Abnormal Development - TORCH Infections: Difference between revisions
| Line 62: | Line 62: | ||
[[File:Hepatitus B.jpg|400px]] | [[File:Hepatitus B.jpg|400px]] | ||
===Hepatitis Transmission Risk to the Fetus=== | |||
* '''Hepatitis A''' - Fetal transmission of virus occurs with extreme rarity. | |||
* '''Hepatitis B''' - Can occur as a consequence of intrapartum exposure, transplacental transmission, and breastfeeding. | |||
20%–30% of HBsAg-positive/HbeAg-negative women will transmit virus to their infants. | |||
90% of HBsAg- and HBeAg-positive women will transmit virus to their infants. | |||
Immunoprophylaxis at birth with both HBIG and Hepatitis B vaccine within 12 hours of birth decreases the risk of transmission. | |||
Passive (HBIG) and active immunization is 85%–95% effective in preventing neonatal HBV infection. | |||
* '''Hepatitis C''' - The overall risk of transmission is approximately 5%–10% with unknown maternal viral titers. | |||
All pregnant women with HCV should have viral titers performed. | |||
Data: Hepatitis and reproduction<ref><pubmed>19007636</pubmed></ref> | |||
==References== | ==References== | ||
Revision as of 17:42, 17 June 2010
Introduction
Materal effects should really be called environmental (in contrast to genetic) removing the association of mother with the deleterious agent. Accepting this caveat, there are several maternal effects from lifestyle, environment and nutrition that can be prevented or decreased by change which is not an option for genetic effects.
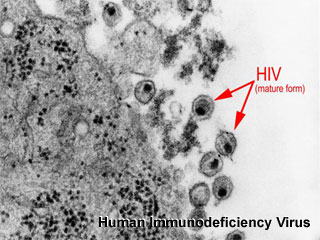
Infections, collectively grouped under the acronym TORCH for Toxoplasmosis, Other organisms (parvovirus, HIV, Epstein-Barr, herpes 6 and 8, varicella, syphilis, enterovirus) , Rubella, Cytomegalovirus and Hepatitis. See related pages on Maternal Hyperthermia and Bacterial infections.
Finally, when studying this topic remember the concept of "critical periods" of development that will affect the overall impact of the above listed factors. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes.
Some Recent Findings
- Seroprevalence of TORCH infections in women of childbearing age in Croatia.[1] "During 2005-2009, a seroepidemiological study was carried out in Croatia to define the population susceptible to common TORCH agents among pregnant and non-pregnant women of childbearing age. The IgG seroprevalence was 29.1% forT. gondii, 94.6% for rubella, 75.3% for cytomegalovirus (CMV), 78.7% for herpes simplex virus type 1 (HSV-1), and 6.8% for HSV-2. Acute toxoplasmosis and CMV infection (positive IgM antibodies with low IgG avidity) were documented in 0.25% and 0.09% women, respectively. IgM prevalence was 1.2% for both HSV-1 and HSV-2. None of the participants showed acute rubella infection. Seropositivity to T. gondii and HSV-2 varied significantly between age groups (p = 0.001 and p = 0.036, respectively). Women residing in rural regions showed a significantly higher seroprevalence rate for T. gondii, CMV, and HSV-1 than urban women (T. gondii: 44.0% vs. 25.4%, p < 0.001; CMV: 85.0% vs. 73.1%, p = 0.018; HSV-1: 86.0% vs. 76.4%, p = 0.041)."
- Routine TORCH screening is not warranted in neonates with subependymal cysts.[2] "Congenital infections are associated with a wide variety of clinical symptoms, including subependymal cysts (SEC). ...The co-occurrence of TORCH congenital infections in infants with SEC is rare. Routine TORCH screening in neonates with SEC does not seem warranted."
