2015 Group Project 6
| 2015 Student Projects | ||||
|---|---|---|---|---|
| 2015 Projects: Three Person Embryos | Ovarian Hyper-stimulation Syndrome | Polycystic Ovarian Syndrome | Male Infertility | Oncofertility | Preimplantation Genetic Diagnosis | Students | ||||
| 2015 Group Project Topic - Assisted Reproductive Technology | ||||
| This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
Preimplantation Genetic Diagnosis and Preimplantation Genetic Screening
Introduction
Preimplantation genetic diagnosis (PGD) and preimplantation genetic screening (PGS) are reproductive options for couples with known family histories of genetic disease or couples undergoing IVF procedures due to infertility issues. PGD can diagnose many genetic disorders caused by known chromosomal abnormalities (number and/or structure) or single gene mutations, and, thus decrease the risk of termination of pregnancy or miscarriages and enable such couples to have an unaffected child[1]. Through major improvements in PGD/S and general laboratory technology, the testing for abnormalities in fetuses has shifted from prenatal diagnosis during the first 2 trimesters [2], testing for overall fetal growth, complications of pregnancy, and birth defects[3], to an embryonic focus especially in advancements in Artificial Reproductive Technologies (ART)[4]. In 1990 PGD for a recessive X-linked disease resulted in the first live birth[5] and has since been incorporated in clinical routine and applied for a variety of genetic diseases, such as sickle cell anemia, thalassemia, or cystic fibrosis[1].
| <html5media height="400" width="533">https://www.youtube.com/watch?v=0e-79qKllqk</html5media> |
| Preimplantation Genetic Diagnosis[6] |
History
The advances in reproductive technology during the second half of the 20th century, led to PGD's first clinical application in 1990 [7].
| Year | |
| 1967 | First PGD on rabbit blastocysts (Gardner& Edwards) [8] |
| 1986 | First Cleavage Biopsy (Wilton & Trounson)[9] |
| 1987 | First Blastocyst Biopsy (Muggleton-Harris, Monk, Rawlings, & Whittingham)[10] |
| 1988 | First Polar Body Biopsy (Yury Verlinksy)[11] |
| 1990 | First Clinical PGD using PCR testing for X-linked (Handyside, Kontogianni, Hardy, & Winston)[12] |
Preimplantation Genetic Diagnosis
PGD is used to test the genetic makeup of embryos to detect single gene disorders, chromosomal abnormalities and mitochondrial disorders. It also has applications in gender selection for diseases with unequal gender distributions [13]. Some diseases commonly involved with PGD include cystic fibrosis, spinal muscular atrophy and beta – thalassaemia [14] [15]. It was first used in the United Kingdom in the 1980s of arenoleucodystrophy and primarily focusing on sex- linked disorders [15][16]. PGD is now capable of detecting single cell defects (molecular) and chromosomal disorders resulting from the inversion, translocation or deletion of chromosomes (cytogenic) [14],[17]. PGD can be applied to the embryo at different stages. That is on polar bodies, blastomeres or blastocyst [14]. Depending on the type of genetic disorder, PGD utilises different methods of genetic testing. These include Fluorescence in situ hybridisation (FISH) which is used for sex – linked disorders and detects chromosomal rearrangements [17][18] and Embryo halotyping which allows the identification of chromosomes causing the inherited disorder through knowledge of the pattern of closely linked markers [14]. Polymerase chain reaction (PCR) is also widely used to detect molecular abnormalities [17]. PGD is tightly regulated and supported by large organisations namely The American Society for Reproductive Medicine, The European Society for Human Reproduction and Embryology (ESHRE), The European Society of Human Genetics and the Preimplantation Genetic Diagnosis International Society [19].
Sex-linked disorders
The determination of a disease as gender specific usually correlates with the presence or absence of specific genes such as SRY on the Y chromosome. It is known that females have two X chromosomes and males have an X and a Y chromosome where abnormalities are more prevalent on the X chromosome. PGD can be used for sex selection where only male embryos are transferred to reduce the chance of inheriting X-linked disorders. However, this does not completely eradicate the problem as male embryos remain susceptible to inheriting an affected X chromosome. Sex determination is only used when the specific mutation is unknown and has yet to be discovered [20].
