2017 Group Project 6
| 2017 Student Projects | |||
|---|---|---|---|
|
Introduction
The cerebellum is a large portion of the brain that functions in coordination, balance and control, and its development occurs both prenatally and postnatally. The cerebellum underlies the occipital and temporal lobes of the cerebral cortex and constitutes to about 10% of the brains weight however contains around 50% of the neurons.
This page will highlight the anatomy of the cerebellum, its developmental process, current research on the structure, animal models and abnormalities associated with it. The anatomy will discuss the distinguishable lobes and zones of the cerebellum. The anatomy of the cerebellum cannot be completely discussed without explaining the vasculature of the structure and hence this page will provide a brief overview of it. The development of the cerebellum discusses the neural development and where the cerebellum forms on the neural tube. The development will highlight how the circuitry of the post-natal cerebellum came to be from neurons(?). Hence purkinje cells, granule cells, deep nuclei cells, glia cells and cerebellar nuclei will be highlighted to discuss the developmental process. A developmental timeline of the formation of the cerebellum is also included on this page. The cerebellum is a topic of continuous research and past findings would not have been done without the use of animal models, hence current research and animal models will be discussed. The abnormalities that could happen if the cerebellum were to be affected is also highlighted.
Basic Anatomy of the Cerebellum
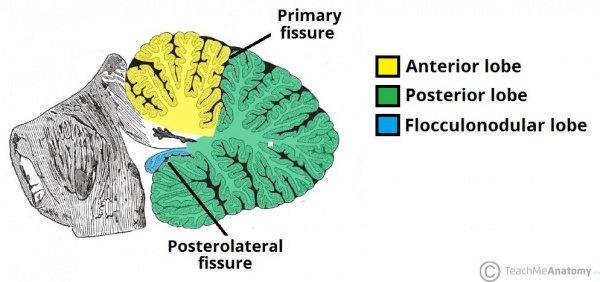
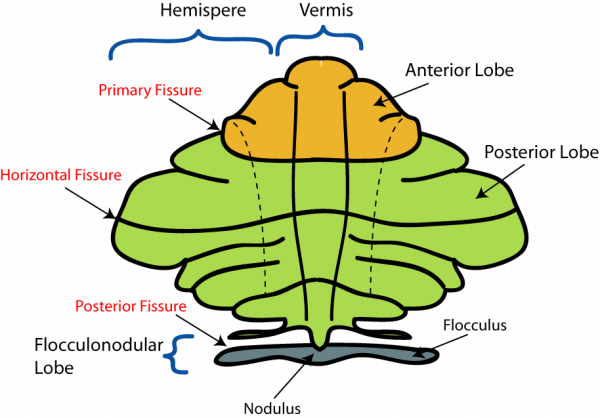
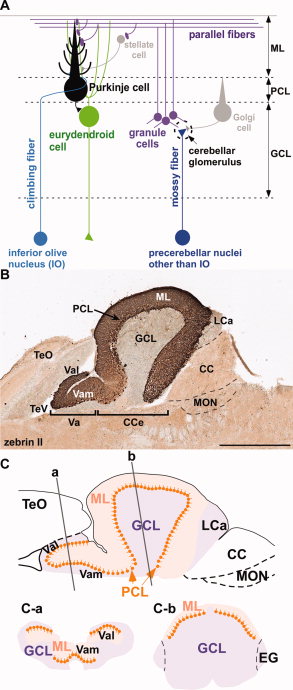
The cerebellum has 3 distinguishable lobes; flocculonodular lobe, anterior lobe and the posterior lobe. The anterior and posterior lobe can be further divided in a midline cerebellar vermis and lateral cerebellar hemispheres (Figure 1) [1]. In a superior cerebellar view, the cerebellum contains a vermis that runs through the middle of the organ and 2 intermediate zones located laterally from the vermis (Figure 2).