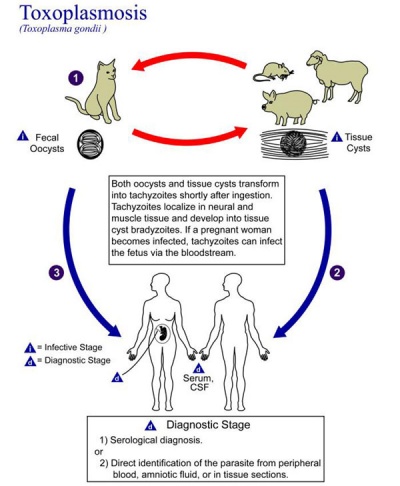
Toxoplasmosis
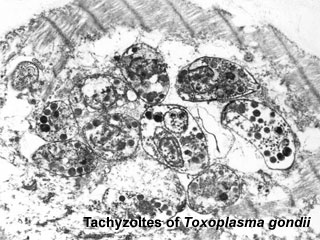
The causal agent of Toxoplasmosis is the protist Toxoplasma gondii. This unicellular eukaryote is a member of the phylum Apicomplexa which includes other parasites responsible for a variety of diseases (malaria, cryptosporidiosis). The diagnosis and timing of an infection are diagnostically based on serological tests.
|
|
| Toxoplasmosis lifecycle | Toxoplasma tachyzoites |
Recent findings suggest that pre-pregnancy immunization against toxoplasmosis may not protect against reinfection by atypical strains.
Other Organisms
A general term covering a ranges of viruses: parvovirus, HIV, Epstein-Barr, herpes 6 and 8, varicella, syphilis, enterovirus.
Links: Abnormal Development - Viral Infection
Rubella
Rubella virus (Latin, rubella = little red) is also known as "German Measles" due to early citation in German medical literature. Infection during pregnancy can cause congenital rubella syndrome (CRS) with serious malformations of the developing fetus. The type and degree of abnormality relates to the time of maternal infection.
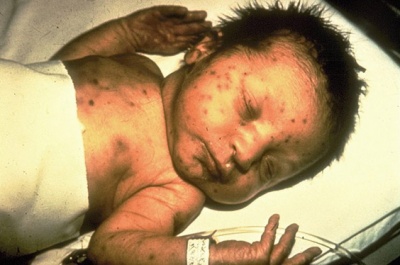
|
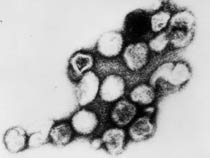
|
| Infant rubella virus | Rubella virus (electron micrograph |
Cytomegalovirus
Human cytomegalovirus (HCMV, Greek, cyto = "cell", megalo = "large") or Human Herpesvirus 5 (HHV-5) is a member of the herpes virus family. A viral infection that causes systemic infection and extensive brain damage and cell death by necrosis. HCMV infection is ranked as one of the most common infections in adults, with the seropositive rates ranging from 60–99% globally. In Western countries, adults with advanced AIDS prior to the introduction of highly active antiretroviral therapy (HAART) this virus also a cause of blindness (CMV retinitis) and death in patients.
Hepatitis
Hepatitis (inflammation of the liver) is caused in humans by one of 7 viruses (A, B, C, D, E) with the 2 additional F has not been confirmed as a distinct genotype; and G is a newly described flavivirus.
"All of these viruses can cause an acute disease with symptoms lasting several weeks including yellowing of the skin and eyes (jaundice); dark urine; extreme fatigue; nausea; vomiting and abdominal pain. It can take several months to a year to feel fit again." (CDC text).
Virus particles measure 42nm in overall diameter and contain a 27nm diameter DNA-based core.
Hepatitis Transmission Risk to the Fetus
- Hepatitis A - Fetal transmission of virus occurs with extreme rarity.
- Hepatitis B - Can occur as a consequence of intrapartum exposure, transplacental transmission, and breastfeeding.
20%–30% of HBsAg-positive/HbeAg-negative women will transmit virus to their infants. 90% of HBsAg- and HBeAg-positive women will transmit virus to their infants. Immunoprophylaxis at birth with both HBIG and Hepatitis B vaccine within 12 hours of birth decreases the risk of transmission. Passive (HBIG) and active immunization is 85%–95% effective in preventing neonatal HBV infection.
- Hepatitis C - The overall risk of transmission is approximately 5%–10% with unknown maternal viral titers.
All pregnant women with HCV should have viral titers performed.
Data: Hepatitis and reproduction[3]
References
Reviews
Articles
Search Pubmed
June 2010 "TORCH Infections" All (183) Review (37) Free Full Text (18)
Search Pubmed: TORCH Infections | maternal infections | teratogens
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 23) Embryology Abnormal Development - TORCH Infections. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_TORCH_Infections
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G