Single gene defects
Single gene defects can be dominant, recessive, autosomal or X-linked. They are commonly diagnosed using PCR and although the PCR available today is complex and capable of combatting a large range of disease the development of new protocols has been proven to be difficult due to the small DNA sample available [21]
Mitochondrial disorders
Mitochondrial disorders also known as oxidative phosphorylation disorders arise from mutations in the nuclear DNA or mitochondrial DNA [22]. They pose as a problem because they are unrecognisable until the mutations in the cell reach a detrimental level [13]. Mitochondrial disorders cause miscarriages and stillbirths as well as death in children and young adults. The effects can either be contained in a single organ or more commonly involve multiple organ failure where organs with high energy demands such as the brain, liver muscle and heart and heavily influenced. They are usually occur spontaneously or result from inheritance from the mother. Since mitochondria are solely inherited from the mother oocyte donations have been used as a solution to combat mitochondrial disorders. Additionally, there has been an increasing use in PGD where embryos that stay under the given threshold of 18% gene-mutations are allowed to be transferred and result in normal development. New technology in the areas of nuclear gene transfer and genome editing are also being experimented with [22].
Chromosomal disorders
Chromosomal disorders can be reciprocal, Robertsonian translocations, inversions, deletions and insertions [21]. . Data from ESHRE collected between 2010 and 2011 has shown that the most common chromosomal abnormality confronted in PGD are reciprocal chromosomal abnormalities [23]. PGD has also been used successfully for Robertsonian translocations (RT), a type of structural translocation. Children who carry RT are phenotypically normal, however in their later years it is found that they will suffer from infertility and repeated miscarriages due to the high frequency of abnormal embryos [24]. PCR and FISH are the two main techniques used for chromosomal disorders. To be able to conduct the examinations the cells are required to be at the metaphase stage [21].

Preimplantation Genetic Screening
PGS involves an array of methods or ideas that aim to segregate embryos that have genetic flaws and those that are healthy [14]. The occurrence of aneuploidy is high around the stages of early embryonic development and they are the most common cause if miscarriages and congenital birth defects [26]. They have little effect on the morphology of the embryo making them difficult to identify thus identification heavily relies on genetic testing [14]. Genetic sampling is most commonly conducted using Microarray Comparative Genomic Hybridisation (aCGH) as well as FISH, Quantitative PCR and Single Nucleotide Polymorphism (SNP) [14]. These methods collectively aim to assess numeral and structural chromosomal errors [26]. Studies have also introduced Next- Generation Sequencing (NGS) [26] and Whole Genome Amplification used to screen imbalances in the complete 24-chromosomes [27] Statistics from ESHRE have shown that the most common indications for PGS is advanced age, followed by repeated implantation failure or recurrent miscarriage and male infertility [28].
Indications
A low percentage of structural abnormalities in chromosomes are responsible for the cause of miscarriages. Despite this, they are the most prevalent type of chromosomal abnormality accounted for in PGD [13]. The presence of specific gene cycles, initiation of embryonic protein synthesis and evident physiological development are all indicative of a successful in vitro fertilisation procedure [29]. To eliminate further errors from occurring during the PGD procedure it is recommended to undertake further prenatal testing such as amniocentesis in the later stages [13].
Advanced maternal age
Data provided by the ESHRE has shown that the mean age of women undergoing PGS is 39 years [28]. Women of advanced age have been shown to have a lower rate of pregnancies reaching childbirth [30]. The highlighted concern revolves around the increased occurrence of aneuploidy following maternal age. The optimal age range for the lowest aneuploidy incidence was found to be between 27 to 37 years of age (6%) then progressively higher in women aged up to 42 (33%) and most common in those 44 and above (53%) [31].
