2014 Group Project 6
| 2014 Student Projects | ||||
|---|---|---|---|---|
| 2014 Student Projects: Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | ||||
| The Group assessment for 2014 will be an online project on Fetal Development of a specific System.
This page is an undergraduate science embryology student and may contain inaccuracies in either description or acknowledgements. | ||||
The Endocrine System
Introduction
The endocrine system consists of a number of organs that influence the development, growth, metabolism, electrolyte balance, reproduction and homeostasis of the human body through their release of hormones into the blood stream. Hormones can exert both a direct influence on target organs (for example the pituitary gland) as well as stimulating or inhibiting the release of hormones from other organs (e.g. hypothalamus) as endocrine organs are highly intertwined with each other[1]. The following page explains the key stages of development of endocrine organs in the human fetus, relevant developmental abnormalities as well as some current research findings.
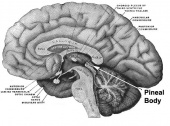
Pineal gland
Fetal development
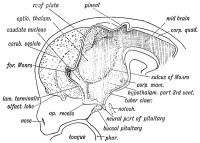
The pineal gland is part of the epithalamus, located in the diencephalon. Like the other neurosecretory glands, it is formed by the neuroectoderm of the neural plate. Its primary function is to regulate circadian cycles postnatally through its secretion of melatonin. The synthesis of melatonin is regulated by a dense network of sympathetic nerve fibers located in the autonomic superior cervical ganglia. In adults, the pineal gland converts sympathetic input into hormonal output by producing melatonin, which has regulatory effects upon reproductive hormones such as luteinizing hormone (LH) and follicle-stimulating (FSH) hormone. The activity of the pineal gland is inhibited by stimulation from the photoreceptors of the retina. This light sensitivity causes melatonin to be produced only in low light or darkness and thus the pineal gland is active most at night. However it is not until after birth that the infant produces significant levels of melatonin to sustain cyclic rhythms associated with day/night and light exposure. During early fetal development before the pineal gland is developed, it is the maternal melatonin that exerts its effect on development after crossing the placental barrier[2].
Melatonin plays an important role during fetal development. Both pineal and placental melatonin have a regulatory effect on maintaining homeostasis in the uterus as well as fetal maturation and reproductive development. The suprachiasmatic nuclei in the hypothalamus acts as the central pacemaker for melatonin production. As the two circuits are already interconnected in the mammalian fetus, intrinsic circadian rhythms are already established prior to birth.
How far has the pineal gland developed by week 8 of gestation?
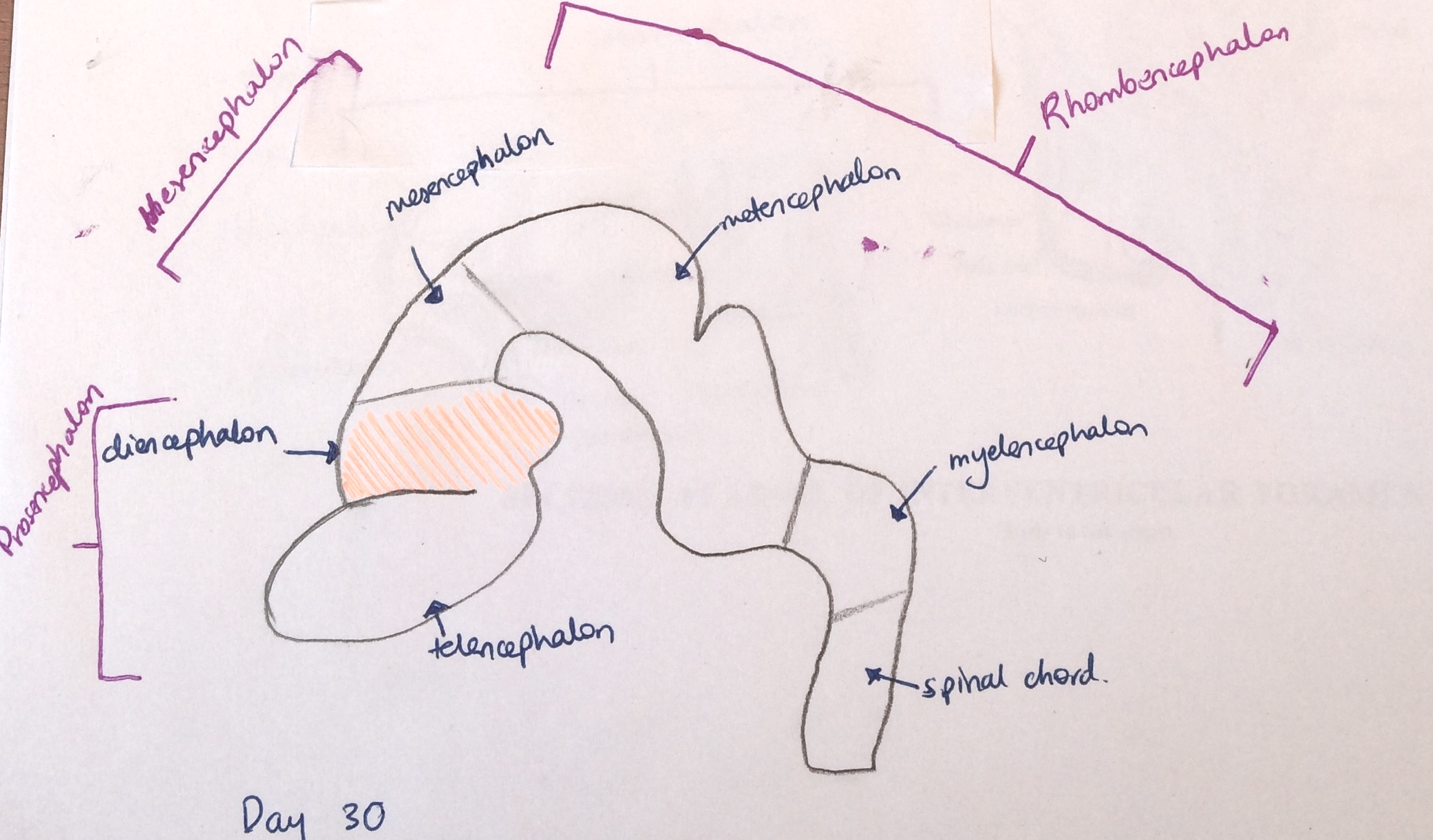
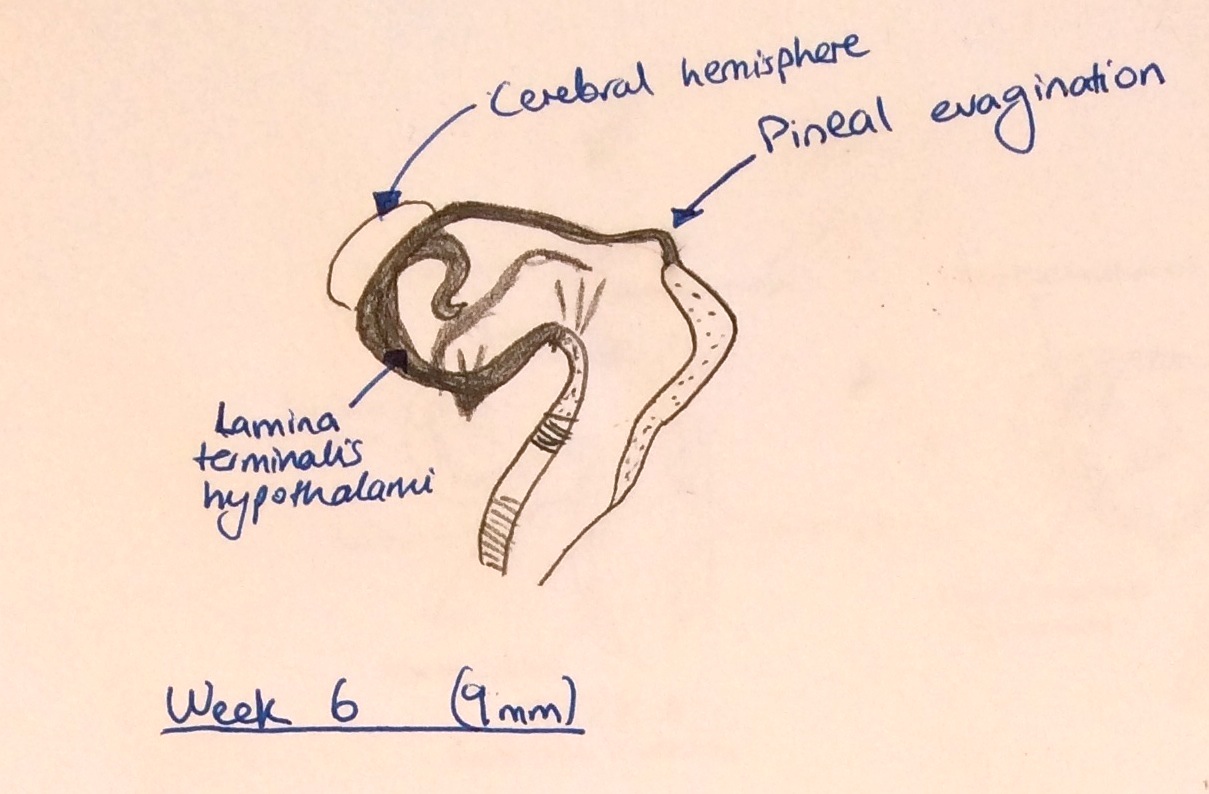
- Week 5 - The pineal gland primarily begins its development after the diencephalon is differentiated from the prosenecephalon and forms on the diencephalic roof of the third ventricle (Image 3).[2]
- Week 6 - A number of hollow diverticula begin formation in a clustered arrangement and an evagination of the pineal gland is visible (shown in image 4).
