2018 Group Project 1
The Contribution of Neural Crest Cells to the Adrenal Medulla
| Projects 2018: 1 Adrenal Medulla | 3 Melanocytes | 4 Cardiac | 5 Dorsal Root Ganglion |
Project Pages are currently being updated (notice removed when completed)
Introduction
Z5014972 (talk) 12:47, 14 August 2018 (AEST)z5014972 good review article https://link.springer.com/article/10.1007%2Fs12022-009-9070-6
History
Describing the neural crest
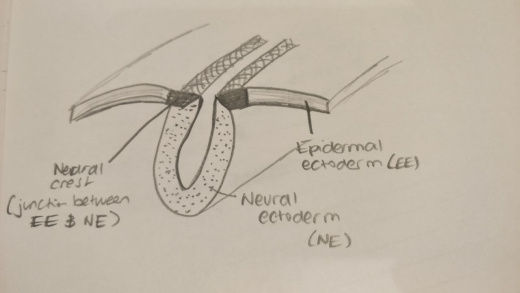
The "neural crest" is a term referring to the junction between the neural and epidermal ectoderm in the embryo. The neural crest contributes a large number of cells of varying structure and function that directly or indirectly contribute to the development of tissues and organs within the body [1]. In 1868, the neural crest cells were first identified in a chick embryo as a collection of cells known as "Zwischenstrang" by Professor Wilhelm His [2]. Wilhelm His was among the first to provide an explanation for the mechanics governing the developing embryo as well as the physiology and the organs that the differing germinal regions would eventually give rise to. In 1878, the term neural ridge was used to describe this collection of cells but the term neural crest was later coined by Arthur Milnes Marshall in 1879 in a paper he worked on disseminating the knowledge from his research on the development of cranial nerves in chicken embryos. This change in the naming of the neural crest was due to the fact that two neural ridges could be seen on either size prior to the closure of the neural tube, resembling the appearance of a crest [1].
This figure shows the neural crest, a fold located between the epidermal ectoderm and the neural ectoderm. The neural ectoderm gives rise to the neural tube in the centre, the epidermal ectoderm, as the name suggests, gives rise to the skin and the neural crest is located between the two and gives rise to many other structures, tissues and organs in the body.
1890s-1950s The neural crest is the point of origin for spinal/cranial ganglia and neurons. This theory received little criticism and was not contested much by other researchers prior to the 1890s. This is owing to the fact that the neural crest is quite closely related to the neural tube and thus this relationship makes sense. This discovery was made by His and Marshall, however, they found this out independently. However, in the 1890s, Julia Platt, claimed that odontoblasts as well as the cartilages forming the facial and pharyngeal arch skeletons came from the ectoderm. This created much debate and was contested by many. This is because this theory completely contradicted the germ-layer theory, whereby skeletal tissues originate at the mesoderm and not the ectoderm. The controversies this finding brought about a slowing down of research, as demonstrated by a gap of almost 40 years between the theory that skeletal tissues originating from the neural crest. Today, it is known that the neural crest dos indeed play a role in the development of the skeleton in vertebrates, particularly, the cranial neural crest. Although these studies were occurring, the focus was still primarily on researching pigment cells and ganglia of the spine up until the 1950s, when Sven Hörstadius' major work on the neural crest was published, titled "The Neural Crest: Its properties and derivatives in the light of experimental research". This work focused on experimental data tracing the development of the cartilaginous skeleton derived from the neural crest [1].
1960s - 1970s
Embryonic origins
Developmental time course
The adrenal gland has two distinct sections, the cortex and the medulla, both with different embryonic origins. The cortex accounts for 90% of the adrenal glands and arises from intermediate mesoderm. The medulla only accounts for 10% of the adult adrenal glands, however it has neural crest embryonic origin. The medulla consists of chromaffinoblast cells which are migrant neuroblast cells from the neural crest, therefore the adrenal medulla is a component of the sympathetic nervous system.[3] Neural crest cells replicate in week 7 and begin to differentiate in week 8, the cells then migrate to the developing adrenal gland in week 9 of development.[4][5] The neural crest cells infiltrate the cortex and the chromaffin cells are scattered islands in the cortex.[6] Throughout development the cells will become more compact in the centre of the gland, during this period of development the adrenal gland is growing rapidly. The gland is abnormally large until the second trimester and the medulla is much thicker prenatally.[5][3]
Developmental/adult function
Tissue/organ structure
Normal Structure and Function of the Adrenal Medulla
The cells of the adrenal medulla are derived from the neural crest as opposed to the mesodermal origins of the cortex. The medulla contains secretory cells called chromaffin cells, due to the agents they produce when oxidised, such as chromate. These cells secrete epinephrine and norepinephrine in response to various substances such as acetylcholine.
