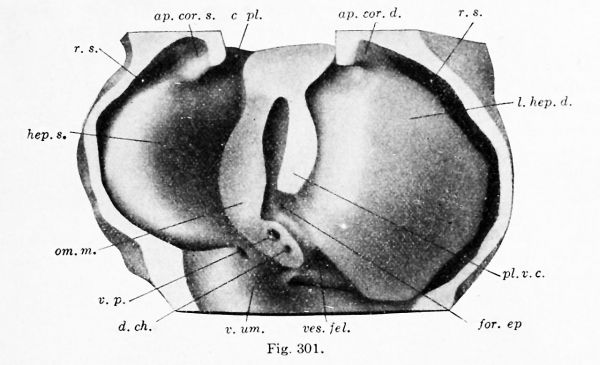
In an embryo of 9.4 mm. (Fig. 301) it is seen that the coronary appendages have fused with the septum transversum and the lateral body wall, thus shutting off the superior lateral recess of the peritoneal cavity (Swaen). The liver now presents a crescentic transverse attachment to the diaphragm, passing from one coronary appendage to the other; this attachment is the coronary ligament. Within its concavity, on either side, are the pleural cavities which communicate below with the peritoneal cavity.
A fundamental feature of the 9.4 mm. embryo is the presence of the plica vence cavce, of Ravn (1889). This is essentially an attachment of the right lobe of the liver to the dorsal body wall, and it has developed downward from the right ala pulmonalis. 17 Through this attachment the right subcardinal vein anastomoses with the veins of the liver, thus giving rise to the vena cava inferior. The portion of the liver between the plica venae cavae and the ventral mesentery, or omentum minus, is the caudate lobe (of Spigelius). The caudate lobe joins the right lobe across the foramen epiploicum (of Winslow). Below the foramen, the portal vein and bile-duct are seen in section. In the lower part of the model the place where the left umbilical vein enters the liver is indicated by a fold. The gall-bladder is on the right of it.
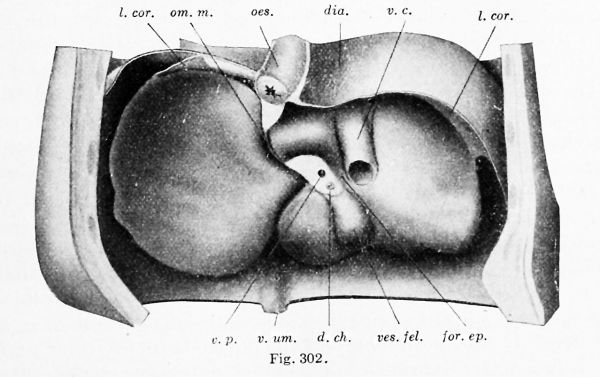
In an embryo of 5 months (Fig. 302) the diaphragm has been completed in the way described in Chapter XIII. The oesophagus, not included in the preceding drawings, is seen passing through it. The thin lateral extensions of each coronary ligament {lig amenta triangularia) mark the position of the former appendages, and filling their dorsal concavity is the portion of the diaphragm which formed last, and which completes the separation of pleural and peritoneal cavities. The vena cava inferior now fills its plica, which has become broad. The lesser omentum is very thin except at the transverse fissure or porta; together with the gall-bladder it marks the true median plane of the liver. The umbilical vein is seen in the ventral abdominal wall, from which it passes to the liver along the free margin of the falciform ligament. It then lies in a deep groove on the under surface of the liver, and with the porta and the gall-bladder it bounds the quadrate lobe. Its extension, the ductus venosus, passes toward the vena cava at the bottom of the fissure of the lesser omentum.
- 10 They were probably included by Lieberkuhn (1876) among the "villi" which occur where the omphalomesenteric veins enter the heart, and which were said to be so related to the developing liver that they contained the first blood vessels of that organ.
- Ala pulmonalis is the term introduced by Ravn for the developing mesodermal portion of each lung. The ala pulmonalis appears on either side of the fore-gut as a wing-like fold, which is flattened dorsoventrally and which has a free lateral margin. The caudal portion of each ala becomes a pulmonary ligament (lig. pulmonale).