Fetal Stage of Pineal Development - from week 8 of gestation onwards:
- Week 8 - Proliferation of cells begins and these cells form the walls of the diverticulum
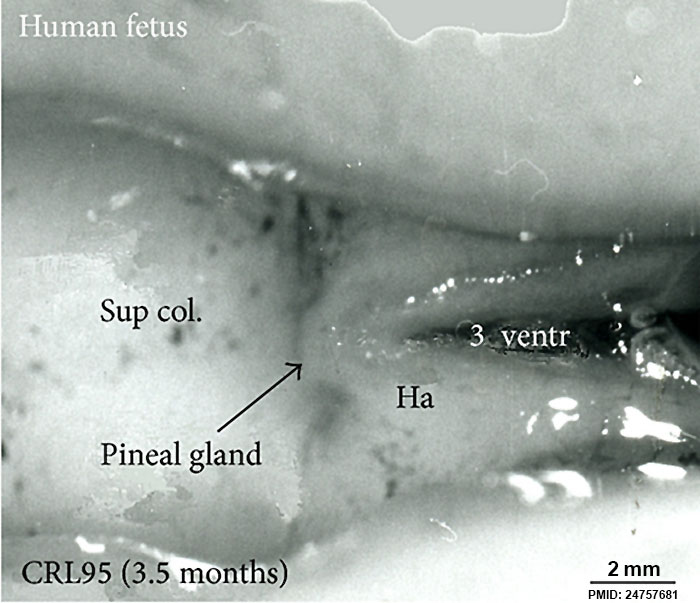
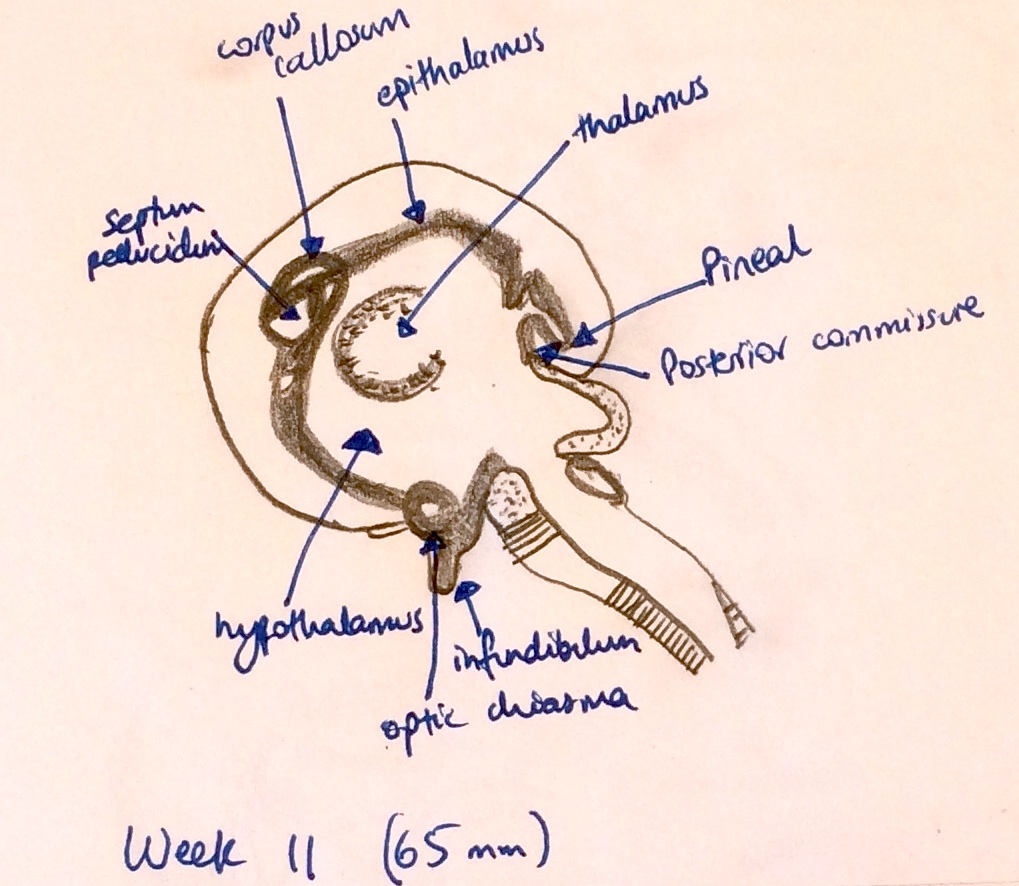
- Week 9-11 - On the caudal roof of the hollow diverticulum, cells continue proliferation and begin to differentiate into pinealoctyes to form the solid epiphysis (shown in Image 5)
- Week 15-17 - By the second trimester small amounts of melatonin production by the fetal pinealocytes has begun, however the pineal gland is not fully functional till post-natally.[2]
Table 1. Summarises the hormones released by the human pineal gland and their role in embryonic and fetal development
| Hormone | Produced by | Function |
|---|---|---|
| Melatonin | Pinealocytes | Contributes to the development of gametes and also helps in maintaining uterine homeostasis through its antioxidant properties.
Melatonin also inhibits the secretion of gonadotrophin releasing hormone (GnRH) until puberty. This mechanism is in place to protect premature initiation of pubertal pubertal activation of reproductive functions that are dependent on plasma gonadotropin levels. [3] |
Abnormalities
- Pineal tumors
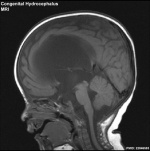
Tumors of the pineal region during fetal development are generally a result of displaced embryonic tissue or chromosomal abnormalities. Fetal pineal tumors are characterized by pigmented (melanin) epithelial cells, small undifferentiated cells, and a fibrovascular stroma. [4] Depending on the size and type of tumor the fetus may survive till birth however infants with pineal gland tumor experience abnormal puberty development and have a high probability of developing hydrocephalus. As the pineal gland sits just superior to the cerebral aqueduct (shown in image 7 below), pineal tumors can compress this aqueduct, resulting a build up of pressure of CSF in the brain, a condition known as obstructive congenital hydrocephalus.[5]
- Pineal hypoplasia
Pineal gland hypoplasia is the underdevelopment of the pineal gland resulting in the production of an insufficient level of melatonin. As discussed, fetal melatonin plays a crucial role in the growth and development of important organs and functions so a lack of melatonin particularly during the second and third trimesters of gestation may have detrimental results on the health of the fetus. Studies have suggested a possible link between neonatal pineal gland hypoplasia and sudden infant death syndrome (SIDS) with almost 95% of deaths occurring within 6 months of birth [6]. Oxidative stress of the brain, as melatonin's hydroxyl radical reducing ability is lost is suggested as a possible cause of SIDS. Additionally, the loss of melatonin's antioxidant activity during the fetal period is proposed as another factor to consider in development of atherosclerotic vascular disease later on in adult life.[6]
Hypothalamus
Fetal development
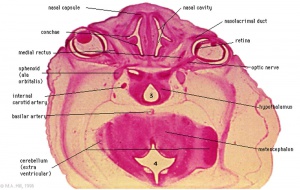
The hypothalamus is part of the diencephalon and plays an important role in the maintenance of homeostasis and the driving of motivated behaviours. Distinct nuclei in the hypothalamus secrete specific hormones that function to regulate thirst, hunger, thermoregulation, circadian rhythms, reproduction and defensive behaviour[7]. The hypothalamus is involved in two main signalling pathways, the Hyothalamus-Pituitary-Adrenal (HPA) and the Hypothalamus-Pituitary-Gonad (HPG) and exerts most of it's influence on the body through these two axes.
The sexually dimorphic nucleus (SDN, intermediate nucleus) is twice as large in young male adults as in young females. Immediately after birth, only 20% of the SDN cell number is present. During the post-natal period up till two to four years of age cell numbers continue to increase rapidly and equally in both sexes. Past this age, cell numbers start to decrease in girls and this is the point of physiological differentiation in sex[8].