Recurrent pregnancy loss / IVF failure
Recurrent pregnancy loss is defined as three or more IVF failures after cumulative transfer of more than 10 good-quality embryos. These are primarily caused by two main factors, reduced endometrium receptivity or embryonic defects. Endometrium receptivity can be negatively influences by instances including uterine pathologies such as thin endometrium, altered expression of adhesive molecules and immunological factors. Additionally, embryonic defects may be due to genetic abnormalities, embryonic aneuploidy or zona hardening. Endometriosis and hydrosalpinx has been known to effect both the endometrium and embryo [32].
Human leukocyte antigen matching
First used in 2001, HLA matching is an option given to parents to save a child with haematological or immunological disease through conceiving another child who would potentially be able to donate cord blood or haematopoietic stem cells from the bone marrow for transplantation. The process namely PGD-HLA has shown to improve haematopoietic stem cell transplant (HSCT). PGD-HLA can only be performed when HSCT is not needed urgently due to the time needed to conceive and delivery the baby [19]. It is most commonly applied to children suffering from relapsed leukaemia [33]. In a case study PGD-HLA was proven to successfully cure 10 diseases including Fanconi anaemia, Diamond-Blackfan anaemia and beta thalassemia [34].
Biopsy Methods
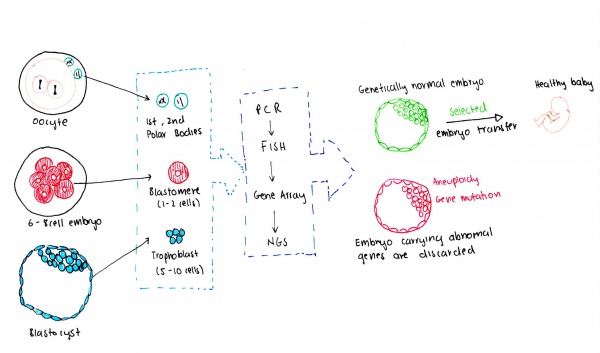
Biopsy, the removal of genetic materials, from oocytes or embryos in the preimplantation stage is the primary step in PGD. For the past two decades these biopsies have been performed at three stages, the polar body, blastomere, and blastocyst, and the methodologies optimized to ensure the embryo’s viability. The most common approach involves biopsies at the cleavage stage. However, polar body and blastocyst biopsies are increasingly more often tested and applied. Approached for opening the zona pellucida involve next to the traditional mechanical and chemical means, novel approaches such as noncontact lasers. Their application may simplify and secure the procedure significantly. The most challenging question about PGD procedures remains at what stage biopsies should be taken. Much controversy has developed around this topic, highlighting the varying disadvantages and advantages of temporal biopsies. [35]
| Time of Biopsy | Advantages | Disadvantages | |
| Polar Body | Day 1 | No harm to oocyte | Only maternal DNA is tested, often needs to be coupled with other biopsies, and there are difficulties distinguishing between the first and second PB. |
| Blastomere | Day 3 | … | … |
| Trophectoderm | Day 5 and 6 | Little harm to the embryo and large amount of genetic material is extracted. | Opening of blastocyst necessary, small time window for procedure |
Polar Body Analysis
Day 1
Description
Polar body (PB) biopsy offers a promising alternative to biopsies performed at the blastomere stage for PGD/S indications on legal and practical grounds. Consequent embryo development does not necessitate the presence of the first and second PB and their removal may not be crucial. PB biopsy requires precise timing. Keeping track of the meiotic cell cycle is necessary to perform a successful biopsy and, therefore, PB biopsy usually is applied in combination with intracytoplasmic sperm injection (ICSI). During the maturation from the germinal vesicle stage to the metaphase-II stage the first PB is formed. A cytoplasmic bridge containing spindle remnants, that are still in contact with the cellular genetic material, links this PB to the oolemma for about 90 minutes after extrusion. It is possible to visualize these remnants by polarization microscopy and it is crucial to not perform the biopsy until the first PB is no longer firmly attached to the oolemma as this indicates an immature embryo.The oocyte tolerates mechanical zona dissection best during hours four until six after ICSI as the oolemma has stabilized by that time because of the cortical granule reaction. Over time the first PB degenerates stressing a temporal biopsy and its optimal extraction time window is four to 12 hours after ICSI. The second PB forms around two to four hours after ICSI. Its optimal time window for biopsy is set to be eight to 16 hours after ICSI due to the second PB being attached to the oolemma with spindle remnants until six hours after ICSI. Biopsy at this point may cause enucleation of the oocyte. Studies have shown that the amplification efficiency of second PB’s DNA is worse if the PB is extracted prior to eight hours after ICSI. Thus, it is possible to perform sequential and simultaneous biopsies for the first and second PB. If performed, sequentially the first PB may be removed four to 12 hours and the second PB eight to 16 hours after ICSI. The optimal time window for a biopsy for both the first and second PB simultaneously is eight to 12 hours after ICSI. It is highly preferred to analyze both PBs due to potential aneuploidies in either PB and crossing overs during meiosis[35].
Procedure
Chemical opening achieved with for example acidic tyrode’s solution of the zona pellucida is not tolerated by the oocyte and may have detrimental effects on the embryo’s development. Therefore, the access to the perivitelline space of the oocyte is provided by mechanical zona dissection or by laser. Both techniques work well if performed by experienced embryologist. However, laser-assisted biopsy is less time consuming when compared to manual dissection. It is critical to consider the size of the introduced opening as it will remain permanent. If it is too large the blastomere may be lost during embryo development and if it is too small it may interfere with hatching of the embryo during blastocyst stage[35].
| <html5media height="300" width="400">https://www.youtube.com/watch?v=JTaQzszVr8o</html5media> |
| Laser-assisted polar body biopsy[38] |
Advantages & Disadvantages
PB biopsies can only investigate the maternal contributions to the embryo as they are of maternal origin. Thus, this procedure is appropriate for PGD solely for monogenetic diseases from the maternal side. Recessive diseases may be evaluated with PB biopsies on the basis that the embryo’s outcome will be determined by the paternal contribution. It may still be applied for PGS as it is a fairly safe biopsy option and most aneuploidies either arise during meiosis or origin from the maternal genome. However, diagnosis error has been reported due to the lack of considering paternal contributions.Because PB biopsies are performed very early it is not yet known whether the oocyte will develop into a viable embryo. Thus, PBs are commonly frozen or fixed after biopsy and depending on the state of the embryo only chosen PBs will be tested. This is primarily an economic issue as many genetic testing procedures are very cost-intensive[35]
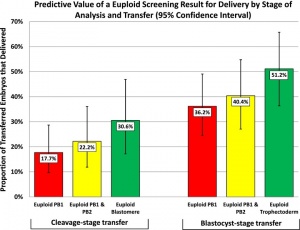
Generally the sustained implantation predictive value of screening of PBs is significantly lower than of for example biopsies of the blastocyst stage[39]. Moreover, it is often difficult to distinguish between the first and the second PB. As the first one degenerates quicker, this may influence diagnostic procedures[40].
Blastomere biopsy
Day 3
Description
Blastomere biopsy has been the prevalent method for PGD and PGS in the last two decades. At least one, but up to two, blastomeres are biopsied on day three of the cleavage stage embryo. The right number of blastomeres removed is a controversial topic as two cells allow for more genetic material and more accurate results. However, removing two cells might be too invasive and damaging to the embryo. As PGS tries to improve implantation rates, whereas PGD sets out to avoid known genetic disorders, for the former only one cell is removed, while for the latter often two need to be removed, to ensure results as correct as possible[40].
Procedure
Initially a hole was drilled into the zona pellucida using acid tyrodes with the consequent aspiration of blastomeres with a pipette. Nowadays the zona pellucida is largely opened using a laser and calcium and magnesium free media have been introduced to decrease junctions between blastomeres, which facilitates the biopsy[40].
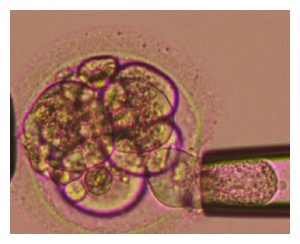
| <html5media height="300" width="400">https://www.youtube.com/watch?v=TbridWVwipI</html5media> |
| Laser-assisted blastomere biopsy[42] |
Advantages & Disadvantages
Trophectoderm biopsy
Day 5 and 6
Description
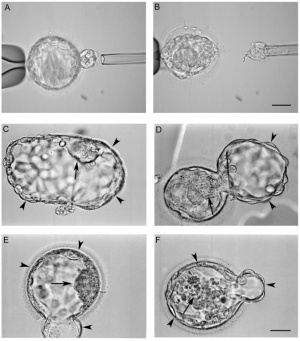
The improvement of embryo culture media allowed the in vitro development of human embryos until the blastocyst stage and opened up the possibility to take blastocyst biopsies. During day three to day five the haploid maternal and paternal genomes work together for the first time to form the genome of the embryo. The maternal epigenetic control lessens significantly while preparing for implantation with precisely arranged events happening. The first event includes a rapid increase in the number of embryonic cells which are active in mitotic divisions and apoptosis of aberrant cells. This is followed by the formation of blastocoel (cavitation) which results from the flattening of cell located on the outside of the blastomere. Now the blastocyst will expand until the embryo hatches by rupturing the zona pellucida. The cells of the blastocyst will differentiate to form two distinct cell lineages, the outer trophectoderm and the inner cell mass[35].
Procedure
Trophectoderm biopsy is usually performed in Hepes buffered biopsy medium and opening approaches include needle cutting which has been replaced by lasers in past years. Different research groups report different timings of the opening to be the most successful. Some open the blastocyst on day three or four by creating a 25 µm which causes the trophectoderm to herniate through this hole and is, thus, accessible for biopsy. Others create this hole about four hours before biopsy which allows sufficient herniation of trophectoderm cells. It is also possible to open the blastocyst immediately before biopsy. This avoids an extra step and the inner cell mass usually is easy to locate. Blastocyst biopsies involve the removal of trophectoderm cells and the ideal time for the procedure is day five. Successful biopsies on day six have been performed, while little is known about the results of biopsies on day seven. However, since the window of implantation in humans is from day eight to ten after ovulation, day seven biopsies appear to be possible[35]. -collapse of trophoblasts -possible: biopsy of hatched blastocyst, holding pipette careful mild suction force to hold embryo near ICM -Cryopreservation
| <html5media height="300" width="400">https://www.youtube.com/watch?v=JI_TQ8d8tNM</html5media> |
| Laser-assisted blastocyst biopsy (at 00:50 min)[44] |
Advantages & Disadvantages
Blastocyst biopsies are believed to be less damaging to the embryo and appear to be unrelated to implantation rates. In addition, this biopsy method allows for a larger extraction of cells for genetic testing. However, since they are performed late in in vitro development, the time window for genetic testing is relatively small. Fast and accurate genetic testing methods are needed to ensure a successful and safe result from this biopsy. Thus, blastocyst biopsies may gain more popularity in the future whn such methods have been developed or improved[35].
Genetic Techniques
Polymerase Chain Reaction
PCR amplifies DNA specific to genetic sequence of interest . PCR was developed by Kay Mullis in the 1980s, for which he was awarded the Nobel prize for chemistry in 1993. [45] This technique enables clinicians to monitor and diagnose diseases using minute samples such as embryonic cells, blood & tissue. [46] PCR has many important applications like DNA fingerprinting, genetic mapping, detection of viruses and bacteria as well as being used to detect genetic disorders, as a part of PGD, in conjunction with IVF. It is used to detect molecular abnormalities such as single gene disorders like Tay Sach, Cycstic fibrosis, Duchenne Muscular Dystrophy, Thalassemia, Huntington disease, Spinal muscular atrophy and many more. Molecular and Genetic analysis require a significant amount of DNA, which we would not have without PCR. It revolutionized the study of DNA, replacing all previous recombinant DNA technology. [47]
| Procedure |
Samples are obtained from the the blastocyst, a polar body biopsy or the blastomeres stages of the embryo.