Figs. 300, 301, and 302. — Dorsal views of the hepatic region. Fig. 300, model from a 4 mm. embryo (Harvard Collection, Series 714), X 66 diam.; Fig. 301, model from a 9.4 mm embryo (Harvard Collection, Series 1005), X 32 diam. ; Fig. 302, dissection of a 5 months' embryo, 220 mm in length, X l'i diam. ap.cor. d., ap. cor. s., right and left coronary appendages; c. pi., pleural part of the pleuroperitoneal cavity; c. per., peritoneal part of the pleuroperitoneal cavity; d. ch., common bile-duct; dia., diaphragm; for. > p., foramen epiploicum; I. cor., coronary ligament; 1. hep, d., I. hep. «., right and left hepatic lobes; mes., mesentery; oes., oesophagus; om. m., lesser omentum; pl. v. c, plica venae cavae; r. s., superior lateral recess of the peritoneal cavity; s.tr. septum transversum; v.c, vena cava; ves. fel., gall-bladder; v. p., portal vein; v. um., umbilical vein.
In the preceding description the embryonic liver has been divided into right and left lobes, separated by the falciform ligament. Rex, after careful comparative studies of adult livers, declares that the only subdivision of the human liver which is a true lobe is the omental or caudate, but he admits that the recognition of the right and left lobes is justified by the distribution of the branches of the portal vein. Some embryologists have found this convenient. The absence of deep clefts, such as mark off the dorsolateral lobes in the embryonic liver of pigs and rabbits, is notable in the human liver. Nevertheless Swaen (1897) considers that corresponding lobes should be recognized, and accordingly he describes the human liver as composed of three lobes, — one median and two lateral. Mall (1906) states that each of the six primary lobules which he finds in an embryo of 11 mm. is to expand into a whole lobe, and Bradley (1908) shows the relation of six lobes to the three which he considers fundamental. Thompson (1899) has described the fissures and clefts which frequently appear on the under surface of the liver, especially of the right lobe. Certain of these were found with considerable regularity. The two most frequently met with, occurring respectively in 83 and 50 per cent, of the cases examined, were believed to form partial boundaries of a lobe which is well defined in the gorilla. The almost entire absence of lobes in the human liver has been emphasized by Rex.
The Liver as a Whole
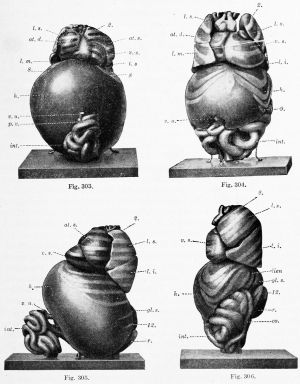
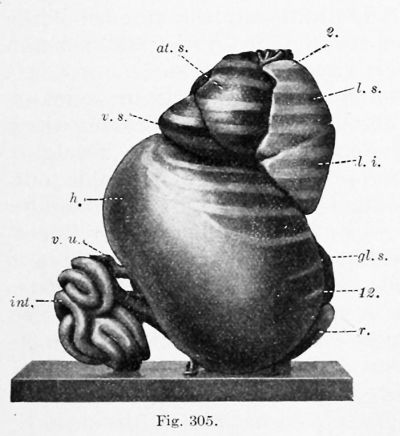
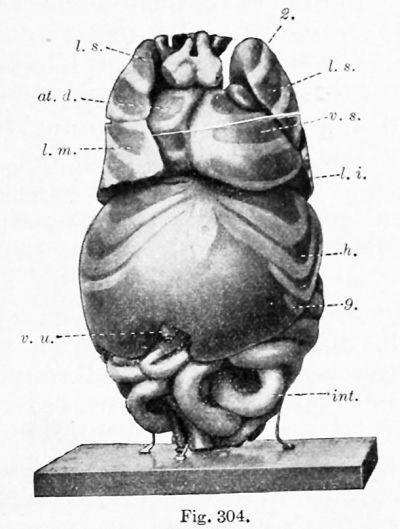
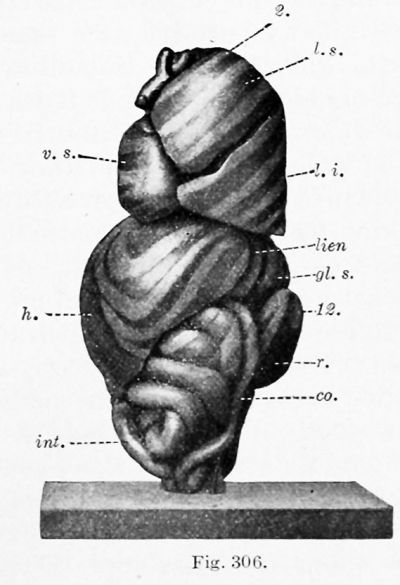
Except for a temporary decrease at birth, associated with the closure of the umbilical vein, the weight of the liver steadily increases. At the end of the second fetal month it weighs .2 gin. ; at birth, 75 gm. ; and in the adult 1500 gm. (Mall). But the volume of the liver, as compared with that of the body, reaches a maximum in a 31 mm. embryo, as determined by Jackson (1909). He found that in an 11 mm. embryo the liver is 4.85 per cent, of the total body volume, or approximately the same as at birth ; in a 17 mm. embryo it is 6.9 per cent. ; and at 31 mm. it is 10.56 per cent. (Pigs. 303 and 305). In a 65 mm. embryo (Figs. 304 and 306) it has apparently decreased to about 5 or 6 per cent., which is the average for the remainder of the fetal period. In this specimen the liver, as indicated by its relation to the ribs, has aco^ired approximately its final position.