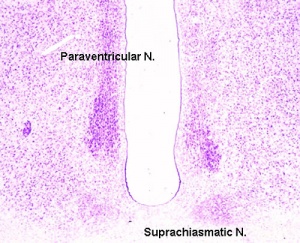
Neurosecretory cells of the supraoptic (SON) and paraventricular nucleus (PVN) project to the neurohypophysis, where they release vasopressin and oxytocin into the blood circulation. These hormones play an important role in fetal development up till and including the birth process[7]. Fetal oxytocin may initiate or accelerate the course of labor whereas vasopressin plays a role in the adaptation to stress caused by the birth process by redistribution of fetal blood flow.
How far has the hypothalamus gland developed by week 8 of gestation?
- Week 5- The development of the CNS has reached the five vesicle stage, where the prosencephalon divides into the diencephalon which is more caudal and in which the hypothalamus is formed, and the telencephalon located more rostrally[8].
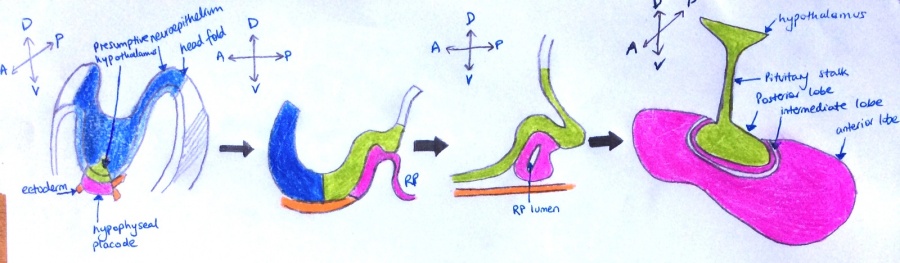
- Week 6- During pre-foetal phase when the head folds begin to take shape, a thickening called the hypophyseal placode (shown in pink in Image 3.a) forms at the midline of the rostral ectoderm, adjacent to the area where the hypothalamus will form on the neural fold.[9]
Fetal Stage of hypothalamus development - from week 8 of gestation onwards:
- Week 9- The hypophyseal placode changes shape as it is pulled upwards, towards the overlying neuroepithelium, to form Rathke's pouch.
- Week 18- By mid-gestation, this simple epithelial invagination separates from the underlying ectoderm to form the definitive Rathke's pouch with lumen.
- Weeks 19-27- Subsequent cell proliferation and differentiation of the intermediate zone allows for the formation of the primordial hypothalamus. The posterior lobe and the pituitary stalk connects the gland to the hypothalamus [9]
- Week 28- Sexual differentiation of hypothalamus is complete
Table 2. Summarises the hormones released by the human hypothalamus and their role in embryonic and fetal development
| Hormone | Abbreviation | Produced by | Function |
|---|---|---|---|
| Vasopressin | ADH | Paraventricular and Magnocellullar neurosecretory neurons | (also known as antidiuretic hormone) Controls the body's water balance electrolyte concentration and blood pressure by increasing water permeability in distal convoluted tubules and in the collecting duct of nephrons in the kidney. Results in higher reabsorption of water in kidneys and thus higher blood volume and pressure. |
| Oxytocin | Magnocellular neurosecretory cells | Stimulates lactation, hormone release during labor causes the muscles of the uterus contract | |
| Thyrotropin-releasing hormone (Prolactin-releasing hormone) | TRH, TRF, or PRH | Parvocellular neurosecretory neurons | Releases thyroid-stimulating hormone (TSH) as well as limited prolactin from anterior pituitary |
| Dopamine (Prolactin-inhibiting hormone) | DA or PIH | Dopamine neurons of the arcuate nucleus | Inhibits release of prolactin from anterior pituitary |
| Growth hormone-releasing hormone | GHRH | Neuroendocrine neurons of the Arcuate nucleus | Stimulates release of growth hormone from anterior pituitary |
| Corticotropin-releasing hormone | CRH or CRF | Parvocellular neurosecretory neurons | Releases adrenocorticotropic hormone (ACTH) from anterior pituitary |
| Gonadotropin-releasing hormone | GnRH or LHRH | Neuroendocrine cells of the Preoptic area | Stimulates release of follicle-stimulating hormone (FSH) as well as luteinizing hormone (LH) from anterior pituitary. |
| Somatostatin (growth hormone-inhibiting hormone) | SS, GHIH, or SRIF | Neuroendocrine cells of the Periventricular nucleus | Stimulates release of growth hormone (GH) from anterior pituitary and also has inhibitory effect on release of thyroid-stimulating hormone (TSH) from the anterior pituitary |
Abnormalities
- Congenital Hypothalamic Hamartoma
A hypothalamic hamartoma arises from the region of the tuber cinereum and is commonly associated with isosexual precocious puberty, i.e. puberty that develops unusually early in infants. It involves the alteration of pathways that modulate gonadotropins and contain releasing hormones within the floor of the third ventricle. .[10] Precocious puberty may either result from a physical manipulation of inhibitory pathways by the hamartoma or a direct neurosecretory process of the hamartoma itself. Seizures are also another common clinical presentation in children with a hypothalamic hamartoma. MR imaging is typically used to identify and monitor the growth of a hypothalamic hamartoma during embryonic and fetal development. Research has shown a trend of congenital hamartomas forming in the embryo within a period of 26 weeks. Because of the known association with other congenital anomalies of the central nervous system, hypothalamic hamartoma is thought to form as early as 4 weeks and may in fact be part of a developmental abnormality spectrum.[10]. Treatment options are currently limited to post-natal deliver of leuprolide acetate to the infant and in general it is relatively effective in managing and treating the condition.
Pituitary gland
Fetal development
How far has the pituitary gland developed by week 8 of gestation?
- Formation of Rathke's Pouch by week 4-5 of gestation
- At the point of the oropharynx in the primitive gut there is an invagination of the ectoderm and this is the origin of the anterior pituitary lobe
- Eventually Rathke's pouch is pinched off and separates from the oral cavity. All the Rathke cells need to migrate down to sit in the sphenoid bone of the skull. Any cells left behind can becomes tumours.
- The posterior pituitary is formed from the downward outgrowth of the third ventricle forming a median eminence
- Together with cells from the mammillary body, a neural stalk forms giving the neurohypophysis Cite error: Invalid
<ref>tag; invalid names, e.g. too many - Anterior pituitary
Fetal stage of pituitary development - from week 8 of gestation onwards:
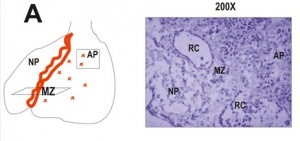
- Week 8 - By week 8 embryological development, the pituitary gland is fully formed and begins functioning as a control center for release of hormones that are vital for subsequent organ development.
- Week 10 - Growth Hormone and ACTH detectable
- Week 21 - Vasopressin hormone synthesis established
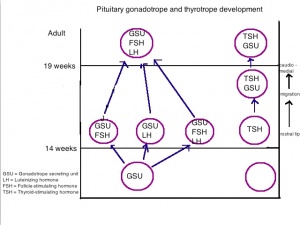
Anterior Pituitary - Adenohypophysis
Table 3. Summarises the hormones secreted by particular cells of the anterior pituitary during the fetal period. The cell types that are present are somatotrophs and gonadotrophs which secrete hormones that cause cell proliferation and activation of gonadal cells respectively. Cite error: Invalid <ref> tag; invalid names, e.g. too many
| Hormone | Abbreviation | Cell Type | Function |
|---|---|---|---|
| Growth Hormone | GH | Somatotroph | No function in fetal development. All postnatal action. |
| Luteinizing Hormone | LH | Gonadotroph |
|
| Follicle Stimulating Hormone | FSH | Gonadotroph |
|
Posterior Pituitary - Neurohypophysis
Table 4. Summarises the hormones secreted by cells of the posterior pituitary which is also known as the neurohypophysis. These hormones contribute to proper brain and central nervous system development. Their neuronal origin are from the hypothalamus and run into the pituitary gland where they are released to take action on various systems and organs. Cite error: Invalid <ref> tag; invalid names, e.g. too many
| Hormone | Neuronal Origin | Function |
|---|---|---|
| Oxytocin | Hypothalamic paraventricular and supraoptic nuclei |
|
| Vasopressin | Hypothalamic paraventricular and supraoptic nuclei |
|
Abnormalities
- Ectopic Posterior Pituitary:
This is an abnormal pituitary development disorder that results from improper migration of the posterior pituitary from the neural tissue that initially forms an invagination at the floor of the third ventricle. This results in the posterior pituitary lobe developing and maturing at the level of the median eminence or in line with the pituitary stalk rather than sitting further down in the cella turcica. Consequently, the releasing factors secreted from the hypothalamus that normally migrate down the portal circulation towards the anterior pituitary can only target their destination using the general vasculature. This leads to growth hormone deficiency and in some cases panhypopituitarism.Cite error: Invalid <ref> tag; invalid names, e.g. too many
- Hypopituitarism:
This is a disorder that results from abnormal production of pituitary hormones like growth hormone, luteinizing hormone, vasopressin and oxytocin. If there are insufficient levels of fetal production of these hormones, there are many postnatal effects. The infant would have irregularities in bone growth, urinary system development and brain growth and would show retinal dysfunction. This is because the pituitary gland plays a pivotal role in the hypothalamic-pituitary axis in maintaining proper body balance and organ growth. Cite error: Invalid <ref> tag; invalid names, e.g. too many
Thyroid
Fetal development
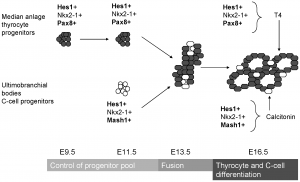
The functional unit of the thyroid gland is the follicle hence it is necessary to understand how the follicle develops in order to understand the important function of this gland. There are progenitor cells in the anterior endoderm that are specified thyroid progenitor cells and contribute to thyroid organogenesis. The proliferation of these cells results in the formation of a placode along the midline of the pharyngeal floor, just below the future tongue. The left and right lobes of the thyroid gland start off as single structures: buds of endoderm surrounded by mesoderm.
The mammalian thyroid gland is unique in the sense that there is a second endocrine cell called the parafollicular C cell. The progenitor cell for this enters the gland at the stage where there is a fusion between the thyroid progenitor proper and the ultimobranchial bodies. The ultimobranchial bodies arise bilaterally in the most inferior pharyngeal arches and are important in the final organ.
Eventually the midline primordium stretches laterally to reach the ultimobranchial bodies and there is a bilobation event resulting in the recognisable bilobed thyroid gland. [11]
How far has the thyroid gland developed by week 8 of gestation?
- Formation of medial anlage (foregut endoderm origin) and 2 lateral anlages (neuroectodermal origin and derivatives of the 4th pharyngeal pouch)
- Budding of ventral pharynx to form thryoid primoridum
- Fusion event of the median anlage with the lateral angales (ultimobranchial bodies) followed by migration of median anlage to it's final pretracheal location
- Sonic hedgehog plays a role in directing correct lobulation of the median anlage into two lobes connected by an isthmus
Fetal Stage of Thyroid Development - from week 8 of gestation onwards:
- Week 7-8- Terminal differentiation of thyroid gland occurs and involves the onset of the gland function
- Week 7-9- This is the Pre-colloid stage where the thyroid gland contains strands of compact unpolarised Thyroid Follicular Cell (TFC) precursors
- Week 10-11- Is the Beginning colloid stage and involves the polarisation of the TFC precursors. This gives the first appearance of small thyroid follicles (the eventual functional unit of the gland)
- Week 12- Progressive follicular growth occurs. At this point the fetal thyroid gland gains the ability to accumulate iodine and begin thyroid hormone synthesis[12]
- Week 16 - 18- Fetal thyroid hormone synthesis occurs. Prior to this, the fetus receives vital maternal thyroid hormones via the placenta
Table 5. Explains the ontogenic action of thyroid hormone on the developing fetus. Thyroid hormones are essential in brain and central nervous system development in the fetus. A lot of neurological disorders are associated with insufficient thyroid hormone synthesis:
| Hormone | Types | Ontogenic Action |
|---|---|---|
| Thyroid Hormone |
|
Brain development:
|
Abnormalities
Hypothyroidism
Hypothyroidism is an abnormality that can occur in fetal development were there is insufficient thyroid hormone action. This can be due to mutations in the thyroid-stimulating hormone receptor on the thyroid gland, hence the hypothalmic-pituitary-thyroid axis communication is lost. This then results in distinct decrease in T3 and T4 levels in the fetus. Follow on effects of this deficiency include abnormal brain and central nervous system development due to lack of neuron branching and myelination. Postnatally, this can cause learning difficulties, issues with olfactory and optic systems and impaired reflex actions. Cite error: Invalid <ref> tag; invalid names, e.g. too many
Parathyroid gland
Fetal development
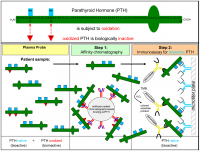
The parathyroid gland is an important endocrine organ that plays an essential role in regulating extracellular calcium homeostasis and hence serves many physiological processes that involve muscle contraction, blood coagulation, and synaptic activity.[13] They detect changes fluctuations of calcium levels in blood which is detected by the calcium-sensing receptor (CasR). This process then stimulates the secretion of parathyroid hormone (PTH) which releases calcium from internal stores such as bone in order to counterbalance any extremities.[14]
How far has the parathyroid gland developed by week 8 of gestation?
- Week 5 - the Parathyroid glands arise from the endodermal third and fourth pharyngeal pouches in cranial portions.
- Cranial third pharyngeal pouches form inferior parathyroids and cranial fourth pharyngeal pouches forms superior parathyroids.
- Pouches are bilateral and hence form four parathyroids
- Parathyroid gland development cannot occur without the transcription factor encoded by Gcm-2.
- Week 6 - diverticulum extends from the pouch which is hollow at first and then solidifies with dorsal cell proliferation
Fetal Stage of parathyroid Development - from week 8 of gestation onwards:
- Active transport regulates high fetal calcium concentrations levels (11-12 mg/dl) from maternal serum via an ATP-dependent calcium pump situated across the syncytiotrophoblast.
- The middle portion of the parathormone related peptide (PTHrP) is secreted via the fetal parathyroid and activates the placental calcium pump.
- Sections 1-34 of the Parathormone (PTH) or PTHrP stimulate PTH/PTHrP receptors causing a fetal skeletal calcium flux. This subsequently leads to the excretion of calcium via the fetal renal 1, 25 (OH) 2 D production also occurs which serves to increase the calcium transport occurring in carrying mothers. Calcium reabsorption from amniotic fluid also takes place through this action.
Table 6. Summarises the hormones produced by the parathyroid gland:
| Hormone | Secreted by | Function | Image |
|---|---|---|---|
| Parathyroid hormone | Chief cells | Regulates calcium levels in the blood by bone resorption releasing calcium in the blood when calcium levels are low. It also increases the ability to absorb calcium from food sources as well as aid the kidney in retaining calcium that would otherwise be lost in the urine. |
Abnormalities
Fetal hyperparathyroidism
Fetal hyperparathyroidism is a condition that is caused when an excess of parathyroid hormone (PTH) is present the bloodstream.[18] This may be due to an overstimulated maternal parathyroid gland out of all the four which transfers the over accumulated PTH to the fetus. Normal fetal development requires 25–30 gms of calcium for bone mineralization and around 80% of this calcium accretion is observed in the third trimester of pregnancy. However the calcium gradient of 1.0:1.4 is no longer maintained when there is hyperparathyroidism present.[19]Instead the gradient is elevated causing fetal parathyroid gland suppression thereby leading to Hypocalcemia. Calcium mobilisation then becomes difficult and hence fetal tetany may occur followed by miscarriage.[20] In the event that the fetus survives, postnatal problems such as the baby having weak bones will be likely and would require calcium treatment throughout most of their lives.
Thymus
Fetal development
The thymus gland is an organ that belongs to two systems of the human body which are the endocrine and immune system. It consists of two distinct but identical lobes which are both encased by a tough and fibrous capsule. Within each lobe are two layers which is the cortex that is superficial to the deep medullary layer in the tissue. Epithelial tissues and lymphatic tissues including macrophages make up majority of the thymus. In terms of its role in the endocrine system, it is responsible for the development of hormone called thymosin. This hormone is needed to tranform white blood cells (lymphocytes) that pass through the thymus gland into T cells, thereby forming the link to aid the immune system. This important gland is located in the upper anterior chest straight behind the sternum and in between the lungs. Other associated hormones of the thymus gland include thymopoietin hormones, thymic humoral factors , thymostimulin and factor thymic serum.
How far has the thymus gland developed by week 8 of gestation?
- It originates primarily from the third pharyngeal pouch.
- The primordia is initially divided into the thymic and parathyroid domains which are both encased in a neural crest-derived mesenchymal capsule.