There are three types of cells in the adult adrenal medulla: 1) Epinephrine cells 2) Norepinephrine cells 3) Small granule-containing cells (SGCs) These cells also produce various other peptides such as substance P and neurotensin. The adrenal medulla also contains presynaptic sympathetic ganglion cells.
Related Anatomy
The tissue and organ structure of the adrenal medulla should be understood with regard to its specialised function within the sympathetic nervous system.
The medullary region of the adrenal glands is tasked with the endocrine secretions of adrenaline and nor-adrenaline in response to environmental stressors that are signalled for by the sympathetic nervous system. That is, the secretion of fight-or-flight response hormones in order to restrict vasculature to the trunk and increase vascular activity in the peripheral musculature.
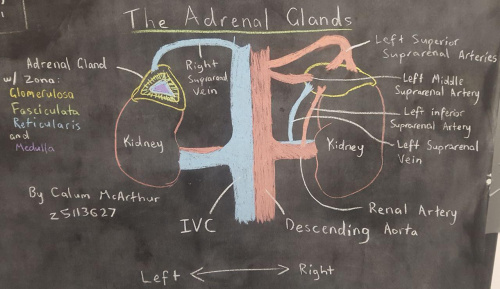
The adrenal glands are supplied by several branches of the great vessels in the abdominal cavity. The secreted catecholamines in the medulla are directly able to pass to the blood stream this way.
These adrenal glands and their contents are retroperitoneal in the adult and varied in shape, with the left often being semilunar and right being pyramidal. Nervous supply of the adrenal glands is achieved by contributions from the splanchnic nerves of the celiac plexus.
Molecular mechanisms/factors/genes
Role of Adrenal Medulla in the Neonate and Adult
Understanding the biochemistry of catecholamines is necessary to see how the adrenal medulla is designed in the stages of embryology and how it is suited for life in the neonate and adult.
The epinephrine and norepinephrine in the neonate and adult work by primarily changing the osmoregulatory state of vasculature in organs. With the exclusion of their neuromodulatory and neurotransmitter functions, epinephrine and norepinephrine are mainly tasked with the control of sympathetic and parasympathetic supplies.
>Norepinephrine is a constrictor of peripheral vasculature by antagonising the action of surface receptors expressed on the endothelium of blood vessels, specifically Alpha-1 and Alpha-2 receptors, such that vascular resistance increases.
>Epinephrine is both a vasoconstrictor and vasodilator, depending on what receptors it attaches to. As a non-selective adrenergic agonist, it acts on Alpha-1, Alpha-2, Beta-1, Beta-2 and Beta-3 receptors that are found throughout the body's tissues, yielding many different physiological responses.
Genes and Transcription Factors Involved with the Adrenal Medulla's Development
Members of the SOX (SRY-Box) gene family have multiple key contributors to Neural Crest in the Adrenal Medulla.
Mouse models have demonstrated that SOX-10 knock-outs (mice whose DNA has been engineered to not express SOX-10) do not have an adrenal medulla. There has also been suggestion that SOX-8,9 and 10 are all strongly related to the migratory patterning of neural crest cells to the developing adrenal glands.
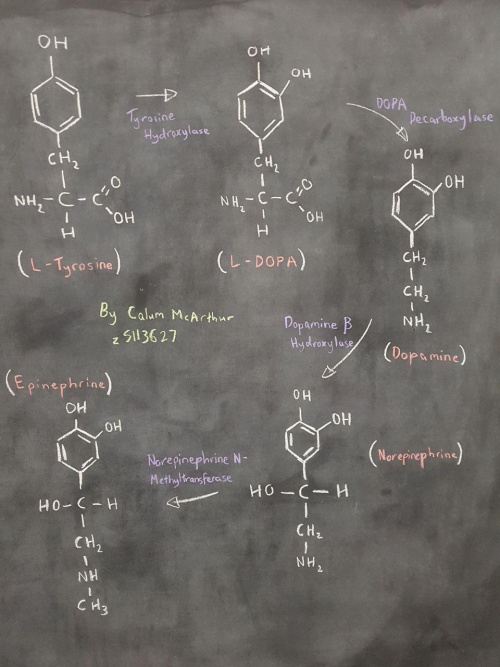
The TH gene (for Tyrosine Hydroxylase) encodes the aforementioned enzyme in the adrenal medulla and central nervous system. Tyrosine Hydroxylase is crucial in biochemical pathways and cascades for the synthesis of catecholamines and other CNS neurotransmitters. The conversion from L-Tyrosine to L-DOPA is dependent upon the catalytic action of Tyrosine Hydroxylase. From there, L-DOPA can make Dopamine and eventually Epinephrine and Norepinephrine as well.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2291442/ https://www.ncbi.nlm.nih.gov/pubmed/19396395
Abnormalities/abnormal development
Animal models
Animal models are an integral part of research in embryology. In particular due to the moral concerns regarding the use of humans in scientific research. As a result different animals are used as substitutes to simulate the same environments and hope to achieve results that would also be able to represent humans as well. Prenatal adrenal gland development has been described in a number of scientific reports dating from the 1900s to studies in more recent years. These involved a variety of animal models such as ox, sheep, swine and most commonly mice.