Figure 1: Anatomical lobes observed in the cerebellum; anterior lobe, posterior lobe and flocculonodular lobe, which is divided by two fissures – the primary fissure and posterolateral fissure [2]
Figure 2: Superior view of the 3 cerebellar zones. The middle is the vermis. Either side of the vermis is the intermediate zone. Lateral to the intermediate zone is the lateral hemispheres. There is no difference in gross structure between the lateral hemispheres and intermediate zones. [3]
Vasculature
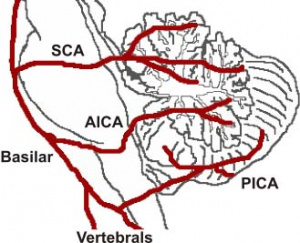
The cerebellum contains 3 bilateral paired arteries which supplies this organ with oxygenated blood. These arteries all originate from the vertebrobasilar system; Superior Cerebellar Artery (SCA), Anterior Inferior Cerebellar Artery (AICA) and the Posterior inferior cerebellar artery (PICA). The SCA and AICA are branches of the basilar artery, which wraps around the anterior aspect of the pons before reaching the cerebellum. The PICA arises from the left and right vertebral artery, which form the basilar artery [5]. The PICA and AICA combine to supply the inferior half of the cerebellum, while the SCA supplies the majority of the superior half. The PICA and SCA combine to supply the vermis [6]. Blood is then drained by superior and inferior cerebellar veins into the superior petrosal and then straight dural venous sinuses. (Figure 3) [7]
Ectoderm
Neural Development

Neural development is one of the earliest systems to begin and the last to be completed after birth due to its highly complex structure. The first step in neural development occurs at the end of week 3 and involves the neural groove fusing to form the neural tube, which then folds to form the cranial and caudal region of the embryo, and ultimately form the cerebellum [9] . There is a high chance of neural dysfunction and defects during the fetal neural development particularly due to the long development time frame and the need of certain nutrients such as folic acid to successfully close the tubes. Neural tube defects (NTDs) such as spina bifida and anencephaly can arise if the tubes do not close effectively.
Microanatomy
Cortical Layers
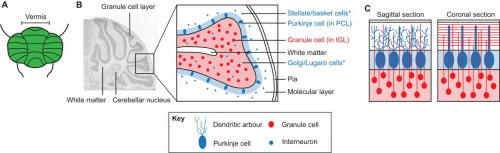
There are 3 major cortical layers of the cerebellum: the molecular layer, the purkinje cell layer, and the granule cell layer. The molecular layer contains basket cells, stellate cells and the purkinje cell and Golgi cell dendrites. The purkinje cell layer contains purkinje cell bodies and Bergmann glia. The granule cell layer contains granule cells, mossy fibers, and Golgi cell bodies. [10]
Purkinje/Pyramidal Cells
Discovered by Jan Evangelista Purkinje in 1837, purkinje cells are inhibitory neurons found in the outside layer of the cerebellum. They receive signals from the granule cell parallel fibers and the superior olive and send inhibitory signals to the deep nuclei in the white matter region via GABA signaling. Purkinje cells have a large branching network of dendrites which allows them to be identified by their morphology.
Granule Cells
Named for their small cell body, cerebellar granule cells of were discovered by Camillo Golgi and Ramon y Cajal in 1899. Cerebellar granule cells are the most numerous cell type in the human brain. They receive signals from mossy fibers of the pons and synapse on the fast network of dendrites of the pyramidal cells. Cerebellar granule cells are glutamatergic and the only excitatory neurons found in the cerebellum. [11]
Deep Nuclei
There are four different deep nuclei of the cerebellum: the dentate, interpositus, fastigial, and vestibular nuclei. The dentate nucleus receives signals from the lateral purkinje cells, the interpositus nucleus receives signals from the intermediate purkinje cells, the fastigial nucleus receives signals from the medial purkinje cells, and the vestibular nucleus receives signals from the flocculonodular purkinje cells. The deep nuclei integrate the inhibitory signals from the purkinje cells and the excitatory signals from the mossy and climbing fibers to determine their output signals. [13] The dentate nucleus in particular is thought to be implicated in higher level cognitive processing. It is enlarged in primates and humans and not observed in non-mammalian species.
Glia
Glial cells of the cerebellum were described by Ramon y Cajal in 1911. He divided them into 3 main categories: the glia of the white matter, the astrocytes of the granule cell layer, and the Bergmann glia of the Purkinje cell layer.
Bergmann Glia
Also known as Goligi epithelial cells, Bergmann glia are unipolar astrocytes that have cell bodies located in the Purkinje cell layer and long processes projecting into the molecular layer. The Bergmann glia's processes interact with the dendrites of Purkinje cells at synapses with parallel and climbing fibers. Bergmann first characterized the long processes of cells he saw in the cerebellum of cats, dogs, and humans in 1857. Ramon y Cajal later described these cells as "epithelial cells with Bergmann fibers," giving the glia their name. https://link.springer.com/content/pdf/10.1046%2Fj.0022-7722.2002.00021.x.pdf
Oligodendrocytes
Oligodendrocytes are glia found primarily in the white matter of the cerebellum. These glial cells form the fatty myelin sheath that gives the white matter its color.