- Week 7 - mid week 8, the thymic part of the primordium migrates ventrally and attach at the pericardium
Fetal Stage of thymus Development - from week 8 of gestation onwards:
- Week 8 - The thymic primordium contains undifferentiated epithelial cells
- Week 8-9- Intrathymic cell types such as mesenchymal, vascular and lymphoid cells begin to develop
- Weeks 8-16 - Medullary development occurs from week 8 and distinct cortical and medullary compartments are formed by week 16
- Weeks 14- 16- Mature lymphocytes begin to migrate from the thymus to seed the peripheral immune system
Table 7. Summarises the hormones produced by the thymus gland:
| Hormone | Function |
|---|---|
| Thymosin | Transforms white blood cells (lymphocytes) that pass through the thymus gland into T cells. |
| Thymopoietin hormones | Belongs to the polypeptide hormone family and is secreted by thymic epithelial cells. These cause differentiation of precursor lymphocytes into thymocytes. |
| Thymic humoral factors | Increase the number of T-cells which enhances cell-mediated immunity. |
| Thymostimulin | Stimulates T cell proliferation and differentiation |
| Factor thymic serum | Involved in T-cell differentiation. |
Abnormalities
- Thymic aplasia
Thymic aplasia is a condition that has a strong correlation to DiGeorge syndrome. This syndrome is a congenital birth defect which occurs when the fetus either inherits an abnormal chromosome 22 or has a mutation of the same chromosome. In particularly, the mutation is a deletion of the genomic information packaged in the chromosomes and normally occurs when there isn’t proper recombination of chromosomal material during fertilisation.[22] As a result of this deletion, the third and fourth pharyngeal pouches fail to develop in the twelfth gestational week, thereby leading to the absence or partial absence of the thymus and parathyroid glands. Other associated abnormalities with thymic aplasia as a result of the deletion of chromosome 22 genetic material are severe cardiac defects, low immunity, hypocalcaemia and facial abnormalities. [23] Statistics suggest that the thymic aplasia may occur in 1:4000 to 1:6395 of fetuses.[24] Treatment is normally difficult for fetuses that exhibit thymic aplasia, however calcium levels can be monitored and regulated and heart problems can be resolved though surgery. Immunity defects can be managed from controlling phosphorus levels in the fetus and protecting them from sources of infection.[25]
- Thymoma
Thymoma is the term given to the neoplasms of the thymus gland. These tumours emerge from the thymic epithelium cells of the thymus gland and do not normally show cytologic characteristics of malignancy, however are considered malignant as they are likely to undergo invasion. Thymomas are classified into different types based on the content and structure of the cells. If the tumour has spindle cell conformation, it is known as type A thymoma and constitutes to 4% to 7% of all thymomas. The tumor is made up of neoplastic thymic epithelial cells that exhibit a spindle oval shape conformation, lack nuclear atypia and have low levels of non-neoplastic lymphocytes. Mixed thymoma, also known as type AB thymoma are similar to a type A thymoma except that the foci is enriched with non-neoplastic lymphocytes. It makes up approximately 28% to 34% of all thymomas.[26]Type B1 thymoma also termed as lymphocytic thymoma is similar to a healthy functional thymus as it contains a large proportion of cells exhibiting features indistinguishable from a healthy thymic cortex with areas similar to thymic medulla. It makes up for approximately 9% to 20% of all thymomas.[27]Cortical thymoma, also known as type B2 thymoma contains neoplastic epithelial cells which appear scattered with vesicular nuclei and distinct nucleoli located in a dense population of non-neoplastic lymphocytes.[28]It is similar to type B1 thymoma but the foci of medullary differentiation are less visible. It makes up for approximately 20% to 36% of all thymomas. Epithelial thymoma, also known as type B3 thymoma is formed mainly from polygonal shaped epithelial cells that show no atypia. The mixture of non-neoplastic lymphocytes gives the tumour a sheet-like appearance. This makes up for approximately 10% to 14% of all thymomas. Since some of these are hard to differentiate from normal thymus tissues, there is poor prognosis.
Thymomas have been closely linked with another disease called myasthenia gravis. It is caused when nerve impulses are disrupted at the neuromuscular junction of skeletal muscles from the lack of binding of acetylcholine (neurotransmitter) to acetylcholine receptors needed to generate muscle contraction. Antibodies produced from the thymus gland destroy the acetylcholine receptors and hence have a strong correlation to myasthenia gravis. [29] Since thymomas contain more cells than normal and are malignant, it is more likely to generate antibodies which target acetylcholine receptors. The formation of these antibodies in B cells is T cell dependent. The T cells involved in this mechanism are generated in the thymus via nontolerogenic thymopoiesis through an abnormal function of thymic epithelial cells.[30] Overall, incorrect signalling messages could be sent out and hence cause an autoimmune attack.
Pancreas
Fetal development
The pancreas is an endocrine organ situated well into the abdomen. It has a dual role and is responsible for digestion via its exocrine function and regulating blood sugar levels through its endocrine function.
How far has the pancreas developed by week 8 of gestation?
- Week 4 – Pancreatic development begin at the septum transversum as dorsal and ventral endodermic buds forms. Dorsal and ventral mesentery are formed by splanchnic mesoderm.
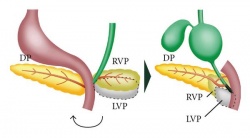
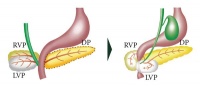
- Dorsal buds normally develop first and form majority of the pancreas whereas the ventral bud only forms a portion of the head and uncinated process of the pancreas.
- Week 6-8 – These buds migrate and fuse from duodenum growth and rotation. In order to make space for the pancreas, the duodenum rotates in to C-shaped conformation. The ventral bud also situates itself dorsally behind the dorsal bud.
- Pancreatic bud endoderm in particularly the ventral bud duct and distal part of dorsal bud differentiates into islet cell clusters which form acini and exocrine ducts needed for exocrine function. At the periphery of these exocrine clusters form the pancreatic islets which serve endocrine function.
Fetal Stage of pancreas development - from week 8 of gestation onwards:
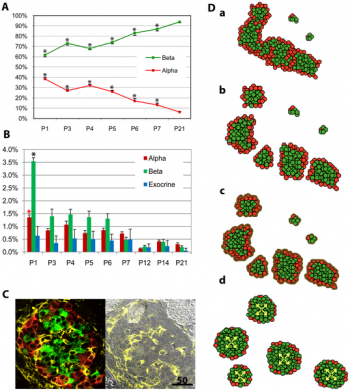
- Week 7 to 20 – Maternal insulin increases exponentially as fetus grows.
- Week 10 – The first cells to differentiate are glucagon (alpha) cells followed by somatostatin (delta), and insulin (beta) cells. Fetus begins to secrete insulin
- Week 15 – Levels of glucagon become noticeable in fetal plasma
Table 8. summarises the hormones produced by the pancreas:
| Hormone | Produced by | Function |
|---|---|---|
| Glucagon | Alpha cells of the islets of Langerhans | Elevates blood sugar levels when blood sugar levels are low. |
| Insulin | Beta cells of the islets of Langerhans | Reduces blood sugar levels when blood sugar levels are too high. It also converts glucose into glycogen to store in the liver for future source of energy. |
| Somatostatin | Delta cells of pancreas | Inhibits the secretion of other pancreatic hormones such as insulin and glucagon. |
| Pancreatic Polypeptide | Pancreatic polypeptide cells | Prevents secretion of somatostatin from the pancreas. |
Abnormalities
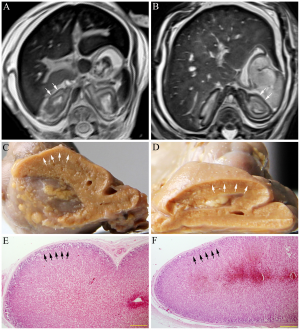
- Annular Pancreas
Annular pancreas is a rare abnormality that can occur during fetal development in which a ring of the pancreatic tissue encircles the growing duodenum.[33] It occurs in 1 out of 7000 birth and is commonly associated with other abnormalities such as esophageal atresia and malrotation of the midgut. It is assumed that annular pancreas occurs when the tip of right ventral bud sticks to the duodenal wall which then forms a ring from the continuous stretching of the wall during rotation.[34] This part of the pancreas can constrict the duodenum thereby restricting the flow of food to the intestines of the gastrointestinal tract.[35] Furthermore, annular pancreas is divided into two forms. Complete annular pancreas is when the pancreatic parenchyma wraps around the 2nd part of the duodenum completely whereas in incomplete annular pancreas, the tissue only surrounds the duodenum partially.[36]Treatment for annular pancreas mainly involves surgery to bypass the constricted portion of the duodenum.
- Gestational diabetes mellitus
Gestational diabetes mellitus (GDM) is a disorder in which women without any history of diabetes show elevated blood glucose levels during pregnancy normally around the third trimester. It is caused when insulin receptors fail to respond correctly due to interference from signalling molecules related to pregnancy such as the interaction between human placental lactogen, prolactin and estradiol with the insulin receptors.[37] As a result, high blood glucose levels are observed. Patients with GDM have high levels of insulin resistance with increased β-cells production of the pancreas since the high levels of blood glucose are not interacting with the abnormal insulin receptors. Glucose is known to diffuse through the placenta via GLUT1 carrier situated in the syncytiotrophoblast of both the microvillus and basal membranes. With GDM, the number of GLUT1 molecules increase and hence placing high levels of glucose to the fetus since insulin itself cannot diffuse across the placenta and counteract this extremity.[38] This mechanism is known to lead to excessive growth at birth with a low glucose environment as it declines and high insulin production overall leading to hypoglycaemia.[39]It can also lead to jaundice and seizures in the new born with mothers having high susceptibility to type 2 diabetes mellitus. GDM can be controlled by monitoring blood sugar levels and controlling levels of glucose and insulin through pharmaceutical drugs.
