2017 Group Project 5: Difference between revisions
| Line 240: | Line 240: | ||
'''3. GATA6''' | '''3. GATA6''' | ||
GATA6 is apart of the GATA family of transcription factors which comprises of a conserved zinc-finger motif | GATA6 is apart of the GATA family of transcription factors which comprises of a conserved zinc-finger motif and modulates gene expression by binding to A/TGATA/G sequence. Through in situ hybridization, it has been shown that GATA6 is important in visceral endoderm differentiation and expression in embryonic bronchial epithelium. To analyse the role of GATA6 in fetal lung development, expression patterns of lungs in early development were examined suggesting that this transcription factor is involved in branching morphogenesis. Chimeric embryos were produced from ES cells and lungs were dissected at different E days. In the chimeric lungs of mouse in earlier E days had smaller and less lung buds compared to non-chimeric lungs and had fewer branches. In defective GATA6 endoderm, the abnormal phenotype displayed was due to abnormal branching morphogenesis as a result of delayed epithelial differentiation. | ||
==Abnormal development== | ==Abnormal development== | ||
Revision as of 15:16, 5 October 2017
| 2017 Student Projects | |||
|---|---|---|---|
|
Lungs
On this page, we will be exploring the development of the lungs through the stages of embryonic growth.
Lung Anatomy
To understand lung embryology, we must first understand what a fully grown adult lung looks and functions like, anatomically and histologically.
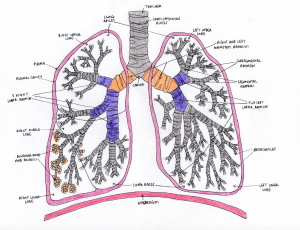
This diagram shows the anatomy of the adult lung. The larger right lung consists of 3 lobes (superior, middle & inferior) whereas the smaller left lung has 2 lobes (superior & inferior). The trachea splits into the left and right bronchi in the lungs. The bronchi are hollow tubes composed for hyaline cartilage and are lined with respiratory epithelium which is ciliated pseudostratified epithelium.
Each lung receives air through these primary bronchi. As these primary bronchi enter the lungs, they branch into smaller secondary bronchi which carry air to each lobe of the lungs.
The respiratory epithelium lining the bronchi contains cilia and goblet cells. The cilia act as hair-like projections and goblet cells secrete mucus to coat the lining of the bronchi. Cilia works to push mucus secreted by goblet cells out of the lungs. In this way, any bacteria or viruses that enter the lungs stick to the mucus and are expelled out of the respiratory tract.
The small bronchioles branch off from the tertiary bronchi. Bronchioles are made of elastin fibres and smooth muscle tissue. The bronchioles branch into even smaller terminal bronchioles. They are the smallest tubes in lungs and terminate at the alveoli. The terminal bronchioles are capable of dilating and contracting to control the amount of airflow to the alveoli.
Lung Histology
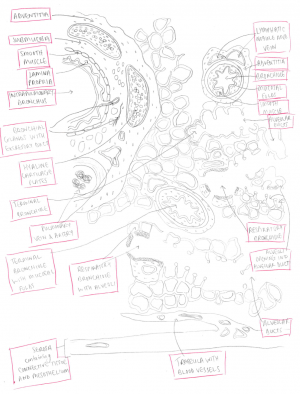
The diagram shows the histology of the lung.
The alveoli are the functional units of the lungs which allow for gas exchange to occur between air in the lungs and blood in the capillaries of the lungs. Alveoli occur in small clusters called alveolar sacs at the end of a terminal bronchiole. The walls of alveoli contain simple squamous epithelial cells called alveolar cells with type I and type II alveolar cells.
Lung Cardiovasculature
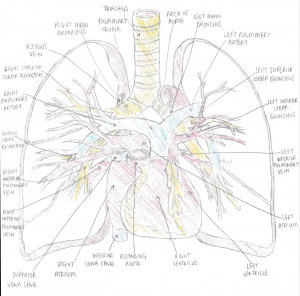
This diagram shows the connection between the heart and the lungs, showing the pulmonary arteries and veins in the lungs. The heart and lungs are interdependent in embryonic development. Since vascular development exists prior to respiratory development, the way cardiovasculature interacts with lung anatomy and functioning is extremely significant. [1]
The main pulmonary artery begins in the right ventricle and divides into two branches: the right and left pulmonary arteries. These arteries are responsible for carrying deoxygenated blood to their respective lobes.