Cerebellar Nuclei
[14].
Early Brain Vesicles
Primary Brain Vesicles
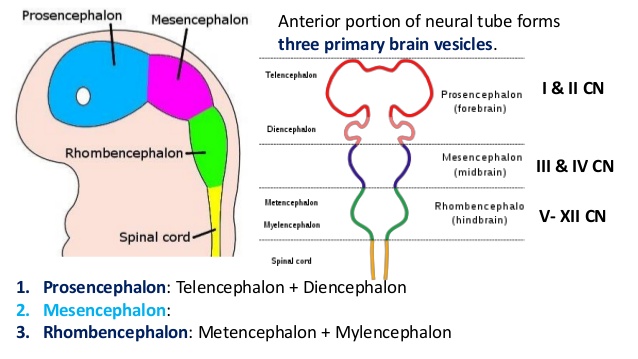
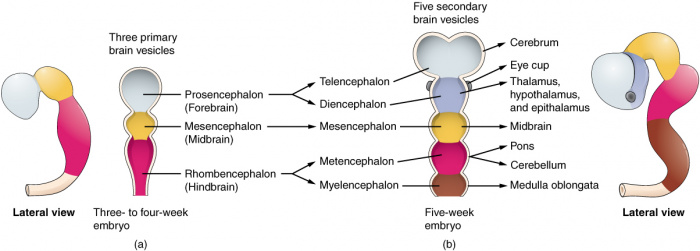
(Week 4) 3 primary brain vesicles are formed; forebrain (prosencephalon) midbrain (mesencephalon), and hindbrain (rhombencephalon) [15]
Secondary
(Week 5) 3 primary vesicles develop into 5 secondary vesicles [16];
- Prosencephalon develops into telencephalon (which includes the endbrain and cerebral hemispheres) and diencephalon (located between the brain and forms an optic outgrowth)
- Mesencephalon does not further develop into a secondary brain vesicle
- Rhombencephalon develops into the metencephalon (behind the brain), and myelencephalon (contains medulla)
Metencephalon
z5113034 The metencephalon refers to the embryonic neural structure that eventually gives rise dorsally, to the cerebellum and ventrally, to the pons. The metencephalon is the anterior part of the rhombencephalon (hindbrain) and differentiates from the posterior part of the rhombencephalon (myencephalon) at week 5 of development.
The dorsal surface is characterised by its highly folded folia separated by grooves termed sulci. The median area is referred to as the vermis, which eventually becomes the most superior aspect of the cerebellum. [17] The first structure that belies the future cerebellum are the rhombic lips that appear on the metencephalon of a 5-6 week old embryo. The rhombic lips are aptly rhombus-shaped and denote the perimeter between the roof plate and the main body of the rhombencephalon. The anterior pair of lips mark the site at which the cerebellum will develop. [18]
Cerebellum Development
(find images of the visualiation of the foetal cerebellum)
As the neural tube folds, the anterior portion develops the three brain vesicles; prosencephalon, mesencephalon and rhombencephalon. The rhombencephalon then further divides into the mesencephalic and myelincephalic vesicles on embryonic day 9. The neural tube failure to close then creates a gap along the dorsal sides and this produces a mouth-like structure as the tube bends to establish the pontine flexure. The pontine flexure further deepens bringing the mesencephalon (midbrain) closer to the primordium of the cerebellum (metencephalon); anterior aspects of the myelincephalon (brain stem) fold underneath developing the cerebellum plate [20] Further development of the cerebellum begins between days 40 and 45 and it arises mostly from the metencephalon however the rhombic lips also contributes. The roof plate which is derived from the dorsal part of the alar plate thickens during development to become the cerebellum. The regulation of patterning involved when he primary fissure deepens by the end of the third month and divides the vermis shows to be particularly important for development. The two lateral bulges are separated into the cranial anterior lob and caudal middle lobe. As the lobes divide further into lobules, fissures are formed and this continues throughout embryonic, fetal and postnatal life, thus increasing the surface area of the cerebellar cortex. The most primitive part of the cerebellum to form is the flocculonodular lobe, which is derived from separation of the first transverse fissure and this functions to keep connections with the vestibular system and it is also concerned with subconsciously controlling equilibrium. The flocculonodular lobe is separated from another crucial part of the cerebellum, corpus cerebelli, by the posterolateral fissure.
The cerebellum is connected to the brain stem via three pairs of peduncles and this allows the afferent and efferent pathways to enter and exit the cerebellum. Cerebellum afferent fibers can be grouped into two major types: mossy fibers and climbing fibres. Mossy fibres contribute to most of the afferent fibres in the cerebellum and they communicate with the cerebellar nuclei neurons and with Purkinje cells through granule cells embryonically, however postnatally they displace from Purkinje cells and synapse with their adult targets, the granule cell dendrites. Whilst mossy fibres originate from numerous sites in the nervous system, climbing fibers originate exclusively from the inferior olivary nucleus. Climbing fibers directly synapse with the cerebellar nuclei and Purkinje cells, relaying information to the cerebellum from several regions. The direction of these afferent fibres to their target neurons early in development are controlled by genes and molecules [21].
The cerebellum has a very basic structure consisting 2 principal classes of neurons and 3 layers, the first is a single layer of inhibitory Purkinje cells which are sandwiched between a dense layer of excitatory granule cells, and another molecular layer of granular cell axons and purkinje cell dendritic fibres. The granule cells receive inputs from outside the cerebellum and project these inputs to purkinje cells where the majority of these inputs are further projected to a variety of cerebellar nuclei in the white matter [22]. Within the layers there are some other main neuronal cell types, the stellate and basket cells are located in the molecular layer, whilst granule, golgi and unipolar brush cells are located in the granular layer (Figure 4) [23]. Among these principal neurons, there is a diverse set of interneurons which are responsible for coordinating the output of the Purkinje cells to the cerebellar nuclei.
The nuclei from the cerebellum are formed by a complex process of neurogenesis and neuronal migration. There are two types of grey matter in the cerebellum, the deep cerebellar nuclei and an external cerebellar cortex. There are 4 deep nuclei formed and the output of the cerebellar cortex are relayed through these nuclei, the ventricular layer produces 4 types of neurons that migrate to the cortex. The adjacent rhombic lips gives rise to cerebellar granule cells. [24]. Granule cells function in coordinating afferent input to and motor output from the cerebellum through excitatory connections with the Purkinje cells. Proper cerebellum function requires well-organised neuronal connections and the integration of afferent and efferent fibres throughout the cerebellar circuit [25]. The cerebellum functions in sensorimotor, balance control and vestibular ocular reflex, however recent studies have come out and shown that the cerebellum has a wide range of cognitive functions which include speech, memory and cognitive functions [26].
z5018156 - https://www.ncbi.nlm.nih.gov/pubmed/21295689
[27]
Cerebellum Developmental Weeks
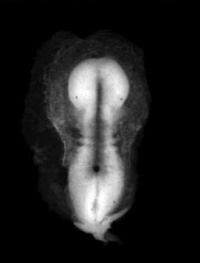
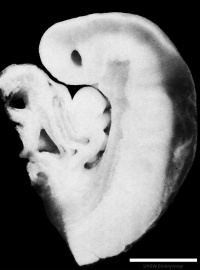 Sagittal view of superior end of embryo [28] | ||
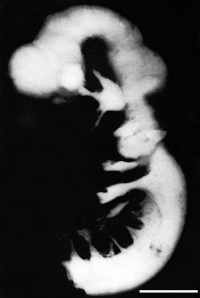 Lateral view of embryo central nervous system at 5 weeks [29] | ||
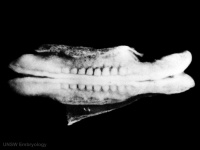
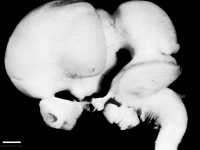 Left lateral view of embryonic CNS[32] 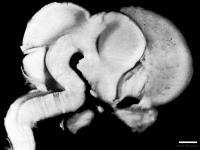 Left medial view of lateral embryonic CNS [33] | ||
Abnormalities
Abnormalities found within the posterior fossa of the cranium may affect the functioning and development of the cerebellum. The abnormalities affecting the cerebellum include Dandy-Walker-Malformation, Joubert Syndrome, Tecto-Cerebllar Dysraphism and Rhombencephalosynapsis (RS).