Adrenal gland
Fetal development
The characteristic zonation of the adult adrenal gland is absent in the fetal gland which is instead arranged in an inner fetal zone and an outer definitive (adult) zone. The inner zone atrophies following birth and contains steroid-secreting cell characteristics while the adult zone contains cells that resemble those present in the adult zona glomerulosa. The adrenal medulla is not a distinct, recogniseable zone during gestation, except for scattered chromaffin cells present throughout the cortex in small clusters. [40]
Fetal Stage of adrenal development - from week 8 of gestation onwards:
- Week 6- Adrenal gland is present at the cranial side of the mesonephric kidney as a condensed mass of coelomic epithelium, appearing as large cells like those of older fetus fetal zones [41]
- Week 8- The definitive or adult zone is formed by a second round of epithelial cell proliferation, where a cap is formed by a narrow rim of cells over the fetal zone. Around the central part of the gland can be seen clumps of medullary cells and neural elements infiltrate it through the vascular pole. [40]
- Weeks 10-20- Rapid growth of the adrenal gland by increased size of the fetal and definitive zones from about 100mg in week 10 to 2g at week 20. Appearance of vasculature and sinusoidal plexuses and increase of medullary cells.
- Weeks 20-30- Gland size doubles with adult-type zonation appearing in the definitive zone around week 30. The zona glomerulosa is delineated by connective tissue stroma and cells appear arranged in a columnar fashion in the developing zona fasciculata. [42]
- Week 30-term- Weight of fetal adrenal gland doubles and 80% of the gland’s volume is made up of the fetal zone. The zona reticularis develops post-natally in year 3 of development, unlike the two other cortical zones.
Table 8. summarises the hormones produced by the adrenal gland:
| Hormone | Type | Produced in | Function |
|---|---|---|---|
| Aldosterone | Mineralocorticoid | Zona glomerulosa | Works on the kidneys, sweat and salivary glands to maintain normal extracellular concentrations of Na+ and K+ and so extracellular volume |
| Cortisol | Glucocorticoid | Zona fasciculata | Restoration of homeostasis following stress; suppresses immune system, increases blood sugar by gluconeogenesis, helps metabolise protein, carbohydrates and fat, activates the CNS. In the foetus/neonate, causes organ development and maturation e.g. lungs. There are high levels of cortisol at childbirth |
| Adrenaline and noradrenaline | Catecholamine | Adrenal medulla | Increase; heart rate contractility, vasoconstriction, ventilation, lipolysis, glycogenolysis and decrease gut motility. Work with the sympathetic nervous system to regulate 'flight or fight' response. |
Abnormalities
- Congenital Adrenal Hyperplasia (CAH)
Congenital Adrenal Hyperplasia (CAH) is the collective term for several autosomal recessive endocrine disorders caused by impaired adrenal steroidogenesis due to mutated steroidogenic enzymes. [43].These mutations inhibit proper synthesis of cortisol from cholesterol, which in turn leads to excess adrenal growth and hyperplasia due to adrenocorticotropic hormone (ACTH) hypersecretion by the pituitary gland. The most commonly affected enzyme (90% of cases) is a 21-hydroxylase deficiency (21-OHD) which clinically presents as cortisol and aldosterone production deficiency. [44] CAH has both a classic (severe) and non-classic (mild) forms with the classic form affecting 1 in 10 000 and non-classic form affecting many more. Females affected by the classic form undergo pre-natal virilisation and have genital ambiguity at birth, whereas virilisation does not occur in the mild form. [45] Males tend to appear asymptomatic but can exhibit oligozoospermia. [43]
- Cushing's Syndrome
Cushing’s Syndrome (CS) is a metabolic disorder characterised by abnormally high production of endogenous cortisol or exogenous levels of corticosteroids, which disrupts the metabolism of macromolecules such as lipids and carbohydrates. It is most common in obese adults aged 20-50 and the most common cause is the therapeutic administration of exogenous ACTH [46]. Cushing’s Syndrome’s two forms are either ACTH-dependent CS (80-85% of patients) or ACTH independent. The etiology of the dependent form is adrenal gland hyperplasia and excessive ACTH secretion by adenomas in the pituitary, however other causes include; small cell lung carcinoma ACTH production ectopically, or medullary thyroid carcinomas. [47] The independent form is however caused by neoplastic growths in the adrenal gland such as carcinomas, adenomas and macronodular hyperplasia. Clinical manifestations of this disorder include; moon facies, cataracts, hyperglycaemia, abnormal fat distribution, poor wounding of the skin, obesity in the central body and muscle wasting in 60% of patients. [47]
Ovary
Fetal development
How far has the ovary developed by week 8 of gestation?
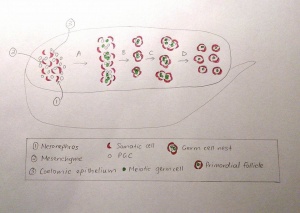
- Week 5- Thickening of mesothelium forms on the mesonephros. Formation of the gonadal ridge results from proliferation of the mesothelium and mesenchymal tissue beneath it [48]
- Projection of finger-like gonadal (epithelial) cords into the mesenchyme. Indifferent gonad segmented medulla )regresses) and cortex (differentiates to form the ovary)
- PGCs migrate through the primitive streak in gastrulation and then reside at the junctional region of the hindgut yolk sac. Migration of the PGCs to the gonadal ridge along the hindgut’s dorsal mesentery [48]
- Week 6- PGCs incorporated into the gonadal cords and are called oogonia, having entered underlying mesenchyme.
Fetal stage of ovarian development - from week 8 of gestation onwards:
- Weeks 8 to 9- Gonad is identifiable as an ovary now due to change in internal structure. Cortical differentiation occurs from the cranial pole to the lower pole. The dense central core spans the mesovarian into the mesonephric organ in a caudal direction, resembling the ‘rete blastema’ which differentiates. Inner ovary; disintegrating blastema surrounded by a thick blastemal layer. Tissue has a cortical region (encloses PGCs) and a central medullary region (reticulum of somatic cells) [48]
- Weeks 10 to 12- Supporting cells grow peripherally and segment the cortex into irregular globules containing rapidly multiplying germ cells and light and dark somatic supporting cells, interspersed with connective tissue [48]
- Week 12- Cortex is penetrated by dark supporting cells, giving the superficial epithelium a ‘dark’ appearance, amongst the original ‘light’ cells of the coelomic epithelium. Oogonia appear in clusters, primordial cells still dominate and oocytes in the premeiotic period exist in small groups. Medulla contains less densely-packed globules, mainly oogonia [48]
- Weeks 14 to 28- In mid-gestation; depletion of the germ cells by apoptosis, highest from weeks 14-28 and decreasing closer to birth [48]
- Week 16- Cortical cords break up into primordial follicles (cell clusters) housing an oogonium each from a PGC. Follicles enclosed by monolayer of flat follicular cells from surface epithelium. Primordial follicle formation is the result of active mitosis of oogonia [48]
Abnormalities
- Congenital gonadal dysgenesis – Turner Syndrome
Female congenital gonadal dysgenesis syndrome refers to a condition where the fetus undergoes abnormal gonadal development with a presentation of streaks of connective tissue on the developing ovary, also referred to as "streak gonads". Streak ovaries extend from the lateral pelvic wall to the attachment of the utero-ovarian ligaments and are essentially sections of functionless tissue. They vary considerably in size but are usually approximately 4 cm in length and 2–3 mm in width. Dysgenetic ovaries are characterized by also absence of follicular structures and oocytes. In women, the most common cause of congenital gonadal dysgenesis is Turner syndrome, 45X which has a prevalence of approximately 1 in 2500 new born girls worldwide[49]. Turner syndrome is a genetic abnormality due to an incomplete or completely missing X chromosome in females. Growth hormones treatments such as estrogen replacement therapy may be used after the child has reached 12-13 years of age to help treat the symptoms[50].
Testis
Fetal development
How far has the testis developed by week 8 of gestation?