The pulmonary veins, however, carry oxygenated blood from the lungs to the rest of the body. There are four pulmonary veins.
Developmental origin overview
A brief summary of the embryonic development of the lungs:[2]
Day 22 - Lung development begins. Respiratory diverticulum forms from the ventral outpouching of the endodermal foregut.
Day 26-28 – Respiratory diverticulum undergoes bifurcation into right and left primary bronchial/lung buds.
Weeks 5-28 – Primary bronchial bugs undergo sixteen rounds of branching to generate the respiratory tree of the lungs. This produces secondary bronchial buds, the lung lobes, tertiary bronchial buds, bronchopulmonary segments of the mature lung, terminal bronchioles and respiratory bronchioles.
Week 36-Birth – Terminal branches of lungs are in a dense network of capillaries and are called terminal sac (primitive alveoli). They then begin to differentiate into mature alveoli which continue to form until 8 years old.
Developmental timeline
| The first stage of fetal lung development is the embryonic stage where the first structural features of the lung can be seen. It is identified by the emergence of lung buds in the ventral wall of the foregut; these lung buds will later divide to form lobar divisions.The endoderm is responsible for the epithelium of the lung buds which will later differentiate into specialised respiratory epithelium. The mesoderm is responsible for the mesenchyme surrounding the lung buds. | Christian Pander, Karl Ernst von Baer, and Heinrich Rathke were among the first to use a new wave of technology in embryology; involving the use of new staining techniques and improved microscopy. Their findings together identified the presence of primary germ layers (ectoderm, endoderm and mesoderm) and the development of early organs. [3] | 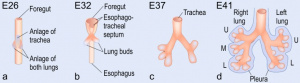 Early lung development demonstrating lung buds and their growth into left and right lungs [4] | |
| The Pseudoglandular stage of lung development is named after its histological appearance which is much like compound tubular glands. This is due to the dichotomous branching of conducting airways in the lung which is repeated up until the 16th week where they will be fully formed [5]. However, these branchings do not yet have functional alveoli; instead, specialised respiratory cells begin to develop. Crucially important to the branching of the airways is the presence of bronchial mesoderm as proven by Spooner & Wessells in 1970 (see history). | In 1970, an experiment by Spooner and Wessells demonstrated the need for the presence of surrounding bronchial mesoderm for adequate bronchial branching. [6]. This was done through in vitro experiments with mice.
Additionally, In 1976 Masters published his work on the importance of epithelial-mesenchymal interactions during lung development. [7] This significance of this work is the discovery that the bronchial epithelium lining the primitive airways interacts with the mesenchyme, to then differentiate into specialised alveolar epithelium. |
 Epithelial structures in Pseudoglandular, Canalicular and Saccular stages of lung development [8] | |
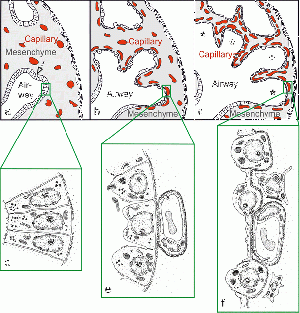
| The canalicular stage is an important step in the development of the lung parenchymal cells. The formation of these cells as well as the vascularization and angiogenesis of surrounding structures is prominent in this stage. The beginning of the process producing surfactant in the lungs starts here but is more evidently seen in the Saccular stage. Additionally, there is differentiation of the specialised pulmonary epithelium into peripheral squamous cells and proximal cuboidal cells which preceeds the formation of the air-blood tissue barrier. [9] At the end of this stage it is possible to distinguish between conducting and respiratory airways. | In 1971, Edward A. Boyden published an article detailing for the first time the detailed structure of a pulmonary acinus. The presence of these acini are important for the distinction between conducting and respiratory airways in the Canalicular stage. | 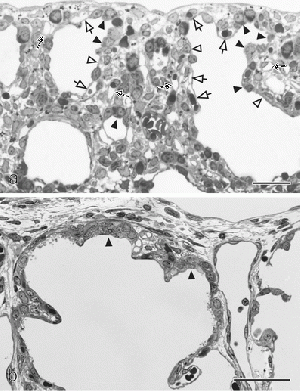 Change in pulmonary epithelium consequently leading to formation of the air-blood barrier in the developing lung [10]
| |
| The Saccular stage shows a marked decrease in the interstitium, making way for growing peripheral airways or saccules. The widening of these saccules marks the regions of future gas exchange in the lung. Capilliaries surround these saccules to form a capillary bilayer in the intersaccular primary septa [11]. Throughout this stage, Type I and Type II pneumocytes can be identified. Type II pneumocytes have lamellar bodies that contain surfactant; a phospholipoprotein which is essential for the inflation of the lungs and to prevent their collapse. It is important for the first breath of a newborn; the higher the number of lamellar bodies corresponds to increased stability in lung functioning at birth. Babies born prematurely that have reached this stage of lung development have a chance of surviving, directly relating to the number of lamellar bodies present.[12] | Historically, it was believed lung structure was fully developed by the time of birth and that all that remained was lung expansion. | image | |
| This is the final stage of lung development that stretches from late fetal to early childhood. This stage involves the formation of a secondary septa and its fusing with the primary septa to produce a more effective single-layered capillary network. This is part of a process called microvascular maturation. [13] Alveolarization occurs from this stage and continues post-natally. | The Alveolarization stage of lung development has been increasingly well documented in the recent years corresponding to increasing technological advances. | image |
Structure of respiratory network
Physiologically, the organ can be divided into two parts:
The Conducting system – consists of all tubular structures including the larynx, trachea and bronchi. (image)
The Functional unit – An alveolus containing specialised epithelial cells where gas exchange occurs.
By week 8 of development, the lungs are in the development of the pseudoglandular stage (refer to developmental timeline). The three germ layers formed in gastrulation all contribute to the development of the lung, including signalling for a cascade of events as well as branching of the bronchiole.
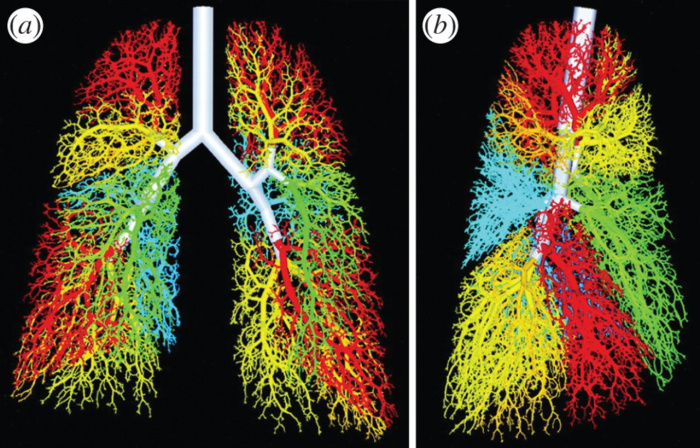
A three-dimensional fractal model of an airway tree with 54 611 branches; branches distal to different segmental bronchi are shown in same colour as segmental bronchus. (a) Anterior view and (b) right lateral view
The Conducting System
Branching morphogenesis is observed in many organ systems, including the lungs. It has been of great interest and has been studied in great detail. The bronchial tree arises from the sequential use of three simple modes of branching.
These modes include:
1. Domain branching
2. Planar bifurcation
3. Orthogonal bifurcation
Trifurcations have also been observed as contributing the backbone of the respiratory tree, however it is not as prevalent.
These branching structures involve are regulated by a network of signalling factors. To form a branched structure, signalling molecules have to form a pattern in space that precedes bud outgrowth. This is assisted with the molecule FGF10 and will be further explored. For direct elongation, FGF10 has a higher concentration at the distal tip of the lung bud allowing direct elongation of the structure. In terminal branching, there is a split localisation of FGF10 and results in a terminally branched structure. For lateral branching, FGF10 is restricted to being a single spots on the side allowing the structure to grow laterally. FGF10 appears to drive outgrowth of lung buds, as well as other organs.
With regards to early lung development, transitions from one mode of branching to the other occur to ultimately build the lung tree. In the domain branching mode, the lung bud elongates and new buds first appear on one side of the stalk, perpendicular to the main axis on either sides of the stalk. Domain branching is used to build the backbone of the respiratory tree.
Planar and orthogonal bifurcations represent two consecutive rounds of branching, however they differ in the second round as they branch in the same plane as planar bifurcations. Planar and orthogonal bifurcations create lobes surfaces and fill the interior. The branching process is controlled by genetic information and is tightly regulated. In the lung, dichotomous branching gives rise to two daughter branches with a smaller diameter than the mother branch.
In earlier studies, lung branching has been proposed to be influenced by the viscosity of amniotic fluid and the mesenchyme, separated by a ‘skin’ of surface tension, the epithelium. However, more recently this has been negated as branching can occur without a mesenchyme, without growth and the robustness of the branching process suggests that it is a highly controlled process. Signalling factors play a key role in branching morphogenesis. A recent study has shown that branching can still occur in the absence of mesenchyme, only if the appropriate signalling factors are added.
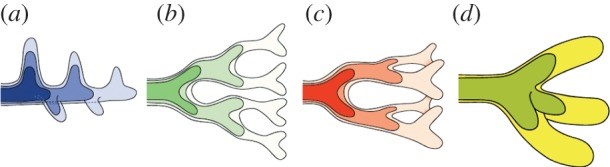
This image is a stylised representation of the modes of lung branching.
a) lateral branching, b) planar bifurcation, c) orthogonal bifurcation, d) trifurcation
This image is a stylised typical developmental branching pattern over time in a lung bud.
Alveolus: the functional unit
The alveolus are small "sac-like structures" in which gas exchange occurs across. Within the alveolus there are two types of cells that line the sacs. These are type I and type II alveolar cells, these cells must differentiate in a process known as alveolar differentiation and are both crucial for gas exchange.
Type I: Type I cells are flat and cover more than 90% of the alveolar surface in which gases diffuse across.
Type II: Type II cells are cuboidal shaped and synthesise pulmonary surfactants that hydrate and prevent the alveolar collapsing into itself by reducing surface tension.
It was previously thought that alveolar type I cells arise from type II cells, however more recent studies suggest that they both arise from a bipotent progenitor. After birth, type I cells are derived from rare, self-renewing, mature, long-living alveolar type II cells, that produce slowly, gradually expanding the the clonal foci of alveolar renewal.
Alveolar differentiation occurs in the distal region of the developing airway tree. Studies have found that over development there is an increase in alveolar differentiation and a decrease in branching morphogenesis as well as alveolar differentiation expanded distally over time.
ECAD (a cell junction protein stain) staining showed that undifferentiated cells at the branch tips were clustered and cuboidal, whereas differentiated alveolar cells (type1 and type 2) were at the non-branch tips and were flat and isolated cuboidal cells. This study suggests that in late lung stages branching continues at the distal edge of the airway tree whilst leaving behind cells undergoing alveolar differentiation. A genetic factor involved in alveolar differentiation is expression of hyperactive Kras, which expands the branching of lungs, however reducing the differentiation of alveolar cells and suppresses cell flattening, a hallmark of alveolar cells.
One study, suggested that mothers who are overweight when pregnant can potentially effect the fetus in terms of lung development and air breathing at birth. The study looked at sheep who over-nourished their young, and found that the after birth, the lambs who had overweight mothers had a reduced capacity for surfactant production as well as altered transport mechanisms within cells. This research can assist with the diagnosing and preventing potentially detrimental conditions that can occur during pregnancy.
Developmental signalling processes
The development of a mammalian lung is a multi-step and highly complex process involving signalling pathways. This hierarchy process, referred to as branching morphogenesis is essential to generate numerous airways and gas-exchanging units, and is critically regulated by interactions of signalling pathways in the epithelium and mesenchyme.
Several growth factors and chemical signals have been identified to influence lung development. The main signalling molecules include:
1. Fibroblast Growth Factor (FGF10) - serves as a signalling cue for epithelium outgrowth in the mesenchyme.
2. Sonic Hedgehog (SHH) - produced in the epithelium and is involved in regulating FGF10 expression, leading to the intertubular mesenchyme to facilitate growth and formation of epithelial buds.
3. Sox9 - promotes proper branching morphogenesis by balancing the proliferation and differentiation of epithelial tip progenitor cells, whilst regulating the extracellular matrix. It is expressed at the distal tips of branching epithelium and controls multiple aspects of lung branching.
4. Heparan Sulfate Glycosaminoglycans (HS-GAG) - interacts with SHH in order to produce epthelial cells and lung morphogenesis. There is still a lack of understanding in the role that HS plays in mammalian lung development.
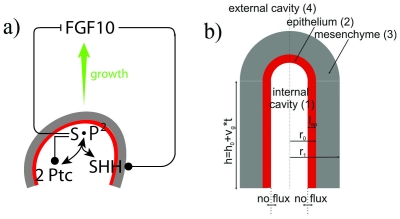
During branching morphogenesis, the distal tips of the branching epithelium contain a distinct population of progenitor cells that give rise to all epithelial types in early lung development, but become developmentally restricted after embryonic day 16.5 in mice. Because this epithelium is highly proliferative population of cells, a balance between differentiation and proliferation must be maintained during lung development. After embryonic day 16.5, Sox9 is down-regulated as differentiation begins, with Sox9 being expressed in the highest concentration where two new buds will form, and lowest where the cleft forms. Two core signalling proteins consist of FGF10 and SHH. FGF10 has been shown to induce outgrowth of lung buds. FGF10 and SHH also engage in a negative feedback loop, in that FGF10 signalling induces SHH expression in the epithelium. This expression of SHH causes a repression in FGF10 expression in the mesenchyme of the tissue.
a) FGF10 is transcribed at high levels in the distal mesenchyme (grey area). FGF10 stimulates SHH expression in the epithelium (red), thus promoting proliferation and outgrowth shown by the green arrow. b) The epithelium and mesenchyme are shown in red and grey respectively, showing the outgrowth of mesenchyme after the signalling pathway between both FGF10 and SHH.
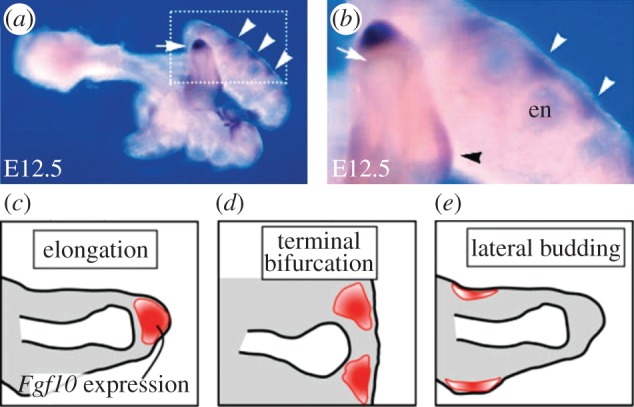
This image shows three spatial locations of where concentrations of FGF10 are expressed, in order to generate different branching modes c) elongation d) terminal bifurcation e) lateral budding. This model is used in modern research, outlining the development of the conducting system of the lung
A recent study found that the production of a biologically active form of SHH was decreased after a loss of HS-GAG in the epithelium, highlighting the crucial requirement of HS-GAG in SHH-producing epithelial cells to maintain SHH signalling activity in the event of lung morphogenesis.
The branching pattern and branching rate are sensitive to the changes in the mesoderm shape and volume. The growth of the epithelial tree is likely to adapt to fill the available space in the mesenchyme.