Dandy-Walker Malformation
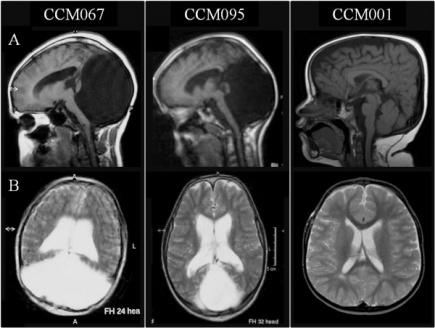
Dandy-Walker-Malformation (DWM) includes the incomplete development of the cerebellar vermis with cystic dilatation of the 4th ventricle and enlargement of the posterior fossa [36]. The cerebellar vermis (found at the medial, cortico-nuclear zone of the cerebellum[37]) may either be elevated or rotated upwards [36]. The communication of the 4th ventricle with the midline posterior fossa cyst can be seen on MRI scans of DWM [36]. Other structures that can be identified on MRI scans include upward displacement of the tentorium and anterolateral shift of cerebellar hemispheres [38].
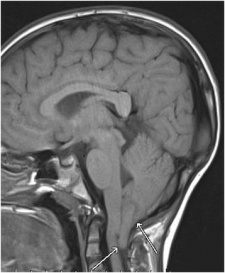
Brain MRIs. Brain imaging of patients with Dandy-Walker malformation (CCM067, CCM095) and patients with normal cerebellum and posterior fossa (CCM001). A: sagittal midline T1-weighted images. B: axial T2-weighted images. A subsection labeled “Reference” and under this the original image source, appropriate reference and all copyright information.[39]
Joubert Syndrome
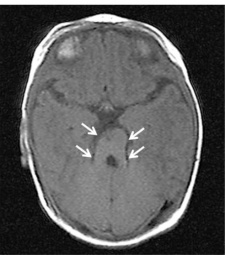
Joubert syndrome (JS) is described as a rare inherited genetic disorder characterised by lack of muscle coordination, intellectual disability, respiratory disturbances and abnormal eye movement [40]. On neuroimaging, the cerebellar vermis is identified to have gone through hypoplasia (underdevelopment) or dysplasia (abnormal development) [36]. It is also described as the "molar tooth sign" on brain imaging, which the "molar tooth" shaped is caused by the defects in the midbrain-hindbrain development.
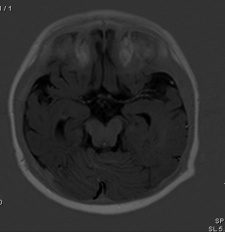
Brain Imaging of Joubert Syndrome: Cranial MRI showing “molar tooth sign” (arrows)[41]
Chiari Syndrome I-III
Chiari Syndrome I-III is when the structures within the posterior cranial fossa protrude into the spinal canal [42] which can affect the development and functioning of the cerebellum in the long term. Chiari Syndrome can be classified into four types. The downward shift of the cerebellar tonsils to beneath the foramen magnum is type 1 and the downward movement of the vermis, medulla oblongata and pons with the fourth ventricle is type 2 [42]. When majority of the cerebellum lies in the foramen magnum it is type 3 and when it is completely in the foramen magnum (and therefore cannot develop normally) it is type 4[42].
Cerebellar tonsils herniation on magnetic resonance imaging: Chiari malformation type I[43]
Rhombencephalosynapsis
Rhombencephalosynapsis is a rare cerebellar defect whereby there is dorsal fusion of the cerebral hemispheres, fusion of dentate nuclei and superior cerebellar peduncles as well as the agenesis of the cerebella vermis [44]. Like other mentioned abnormalities there is development delay because of the underdeveloped cerebellum.
Partial Rhombencephalosynapsis with fused upper parts of the cerebellum[45]
Current Research
Animal Models
Isthmic Organiser
Throughout history there have been many investigations on the cerebellum and how it has developed through research involving chicken embryos and mice. As previously mentioned the neural plate closes to form the neural tubes which have anterior-posterior (AP) and dorsal-ventral (DV) axes. Earlier experiments involving chick-quail chimera suggested that the cerebellum was derived from both midbrain and hindbrain. However, through successive gene expression and fate mapping studies, it was discovered that the anterior-most rhombomere of the hindbrain is where the cerebellum is formed from [46]. Once the axes are formed the isthmic organiser (IsO) is formed which plays a vital role in establishing the anterior limit of the cerebellar territory. The IsO in other words is the mid-hindbrain boundary [47]. However, the IsO does not position itself without the help of transcription factors. Studies of mouse and chicken embryos have shown that two homeo domain-containing transcription factors Otx2 and Gbx2 have an important role in positioning the isthmic organiser [47]. Surgical movement of the isthmic tissue to more anterior or posterior regions of the neural tube of 10-somite stage chick embryos led to ectopic midbrain and cerebellar structures[46], indicating that where the IsO is placed is an important factor in establishing where the cerebellum positions. z5018156
Historic Images
Glossary
References
- ↑ http://www.sciencedirect.com/science/article/pii/S1364661398012108
- ↑ http://teachmeanatomy.info/neuro/structures/cerebellum
- ↑ https://commons.wikimedia.org/wiki/File:CerebellumDiv.png
- ↑ http://teachmeanatomy.info/neuro/structures/cerebellum/
- ↑ https://www.ncbi.nlm.nih.gov/books/NBK11042/
- ↑ https://www.ncbi.nlm.nih.gov/pubmed/2535662
- ↑ https://www.researchgate.net/publication/309323728_Arteries_and_Veins_of_the_Cerebellum
- ↑ Alberts B, Johnson A, Lewis J, et al. Molecular Biology of the Cell. 4th edition. New York: Garland Science; 2002. Neural Development.