Prenatal adrenal gland development has been described in numerous published reports covering a wide variety of species, including the ox (Katznelson, 1966; Wrobel and Suss, 1999), sheep (Davies, 1950; Wintour et al., 1975; Upadhyay and Zamboni, 1982; Naaman‐Reperant and Durand, 1997; Grino, 2004), swine (Sokolov et al., 2006) and mouse (Waring, 1935; Ikeda et al., 1994; Sass, 1996; Nyska and Maronpot, 1999; Bland et al., 2003; Val et al., 2007). Adrenal gland development also has been extensively described for humans as well (Kolliker, 1879; Minot, 1892; Zuckerkandl, 1912; Wieman, 1920; Ikeda et al., 1981; Langlois et al., 2002; Hanley and Arit, 2006).
https://www.sciencedirect.com/science/article/pii/S0925477302004550 http://dev.biologists.org/content/develop/126/13/2935.full.pdf
Current research (labs)
Adrenomedullary chromaffin cells are catecholamine (CA)-producing cells originating from trunk neural crest (NC) via sympathoadrenal progenitors (SAPs). We generated NC and SAPs from human embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) in vitro via BMP2/FGF2 exposure, ascertained by qPCR and immunoexpression of SOX10, ASCL1, TFAP2α, and PHOX2B, and by fluorescence-activated cell sorting selection for p75NTR and GD2, and confirmed their trunk-like HOX gene expression. We showed that continuing BMP4 and curtailing FGF2 in vitro, augmented with corticosteroid mimetic, induced these cells to upregulate the chromaffin cell-specific marker PNMT and other CA synthesis and storage markers, and we demonstrated noradrenaline and adrenaline by Faglu and high-performance liquid chromatography. We showed these human cells' SAP-like property of migration and differentiation into cells expressing chromaffin cell markers by implanting them into avian embryos in vivo and in chorio-allantoic membrane grafts. These cells have the potential for investigating differentiation of human chromaffin cells and for modeling diseases involving this cell type.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5768882/
Glossary
Reference list
- ↑ 1.0 1.1 1.2 Hall BK. (2008). The neural crest and neural crest cells: discovery and significance for theories of embryonic organization. J. Biosci. , 33, 781-93. PMID: 19179766
- ↑ Bronner ME. (2012). Formation and migration of neural crest cells in the vertebrate embryo. Histochem. Cell Biol. , 138, 179-86. PMID: 22820859 DOI.
- ↑ 3.0 3.1 El-Nahla SM, Imam HM, Moussa EA, Elsayed AK & Abbott LC. (2011). Prenatal development of the adrenal gland in the one-humped camel (Camelus dromedarius). Anat Histol Embryol , 40, 169-86. PMID: 21175739 DOI.
- ↑ Ross IL & Louw GJ. (2015). Embryological and molecular development of the adrenal glands. Clin Anat , 28, 235-42. PMID: 25255746 DOI.
- ↑ 5.0 5.1 Kempná P & Flück CE. (2008). Adrenal gland development and defects. Best Pract. Res. Clin. Endocrinol. Metab. , 22, 77-93. PMID: 18279781 DOI.
- ↑ Quinn TA, Ratnayake U, Dickinson H, Nguyen TH, McIntosh M, Castillo-Melendez M, Conley AJ & Walker DW. (2013). Ontogeny of the adrenal gland in the spiny mouse, with particular reference to production of the steroids cortisol and dehydroepiandrosterone. Endocrinology , 154, 1190-201. PMID: 23354096 DOI.