Current understandings and areas of research
include any relevant articles
Movies
|
Early embryo (stage 13)
|
|
Late embryo (stage 22)
|
Animal models
Mouse models
Key pathways that maintain the mesenchymal-epithelial interactions that allow normal embryological lung development: VEGF, Bmp, Wnt.... Sox 2
1. Wnt7b/Fzd2
Wnt signaling pathway involves the binding of the Wnt ligand to the Frizzled family receptor that can elicit different effects depending on the type of Wnt pathway (noncanonical/calcium, canonical, noncanonical planar cell polarity). Canonical Wnt pathway has a signalling role in embryonic lung development and with the action of β-Catenin by trans-activating the LEF/TCF pathway, which advances epithelial proliferation, differentiation and tissue development. On the other hand, the noncanonical pathway has been suggested to be a modulator of epithelial cell shape and cytoskeletal structure. To examine the function of Fzd2, mouse models were developed by in attempt to delete the Fzd2 gene by crossing over Fzd2 flox/flox allele mice with Shh cre allele mice. In the embryonic chimeric mice, at day 12.5, expression of Fzd2 was absent and by day 14.5 large cysts were observed in the distal lung region. It was also noted that in Fzd2 mutant mice, airways were shorter and wider and presence of planar bifurcation, leading to large cysts rather than distal branching. There were also molecular changes in signalling processes for lung branching morphogenesis involving the factors Fgf10, Bmp4, Fgfr2, and Shh. From the data, it is evident of Wnt7b/Fzd2's role in the modulation of lung epithelial cell shape and initiation of branch points.
2. Sox 2
Sox 2 - branching morphogenesis and epithelial cell differentiation. - Sox 2 is a Sry related HMG - There is an increased expression of Sox 2 in unbranched lung epithelium and is absent in branching regions - Development of a transgenic mice with doxycycline inducible Sox2 continuously expressed in their lung epithelium - Results showed a diminution of branching airways and alveoli bronchiolization - Driving precursor-like cells to a committed state (cGRP positive neuroendocrine cells and ΔNp63 isoform expressing (pre-) basal cells)
3. GATA6
GATA6 is apart of the GATA family of transcription factors which comprises of a conserved zinc-finger motif and modulates gene expression by binding to A/TGATA/G sequence. Through in situ hybridization, it has been shown that GATA6 is important in visceral endoderm differentiation and expression in embryonic bronchial epithelium. To analyse the role of GATA6 in fetal lung development, expression patterns of lungs in early development were examined suggesting that this transcription factor is involved in branching morphogenesis. Chimeric embryos were produced from ES cells and lungs were dissected at different E days. In the chimeric lungs of mouse in earlier E days had smaller and less lung buds compared to non-chimeric lungs and had fewer branches. In defective GATA6 endoderm, the abnormal phenotype displayed was due to abnormal branching morphogenesis as a result of delayed epithelial differentiation.
Abnormal development
Newborn Respiratory Distress Syndrome
Newborn respiratory distress syndrome (NRDS) is usually seen in preterm infants that are born before 34 weeks' gestation. They usually present with respiratory distress symptoms such as tachypnea, grunting and cyanosis immediately after birth. A chest radiography would show a diffuse ground-glass appearance with air bronchograms and hypoexpansion. Usually, with treatment, infants with NDRS can recover wihout much long-term effects. In certain cases, bronchopulmonary dysplasia can occur and this will be talked about more in the next section.
Hyaline Membrane Disease
NRDS is often also called Hyaline Membrane Disease (HMD), as NRDS is usually due to a deficiency of pulmonary surfactant [14] and the terminal bronchioles and alveolar ducts are seen to be lined with hyaline membranes. If surfactant is absent in infants, the alveoli would be more likely to collapse and this would explain most of the clinical features of NRDS. [15]
Prematurity is the most significant cause of the deficiency in surfactant. At around 23 weeks of gestation, surfactant is first seen in small amounts, and only at 30 weeks can surfactant in the lungs be easily detectable. Usually before 34 weeks' gestation, there is insufficient surfactant for the lungs to work properly. [15] Also, the mechanism to replace surfactant might be too immature in preterm babies so when surfactant is lost during breathing, there is a lack of production to replace it.[15] Another important cause is destruction of surfactant in the lungs by asphyxia before, during or after birth. This results in either the destruction of surfactant-producing alveolar cells or allow for the secretion of fibrinogen, which is known to inactivate surfactant. [15]
Bronchopulmonary dysplasia