- ↑ https://discovery.lifemapsc.com/library/review-of-medical-embryology/chapter-26-embryonic-folding-and-flexion-of-the-embryo'
- ↑ http://neurotransporter.org/Cerebellum.html
- ↑ https://link.springer.com/referenceworkentry/10.1007%2F978-94-007-1333-8_31
- ↑ <pubmed>25336734</pubmed>
- ↑ http://www.neuroanatomy.wisc.edu/cere/text/P5/intro.htm
- ↑ Knierim, J. (n.d.). Chapter 5: Cerebellum. Retrieved 10 3, 2017, from Neuroscience: http://neuroscience.uth.tmc.edu/s3/chapter05.html
- ↑ https://www.karger.com/Article/FullText/334842
- ↑ http://onlinelibrary.wiley.com/doi/10.1111/j.1447-0756.2008.00964.x/full
- ↑ Gerardo De Iuliis PhD, Dino Pulerà MScBMC, CMI, in The Dissection of Vertebrates (Second Edition), 2011
- ↑ Bruce M. Carlson MD, PhD, in Human Embryology and Developmental Biology (Fifth Edition), 2014
- ↑ <pubmed>21309081</pubmed>
- ↑ <pubmed>7605067</pubmed>
- ↑ <pubmed>4552263</pubmed>
- ↑ <pubmed>25336734</pubmed>
- ↑ <pubmed>4552263</pubmed>
- ↑ Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R. and Francis-West, P.H., 2014. Larsen's Human Embryology E-Book. Elsevier Health Sciences.
- ↑ <pubmed>4552263</pubmed>
- ↑ <pubmed>3213765</pubmed>
- ↑ <pubmed>2775156</pubmed>
- ↑ Hill, M.A. 2017 Embryology Human Stage13 sagittal upper half01.jpg. Retrieved October 4, 2017, from https://embryology.med.unsw.edu.au/embryology/index.php/File:Human_Stage13_sagittal_upper_half01.jpg
- ↑ M.A. 2017 Embryology Human Stage14 neural01.jpg. Retrieved October 4, 2017, from https://embryology.med.unsw.edu.au/embryology/index.php/File:Human_Stage14_neural01.jpg
- ↑ <pubmed>21380713</pubmed>
- ↑ <pubmed> 2252222</pubmed>
- ↑ Hill, M.A. 2017 Embryology Human Stage21 neural01.jpg. Retrieved October 4, 2017, from https://embryology.med.unsw.edu.au/embryology/index.php/File:Human_Stage21_neural01.jpg
- ↑ Hill, M.A. 2017 Embryology Human Stage21 neural02.jpg. Retrieved October 4, 2017, from https://embryology.med.unsw.edu.au/embryology/index.php/File:Human_Stage21_neural02.jpg
- ↑ <pubmed>21380713</pubmed>
- ↑ <pubmed>21380713</pubmed>
- ↑ 36.0 36.1 36.2 36.3 <pubmed>22108217</pubmed>
- ↑ <pubmed> PMC3179064</pubmed>
- ↑ <pubmed> 21093738</pubmed>
- ↑ <pubmed> PMC3667004</pubmed>
- ↑ <pubmed>PMC2913941</pubmed>
- ↑ <pubmed>PMC3896311</pubmed>
- ↑ 42.0 42.1 42.2 <pubmed>18762395</pubmed>
- ↑ <pubmed>PMC4279813</pubmed>
- ↑ <pubmed>18155944</pubmed>
- ↑ <pubmed>PMC3447431</pubmed>
- ↑ 46.0 46.1 <pubmed> 14567957</pubmed>
- ↑ 47.0 47.1 <pubmed>21309081</pubmed>
https://link.springer.com/referenceworkentry/10.1007%2F978-94-007-1333-8_9