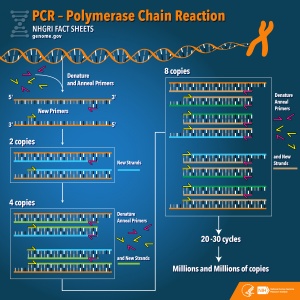
- Stage 1 Denaturing: separating the target strands of DNA
The obtained sample is heated to roughly 90 degrees celcius, this heat breaks the relatively weak bonds between nucleotides that form DNA. The double stranded DNA is split into two single strands of DNA that are used as templates.
- Stage 2 Annealing: Binding the Primers to the target DNA sequence
PCR will only copy the target sequence of DNA specified by specific PCR primer. These synthesized primers oligonucleotides are small artificial pieces of DNA. TAQ polymerase enzyme synthesize two new strands of DNA duplicate to the single sample DNA stand template indicated by these primers. During this stage the reaction is cooled to a temperature between 40-60 degrees Celsius.
- Stage 3- Extension- making copies
Each of these two copies are then used again as templates generating two further replications This cycle can occur as many 30 -40 times within a couple hours leading to billions of extra copies of the original DNA segment. The optimal temperature for the further replication is roughly 72 degrees Celsius. This process mediated by a thermocycler machine that is programmed to alter the temperature of the reaction every couple of minutes, perpetuating the cycle of DNA denaturing and synthesis. This process, generates exponentially exact copies of the original template DNA sequence.
PCR cycles
Fluorescent In Situ HybridisationFISH is the one of the most effective and rapid [52] method of conducting PGD. This techniques used to locate a specific DNA sequences within a chromosome. FISH facilitates the clinical diagnosis of chromosomal abnormalities indicated by sequential duplications, deletions and rearrangements of chromosome, that are usually missed with microscopic analysis. This technique is especially relevant for female embryos with X-linked diseases,[53] that have no other mutation specific tests [20] As part of PGD, samples are collected from various stages of the embyro,and they are able to conducted tests on the blastocyst, a polar body biopsy and the blastomeres [40]. FISH is 99% effective when used in conjunction with competitive Genomic Hybridisation (CGH) to diagnose chromosomal abnormalities[52].
Samples are collected from either the blastocyst, a polar body biopsy or the blastomeres stages of the embryo [40]. DNA strands are heated and denatured causing their the individual DNA strands to break apart Probes are single complimentary stands of DNA a that have been tagged with small chemical agents that glow brightly in the presence of a specific region on a chromosome. These specific probes then hybridize and join to their complementary DNA strand. The fluorescent tags enable the researchers to correctly identify the presence or lack thereof and location of the specific chromosomes that they are testing for. [18] Researcher analyse the results by identifying the number and the relative location of the fluorescent dots generated by the FISH images.[54] The probe will not fully hybridise if there has been a duplication or a deletion of the DNA - indicating chromosomal and sex chromosomal anomalies like trisomies and aneuploidies. [55] Different probes are used for different purposes: [56]
of genes within a chromosome
 FISH We are able to tag Specific DNA sequences using Probes which have been tagged with fluorescent Labels.This technique enables the clinical diagnosis of chromosomal abnormalities, indicated by sequential duplication, deletions and rearrangements of chromosome [57]
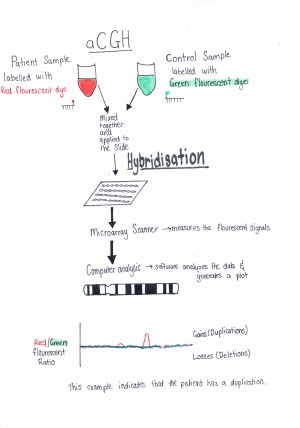
Array Comparative Genomic Hybridisation (aCGH)aCGH also known as Microarray analysis efficiently scans the entire genome for chromosomal imbalances. CGH was initially developed to detect the number of changes in a solid tumor mass. It uses 2 genomes comparing the sample to the control, with each labeled in a different fluorescent dye[65]. Earlier CGH techniques were limited by the resolution of the imaging [66] , these initial limitations were overcome by using Microarrays in conjunction with CGh to improve the resolution of the imaging, Array Comparative Genomic Hybridisation (aCGH). This method compares sample and control microarrayed slides containing small segments of DNA (probes). [67] the Probes used will vary according from the small (25-85 base pairs) oligonucleotides manufactured to highlight different target sequences, to the very large genomic clones (80,000- 200,00 base pairs), and as these are significantly smaller than the traditional metaphase chromosomes used for CGH, generating a higher resolution of image. [68].aCGH is used as a diagnostic tool for prenatal detection f chromosomal abnormalities [69].