Bronchopulmoary dysplasia (BPD) is described as a chronic lung condition derived from the disruption of normal pulmonary vascular and alveolar growth in infants born less than 28 weeks' gestation.[16] Initially, BPD was said to be caused by treatment of NDRS by using mechanical ventilation. This ventilation was used to provide oxygen to infants who could not breathe on their own. However, a variety of factors from ventilation, such as infection, inflammation and oxygen toxicity, could stunt the postnatal maturation of the lungs, resulting in blunted alveolarization and dysmorphic pulmonary vasculature.[17] In recent times, antenatal and perinatal factors that might disrupt lung development in extremely premature infants are also said to be the cause of BPD.[18]
Meconium Aspiration Syndrome
Meconium aspiration syndrome (MAS) occurs in about 1% of all deliveries and usually affects term and post-term newborns. Meconium is a mixture of various substances that arises from the gastrointestinal tract such as digestive juices, bile, mucus, and cellular debris.[19] It is usually sterile but can result in inflammation, infection and pneumonia. Since meconium is usually hidden from the infant's immune system in the digestive tract, it is not recognised by the immune cells when aspirated into the lungs. Instead, with many endogenous signals from cellular debris in the meconium, these immune cells recognise them as "damaged self", resulting in inflammation in the lungs. [20]
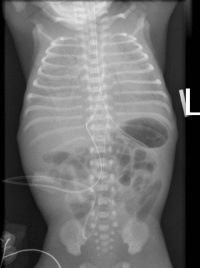
Meconium-induced inflammation is often made worst when bigger particles in the meconium cause partial or complete obstruction of the airways, resulting in severe respiratory failure. Symptoms of MAS are similar to that of NDRS with tachypnea, grunting and cyanosis at birth.[21] Chest radiography often shows diffused bilateral fluffy densities as seen in the Image.[19][21]
CPAM (Congenital Pulmonary Airway Malformation)
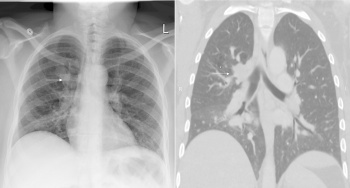
Congenital Pulmonary Airway Malformation (CPAM), previously known as congenital cystic adenomatoid malformation (CCAM), is a developmental abnormality of the lower respiratory tract.[22] It affects approximately 1 in 30,000 live births and is the most common lung lesion that is diagnosed at 18-20 weeks of gestation by prenatal screening.[23] CPAM is described as the absence of normal alveoli and extreme proliferation and cystic dilation of the terminal respiratory bronchioles. These cysts located in the bronchioles are often lined with different types of epithelial lining, such as ciliated, cuboidal or columnar cells. They lack normal structure and there is usually an absence of cartilage in the bronchioles.[24] The appearance of these cysts are said to be a result of unusual airway patterning and branching during lung morphogenesis.[23]
There are 5 types of CPAM, with Type 3 being adenomatoid and Types 1, 2 and 4 being cystic.[24] They are usually characterised in the following way.[23]
- Type 0- Acinar dysplasia
- Type I- Multiple large cysts or a single dominate cyst
- Type II- Multiple evenly spaced (sponge-like appearance)
- Type III- Bulky firm mass (adenomatoid appearance)
- Type IV- Peripheral cyst type
Out of all, Type II CPAM is more commonly seen to be associated with other congenital defects.[24] Most people who have CPAM are usually diagnosed in the perinatal period when they present with respiratory distress symptoms such as those seen in babies with NRDS.[22] In rare cases, they go undiagnosed until adulthood where symptoms arise as seen in the image of the chest radiography of a 36-year old woman diagnosed with CPAM.
Cystic Fibrosis
Cystic fibrosis (CF) is a disease caused an extreme functional deficiency of the cystic fibrosis trans-membrane conductance regulator (CFTR) protein. It is found in high numbers in epithelial cells that line the cylindrical structures of tissue that often secrete fluids rich in mucus, such as the pancreas and the lungs. Out of all the tissues, the airways have one of the highest amount of CFTR. With the absence of functional CFTR in the lungs, there is defective chloride and bicarbonate secretion into the alveolar secretions. This causes the pH of the airway surface fluid to drop and also prevents secretion of mucus strands from glands. This results in a condition known as CF lung disease. [25]
Cystic Fibrosis Lung Disease
This disease is identified by early colonisation and infection of the airways. Due to the acidic conditions of the CF airway surfaces, there is often a significant flaw in the baby's anti-bacterial defences. This leads to a very rapid onset of infection and the resulting inflammatory response is very critical.[25] The usual culprits are Staphylococcus aureus and Pseusdomonas aeruginosa, which when mucoid, becomes the dominant pathogen that attacks CF lung tissues.[26] The chronic airway and systemic inflammation that occurs as a result would then lead to tissue destruction and respiratory failure.[25] In fact, respiratory failure secondary to lung damage is the usually cause of death for CF patients with most of the damage caused arising from the inflammation instead of the actual bacteria.[26]
Future questions
Glossary
References
[27] [28] [29] [30] [31] [32] [33]
- ↑ <pubmed>PMC3641728</pubmed>
- ↑ Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R., Francis-West, P.H. & Philippa H. (2015). Larsen's human embryology (5th ed.). New York; Edinburgh: Churchill Livingstone.