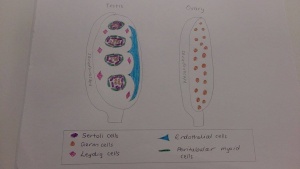
- By week 8, masculine differentiation is induced in the mesonephric duct and external genitalia. This is caused by the interstitial cells (Leydig cells) in the mesenchymal tissue surrounding seminiferous tubules beginning secretion of the androgens androstenedione and testosterone, which is stimulated by human chorionic gonadotropin, peaking in weeks 8-12. [51]
- Antimulleran hormone, AMH (or mullerian-inhibiting substance, MIS), a glycoprotein, is produced by sustentacular (Sertoli) cells. This causes mesonephric duct suppression to prevent formation of the fallopian tubes and uterus in the developing male. [52]
- The majority of the seminiferous epithelium of the fetal testes is composed of Sertoli cells and this epithelium later flattens forming external mesothelium. 15-20 mesonephric tubules are continuous with the rete testes later form efferent ductules, connected to the mesonephric duct to form the epididymis duct. [51]
Fetal stage of testicular development - from week 8 of gestation onwards
- Week 9- Yellowish elongated tube with mesonephros laterally and urinary bladder inferomedially
- Week 13- Testes have acquired the ellipsoidal shape found in the adult and are located above the inguinal canal in the groin region. Convexly curved across all but posterior surface [52]
- Week 16- Located above deep inguinal ring, convexly curved anterior border and flat posterior border covered by epididymis
- Week 17- Migration of testes begins
- Week 24- Testes grow in size and still located about the deep inguinal ring or inside inguinal canal. The testicular poles seem more prominent. Approximately 90% of testes still remain in the abdomen, with migration accelerating in weeks 24-26 [53]
- Weeks 26-28- Arrival of testes in the inguinal canal within a couple of days through the deep inguinal ring, helped by the gubernaculum [53]
- Week 30- Testes sinus is towards lower portion of testes and epididymis tail continuous with vas deferens. Testes are located in the scrotum having passed through the superficial inguinal ring
- Week 36- Testes located within scrotal sac, they enlarge in size and are maximally convex [53]
Abnormalities
Klinefelter’s syndrome
Klinefelter’s syndrome is the common form of congenital primary hypogonadism with a prevalence of an estimated 1 in 1000 men. Male hypogonadism is defined as a testicular dysfunction resulting in decreased sperm and testosterone production[54].. This genetic disorder occurs when a male is born with an additional X chromosome causing complications and a general underdevelopment of structures such as the seminiferous tubules and Leydig cells within the testis. As a consequence, the size of the testis is abnormally small and the patient has a low sperm count and testosterone levels. In addition to infertility caused by testosterone deficiency, men born with this disorder frequently have increased length of their long bones, including those in the arms, legs, and hands. Many other complications exist independent of the testosterone deficiency seen with these patients[54].
Placenta
Fetal development
The placenta is a highly specialised, transient and autonomous organ of pregnancy that plays an essential role in normal fetal development. Its functions include; the transfer of nutrients and oxygen to the developing fetus, removal of carbon dioxide, release of hormones into the materno-fetal circulation, and exchange of metabolic products to protect the fetus from infection, maternal diseases and xenobiotic molecules [55]
How far has the placenta developed by week 8 of gestation?
- Days 6-7- Placental development starts with invasion of the endometrium by the blastocyst. The outer cover of the blastocyst (trophectoderm) and vascular components are allantois-derived. The trophoblast differentiates into the syncytiotrophoblast following invasion of the uterine mucosa. [56]
- Days 8-13- The chorionic villi are developed, arising in the syncytiotrophoblast and composed of trabeculae and lacunae. The primary chorionic plate surrounds this system
- From day 12- Establishment of the primitive maternal circulation from trophoblastic shell cels. Mesenchymal cells invade the primary villi to form secondary villi.
- By the end of week 5- The intra-placental fetal circulation is fully established. [56]
Fetal stage of placental development - from week 8 of gestation onwards:
- Until beginning of week 8- Chorionic villi cover the whole chorionic sac
- After week 8- Syncytiotrophoblast production of placental progesterone is enough to maintain pregnancy in spite of ovariectomy
- By about week 10- The fetal-placental maternal circulation is fully established. Exchange of blood to the embryo occurs via diffusion until week 10. Peak levels of hCG are produced in maternal serum. [55]
- Until week 18- Rapid growth in the size and thickness of the placenta. Fully mature placenta comprises one sixth of the fetal weight.
- Week 22-24- Decidua capillaris degenerates due to lack of blood supply. There is fusion of the chorionic sac with the decidua parietalis [56].
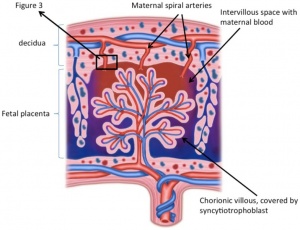
Both fetal tissue from the chorionic sac and endometrium-derived maternal tissue contribute to form the utero-placental unit.The two parts of the mature placenta are; the chorionic plate (fetal part) containing chorionic blood vessels and the basal plate (maternal part) formed by the decidua basalis. In between these two parts is the intervillous space containing the villous structures of fetal blood vessels. Maternal-fetal exchange occurs at the terminal regions of these chorionic villi. [57]
Table 9. summarises the hormones produced by the placenta:
| Hormone | Type | Produced in | Function |
|---|---|---|---|
| Human chorionic gonadotropin (hCG) | Glycoprotein | Trophectoderm | Maintenance of the corpus luteum in first 8 weeks of pregnancy, enhancement of spontaneous cytotrophoblast differentiation into syncytiotrophoblasts |
| Placental Growth Hormone (PGH) | Trophoblast hormone | Syncytiotrophoblast layer | Controls levels of maternal insulin-like growth factor I (IGF-I), prevents variations in maternal blood glucose and allows maternal pregnancy metabolic adaptation |
| Progesterone and estrogens | Steroid hormones | Syncytiotrophoblast | Maintenance of pregnancy even after ovary removal |
Abnormalities
- Pre-eclampsia
Pre-eclampsia is a common abnormality of human pregnancy characterised by significant proteinuria (presence of protein in the urine) and systemic hypertension. There occurs significantly reduced uteroplacental blood flow due to; angiogenic and antiangiogenic factors, hypoxia and inflammation. [58]. Its incidence is about 2-8% of pregnancies, usually in women after gestational week 20 and it contributes to high maternal morbidity rates worldwide, with 50 000 deaths each year. [55] There is evidence that the cause of pre-eclampsia is a combination of an abnormal inflammatory response cascade, damage to endothelial cells and impaired immune function of the maternal placenta. Soluble factors are released into the maternal placenta from the ischemic placenta, leading to significant endothelial dysfunction. It has also been suggested that abnormal differentiation and invasion of cytotrophoblasts into the uterus have an aetiological role, as does failure of arterial remodelling in the uterus. [55]
- Placenta accreta
Placenta accreta (PA )is a rare obstetric abnormality characterised by abnormal insertion of the placenta to the myometrium of the uterus. The 3 types of PA are classified based on the depth of invasion; placenta accreta (superficial- chorionic villi attach to myometrium rather than just decidua basalis), placenta increta (middle layer) or placenta perceta (deep) being the most dire with 1 in 7000 incidence. [59] PA has an incidence of 1 in 2500 deliveries and is associated with grand-scale haemorrhage and requires hysterectomy and blood transfusion. [60] The condition normally occurs in the lower segment of the uterus which is prone to bleeding being poorly contractile and a site of angiogenesis. Prenatal diagnosis of PA is based on second and third trimester ulstrasound findings indicating; retroplacental zone loss, many lucanue in the vasculature and a very thin (<1mm) retroplacental thickness of the myometrium.[60] PA has a 40% likelihood in women aged over 35 who have had Caesarian section with placenta previa in their uterine scars. [59]
Recent Findings
|
Localised inhibition of FGF signalling in the third pharyngeal pouch is required for normal thymus and parathyroid organogenesis[61] Jennifer R Gardiner, Abigail L Jackson, Julie Gordon, Heiko Lickert, Nancy R Manley, M Albert Basson Localised inhibition of FGF signalling in the third pharyngeal pouch is required for normal thymus and parathyroid organogenesis. Development: 2012, 139(18);3456-66 PMID: 22912418