[70] The sample is obtained (skin, blood or fetal cells) and DNA is obtained. As a part of PGD fetal cell samples are collected from; the fertilized egg polar bodies, the blastomere (day 3 embryo) or the blastocyst/tropoectoderm stage (day 5 embryo). Sample DNA is labeled with one fluorescent dye, and the control DNA is labeled with a different colored fluorescent dye. the control DNA is used as the base point of reference. heated and denatured single DNA strands then hybridize to their complementary single strand probes, which are then combined and applied to a microarray and the results are run through a computer program and a digital imaging system is used to quantify the results( fluorescent intensities of the labeled probes). The fluorescent ratio and the hybridization signal at different locations on the genome of the control DNA are used to identify any variances present in the sample DNA. aCGH facilitates the clinical diagnosis of submicroscopic chromosomal duplication, deletion and rearrangements indicative of chromosomal disorders such as trisomies 1-22and specific sex linked disorders. Duplications in the DNA are displayed by the computer program as spikes/ peaks over an established threshold and deletions in DNA are displayed by the computer program as spikes/ toughs beneath this threshold
Next Generation SequencingThe development and advances within ART in the past 20 years, as well as the increasing popularity of IV, has lead to an influx of new technologies developed to screen embryos for chromosomal anomalies, which are covered by the umbrella term of Next generation Sequencing (NGS). NGS is a general term used to describe all of the new and emerging screening techniques currently being introduced and used as part of PGD for IVF . NGS screens for single gene disorders as well as conducting extensive and very comprehensive chromosome diagnosis by sequencing, counting, and accurately assembling millions of DNA reads, simultaneously. [82] There is a movement for NGS to replace the other limited testing techniques and be used as the standard.
Samples are collected from either blastomere or the blastocyst/tropoectoderm and processed for analysis by a computer system. The methodology of each process is unique to the technique being used.
DiagnosisThere are a large amount of diseases that PGD can apply to, below are descriptions of the diseases that PGD are more commonly used for. Utilization of Diseased Cell Lines Cystic FibrosisCystic fibrosis is a single gene disorder. It is autosomal recessive and involves mutations in the cystic fibrosis transmembrane conductance regulator (CTFR) gene. The CTFR gene is normally responsible for the decrease in chloride and the transport of bicarbonate in epithelial cells thus playing a major physiological role. In PGD procedures, identification of the gene is assisted by microsatellite markers that have similar composition to the CTFR gene itself. Biopsy of two cells at the blastocyst stage is recommended if markers are not available. There are many variations of cystic fibrosis the most common being P.Phe508del [87]. Duchenne Muscular DystrophyDuchenne Muscular Dystrophy is an X-linked recessive disease. It involves the Xp21 gene where majority of the mutations are chromosomal deletions with a smaller percentage resulting from duplications [88]. Autosomal Recessive MEckel-Gruber SyndromeAutosomal recessive genetic defect caused by the TMEM67 gene. It results in cystic dysplasia of the kidneys with fibrotic change in the liber and occipital encephalocele. Other malformations could also be present in the central nervous system. In PGD whole genome amplification of single blastomeres are taken to identify the gene, this is also coupled with PCR techniques. Maternal plasma can also be extracted for PGS. [89] β – Thalassemiaβ – Thalassemia is known as the most common type of autosomal recessive inherited disorder among haemoglobinopathies. It involves the adult β-globin gene and is associated with the absent or decreased expression of the gene. This is commonly caused by a single nucleotide change in the gene. PGD has been used successfully worldwide to identify the β-globin gene where its application is usually performed on a single blastomere or polar body [90].
Laws & Legal status
Future/Current ResearchNoninvasive Preimplantation Genetic Testing without Embryo BiopsyDifferent parameters of gametes, zygotes, embryos (“vacuoles in sperm heads, spindle position in mature oocytes, cleavage intervals of zygote, and embryo developmental dynamics”) may correlate with aneuploidy rates. This knowledge may be applied in potential noninvasive preimplantation diagnostic methods. Several methods have been proposed and are currently further researched[41]. Sperm SelectionIntracytoplasmic morphologically selected sperm injection (IMSI) is common procedure in IVF treatment to improve fertilization rates in patients with poor semen quality. In addition, studies have found that IMSI improves embryo development and that spermatozoa with large vacuoles in their heads correlate with increased aneuploidy rates and disturbed chromosomal structures. Thus, selecting spermatozoa based on morphological hallmarks may decrease aneuploidy rates in the fertilized embryos. As with polar body biopsies, however, this approach will solely be applicable if evidence for severe male detrimental contribution is given[41]. Blastocoel Fluid ExtractionIn addition, a less invasive retrieval of material for diagnosis could include the extraction of the blastocoel fluid. The cavity of the blastocyst, lined by the trophectoderm, is filled with this blastocoel fluid, which contains metabolites of both trophectoderm and inner cell mass origin. The retrieval does not require a biopsy but merely a small opening to extract the fluid and, thus, causes less harm to the embryo. Multiple studies have applied this method[98] [99] and it may in the future become of clinical relevance.
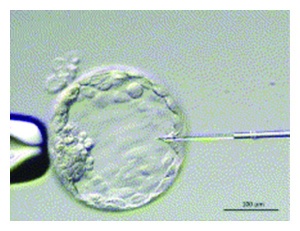 Aspiration of the Blastocoel Fluid using a ICSI pipette[41].
ProteomicsThe protein secretome of blastocysts may be representative of its chromosome constitution Recent studies have found biomarkers such as lipocalin-1, interleukin-10, tumor necrosis factor, stem cell factor, and chemokine ligand 13 to be differently secreted by aneuploid blastocyst than by euploid ones. The most significant biomarker appears to be interleukin-10. Paired with novel proteomic technologies and mass spectrometry this knowledge when extended may contribute to a new invasive PGD method[41]. Embryo MorphologyUsing time lapse imaging the embryo’s morphology can be closely observed and potential aneuploidy characteristics detected. Such characteristics may include the time of division to five cells, the time between the division from three to four cells, and the duration of the division from one to two and subsequently to three. In addition, the morphological quality of ICM an TE has been positively associated with aneuploidy or euploidy prognosis[100]. These may give rise to an embryo quality screening prior to implantation[41]. GlossaryCTFR Cystic Fibrosis Transmembrane Conductance Regulator ESHREThe European Society for Human Reproduction and Embryology FISH Fluorescence in situ hybridisation ICSI Intracytoplasmic Sperm Injection IMSI Intracytoplasmic Morphologically Selected Sperm Injection NGS Next Generation Sequencing PB Polar Body PGD Preimplantation Genetic Diagnosis PGS Preimplantation Genetic Screening PCR Polymerase Chain Reaction RT Robertsonian translocations SNP' Single Nucleotide Polymorphism References
Glossary Links |
|---|