- ↑ Gilbert SF. Developmental Biology. 6th edition. Sunderland (MA): Sinauer Associates; 2000. Comparative Embryology. Available from: https://www.ncbi.nlm.nih.gov/books/NBK9974/
- ↑ Schittny, J. C. (2017). Development of the lung. Cell and Tissue Research, 367(3), 427–444. http://doi.org/10.1007/s00441-016-2545-0
- ↑ DiFiore, J., & Wilson, J. (1994). Lung Development. Seminars in pediatric surgery, 3(4), 221.
- ↑ Spooner, B. S., & Wessells, N. K. (1970). Mammalian lung development: Interactions in primordium formation and bronchial morphogenesis. Journal of Experimental Zoology, 175(4), 445-454. doi:10.1002/jez.1401750404
- ↑ Masters, J. R. W. (1976). Epithelial-mesenchymal interaction during lung development: The effect of mesenchymal mass. Developmental Biology, 51(1), 10. doi:https://doi.org/10.1016/0012-1606(76)90125-1
- ↑ Schittny, J. C. (2017). Development of the lung. Cell and Tissue Research, 367(3), 427–444. http://doi.org/10.1007/s00441-016-2545-0
- ↑ Warburton, D., El-Hashash, A., Carraro, G., Tiozzo, C., Sala, F., Rogers, O., … Jesudason, E. (2010). Lung Organogenesis. Current Topics in Developmental Biology, 90, 73–158. http://doi.org/10.1016/S0070-2153(10)90003-3
- ↑ Schittny, J. C. (2017). Development of the lung. Cell and Tissue Research, 367(3), 427–444. http://doi.org/10.1007/s00441-016-2545-0
- ↑ https://www.ncbi.nlm.nih.gov/pubmed/6370120
- ↑ http://www.columbia.edu/itc/hs/medical/humandev/2004/Chpt12-LungDev.pdf
- ↑ Schittny, J. C. (2017). Development of the lung. Cell and Tissue Research, 367(3), 427–444. http://doi.org/10.1007/s00441-016-2545-0
- ↑ <pubmed>4984152</pubmed>
- ↑ 15.0 15.1 15.2 15.3 <pubmed>PMC1840752</pubmed>
- ↑ <pubmed>24666156</pubmed>
- ↑ <pubmed>26361876</pubmed>
- ↑ <pubmed>4469359</pubmed>
- ↑ 19.0 19.1 <pubmed>27486480</pubmed> Cite error: Invalid
<ref>tag; name 'PMID26760414' defined multiple times with different content - ↑ <pubmed>25721501</pubmed>
- ↑ 21.0 21.1 <pubmed>PMC2666857</pubmed>
- ↑ 22.0 22.1 <pubmed>PMC4821328</pubmed>
- ↑ 23.0 23.1 23.2 <pubmed>27070354</pubmed>
- ↑ 24.0 24.1 24.2 <pubmed>24715554</pubmed>
- ↑ 25.0 25.1 25.2 <pubmed>25814049</pubmed>
- ↑ 26.0 26.1 <pubmed>26003065</pubmed>
- ↑ <pubmed>24499815</pubmed>
- ↑ <pubmed>PMC3787747</pubmed>
- ↑ <pubmed>25114215</pubmed>
- ↑ <pubmed>18374910</pubmed>
- ↑ <pubmed>11171334</pubmed>
- ↑ <pubmed>PMC3839746</pubmed>
- ↑ <pubmed>28759122</pubmed>