There has been research showing that fibroblast growth factor (FGF) signalling controls the organogenesis processes of the pharyngeal region. Mutations in FGF signalling are known to impact the thymus and parathyroid organs by causing aplasia or hypoplasia. FGF feedback antagonists of the sprouty gene family are key regulators in the formation of the mentioned organs. Localised inhibition of FGF signalling by sprouty proteins has shown a favourable result in the organogenesis of the thymus and parathyroid organs by inducing differentiating factors Gcm2, Bmp4 and Foxn1 expression in the third pouch as well as apoptosis. In this study, mouse embryos are used to show how FGF feedback antagonists that inhibit FGF can result in proper organogenesis of the thymus and parathyroid organs. Mouse embryos that contained the two FGF feedback antagonists, Spry1 and Spry2 and those that lacked them were studied. Data revealed that there was increased FGF signalling when Spry1 and Spry2 were not present, thereby leading to lack of Gcm2 expression and hence parathyroid hypoplasia. Similarly, levels of Bmp4 expression was also decreased in the thymus domain when Spry1 and Spry2 were absent, leading to thymus hypoplasia. It was also found that FGF Ligands were upregulated in the third pouch which resulted in the deletion of the sprouty gene. Consequently, the markers needed for thymus and parathyroid organogenesis was altered and apoptosis was inhibited leaving the organs still attached to the pharynx. Therefore, it is established that localised inhibition of FGF signalling present in the third pharyngeal pouch is vital for the organogenesis of the thymus and parathyroid organs. |
| Mouse testis development and function are differently regulated by follicle-stimulating hormone receptors signaling during fetal and prepubertal life[62]
Stéphanie Migrenne, Evelyne Moreau, Pirjo Pakarinen, Andrée Dierich, Jorge Merlet, René Habert, Chrystèle Racine Mouse testis development and function are differently regulated by follicle-stimulating hormone receptors signaling during fetal and prepubertal life. PLoS ONE: 2012, 7(12);e53257 PMID: 23300903 This article investigates the role of Follicle Stimulating Hormone (FSH) on Sertoli cell development in the fetal and post-natal period using a mouse model. Although the role of FSH is clear in the rat, there remains a lack of understanding in the mouse. They confirm the current understanding that Sertoli cell formation occurs during fetal life at 11.5-12.5 days post conception (dpc) in the mouse and proliferation is fully established at day 17. In Rathke’s pouch, detection of the alpha-gycoprotein subunit occurs 11.5dpc and the number of Sertoli cells reduces between 18.5dpc and birth in hypogonadal mice. Through performing morphometric studies on FSH+/+, FSH+/- and FSH-/- mice, at days 0, 6, 8 and 10, the study found that FSH-/- mice contained 22% less fetal Sertoli cells than wild-type mice. Assessment of the transcription factor GATA-6 was performed to establish the expression of Sertoli cell specific markers and the study concluded that GATA-6 was expressed less than wild-type mice by 30% compared to FSH-R-/- mice. Also, a molecule involved in testis organogenesis, Claudin 11, was found to be expressed at significantly less levels in FSH-/- testes.The expression of testicular steroidogenesis genes such as P450scc and StAR revealed that these molecules had greater expression in FSH-/- mice than control ones. The study thus provides light on the fact that Sertoli cells have a paracrine mechanism of action to regulate FSH-R function of Leydig cells in the fetal period and that FSH controls Sertoli cell development in both the mouse and rat. |
| Neuropeptide Y in the Adult and Fetal Human Pineal Gland[63]
Morten Møller, Pansiri Phansuwan-Pujito, Corin Badiu Neuropeptide Y in the adult and fetal human pineal gland. Biomed Res Int: 2014, 2014;868567 PMID: 24757681 Neuropeptide Y was isolated from the porcine brain in 1982 and shown to be closely linked to noradrenaline in sympathetic nerve terminals. In a number of studies over the years the peptide has been detected in sympathetic nerve fibers innervating the pineal gland in many mammalian species. The investigation sets out to identify the presence of neuropeptide Y in the adult and fetal human pineal gland. Neuropeptide Y-containing nerve fibers were confirmed as being present in the adult pineal and could be also be detected as early as in the pineal of four- to five-month-old fetuses. This early innervation of the human pineal is markedly different from studies of rodents models, where the innervation starts postnatally which is an important factor to consider when applying animal models to research of the human pineal gland. Another variation from rodent models were the numerous NPY-immunoreactive nerve fibers endowed with large boutons en passage (shown in image 8) that were present in a perifollicular position unlike in rodents where immunoreactive nerve fibers penetrate and densely innervate the follicle itself. |
References
- ↑ <pubmed>15706790</pubmed>
- ↑ 2.0 2.1 2.2 J.N. Bruce, E.M. Housepian, ‘Pineal Tumors’, Medscape:2013, http://emedicine.medscape.com/article/249945-overview
- ↑ <pubmed>15119946 </pubmed>
- ↑ <pubmed>851944</pubmed>
- ↑ <pubmed>1652883</pubmed>
- ↑ 6.0 6.1 C.P. Maurizi, 'Could exogenous melatonin prevent sudden infant death syndrome?', Medical Hypotheses:1997, 49(5); 425-427, http://www.sciencedirect.com/science/article/pii/S0306987797900918
- ↑ 7.0 7.1 <pubmed>7643957</pubmed>
- ↑ 8.0 8.1 <pubmed>11954031</pubmed>
- ↑ 9.0 9.1 K. Rizzoti, R. Lovell-Badge, ‘Regenerative Medicine: organ recital in a dish’, Nature:2011, http://www.nature.com/nature/journal/v480/n7375/full/480044a.html
- ↑ 10.0 10.1 T. Booth, C. Timmons, K. Shapiro, N.K. Rollins, ‘Pre- and Postnatal MR Imaging of Hypothalamic Hamartomas Associated with Arachnoid Cysts’, Americal Jounral of Neuroradiology:2004, http://www.ajnr.org/content/25/7/1283.full
- ↑ <pubmed>24290349</pubmed>
- ↑ <pubmed>10.1016/j.beem.2013.08.005</pubmed>
- ↑ <pubmed>7835276</pubmed>
- ↑ <pubmed>21904825</pubmed>
- ↑ <pubmed>22808183</pubmed>
- ↑ <pubmed>21881196</pubmed>
- ↑ <pubmed>7344740</pubmed>
- ↑ <pubmed> 1745971</pubmed>
- ↑ <pubmed> 12140371</pubmed>
- ↑ <pubmed> 25327435</pubmed>
- ↑ <pubmed>512270</pubmed>
- ↑ <pubmed>4166528</pubmed>
- ↑ <pubmed>5057550</pubmed>
- ↑ <pubmed> 10861201</pubmed>
- ↑ <pubmed>1454183</pubmed>
- ↑ <pubmed>25143131</pubmed>
- ↑ <pubmed>24672784</pubmed>
- ↑ <pubmed>2698424</pubmed>
- ↑ <pubmed>21747138</pubmed>
- ↑ <pubmed> 20402580</pubmed>
- ↑ <pubmed>22761699</pubmed>
- ↑ <pubmed>19893748</pubmed>
- ↑ <pubmed>25124266</pubmed>
- ↑ <pubmed> 25165593</pubmed>
- ↑ <pubmed>24890427</pubmed>
- ↑ <pubmed> 24741860</pubmed>
- ↑ <pubmed>2141655</pubmed>
- ↑ <pubmed>25333246</pubmed>
- ↑ <pubmed>25315294</pubmed>
- ↑ 40.0 40.1 <pubmed>7011178</pubmed>
- ↑ <pubmed>24116052</pubmed>
- ↑ <pubmed>PMC3365797</pubmed>
- ↑ 43.0 43.1 <pubmed>25227725</pubmed>
- ↑ <pubmed>10748766</pubmed>
- ↑ <pubmed>15838095</pubmed>
- ↑ <pubmed>24656295</pubmed>
- ↑ 47.0 47.1 <pubmed>24365350</pubmed>
- ↑ 48.0 48.1 48.2 48.3 48.4 48.5 48.6 17237341<pubmed>17237341</pubmed> Cite error: Invalid
<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content Cite error: Invalid<ref>tag; name 'PMID' defined multiple times with different content - ↑ <pubmed>15475933</pubmed>
- ↑ <pubmed>0001417</pubmed>
- ↑ 51.0 51.1 <pubmed>1769902</pubmed>
- ↑ 52.0 52.1 <pubmed>10510117</pubmed>
- ↑ 53.0 53.1 53.2 Subhalakshmi Wahengbam, S. Arunchandra Singh, Ningthoujam Damayanti Development and Morphogenesis of Testis in Human Fetuses. Journal of Anatomical Society of India: 2011, 60(2); 160-167, http://www.sciencedirect.com/science/article/pii/S0003277811800171
- ↑ 54.0 54.1 E. Sexson, J. Knezevich, ‘Male Hypogonadism: A Review of the Disease and Its Treatment’, U.S. Pharmacist:2010, http://www.uspharmacist.com/content/d/health%20systems/c/21150/
- ↑ 55.0 55.1 55.2 55.3 Danielle Evain-Brion, Malassine Andre Human placenta as an endocrine organ. Proceedings of the 34th International Symposium on Growth Hormone and Growth Factors in Endocrinology and Metabolism: 2003, 13;S37-S37, http://www.sciencedirect.com/science/article/pii/S1096637403000534# Cite error: Invalid
<ref>tag; name 'Evain-Brion' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Evain-Brion' defined multiple times with different content Cite error: Invalid<ref>tag; name 'Evain-Brion' defined multiple times with different content - ↑ 56.0 56.1 56.2 <pubmed>17624715</pubmed>
- ↑ Neil M Gude, Claire T Roberts, Bill Kalionis, Roger G King Growth and function of the normal human placenta. Thrombosis Research: 2004, 114(5-6); 397-407, http://www.sciencedirect.com/science/article/pii/S0049384804003421#
- ↑ <pubmed>25071761</pubmed>
- ↑ 59.0 59.1 <pubmed>23127895</pubmed>
- ↑ 60.0 60.1 <pubmed>21280984</pubmed>
- ↑ <pubmed>22912418</pubmed>
- ↑ <pubmed>23300903</pubmed>
- ↑ <pubmed>24757681</pubmed>
- ↑ 64.0 64.1 A. Peruffoa, M. Giacomellob, S. Montellia, M. Panina, B. Cozzia, ‘Expression profile of the pore-forming subunits α1A and α1D in the foetal bovine hypothalamus: A mammal with a long gestation.’ Neuroscience Letters. 556; pp 124–128 http://www.sciencedirect.com/science/article/pii/S0304394013009300