During its development, certain portions of the liver atrophy, while other parts increase. The most extensive degeneration is in the peripheral part of the left lobe. In the 31 mm. embryo the two lobes are still nearly symmetrical, and the left lobe extends between the spleen and the body wall (Fig. 305). At 65 mm. "the liver has partly retracted, so that it covers only the anterior portion of the external splenic surface " (Fig. 306). In the adult any contact between the liver and spleen is exceptional. The decrease in the size of the left lobe is generally ascribed to pressure from adjacent organs. Pougnault (1905) notes that in cases of umbilical hernia the symmetry may be retained, and Jackson's models indicate that the decrease occurs when the intestines enter the abdomen. It may also be associated with the expansion of the gastric fundus. As a result of this degeneration, the left portion of the coronary ligament (the appendix fibrosa) contains a network of anastomosing ducts, discovered by Ferrein and described by Kiernan as a " rudimental liver. ' ' Both of these anatomists recognized similar tissue around the vena cava. Usually this vein occupies a fissure on the dorsal surface of the liver, but Kiernan states that the fissure is frequently converted into a canal, either by hepatic parenchyma or by a ligamentous band containing ducts and blood-vessels. He found that similarly the umbilical vein may be completely surrounded by hepatic tissue, or bridged by a band of the same structure as the lig amentum venae cavce. Hepatic trabecular may also invade the diaphragm, and at birth they have been reported as extending into the falciform ligament as far as the umbilicus. In all of these situations, and also near the expanding gall-bladder, the hepatic cells may degenerate, leaving aberrant ducts and blood-vessels. These have been studied through injections by Toldt and Zuckerkandl (1875). The marked variations in the form of the fetal liver have been tabulated by Ruge (1907).
| Figs. 303 and 305. — Ventral and lateral views of a model of the viscera from a 31 mm. embryo. X 4.5 diam. | |
| Figs. 304 and 306. Similar views of a model of the viscera from a 65 mm. embryo. X2 diam. (After C. M. Jackson.) 2, 8, q, 12, ribs; at. d., <it. 8., right and left atria: co., colon; nL s., left suprarenal gland; h., liver; int., small intestine; lien, spleen; /. £., /. m., I. 8., inferior, middle, and superior lobes of the lung; p. v., vermiform process and cecum; r., kidney; v. 8., left ventricle; v. u., umbilical vein. | |
Anomalies of the Liver
The total absence of the gall-bladder, according to Meckel (1812), is not very unusual. In these cases the hepatic diverticulum has presumably developed normally, but has failed to produce the secondary subdivision which gives rise to the gall-bladder. Sometimes, in addition to the absence of the gall-bladder, there is no trace of the hepatic, cystic, and common bile-ducts. Kirmisson and Hebert have reported such a case in a child of one month, and they found two similar instances in the literature. These are probably due to obliterative processes which begin after the extra-hepatic bile-ducts have developed. Two gallbladders may be present, perhaps produced by a double outpocketing of the diverticulum. Sometimes when the gall-bladder is single there are two cystic ducts, as in a case reported by Dreesman. The two ducts arose from the gall-bladder 1 cm. apart, and united before entering the common bile-duct. Fig. 292, B, indicates how such an anomaly may develop. Beneke (1907) has studied congenital atresia of the bile-ducts. Multiple hepatic ducts have been recorded, sometimes opening separately into the duodenum.
Congenital cysts of the liver generally arise from the ducts in the connective tissue, but they may occur within the hepatic parenchyma (Moschcowitz, 1906). Sometimes they attain very large size (Sanger and Klopp, 1880). The subdivision of the liver into multiple lobes is quite common, and the occurrence of accessory livers, more or less isolated from the central mass, is well known (Toldt and Zuckerkandl, 1875). An excessive atrophy of the left lobe, leading to its "entire absence," has been recorded by Kantor (1903).
| Embryology - 24 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Grosser O. Lewis FT. and McMurrich JP. The Development of the Digestive Tract and of the Organs of Respiration. (1912) chapter 17, vol. 2, in Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |